Potential of Impedance Flow Cytometry to Assess the Viability and Quantity of Cannabis sativa L. Pollen
Abstract
:1. Introduction
2. Results and Discussion
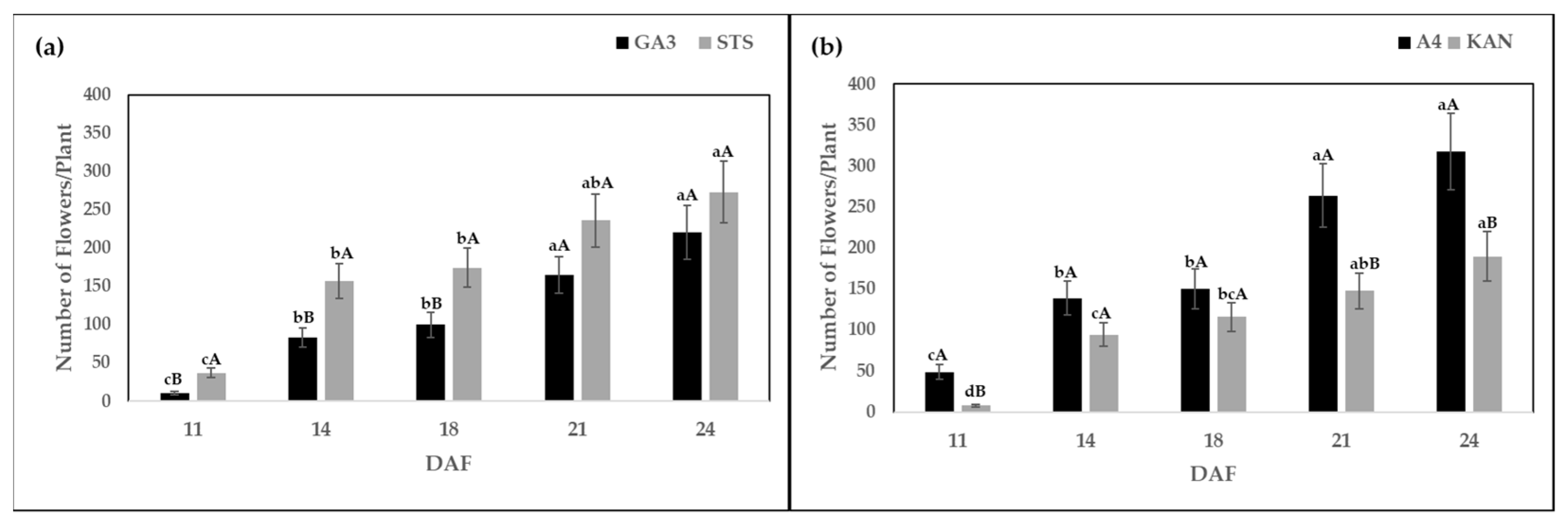
2.1. Number of Flowers Per Plant
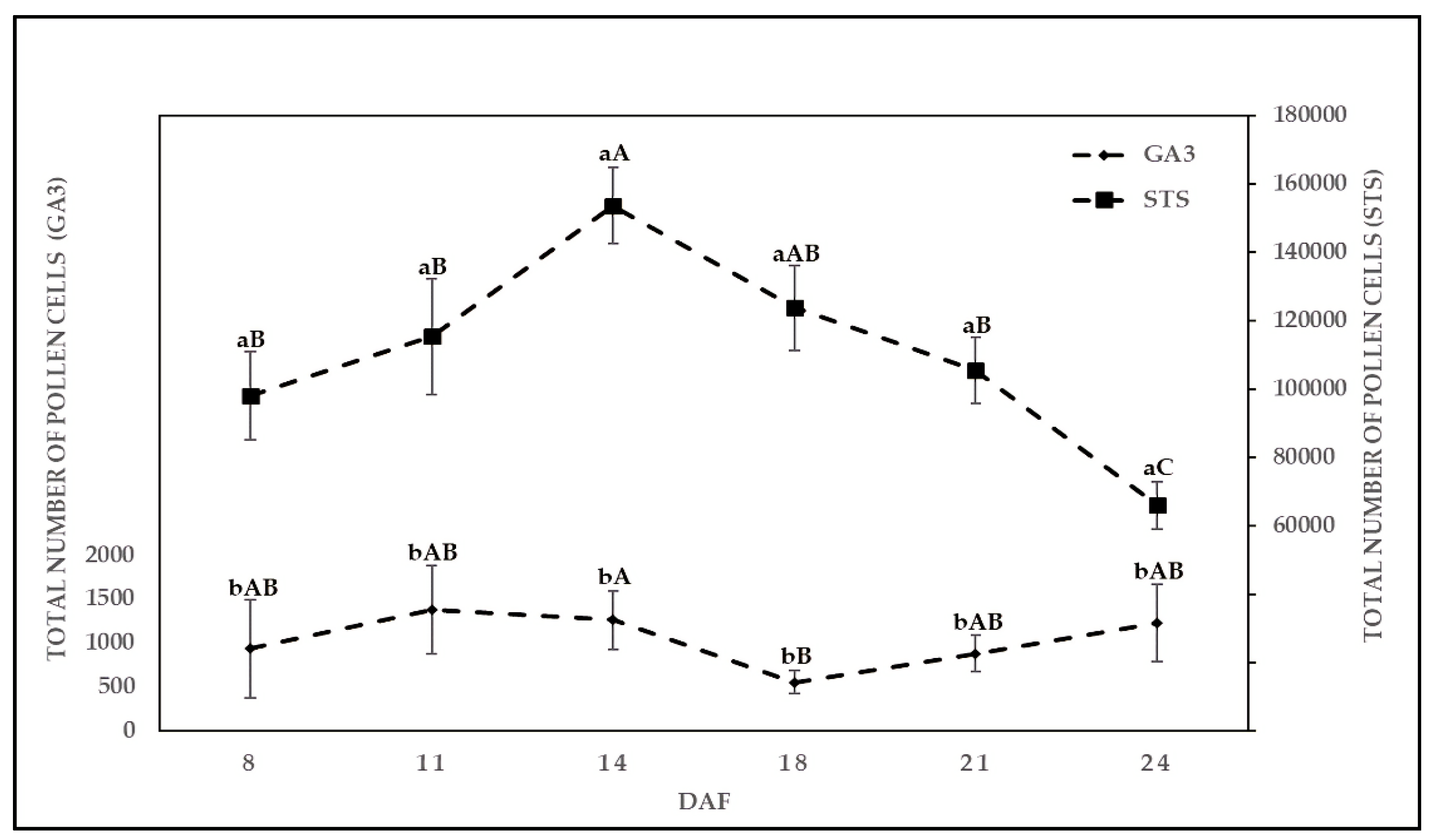
2.2. Effect of Chemical Solution on TPC at Various Sampling Dates
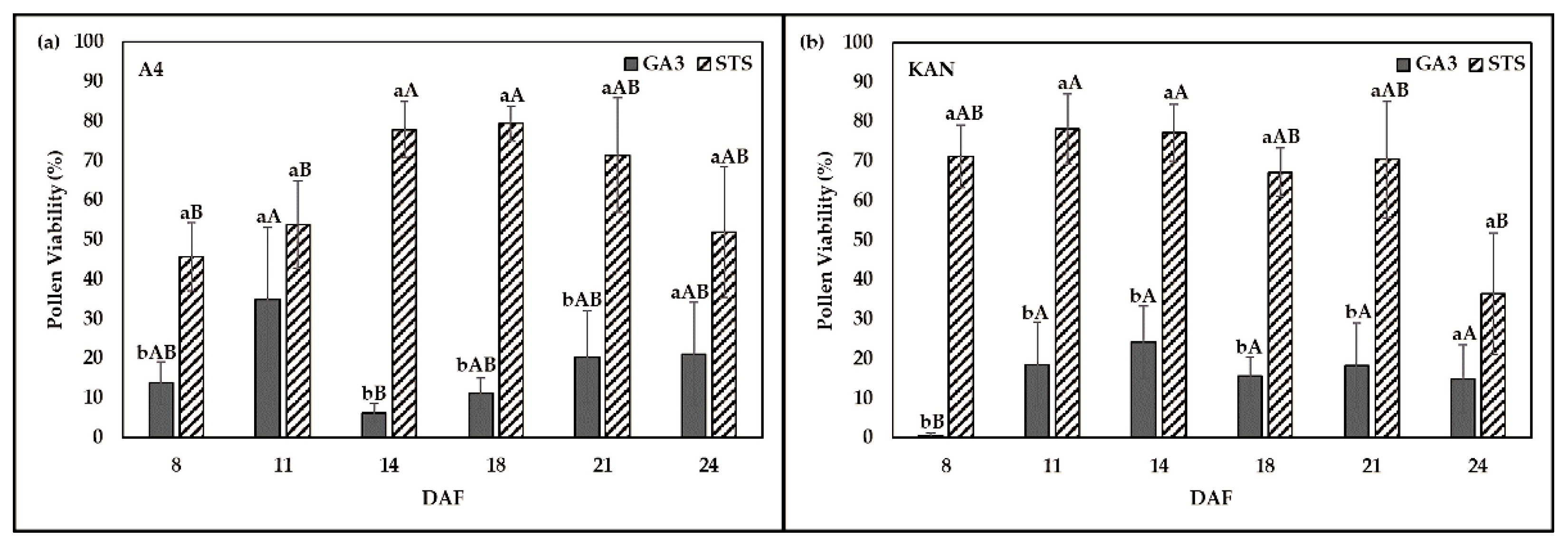
2.3. Effect of Chemical Solution on PV at Various Sampling Dates
3. Materials and Methods
3.1. Greenhouse Cultivation System
3.2. Preparation of Chemical Solutions
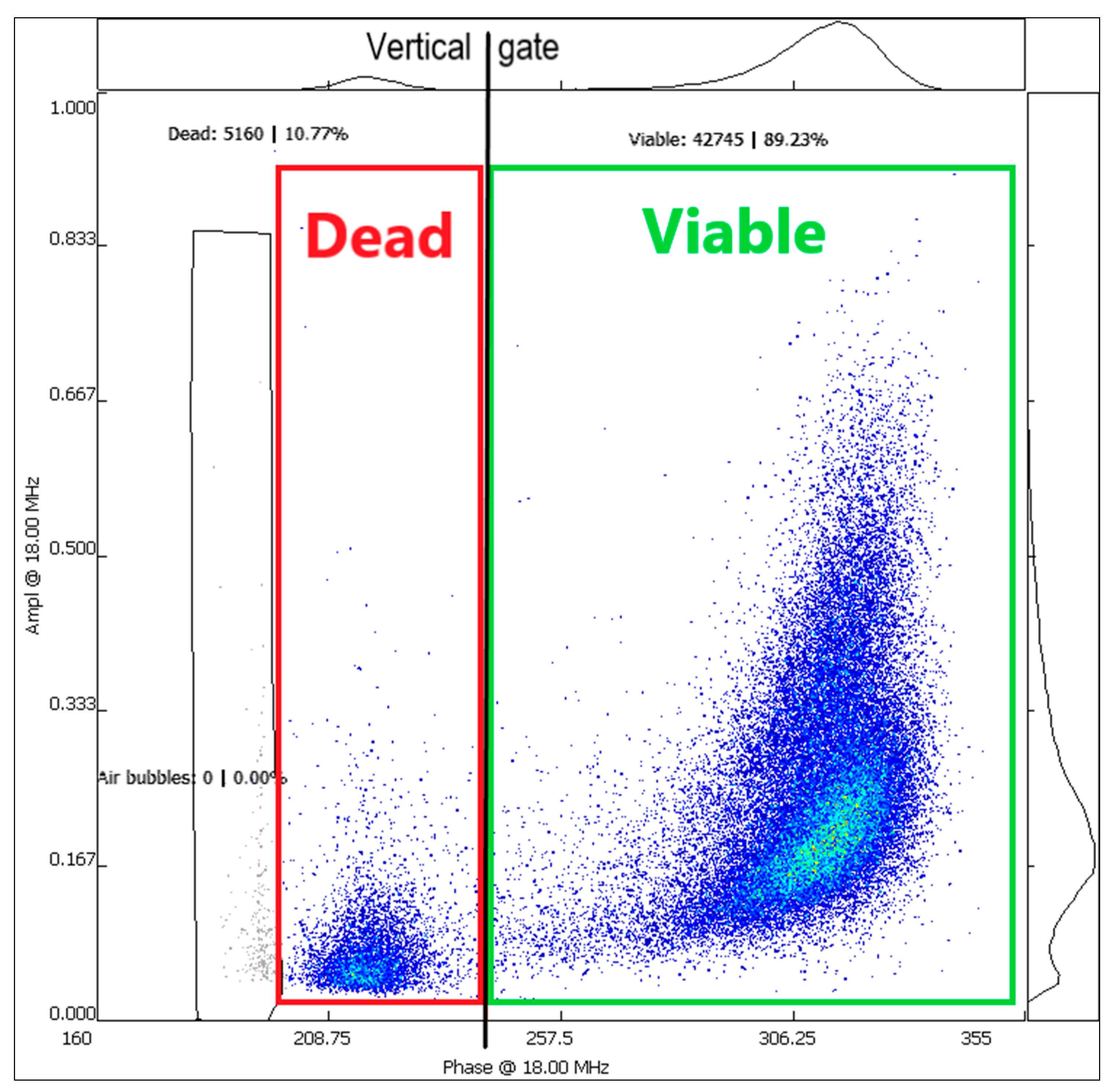
3.3. Pollen Viability Analysis with IFC
3.4. Sample Preparation and Measurements
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salentijn, E.M.J.; Zhang, Q.; Amaducci, S.; Yang, M.; Trindade, L.M. New developments in fiber hemp (Cannabis sativa L.) breeding. Ind. Crop Prod. 2015, 68, 32–41. [Google Scholar] [CrossRef]
- Martínez, V.; Iriondo De-Hond, A.; Borrelli, F.; Capasso, R.; Del Castillo, M.D.; Abalo, R. Cannabidiol and Other Non-Psychoactive Cannabinoids for Prevention and Treatment of Gastrointestinal Disorders: Useful Nutraceuticals? Int. J. Mol. Sci. 2020, 21, 3067. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Henry, P.; Shan, J.; Chen, J. Classification of cannabis strains in the Canadian market with discriminant analysis of principal components using genome-wide single nucleotide polymorphisms. PLoS ONE 2021, 16, e0253387. [Google Scholar] [CrossRef]
- Clarke, R.C.; Merlin, M.D. Cannabis Domestication, Breeding History, Present-day Genetic Diversity, and Future Prospects. Crit. Rev. Plant Sci. 2016, 35, 293–327. [Google Scholar] [CrossRef]
