Improving the Health-Benefits of Kales (Brassica oleracea L. var. acephala DC) through the Application of Controlled Abiotic Stresses: A Review
Abstract
1. Introduction
2. Kale as a Novel Source of Nutraceuticals
2.1. Glucosinolates
Glucosinolates as Bioactive Compounds
2.2. Phenolic Compounds
Phenolic Compounds as Bioactive Compounds
2.3. Carotenoids
Carotenoids as Bioactive Compounds
3. Application of Controlled Abiotic Stresses as a Tool to Induce an Increase in the Content of Bioactive Compounds in Kale
3.1. Saline Stress Conditions
3.1.1. Sulfur as an Abiotic Stressor
3.1.2. Selenium as an Abiotic Stressor
3.1.3. NaCl as an Abiotic Stressor
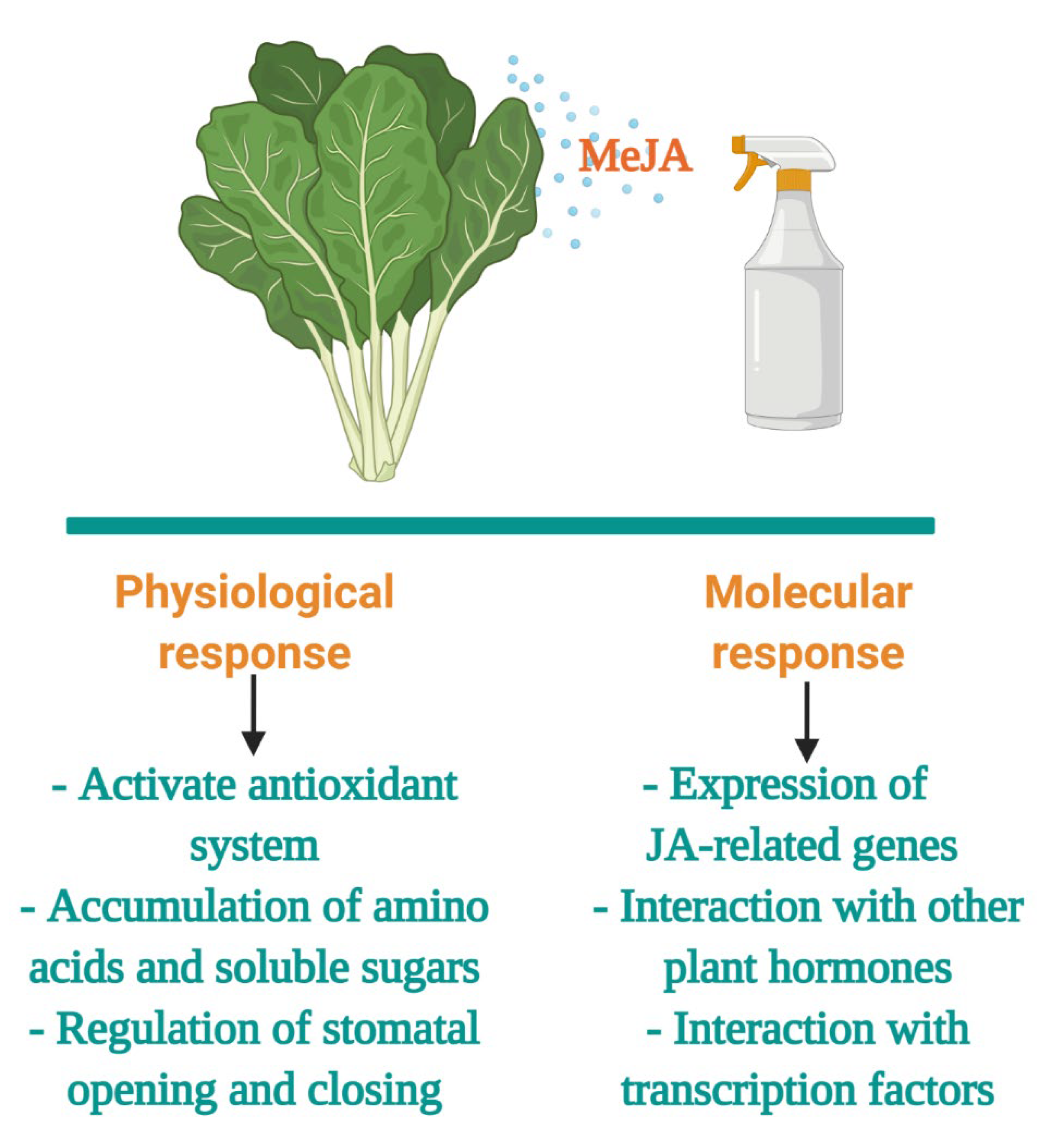
3.2. Methyl Jasmonate as an Abiotic Stressor
3.3. Drought as an Abiotic Stressor
3.4. Temperature as an Abiotic Stressor
3.5. Radiation as an Abiotic Stressor
4. Drawbacks of Using Abiotic Stresses at a Large Scale to Increase the Content of Bioactive Compounds in Kale
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vidal, N.P.; Pham, H.T.; Manful, C.; Pumphrey, R.; Nadeem, M.; Cheema, M.; Galagedara, L.; Leke-Aladekoba, A.; Abbey, L.; Thomas, R. The Use of Natural Media Amendments to Produce Kale Enhanced with Functional Lipids in Controlled Environment Production System. Sci. Rep. 2018, 8, 14771. [Google Scholar] [CrossRef]
- Šamec, D.; Urlić, B.; Salopek-Sondi, B. Kale (Brassica oleracea var. acephala) as a Superfood: Review of the Scientific Evidence behind the Statement. Crit. Rev. Food Sci. Nutr. 2019, 59, 2411–2422. [Google Scholar] [CrossRef] [PubMed]
- USDA. Agricultural Research Service: Kale Raw. 2017. Available online: https://fdc.nal.usda.gov/fdc-app.html#/food-details/323505/nutrients (accessed on 3 October 2020).