- Ming, R.; Bendahmane, A.; Renner, S.S. Sex Chromosomes in Land Plants. Annu. Rev. Plant Biol. 2011, 62, 485–514. [Google Scholar] [CrossRef] [Green Version]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [Green Version]
- Parsons, J.L.; Martin, S.L.; James, T.; Golenia, G.; Boudko, E.A.; Hepworth, S.R. Polyploidization for the Genetic Improvement of Cannabis sativa. Front. Plant Sci. 2019, 10, 476. [Google Scholar] [CrossRef]
- Campbell, L.G.; Peach, K.; Wizenberg, S.B. Dioecious hemp (Cannabis sativa L.) plants do not express significant sexually dimorphic morphology in the seedling stage. Sci. Rep. 2021, 11, 16–25. [Google Scholar] [CrossRef]
- Kaushal, S. Impact of Physical and Chemical Mutagens on Sex Expression in Cannabis sativa. Indian J. Fundament. Appl. Life Sci. 2012, 2, 97–103. [Google Scholar]
- Punja, Z.K.; Holmes, J.E. Hermaphroditism in Marijuana (Cannabis sativa L.) Inflorescences—Impact on Floral Morphology, Seed Formation, Progeny Sex Ratios, and Genetic Variation. Front. Plant Sci. 2020, 11, 718. [Google Scholar] [CrossRef] [PubMed]
- Ram, H.Y.M.; Jaiswal, V.S. Induction of male flowers on female plants of Cannabis sativa by gibberellins and its inhibition by abscisic acid. Planta 1972, 105, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Ram, H.Y.M.; Sett, R. Induction of fertile male flowers in genetically female Cannabis sativa plants by silver nitrate and silver thiosulphate anionic complex. Theor. Appl. Genet. 1982, 62, 369–375. [Google Scholar] [CrossRef]
- Rosenthal, E. Marijuana Grower’s Handbook; And/Or Press: Berkeley, CA, USA, 2009; ISBN 978-0-932551-46-7. [Google Scholar]
- Lubell, J.D.; Brand, M.H. Foliar Sprays of Silver Thiosulfate Produce Male Flowers on Female Hemp Plants. HortTechnology 2018, 28, 743–747. [Google Scholar] [CrossRef] [Green Version]
- McKernan, K.J.; Helbert, Y.; Kane, L.T.; Ebling, H.; Zhang, L.; Liu, B.; Eaton, Z.; McLaughlin, S.; Kingan, S.; Baybayan, P.; et al. Sequence and Annotation of 42 Cannabis Genomes Reveals Extensive Copy Number Variation in Cannabinoid Synthesis and Pathogen Resistance Genes. bioRxiv 2020, bioRxiv:2020.01.03.894428. [Google Scholar]
- García, C.C.; Nepi, M.; Pacini, E. It is a matter of timing: Asynchrony during pollen development and its consequences on pollen performance in angiosperms—A review. Protoplasma 2017, 254, 57–73. [Google Scholar] [CrossRef]
- Luria, G.; Rutley, N.; Lazar, I.; Harper, J.F.; Miller, G. Direct analysis of pollen fitness by flow cytometry: Implications for pollen response to stress. Plant J. 2019, 98, 942–952. [Google Scholar] [CrossRef]
- Rieu, I.; Twell, D.; Firon, N. Pollen Development at High Temperature: From Acclimation to Collapse. Plant Physiol. 2017, 173, 1967–1976. [Google Scholar] [CrossRef] [Green Version]
- Brunet, J.; Ziobro, R.; Osvatic, J.; Clayton, M.K. The effects of time, temperature and plant variety on pollen viability and its implications for gene flow risk. Plant Biol. 2019, 21, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Heslop-Harrison, J.; Heslop-Harrison, Y.; Shivanna, K.R. The evaluation of pollen quality, and a further appraisal of the fluorochromatic (FCR) test procedure. Theor. Appl. Genet. 1984, 67, 367–375. [Google Scholar] [CrossRef]
- Impe, D.; Reitz, J.; Köpnick, C.; Rolletschek, H.; Börner, A.; Senula, A.; Nagel, M. Assessment of Pollen Viability for Wheat. Front. Plant Sci. 2020, 10, 1588. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.M.; Gillings, M.R.; Altavilla, N.; Beattie, A.J. Potential problems with fluorescein diacetate assays of cell viability when testing natural products for antimicrobial activity. J. Microbiol. Methods 2001, 46, 261–267. [Google Scholar] [CrossRef]