- Lemos, M.; Santin, J.R.; Júnior, L.C.K.; Niero, R.; de Andrade, S.F. Gastroprotective Activity of Hydroalcoholic Extract Obtained from the Leaves of Brassica oleracea var. acephala DC in Different Animal Models. J. Ethnopharmacol. 2011, 138, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Kuerban, A.; Yaghmoor, S.S.; Almulaiky, Y.Q.; Mohamed, Y.A.; Razvi, S.S.I.; Hasan, M.N.; Moselhy, S.S.; Al-Ghafari, A.B.; Alsufiani, H.M.; Kumosani, T.A.; et al. Therapeutic Effects of Phytochemicals of Brassicaceae for Management of Obesity. J. Pharm. Res. Int. 2017, 19, 1–11. [Google Scholar] [CrossRef]
- Šamec, D.; Salopek-Sondi, B. Chapter 3.11—Cruciferous (Brassicaceae) Vegetables. In Nonvitamin and Nonmineral Nutritional Supplements; Nabavi, S.M., Silva, A.S., Eds.; Academic Press: San Diego, CA, USA, 2019; pp. 195–202. ISBN 978-0-12-812491-8. [Google Scholar]
- Abellán, Á.; Domínguez-Perles, R.; Moreno, D.A.; García-Viguera, C. Sorting out the Value of Cruciferous Sprouts as Sources of Bioactive Compounds for Nutrition and Health. Nutrients 2019, 11, 429. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Keum, Y.-S. Significance of Genetic, Environmental, and Pre- and Postharvest Factors Affecting Carotenoid Contents in Crops: A Review. J. Agric. Food Chem. 2018, 66, 5310–5324. [Google Scholar] [CrossRef] [PubMed]
- Cisneros-Zevallos, L. The Use of Controlled Postharvest Abiotic Stresses as a Tool for Enhancing the Nutraceutical Content and Adding-Value of Fresh Fruits and Vegetables. J. Food Sci. 2003, 68, 1560–1565. [Google Scholar] [CrossRef]
- Jacobo-Velázquez, D.A.; González-Agüero, M.; Cisneros-Zevallos, L. Cross-Talk between Signaling Pathways: The Link between Plant Secondary Metabolite Production and Wounding Stress Response. Sci. Rep. 2015, 5, 8608. [Google Scholar] [CrossRef]
- Dudeja, P.; Gupta, R.K. Chapter 40—Nutraceuticals. In Food Safety in the 21st Century; Gupta, R.K., Dudeja, S.M., Eds.; Academic Press: San Diego, CA, USA, 2017; pp. 491–496. ISBN 978-0-12-801773-9. [Google Scholar]
- Biegańska-Marecik, R.; Radziejewska-Kubzdela, E.; Marecik, R. Characterization of Phenolics, Glucosinolates and Antioxidant Activity of Beverages Based on Apple Juice with Addition of Frozen and Freeze-Dried Curly Kale Leaves (Brassica oleracea L. var. acephala L.). Food Chem. 2017, 230, 271–280. [Google Scholar] [CrossRef]
- Johnson, M.; McElhenney, W.H.; Egnin, M. Influence of Green Leafy Vegetables in Diets with an Elevated ω-6:ω-3 Fatty Acid Ratio on Rat Blood Pressure, Plasma Lipids, Antioxidant Status and Markers of Inflammation. Nutrients 2019, 11, 301. [Google Scholar] [CrossRef]
- Olsen, H.; Grimmer, S.; Aaby, K.; Saha, S.; Borge, G.I.A. Antiproliferative Effects of Fresh and Thermal Processed Green and Red Cultivars of Curly Kale (Brassica oleracea L. convar. acephala var. sabellica). J. Agric. Food Chem. 2012, 60, 7375–7383. [Google Scholar] [CrossRef]
- Luang-In, V.; Saengha, W.; Buranrat, B.; Chantiratikul, A.; Ma, N.; Ma, N. Cytotoxicity of Selenium-Enriched Chinese Kale (Brassica oleracea var. alboglabra L.) Seedlings Against Caco-2, MCF-7 and HepG2 Cancer Cells. Pharmacogn. J. 2020, 12, 674–681. [Google Scholar] [CrossRef]
- Sikora, E.; Bodziarczyk, I. Influence of Diet with Kale on Lipid Peroxides and Malondialdehyde Levels in Blood Serum of Laboratory Rats over Intoxication with Paraquat. Acta Sci. Pol. Technol. Aliment. 2013, 12, 91–99. [Google Scholar]
- Horst, M.A.; Ong, T.P.; Jordão, A.A., Jr.; Vannucchi, H.; Moreno, F.S.; Lajolo, F.M. Water Extracts of Cabbage and Kale Inhibit Ex Vivo H2O2-Induced DNA Damage but Not Rat Hepatocarcinogenesis. Braz. J. Med. Biol. Res. 2010, 43, 242–248. [Google Scholar] [CrossRef]
- Chung, E.J.; Kim, S.Y.; Nam, Y.J.; Park, J.H.; Hwang, H.J.; Lee-Kim, Y.C. Effects of Kale Juice Powder on Serum Lipids, Folate and Plasma Homocysteine Levels in Growing Rats. J. Korean Soc. Food Sci. Nutr. 2005, 34, 1175–1181. [Google Scholar]
- Giacoppo, S.; Galuppo, M.; De Nicola, G.R.; Iori, R.; Bramanti, P.; Mazzon, E. Tuscan Black Kale Sprout Extract Bioactivated with Myrosinase: A Novel Natural Product for Neuroprotection by Inflammatory and Oxidative Response during Cerebral Ischemia/Reperfusion Injury in Rat. BMC Complement. Altern. Med. 2015, 15, 397. [Google Scholar] [CrossRef]
- Lima de Albuquerque, C.; Comalada, M.; Camuesco, D.; Rodríguez-Cabezas, M.E.; Luiz-Ferreira, A.; Nieto, A.; Monteiro de Souza Brito, A.R.; Zarzuelo, A.; Gálvez, J. Effect of Kale and Papaya Supplementation in Colitis Induced by Trinitrobenzenesulfonic Acid in the Rat. E-SPEN Eur. E-J. Clin. Nutr. Metab. 2010, 5, e111–e116. [Google Scholar] [CrossRef]
- Ide, T.; Suzuki, A.; Kurokawa, M.; Minagawa, N.; Inuzuka, H.; Ichien, G. Analysis Of Effects Of Kale Juice Consumption Among Subjects With Potential Metabolic Syndrome: A Prospective Single-Arm Clinical Study. J. Hypertens. Cardiol. 2016, 2, 25–38. [Google Scholar] [CrossRef][Green Version]
- Kondo, S.; Suzuki, A.; Kurokawa, M.; Hasumi, K. Intake of Kale Suppresses Postprandial Increases in Plasma Glucose: A Randomized, Double-Blind, Placebo-Controlled, Crossover Study. Biomed. Rep. 2016, 5, 553–558. [Google Scholar] [CrossRef]
- Kim, S.Y.; Yoon, S.; Kwon, S.M.; Park, K.S.; Lee-Kim, Y.C. Kale Juice Improves Coronary Artery Disease Risk Factors in Hypercholesterolemic Men. Biomed. Environ. Sci. 2008, 21, 91–97. [Google Scholar] [CrossRef]
- Possenti, M.; Baima, S.; Raffo, A.; Durazzo, A.; Giusti, A.M.; Natella, F. Glucosinolates in Food. In Glucosinolates; Mérillon, J.-M., Ramawat, K.G., Eds.; Series in Phytochemistry; Springer International Publishing: Cham, Switzerland, 2017; pp. 87–132. ISBN 978-3-319-25462-3. [Google Scholar]