- Peterson, R.; Slovin, J.P.; Chen, C. A simplified method for differential staining of aborted and non-aborted pollen grains. Int. J. Plant Biol. 2010, 1, 66–69. [Google Scholar] [CrossRef] [Green Version]
- Heidmann, I.; Schade-Kampmann, G.; Lambalk, J.; Ottiger, M.; Di Berardino, M. Impedance Flow Cytometry: A Novel Technique in Pollen Analysis. PLoS ONE 2016, 11, e0165531. [Google Scholar] [CrossRef]
- Rodríguez-Riaño, T.; Dafni, A. A new procedure to asses pollen viability. Sex. Plant Reprod. 2000, 12, 241–244. [Google Scholar] [CrossRef]
- Heidmann, I.; Di Berardino, M. Impedance flow cytometry as a tool to analyze microspore and pollen quality. In Plant Germline Development; Humana Press: New York, NY, USA, 2017; pp. 339–354. [Google Scholar] [CrossRef]
- Ascari, L.; Cristofori, V.; Macrì, F.; Botta, R.; Silvestri, C.; De Gregorio, T.; Huerta, E.S.; Di Berardino, M.; Kaufmann, S.; Siniscalco, C. Hazelnut Pollen Phenotyping Using Label-Free Impedance Flow Cytometry. Front. Plant Sci. 2020, 11, 615922. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Parvatam, G.; Ravishankar, G.A. AgNO3—a potential regulator of ethylene activity and plant growth modulator. Electron. J. Biotechnol. 2009, 12, 8–9. [Google Scholar] [CrossRef] [Green Version]
- Adal, A.M.; Doshi, K.; Holbrook, L.; Mahmoud, S.S. Comparative RNA-Seq analysis reveals genes associated with masculinization in female Cannabis sativa. Planta 2021, 253, 1–17. [Google Scholar] [CrossRef]
- Flajšman, M.; Slapnik, M.; Murovec, J. Production of Feminized Seeds of High CBD Cannabis sativa L. by Manipulation of Sex Expression and Its Application to Breeding. Front. Plant Sci. 2021, 12, 2380. [Google Scholar] [CrossRef]
- Nguyen, V.; Fleury, D.; Timmins, A.; Laga, H.; Hayden, M.; Mather, D.; Okada, T. Addition of rye chromosome 4R to wheat increases anther length and pollen grain number. Theor. Appl. Genet. 2015, 128, 953–964. [Google Scholar] [CrossRef]
- Hayes, J.D. Hybrid Wheat—Results and Problems, by A. A. Pickett. 259 pp. Berlin, Hamburg: Paul Parey (1993). DM 96.00 (paperback). ISBN 3 489 53510 3. J. Agric. Sci. 1993, 121, 294. [Google Scholar] [CrossRef]
- Green, G. The Cannabis Breeder’s Bible: The Definitive Guide to Marijuana Genetics, Cannabis: Botany and Creating Strains for the Seed Market; Green Candy Press: San Francisco, CA, USA, 2005; ISBN 978-1-931160-27-8. [Google Scholar]
- Anfinrud, M.N. Planting Hybrid Seed Production and Seed Quality Evaluation. Sunflower Technol. Prod. 2015, 35, 697–708. [Google Scholar] [CrossRef]
- Gaudet, D.; Yadav, N.S.; Sorokin, A.; Bilichak, A.; Kovalchuk, I. Development and Optimization of a Germination Assay and Long-Term Storage for Cannabis sativa Pollen. Plants 2020, 9, 665. [Google Scholar] [CrossRef]
- Reichel, P.; Munz, S.; Hartung, J.; Präger, A.; Kotiranta, S.; Burgel, L.; Schober, T.; Graeff-Hönninger, S. Impact of Three Different Light Spectra on the Yield, Morphology and Growth Trajectory of Three Different Cannabis sativa L. Strains. Plants 2021, 10, 1866. [Google Scholar] [CrossRef] [PubMed]
- Burgel, L.; Hartung, J.; Graeff-Hönninger, S. Impact of Different Growing Substrates on Growth, Yield and Cannabinoid Content of Two Cannabis sativa L. Genotypes in a Pot Culture. Horticulturae 2020, 6, 62. [Google Scholar] [CrossRef]