- Ishida, M.; Hara, M.; Fukino, N.; Kakizaki, T.; Morimitsu, Y. Glucosinolate Metabolism, Functionality and Breeding for the Improvement of Brassicaceae Vegetables. Breed. Sci. 2014, 64, 48–59. [Google Scholar] [CrossRef]
- Narbad, A.; Rossiter, J.T. Gut Glucosinolate Metabolism and Isothiocyanate Production. Mol. Nutr. Food Res. 2018, 62, 1700991. [Google Scholar] [CrossRef]
- Barba, F.J.; Nikmaram, N.; Roohinejad, S.; Khelfa, A.; Zhu, Z.; Koubaa, M. Bioavailability of Glucosinolates and Their Breakdown Products: Impact of Processing. Front. Nutr. 2016, 3, 24. [Google Scholar] [CrossRef] [PubMed]
- Chhajed, S.; Mostafa, I.; He, Y.; Abou-Hashem, M.; El-Domiaty, M.; Chen, S. Glucosinolate Biosynthesis and the Glucosinolate–Myrosinase System in Plant Defense. Agronomy 2020, 10, 1786. [Google Scholar] [CrossRef]
- Keck, A.-S.; Finley, J.W. Cruciferous Vegetables: Cancer Protective Mechanisms of Glucosinolate Hydrolysis Products and Selenium. Integr. Cancer Ther. 2004, 3, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic Compounds in Brassica Vegetables. Molecules 2010, 16, 251–280. [Google Scholar] [CrossRef]
- Bhandari, S.R.; Jo, J.S.; Lee, J.G. Comparison of Glucosinolate Profiles in Different Tissues of Nine Brassica Crops. Molecules 2015, 20, 15827–15841. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Tian, Y.-X.; Chen, Q.; Zhang, Y.; Luo, Y.; Wang, Y.; Li, M.-Y.; Gong, R.-G.; Wang, X.-R.; Zhang, F.; et al. Variations in the Glucosinolates of the Individual Edible Parts of Three Stem Mustards (Brassica Juncea). R. Soc. Open Sci. 2019, 6, 182054. [Google Scholar] [CrossRef]
- Hahn, C.; Müller, A.; Kuhnert, N.; Albach, D. Diversity of Kale (Brassica oleracea var. sabellica): Glucosinolate Content and Phylogenetic Relationships. J. Agric. Food Chem. 2016, 64, 3215–3225. [Google Scholar] [CrossRef]
- Mazumder, A.; Dwivedi, A.; du Plessis, J. Sinigrin and Its Therapeutic Benefits. Molecules 2016, 21, 416. [Google Scholar] [CrossRef]
- Lopez-Rodriguez, N.A.; Gaytán-Martínez, M.; de la Luz Reyes-Vega, M.; Loarca-Piña, G. Glucosinolates and Isothiocyanates from Moringa Oleifera: Chemical and Biological Approaches. Plant Foods Hum. Nutr. Dordr. Neth. 2020, 75, 447–457. [Google Scholar] [CrossRef]
- Dufour, V.; Stahl, M.; Baysse, C. The Antibacterial Properties of Isothiocyanates. Microbiol. Read. Engl. 2015, 161, 229–243. [Google Scholar] [CrossRef]
- Kołodziejski, D.; Piekarska, A.; Hanschen, F.S.; Pilipczuk, T.; Tietz, F.; Kusznierewicz, B.; Bartoszek, A. Relationship between Conversion Rate of Glucosinolates to Isothiocyanates/Indoles and Genotoxicity of Individual Parts of Brassica Vegetables. Eur. Food Res. Technol. 2019, 245, 383–400. [Google Scholar] [CrossRef]
- Yin, T.-F.; Wang, M.; Qing, Y.; Lin, Y.-M.; Wu, D. Research Progress on Chemopreventive Effects of Phytochemicals on Colorectal Cancer and Their Mechanisms. World J. Gastroenterol. 2016, 22, 7058–7068. [Google Scholar] [CrossRef]
- Mantso, T.; Anestopoulos, I.; Lamprianidou, E.; Kotsianidis, I.; Pappa, A.; Panayiotidis, M.I. Isothiocyanate-Induced Cell Cycle Arrest in a Novel In Vitro Exposure Protocol of Human Malignant Melanoma (A375) Cells. Anticancer Res. 2019, 39, 591–596. [Google Scholar] [CrossRef]
- Jaafaru, M.S.; Abd Karim, N.A.; Mohamed Eliaser, E.; Maitalata Waziri, P.; Ahmed, H.; Mustapha Barau, M.; Kong, L.; Abdull Razis, A.F. Nontoxic Glucomoringin-Isothiocyanate (GMG-ITC) Rich Soluble Extract Induces Apoptosis and Inhibits Proliferation of Human Prostate Adenocarcinoma Cells (PC-3). Nutrients 2018, 10, 1174. [Google Scholar] [CrossRef]
- Lin, J.; Xu, Y.; Zhao, X.; Qiu, Z. Anticancer Activity of Sulforaphane against Human Hepatoblastoma Cells Involves Apoptosis, Autophagy and Inhibition of β-Catenin Signaling Pathway. Arch. Med. Sci. 2020, 16, 1–9. [Google Scholar] [CrossRef]
- Serini, S.; Guarino, R.; Ottes Vasconcelos, R.; Celleno, L.; Calviello, G. The Combination of Sulforaphane and Fernblock® XP Improves Individual Beneficial Effects in Normal and Neoplastic Human Skin Cell Lines. Nutrients 2020, 12, 1608. [Google Scholar] [CrossRef]
- Georgikou, C.; Buglioni, L.; Bremerich, M.; Roubicek, N.; Yin, L.; Gross, W.; Sticht, C.; Bolm, C.; Herr, I. Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects. Biomolecules 2020, 10, 769. [Google Scholar] [CrossRef]
- Mitsiogianni, M.; Trafalis, D.T.; Franco, R.; Zoumpourlis, V.; Pappa, A.; Panayiotidis, M.I. Sulforaphane and Iberin Are Potent Epigenetic Modulators of Histone Acetylation and Methylation in Malignant Melanoma. Eur. J. Nutr. 2021, 60, 147–158. [Google Scholar] [CrossRef]
- Pocasap, P.; Weerapreeyakul, N.; Thumanu, K. Alyssin and Iberin in Cruciferous Vegetables Exert Anticancer Activity in HepG2 by Increasing Intracellular Reactive Oxygen Species and Tubulin Depolymerization. Biomol. Ther. 2019, 27, 540–552. [Google Scholar] [CrossRef]
- Esteve, M. Mechanisms Underlying Biological Effects of Cruciferous Glucosinolate-Derived Isothiocyanates/Indoles: A Focus on Metabolic Syndrome. Front. Nutr. 2020, 7, 111. [Google Scholar] [CrossRef]
- Nagata, N.; Xu, L.; Kohno, S.; Ushida, Y.; Aoki, Y.; Umeda, R.; Fuke, N.; Zhuge, F.; Ni, Y.; Nagashimada, M.; et al. Glucoraphanin Ameliorates Obesity and Insulin Resistance Through Adipose Tissue Browning and Reduction of Metabolic Endotoxemia in Mice. Diabetes 2017, 66, 1222–1236. [Google Scholar] [CrossRef]
- Chen, J.; Bao, C.; Kim, J.T.; Cho, J.S.; Qiu, S.; Lee, H.J. Sulforaphene Inhibition of Adipogenesis via Hedgehog Signaling in 3T3-L1 Adipocytes. J. Agric. Food Chem. 2018, 66, 11926–11934. [Google Scholar] [CrossRef]
- Chae, S.Y.; Seo, S.G.; Yang, H.; Yu, J.G.; Suk, S.J.; Jung, E.S.; Ji, H.; Kwon, J.Y.; Lee, H.J.; Lee, K.W. Anti-Adipogenic Effect of Erucin in Early Stage of Adipogenesis by Regulating Ras Activity in 3T3-L1 Preadipocytes. J. Funct. Foods 2015, 19, 700–709. [Google Scholar] [CrossRef]