- Burgel, L.; Hartung, J.; Schibano, D.; Graeff-Hönninger, S. Impact of Different Phytohormones on Morphology, Yield and Cannabinoid Content of Cannabis sativa L. Plants 2020, 9, 725. [Google Scholar] [CrossRef]
| Source of Variation | Number of Flowers/Plant | TPC | PV |
|---|---|---|---|
| Genotype | <0.0001 | 0.2612 | 0.5753 |
| Solution | <0.0001 | <0.0001 | <0.0001 |
| DAF | <0.0001 | 0.0657 | 0.0194 |
| Genotype × Solution | 0.6967 | 0.3260 | 0.2789 |
| Genotype × DAF | 0.0003 | 0.1785 | 0.2582 |
| Solution × DAF | 0.0443 | 0.0266 | 0.0711 |
| Genotype × Solution × DAF | 0.0708 | 0.1666 | 0.0183 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rafiq, H.; Hartung, J.; Burgel, L.; Röll, G.; Graeff-Hönninger, S. Potential of Impedance Flow Cytometry to Assess the Viability and Quantity of Cannabis sativa L. Pollen. Plants 2021, 10, 2739. https://doi.org/10.3390/plants10122739
Rafiq H, Hartung J, Burgel L, Röll G, Graeff-Hönninger S. Potential of Impedance Flow Cytometry to Assess the Viability and Quantity of Cannabis sativa L. Pollen. Plants. 2021; 10(12):2739. https://doi.org/10.3390/plants10122739
Chicago/Turabian StyleRafiq, Hamza, Jens Hartung, Lisa Burgel, Georg Röll, and Simone Graeff-Hönninger. 2021. "Potential of Impedance Flow Cytometry to Assess the Viability and Quantity of Cannabis sativa L. Pollen" Plants 10, no. 12: 2739. https://doi.org/10.3390/plants10122739
APA StyleRafiq, H., Hartung, J., Burgel, L., Röll, G., & Graeff-Hönninger, S. (2021). Potential of Impedance Flow Cytometry to Assess the Viability and Quantity of Cannabis sativa L. Pollen. Plants, 10(12), 2739. https://doi.org/10.3390/plants10122739