- Yang, H.; Seo, S.G.; Shin, S.H.; Min, S.; Kang, M.J.; Yoo, R.; Kwon, J.Y.; Yue, S.; Kim, K.H.; Cheng, J.-X.; et al. 3,3′-Diindolylmethane Suppresses High-Fat Diet-Induced Obesity through Inhibiting Adipogenesis of Pre-Adipocytes by Targeting USP2 Activity. Mol. Nutr. Food Res. 2017, 61, 1700119. [Google Scholar] [CrossRef]
- Chang, H.-P.; Wang, M.-L.; Hsu, C.-Y.; Liu, M.-E.; Chan, M.-H.; Chen, Y.-H. Suppression of Inflammation-Associated Factors by Indole-3-Carbinol in Mice Fed High-Fat Diets and in Isolated, Co-Cultured Macrophages and Adipocytes. Int. J. Obes. 2011, 35, 1530–1538. [Google Scholar] [CrossRef][Green Version]
- Yao, A.; Shen, Y.; Wang, A.; Chen, S.; Zhang, H.; Chen, F.; Chen, Z.; Wei, H.; Zou, Z.; Shan, Y.; et al. Sulforaphane Induces Apoptosis in Adipocytes via Akt/P70s6k1/Bad Inhibition and ERK Activation. Biochem. Biophys. Res. Commun. 2015, 465, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.-M.; Lee, Y.-S.; Kim, W.; Kim, S.J.; Shin, K.-O.; Yu, J.-Y.; Lee, M.K.; Lee, Y.-M.; Hong, J.T.; Yun, Y.-P.; et al. Sulforaphane Attenuates Obesity by Inhibiting Adipogenesis and Activating the AMPK Pathway in Obese Mice. J. Nutr. Biochem. 2014, 25, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Chuang, W.-T.; Liu, Y.-T.; Huang, C.-S.; Lo, C.-W.; Yao, H.-T.; Chen, H.-W.; Lii, C.-K. Benzyl Isothiocyanate and Phenethyl Isothiocyanate Inhibit Adipogenesis and Hepatosteatosis in Mice with Obesity Induced by a High-Fat Diet. J. Agric. Food Chem. 2019, 67, 7136–7146. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-X.; Gao, J.-G.; Wan, X.-Y.; Chen, Y.; Xu, C.-F.; Feng, Z.-M.; Zeng, H.; Lin, Y.-M.; Ma, H.; Xu, P.; et al. Allyl Isothiocyanate Ameliorates Lipid Accumulation and Inflammation in Nonalcoholic Fatty Liver Disease via the Sirt1/AMPK and NF-κB Signaling Pathways. World J. Gastroenterol. 2019, 25, 5120–5133. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Tohidi, M.; Nazeri, P.; Mehran, M.; Azizi, F.; Mirmiran, P. Effect of Broccoli Sprouts on Insulin Resistance in Type 2 Diabetic Patients: A Randomized Double-Blind Clinical Trial. Int. J. Food Sci. Nutr. 2012, 63, 767–771. [Google Scholar] [CrossRef]
- López-Chillón, M.T.; Carazo-Díaz, C.; Prieto-Merino, D.; Zafrilla, P.; Moreno, D.A.; Villaño, D. Effects of Long-Term Consumption of Broccoli Sprouts on Inflammatory Markers in Overweight Subjects. Clin. Nutr. 2019, 38, 745–752. [Google Scholar] [CrossRef]
- Kikuchi, M.; Ushida, Y.; Shiozawa, H.; Umeda, R.; Tsuruya, K.; Aoki, Y.; Suganuma, H.; Nishizaki, Y. Sulforaphane-Rich Broccoli Sprout Extract Improves Hepatic Abnormalities in Male Subjects. World J. Gastroenterol. 2015, 21, 12457–12467. [Google Scholar] [CrossRef]
- Shapiro, T.A.; Fahey, J.W.; Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Stephenson, K.K.; Wade, K.L.; Ye, L.; Talalay, P. Safety, Tolerance, and Metabolism of Broccoli Sprout Glucosinolates and Isothiocyanates: A Clinical Phase I Study. Nutr. Cancer 2006, 55, 53–62. [Google Scholar] [CrossRef]
- Yagi, M.; Nakatsuji, Y.; Maeda, A.; Ota, H.; Kamikubo, R.; Miyoshi, N.; Nakamura, Y.; Akagawa, M. Phenethyl Isothiocyanate Activates Leptin Signaling and Decreases Food Intake. PLoS ONE 2018, 13, e0206748. [Google Scholar] [CrossRef]
- Jayakumar, P.; Pugalendi, K.V.; Sankaran, M. Attenuation of Hyperglycemia-Mediated Oxidative Stress by Indole-3-Carbinol and Its Metabolite 3,3′- Diindolylmethane in C57BL/6J Mice. J. Physiol. Biochem. 2014, 70, 525–534. [Google Scholar] [CrossRef]
- Chinni, S.R.; Sarkar, F.H. Akt Inactivation Is a Key Event in Indole-3-Carbinol-Induced Apoptosis in PC-3 Cells. Clin. Cancer Res. 2002, 8, 1228–1236. [Google Scholar]
- Chen, D.Z.; Qi, M.; Auborn, K.J.; Carter, T.H. Indole-3-Carbinol and Diindolylmethane Induce Apoptosis of Human Cervical Cancer Cells and in Murine HPV16-Transgenic Preneoplastic Cervical Epithelium. J. Nutr. 2001, 131, 3294–3302. [Google Scholar] [CrossRef]
- Choi, Y.; Kim, Y.; Park, S.; Lee, K.W.; Park, T. Indole-3-Carbinol Prevents Diet-Induced Obesity through Modulation of Multiple Genes Related to Adipogenesis, Thermogenesis or Inflammation in the Visceral Adipose Tissue of Mice. J. Nutr. Biochem. 2012, 23, 1732–1739. [Google Scholar] [CrossRef]
- Meng, Q.; Yuan, F.; Goldberg, I.D.; Rosen, E.M.; Auborn, K.; Fan, S. Indole-3-Carbinol Is a Negative Regulator of Estrogen Receptor-Alpha Signaling in Human Tumor Cells. J. Nutr. 2000, 130, 2927–2931. [Google Scholar] [CrossRef]
- Garikapaty, V.P.S.; Ashok, B.T.; Chen, Y.G.; Mittelman, A.; Iatropoulos, M.; Tiwari, R.K. Anti-Carcinogenic and Anti-Metastatic Properties of Indole-3-Carbinol in Prostate Cancer. Oncol. Rep. 2005, 13, 89–93. [Google Scholar] [CrossRef]
- Ayad, R.; Akkal, S. Chapter 12—Phytochemistry and Biological Activities of Algerian Centaurea and Related Genera. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Bioactive Natural Products; Elsevier: Amsterdam, The Netherlands, 2019; Volume 63, pp. 357–414. [Google Scholar]
- Lu, L.; Xu, L.; Guo, Y.; Zhang, D.; Qi, T.; Jin, L.; Gu, G.; Xu, L.; Xiao, M. Glycosylation of Phenolic Compounds by the Site-Mutated β-Galactosidase from Lactobacillus Bulgaricus L3. PLoS ONE 2015, 10, e0121445. [Google Scholar] [CrossRef]
- Hussain, M.B.; Hassan, S.; Waheed, M.; Javed, A.; Farooq, M.A.; Tahir, A. Bioavailability and Metabolic Pathway of Phenolic Compounds; IntechOpen: London, UK, 2019; ISBN 978-1-78984-034-6. [Google Scholar]
- Botelho, G.; Canas, S.; Lameiras, J. 14—Development of Phenolic Compounds Encapsulation Techniques as a Major Challenge for Food Industry and for Health and Nutrition Fields. In Nutrient Delivery; Grumezescu, A.M., Ed.; Nanotechnology in the Agri-Food Industry; Academic Press: San Diego, CA, USA, 2017; pp. 535–586. ISBN 978-0-12-804304-2. [Google Scholar]
- Akdaş, Z.Z.; Bakkalbaşı, E. Influence of Different Cooking Methods on Color, Bioactive Compounds, and Antioxidant Activity of Kale. Int. J. Food Prop. 2017, 20, 877–887. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Hu, L.-P.; Liu, G.-M.; Zhang, D.-S.; He, H.-J. Evaluation of the Nutritional Quality of Chinese Kale (Brassica alboglabra bailey) Using UHPLC-Quadrupole-Orbitrap MS/MS-Based Metabolomics. Molecules 2017, 22, 1262. [Google Scholar] [CrossRef]
- Jeon, J.; Kim, J.K.; Kim, H.; Kim, Y.J.; Park, Y.J.; Kim, S.J.; Kim, C.; Park, S.U. Transcriptome Analysis and Metabolic Profiling of Green and Red Kale (Brassica oleracea var. acephala) Seedlings. Food Chem. 2018, 241, 7–13. [Google Scholar] [CrossRef]
- Ferioli, F.; Giambanelli, E.; D’Antuono, L.F.; Costa, H.S.; Albuquerque, T.G.; Silva, A.S.; Hayran, O.; Koçaoglu, B. Comparison of Leafy Kale Populations from Italy, Portugal, and Turkey for Their Bioactive Compound Content: Phenolics, Glucosinolates, Carotenoids, and Chlorophylls. J. Sci. Food Agric. 2013, 93, 3478–3489. [Google Scholar] [CrossRef] [PubMed]
- Serçe, A.; Toptancı, B.Ç.; Tanrıkut, S.E.; Altaş, S.; Kızıl, G.; Kızıl, S.; Kızıl, M. Assessment of the Antioxidant Activity of Silybum Marianum Seed Extract and Its Protective Effect against DNA Oxidation, Protein Damage and Lipid Peroxidation. Food Technol. Biotechnol. 2016, 54, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kang, M.J.; Hur, G.; Lee, T.K.; Park, I.S.; Seo, S.G.; Yu, J.G.; Song, Y.S.; Park, J.H.Y.; Lee, K.W. Sulforaphene Suppresses Adipocyte Differentiation via Induction of Post-Translational Degradation of CCAAT/Enhancer Binding Protein Beta (C/EBPβ). Nutrients 2020, 12, E758. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.J.; Kwon, Y.S.; Park, S.M.; Shin, T.; Park, J.H.; Kim, H.C.; Kwon, M.S.; Wie, M.B. Quercetin Attenuates Oxygen-Glucose Deprivation- and Excitotoxin-Induced Neurotoxicity in Primary Cortical Cell Cultures. Biol. Pharm. Bull. 2003, 26, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Perez, A.; Gonzalez-Manzano, S.; Jimenez, R.; Perez-Abud, R.; Haro, J.M.; Osuna, A.; Santos-Buelga, C.; Duarte, J.; Perez-Vizcaino, F. The Flavonoid Quercetin Induces Acute Vasodilator Effects in Healthy Volunteers: Correlation with Beta-Glucuronidase Activity. Pharmacol. Res. 2014, 89, 11–18. [Google Scholar] [CrossRef]
- Hämäläinen, M.; Nieminen, R.; Asmawi, M.Z.; Vuorela, P.; Vapaatalo, H.; Moilanen, E. Effects of Flavonoids on Prostaglandin E2 Production and on COX-2 and MPGES-1 Expressions in Activated Macrophages. Planta Med. 2011, 77, 1504–1511. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, S.; Mei, H.; Xuan, J.; Guo, X.; Couch, L.; Dobrovolsky, V.N.; Guo, L.; Mei, N. Ginkgo Biloba Leaf Extract Induces DNA Damage by Inhibiting Topoisomerase II Activity in Human Hepatic Cells. Sci. Rep. 2015, 5, 14633. [Google Scholar] [CrossRef]
- Shimojo, Y.; Ozawa, Y.; Toda, T.; Igami, K.; Shimizu, T. Probiotic Lactobacillus Paracasei A221 Improves the Functionality and Bioavailability of Kaempferol-Glucoside in Kale by Its Glucosidase Activity. Sci. Rep. 2018, 8, 9239. [Google Scholar] [CrossRef]
- Ayaz, F.A.; Hayırlıoglu-Ayaz, S.; Alpay-Karaoglu, S.; Grúz, J.; Valentová, K.; Ulrichová, J.; Strnad, M. Phenolic Acid Contents of Kale (Brassica oleraceae L. var. acephala DC.) Extracts and Their Antioxidant and Antibacterial Activities. Food Chem. 2008, 107, 19–25. [Google Scholar] [CrossRef]
- Cuevas, E.; Limón, D.; Pérez-Severiano, F.; Díaz, A.; Ortega, L.; Zenteno, E.; Guevara, J. Antioxidant Effects of Epicatechin on the Hippocampal Toxicity Caused by Amyloid-Beta 25-35 in Rats. Eur. J. Pharmacol. 2009, 616, 122–127. [Google Scholar] [CrossRef]
- Li, Y.-L.; Guo, H.; Zhao, Y.-Q.; Li, A.-F.; Ren, Y.-Q.; Zhang, J.-W. Quercetin Protects Neuronal Cells from Oxidative Stress and Cognitive Degradation Induced by Amyloid β-Peptide Treatment. Mol. Med. Rep. 2017, 16, 1573–1577. [Google Scholar] [CrossRef]
- Rubio-Perez, J.M.; Morillas-Ruiz, J.M. A Review: Inflammatory Process in Alzheimer’s Disease, Role of Cytokines. Sci. World J. 2012, 2012, 756357. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Lozano-Sepúlveda, S.A.; Rincón-Sanchez, A.R.; Rivas-Estilla, A.M. Antioxidants Benefits in Hepatitis C Infection in the New DAAs Era. Ann. Hepatol. 2019, 18, 410–415. [Google Scholar] [CrossRef]
- Rufino, A.T.; Costa, V.M.; Carvalho, F.; Fernandes, E. Flavonoids as Antiobesity Agents: A Review. Med. Res. Rev. 2021, 41, 556–585. [Google Scholar] [CrossRef]
- Steffen, Y.; Gruber, C.; Schewe, T.; Sies, H. Mono-O-Methylated Flavanols and Other Flavonoids as Inhibitors of Endothelial NADPH Oxidase. Arch. Biochem. Biophys. 2008, 469, 209–219. [Google Scholar] [CrossRef]
- Nićiforović, N.; Abramovič, H. Sinapic Acid and Its Derivatives: Natural Sources and Bioactivity. Compr. Rev. Food Sci. Food Saf. 2014, 13, 34–51. [Google Scholar] [CrossRef]
- Maoka, T. Carotenoids as Natural Functional Pigments. J. Nat. Med. 2020, 74, 1–16. [Google Scholar] [CrossRef]
- Canene-Adams, K.; Erdman, J.W. Absorption, Transport, Distribution in Tissues and Bioavailability. Carotenoids: Volume 5: Nutrition and HealthBritton, G., Pfander, H., Liaaen-Jensen, S., Eds.; CarotenoidsBirkhäuser: Basel, Switzerland, 2009; pp. 115–148. ISBN 978-3-7643-7501-0. [Google Scholar]
- Wingerath, T.; Sies, H.; Stahl, W. Xanthophyll Esters in Human Skin. Arch. Biochem. Biophys. 1998, 355, 271–274. [Google Scholar] [CrossRef]
- Muzhingi, T.; Yeum, K.-J.; Bermudez, O.; Tang, G.; Siwela, A.H. Peanut Butter Increases the Bioavailability and Bioconversion of Kale β-Carotene to Vitamin A. Asia Pac. J. Clin. Nutr. 2017, 26, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Moreno, A.; Alanís-Garza, P.A.; Mora-Nieves, J.L.; Mora-Mora, J.P.; Jacobo-Velázquez, D.A. Kale: An Excellent Source of Vitamin C, pro-Vitamin A, Lutein and Glucosinolates. CyTA J. Food 2014, 12, 298–303. [Google Scholar] [CrossRef]
- Lefsrud, M.; Kopsell, D.; Wenzel, A.; Sheehan, J. Changes in Kale (Brassica oleracea L. var. acephala) Carotenoid and Chlorophyll Pigment Concentrations during Leaf Ontogeny. Sci. Hortic. 2007, 112, 136–141. [Google Scholar] [CrossRef]
- Kim, M.J.; Chiu, Y.-C.; Ku, K.-M. Glucosinolates, Carotenoids, and Vitamins E and K Variation from Selected Kale and Collard Cultivars. J. Food Qual. 2017, 2017, e5123572. [Google Scholar] [CrossRef]
- Rafi, M.M.; Kanakasabai, S.; Gokarn, S.V.; Krueger, E.G.; Bright, J.J. Dietary Lutein Modulates Growth and Survival Genes in Prostate Cancer Cells. J. Med. Food 2015, 18, 173–181. [Google Scholar] [CrossRef]
- Ma, L.; Lin, X.-M.; Zou, Z.-Y.; Xu, X.-R.; Li, Y.; Xu, R. A 12-Week Lutein Supplementation Improves Visual Function in Chinese People with Long-Term Computer Display Light Exposure. Br. J. Nutr. 2009, 102, 186–190. [Google Scholar] [CrossRef]
- Sun, Y.-X.; Liu, T.; Dai, X.-L.; Zheng, Q.-S.; Hui, B.-D.; Jiang, Z.-F. Treatment with Lutein Provides Neuroprotection in Mice Subjected to Transient Cerebral Ischemia. J. Asian Nat. Prod. Res. 2014, 16, 1084–1093. [Google Scholar] [CrossRef]
- Binawade, Y.; Jagtap, A. Neuroprotective Effect of Lutein against 3-Nitropropionic Acid-Induced Huntington’s Disease-like Symptoms: Possible Behavioral, Biochemical, and Cellular Alterations. J. Med. Food 2013, 16, 934–943. [Google Scholar] [CrossRef]
- Qiu, X.; Gao, D.-H.; Xiang, X.; Xiong, Y.-F.; Zhu, T.-S.; Liu, L.-G.; Sun, X.-F.; Hao, L.-P. Ameliorative Effects of Lutein on Non-Alcoholic Fatty Liver Disease in Rats. World J. Gastroenterol. 2015, 21, 8061–8072. [Google Scholar] [CrossRef]
- Sowmya Shree, G.; Yogendra Prasad, K.; Arpitha, H.S.; Deepika, U.R.; Nawneet Kumar, K.; Mondal, P.; Ganesan, P. β-Carotene at Physiologically Attainable Concentration Induces Apoptosis and down-Regulates Cell Survival and Antioxidant Markers in Human Breast Cancer (MCF-7) Cells. Mol. Cell. Biochem. 2017, 436, 1–12. [Google Scholar] [CrossRef]
- Levy, Y.; Zaltsberg, H.; Ben-Amotz, A.; Kanter, Y.; Aviram, M. Dietary Supplementation of a Natural Isomer Mixture of Beta-Carotene Inhibits Oxidation of LDL Derived from Patients with Diabetes Mellitus. Ann. Nutr. Metab. 2000, 44, 54–60. [Google Scholar] [CrossRef]
- Boccardi, V.; Arosio, B.; Cari, L.; Bastiani, P.; Scamosci, M.; Casati, M.; Ferri, E.; Bertagnoli, L.; Ciccone, S.; Rossi, P.D.; et al. Beta-Carotene, Telomerase Activity and Alzheimer’s Disease in Old Age Subjects. Eur. J. Nutr. 2020, 59, 119–126. [Google Scholar] [CrossRef]
- Arnold, C.; Jentsch, S.; Dawczynski, J.; Böhm, V. Age-Related Macular Degeneration: Effects of a Short-Term Intervention with an Oleaginous Kale Extract—A Pilot Study. Nutrition 2013, 29, 1412–1417. [Google Scholar] [CrossRef]
- Shi, J.; Ho, C.-T.; Shahidi, F. Functional Foods of the East; CRC Press: Boca Raton, FL, USA, 2010; ISBN 978-1-4200-7193-1. [Google Scholar]
- Pagliaro, B.; Santolamazza, C.; Simonelli, F.; Rubattu, S. Phytochemical Compounds and Protection from Cardiovascular Diseases: A State of the Art. BioMed. Res. Int. 2015, 2015, 918069. [Google Scholar] [CrossRef]
- Ranard, K.M.; Jeon, S.; Mohn, E.S.; Griffiths, J.C.; Johnson, E.J.; Erdman, J.W. Dietary Guidance for Lutein: Consideration for Intake Recommendations Is Scientifically Supported. Eur. J. Nutr. 2017, 56, 37–42. [Google Scholar] [CrossRef]
- Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A.; Pech, J.C.; Koiwa, H. Signaling Molecules Involved in the Postharvest Stress Response of Plants: Quality Changes and Synthesis of Secondary Metabolites. In Handbook of Plant and Crop Physiology; Pessarakli, M., Ed.; CRC Press: Boca Raton, FL, USA, 2014; pp. 259–276. [Google Scholar]
- Cao, M.-J.; Wang, Z.; Zhao, Q.; Mao, J.-L.; Speiser, A.; Wirtz, M.; Hell, R.; Zhu, J.-K.; Xiang, C.-B. Sulfate Availability Affects ABA Levels and Germination Response to ABA and Salt Stress in Arabidopsis Thaliana. Plant J. Cell Mol. Biol. 2014, 77, 604–615. [Google Scholar] [CrossRef]
- Jacobo-Velázquez, D.A.; Cisneros-Zevallos, L. An Alternative Use of Horticultural Crops: Stressed Plants as Biofactories of Bioactive Phenolic Compounds. Agriculture 2012, 2, 259–271. [Google Scholar] [CrossRef]
- Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Controlled Abiotic Stresses Revisited: From Homeostasis through Hormesis to Extreme Stresses and the Impact on Nutraceuticals and Quality during Pre- and Postharvest Applications in Horticultural Crops. J. Agric. Food Chem. 2020, 68, 11877–11879. [Google Scholar] [CrossRef]
- Hideg, É.; Jansen, M.A.K.; Strid, Å. UV-B Exposure, ROS, and Stress: Inseparable Companions or Loosely Linked Associates? Trends Plant Sci. 2013, 18, 107–115. [Google Scholar] [CrossRef]
- Carillo, P.; Soteriou, G.A.; Kyriacou, M.C.; Giordano, M.; Raimondi, G.; Napolitano, F.; Di Stasio, E.; Mola, I.D.; Mori, M.; Rouphael, Y. Regulated Salinity Eustress in a Floating Hydroponic Module of Sequentially Harvested Lettuce Modulates Phytochemical Constitution, Plant Resilience, and Post-Harvest Nutraceutical Quality. Agronomy 2021, 11, 1040. [Google Scholar] [CrossRef]
- Vargas-Hernandez, M.; Macias-Bobadilla, I.; Guevara-Gonzalez, R.G.; Romero-Gomez, S.D.J.; Rico-Garcia, E.; Ocampo-Velazquez, R.V.; Alvarez-Arquieta, L.d.L.; Torres-Pacheco, I. Plant Hormesis Management with Biostimulants of Biotic Origin in Agriculture. Front. Plant Sci. 2017, 8, 1762. [Google Scholar] [CrossRef]
- Rouphael, Y.; Kyriacou, M.C.; Carillo, P.; Pizzolongo, F.; Romano, R.; Sifola, M.I. Chemical Eustress Elicits Tailored Responses and Enhances the Functional Quality of Novel Food Perilla Frutescens. Molecules 2019, 24, 185. [Google Scholar] [CrossRef]
- Pandey, N.; Iqbal, Z.; Pandey, B.K.; Sawant, S.V. Phytohormones and Drought Stress: Plant Responses to Transcriptional Regulation. In Mechanism of Plant Hormone Signaling under Stress; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2017; pp. 477–504. ISBN 978-1-118-88902-2. [Google Scholar]
- Hirani, A.H.; Li, G.; Zelmer, C.D.; McVetty, P.B.E.; Asif, M.; Goyal, A. Molecular Genetics of Glucosinolate Biosynthesis in Brassicas: Genetic Manipulation and Application Aspects; IntechOpen: London, UK, 2012; ISBN 978-953-51-0527-5. [Google Scholar]
- Groenbaek, M.; Jensen, S.; Neugart, S.; Schreiner, M.; Kidmose, U.; Kristensen, H.L. Nitrogen Split Dose Fertilization, Plant Age and Frost Effects on Phytochemical Content and Sensory Properties of Curly Kale (Brassica oleracea L. var. sabellica). Food Chem. 2016, 197, 530–538. [Google Scholar] [CrossRef]
- Park, Y.-J.; Lee, H.-M.; Shin, M.; Arasu, M.V.; Chung, D.Y.; Al-Dhabi, N.A.; Kim, S.-J. Effect of Different Proportion of Sulphur Treatments on the Contents of Glucosinolate in Kale (Brassica oleracea var. acephala) Commonly Consumed in Republic of Korea. Saudi J. Biol. Sci. 2018, 25, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Kopsell, D.E.; Kopsell, D.A.; Randle, W.M.; Coolong, T.W.; Sams, C.E.; Curran-Celentano, J. Kale Carotenoids Remain Stable While Flavor Compounds Respond to Changes in Sulfur Fertility. J. Agric. Food Chem. 2003, 51, 5319–5325. [Google Scholar] [CrossRef] [PubMed]
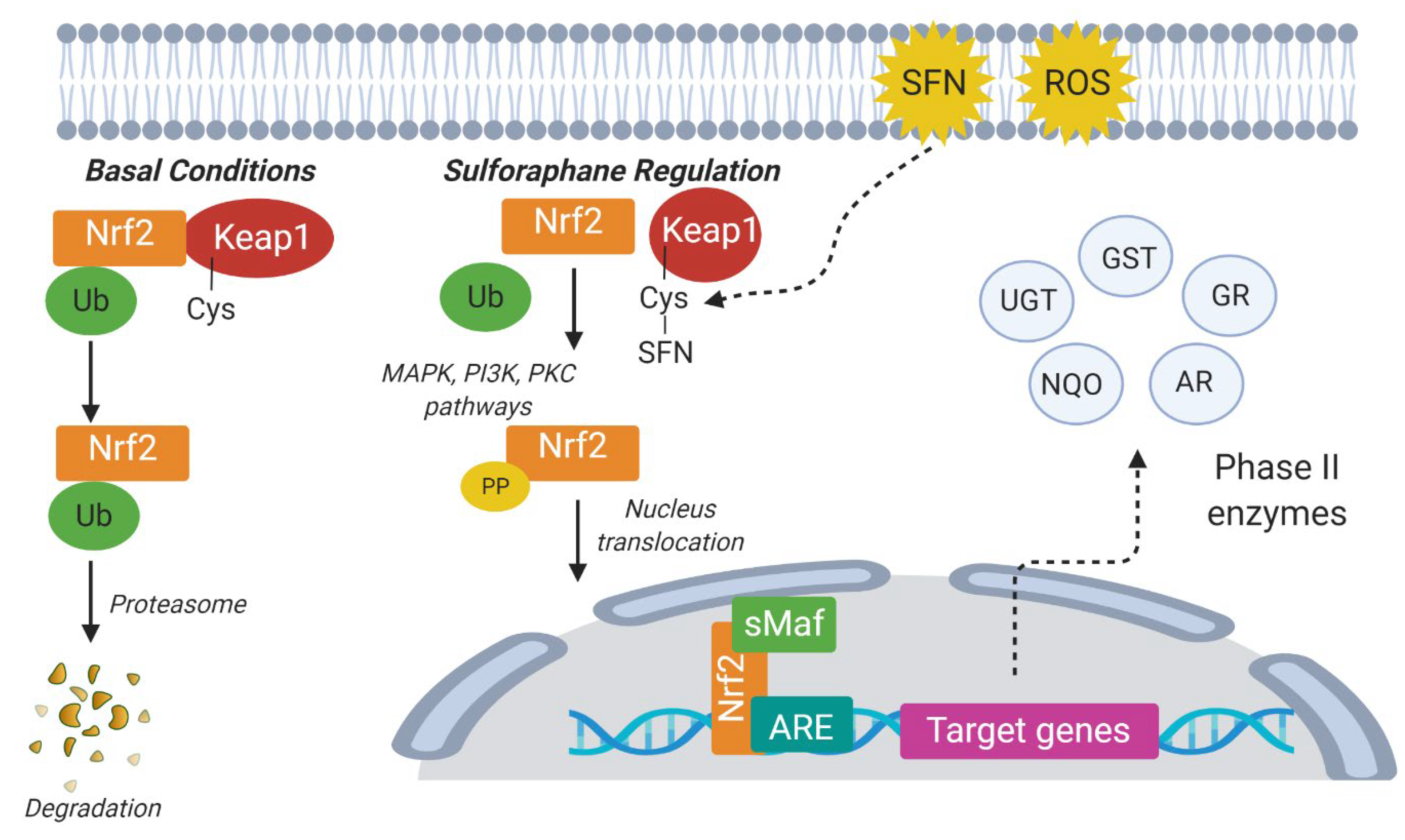
- Kim, S.Y.; Park, J.-E.; Kim, E.O.; Lim, S.J.; Nam, E.J.; Yun, J.H.; Yoo, G.; Oh, S.-R.; Kim, H.S.; Nho, C.W. Exposure of Kale Root to NaCl and Na2SeO3 Increases Isothiocyanate Levels and Nrf2 Signalling without Reducing Plant Root Growth. Sci. Rep. 2018, 8, 3999. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Cai, C.; Lin, J.; Tao, H.; Zeng, W.; Zhang, F.; Miao, H.; Sun, B.; Wang, Q. Combined Treatment of Epi-Brassinolide and NaCl Enhances the Main Phytochemicals in Chinese Kale Sprouts. Food Chem. 2020, 315, 126275. [Google Scholar] [CrossRef] [PubMed]
- Linić, I.; Šamec, D.; Grúz, J.; Vujčić Bok, V.; Strnad, M.; Salopek-Sondi, B. Involvement of Phenolic Acids in Short-Term Adaptation to Salinity Stress Is Species-Specific among Brassicaceae. Plants 2019, 8, 155. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Yan, H.; Zhang, F.; Wang, Q. Effects of Plant Hormones on Main Health-Promoting Compounds and Antioxidant Capacity of Chinese Kale. Food Res. Int. 2012, 48, 359–366. [Google Scholar] [CrossRef]
- Ku, K.M.; Juvik, J.A. Environmental Stress and Methyl Jasmonate-Mediated Changes in Flavonoid Concentrations and Antioxidant Activity in Broccoli Florets and Kale Leaf Tissues. HortScience 2013, 48, 996–1002. [Google Scholar] [CrossRef]
- Yi, G.-E.; Robin, A.H.K.; Yang, K.; Park, J.-I.; Hwang, B.H.; Nou, I.-S. Exogenous Methyl Jasmonate and Salicylic Acid Induce Subspecies-Specific Patterns of Glucosinolate Accumulation and Gene Expression in Brassica oleracea L. Molecules 2016, 21, 1417. [Google Scholar] [CrossRef]
- Barickman, T.C.; Ku, K.-M.; Sams, C.E. Differing Precision Irrigation Thresholds for Kale (Brassica oleracea L. var. acephala) Induces Changes in Physiological Performance, Metabolites, and Yield. Environ. Exp. Bot. 2020, 180, 104253. [Google Scholar] [CrossRef]
- Yoon, H.I.; Zhang, W.; Son, J.E. Optimal Duration of Drought Stress Near Harvest for Promoting Bioactive Compounds and Antioxidant Capacity in Kale with or without UV-B Radiation in Plant Factories. Plants 2020, 9, 295. [Google Scholar] [CrossRef]
- Lee, J.-H.; Oh, M.-M. Short-Term Low Temperature Increases Phenolic Antioxidant Levels in Kale. Hortic. Environ. Biotechnol. 2015, 56, 588–596. [Google Scholar] [CrossRef]
- Jurkow, R.; Wurst, A.; Kalisz, A.; Sękara, A.; Cebula, S. Cold Stress Modifies Bioactive Compounds of Kale Cultivars during Fall–Winter Harvests. Acta Agrobot. 2019, 72, 1761. [Google Scholar] [CrossRef]
- Hwang, S.-J.; Chun, J.-H.; Kim, S.-J. Effect of Cold Stress on Carotenoids in Kale Leaves (Brassica oleracea). Korean J. Environ. Agric. 2017, 36, 106–112. [Google Scholar] [CrossRef]
- Lee, M.-J.; Lim, S.; Kim, J.; Oh, M.-M. Heat Shock Treatments Induce the Accumulation of Phytochemicals in Kale Sprouts. Hortic. Sci. Technol. 2012, 30, 509–518. [Google Scholar] [CrossRef]
- Alegre, S.; Pascual, J.; Trotta, A.; Gollan, P.J.; Yang, W.; Yang, B.; Aro, E.-M.; Burow, M.; Kangasjärvi, S. Growth under High Light and Elevated Temperature Affects Metabolic Responses and Accumulation of Health-Promoting Metabolites in Kale Varieties. bioRxiv 2019. [Google Scholar] [CrossRef]
- Yoon, H.I.; Kim, D.; Son, J.E. Spatial and Temporal Bioactive Compound Contents and Chlorophyll Fluorescence of Kale (Brassica oleracea L.) Under UV-B Exposure Near Harvest Time in Controlled Environments. Photochem. Photobiol. 2020, 96, 845–852. [Google Scholar] [CrossRef]
- Klopsch, R.; Baldermann, S.; Voss, A.; Rohn, S.; Schreiner, M.; Neugart, S. Narrow-Banded UVB Affects the Stability of Secondary Plant Metabolites in Kale (Brassica oleracea var. sabellica) and Pea (Pisum Sativum) Leaves Being Added to Lentil Flour Fortified Bread: A Novel Approach for Producing Functional Foods. Foods 2019, 8, 427. [Google Scholar] [CrossRef]
- Schreiner, M.; Krumbein, A.; Mewis, I.; Ulrichs, C.; Huyskens-Keil, S. Short-Term UV-B Radiation Effects on Secondary Metabolism in Different Organs of Tropaeolum majus L. Innov. Food Sci. Emerg. Technol. 2009, 10, 93–96. [Google Scholar] [CrossRef]
- Jaganjac, M.; Milkovic, L.; Sunjic, S.B.; Zarkovic, N. The NRF2, Thioredoxin, and Glutathione System in Tumorigenesis and Anticancer Therapies. Antioxidants 2020, 9, 1151. [Google Scholar] [CrossRef]
- Brodowska, M.S.; Kurzyna-Szklarek, M.; Haliniarz, M. Selenium in the environment. J. Elem. 2016, 17, 1173–1185. [Google Scholar] [CrossRef]
- Guardado-Félix, D.; Antunes-Ricardo, M.; Rocha-Pizaña, M.R.; Martínez-Torres, A.-C.; Gutiérrez-Uribe, J.A.; Serna Saldivar, S.O. Chickpea (Cicer arietinum L.) Sprouts Containing Supranutritional Levels of Selenium Decrease Tumor Growth of Colon Cancer Cells Xenografted in Immune-Suppressed Mice. J. Funct. Foods 2019, 53, 76–84. [Google Scholar] [CrossRef]
- Cox, D.N.; Bastiaans, K. Understanding Australian Consumers’ Perceptions of Selenium and Motivations to Consume Selenium Enriched Foods. Food Qual. Prefer. 2007, 18, 66–76. [Google Scholar] [CrossRef]
- Schiavon, M.; Pilon-Smits, E.A.H. The Fascinating Facets of Plant Selenium Accumulation—Biochemistry, Physiology, Evolution and Ecology. New Phytol. 2017, 213, 1582–1596. [Google Scholar] [CrossRef] [PubMed]
- Guardado-Félix, D.; Serna-Saldivar, S.O.; Cuevas-Rodríguez, E.O.; Jacobo-Velázquez, D.A.; Gutiérrez-Uribe, J.A. Effect of Sodium Selenite on Isoflavonoid Contents and Antioxidant Capacity of Chickpea (Cicer arietinum L.) Sprouts. Food Chem. 2017, 226, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Feng, R.; Wei, C.; Tu, S. The Roles of Selenium in Protecting Plants against Abiotic Stresses. Environ. Exp. Bot. 2013, 87, 58–68. [Google Scholar] [CrossRef]
- Brosché, M.; Strid, Å. Molecular Events Following Perception of Ultraviolet-B Radiation by Plants. Physiol. Plant. 2003, 117, 1–10. [Google Scholar] [CrossRef]
- Barickman, T.C.; Kopsell, D.A.; Sams, C.E. Selenium Influences Glucosinolate and Isothiocyanates and Increases Sulfur Uptake in Arabidopsis Thaliana and Rapid-Cycling Brassica oleracea. J. Agric. Food Chem. 2013, 61, 202–209. [Google Scholar] [CrossRef]
- Bartels, D.; Sunkar, R. Drought and Salt Tolerance in Plants. Crit. Rev. Plant Sci. 2005, 24, 23–58. [Google Scholar] [CrossRef]
- Chaman, M.E. Variaciones en el Contenido Relativo de Agua y la Concentración de Prolina en Capsicum annuum L. Inducido por NaCl. Tesis de Doctorado en Ciencias Biológicas; Universidad Nacional de Trujillo: Trujillo, Perú, 2008. [Google Scholar]
- Sudhakar, C.; Lakshmi, A.; Giridarakumar, S. Changes in the Antioxidant Enzyme Efficacy in Two High Yielding Genotypes of Mulberry (Morus alba L.) under NaCl Salinity. Plant Sci. 2001, 161, 613–619. [Google Scholar] [CrossRef]
- Dat, J.; Vandenabeele, S.; Vranová, E.; Van Montagu, M.; Inzé, D.; Van Breusegem, F. Dual Action of the Active Oxygen Species during Plant Stress Responses. Cell. Mol. Life Sci. 2000, 57, 779–795. [Google Scholar] [CrossRef]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.-K.; Bohnert, H.J. Plant Cellular and Molecular Responses to High Salinity. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor Signal Transduction Leading to Production of Plant Secondary Metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef]
- Park, W.T.; Kim, Y.B.; Seo, J.M.; Kim, S.-J.; Chung, E.; Lee, J.-H.; Park, S.U. Accumulation of Anthocyanin and Associated Gene Expression in Radish Sprouts Exposed to Light and Methyl Jasmonate. J. Agric. Food Chem. 2013, 61, 4127–4132. [Google Scholar] [CrossRef]
- Augustine, R.; Bisht, N.C. Regulation of Glucosinolate Metabolism: From Model Plant Arabidopsis Thaliana to Brassica Crops. In Glucosinolates; Mérillon, J.-M., Ramawat, K.G., Eds.; Series in Phytochemistry; Springer International Publishing: Cham, Switzerland, 2017; pp. 163–199. ISBN 978-3-319-25462-3. [Google Scholar]
- Skirycz, A.; Reichelt, M.; Burow, M.; Birkemeyer, C.; Rolcik, J.; Kopka, J.; Zanor, M.I.; Gershenzon, J.; Strnad, M.; Szopa, J.; et al. DOF Transcription Factor AtDof1.1 (OBP2) Is Part of a Regulatory Network Controlling Glucosinolate Biosynthesis in Arabidopsis. Plant J. Cell Mol. Biol. 2006, 47, 10–24. [Google Scholar] [CrossRef]
- Mikkelsen, M.D.; Petersen, B.L.; Glawischnig, E.; Jensen, A.B.; Andreasson, E.; Halkier, B.A. Modulation of CYP79 Genes and Glucosinolate Profiles in Arabidopsis by Defense Signaling Pathways. Plant Physiol. 2003, 131, 298–308. [Google Scholar] [CrossRef]
- Baskar, V.; Gururani, M.A.; Yu, J.W.; Park, S.W. Engineering Glucosinolates in Plants: Current Knowledge and Potential Uses. Appl. Biochem. Biotechnol. 2012, 168, 1694–1717. [Google Scholar] [CrossRef]
- Chiu, Y.-C.; Juvik, J.A.; Ku, K.-M. Targeted Metabolomic and Transcriptomic Analyses of “Red Russian” Kale (Brassicae napus var. pabularia) Following Methyl Jasmonate Treatment and Larval Infestation by the Cabbage Looper (Trichoplusia Ni Hübner). Int. J. Mol. Sci. 2018, 19, 1058. [Google Scholar] [CrossRef]
- Cruz de Carvalho, M.H. Drought Stress and Reactive Oxygen Species: Production, Scavenging and Signaling. Plant Signal. Behav. 2008, 3, 156–165. [Google Scholar] [CrossRef]
- Claeys, H.; Inzé, D. The Agony of Choice: How Plants Balance Growth and Survival under Water-Limiting Conditions. Plant Physiol. 2013, 162, 1768–1779. [Google Scholar] [CrossRef]
- Fischer, B.B.; Hideg, É.; Krieger-Liszkay, A. Production, Detection, and Signaling of Singlet Oxygen in Photosynthetic Organisms. Antioxid. Redox Signal. 2013, 18, 2145–2162. [Google Scholar] [CrossRef]
- Nelson, N.; Junge, W. Structure and Energy Transfer in Photosystems of Oxygenic Photosynthesis. Annu. Rev. Biochem. 2015, 84, 659–683. [Google Scholar] [CrossRef]
- Issarakraisila, M.; Ma, Q.; Turner, D.W. Photosynthetic and Growth Responses of Juvenile Chinese Kale (Brassica oleracea var. alboglabra) and Caisin (Brassica rapa subsp. parachinensis) to Waterlogging and Water Deficit. Sci. Hortic. 2007, 111, 107–113. [Google Scholar] [CrossRef]
- Zhu, Z.; Sun, B.; Xu, X.; Chen, H.; Zou, L.; Chen, G.; Cao, B.; Chen, C.; Lei, J. Overexpression of AtEDT1/HDG11 in Chinese Kale (Brassica oleracea var. alboglabra) Enhances Drought and Osmotic Stress Tolerance. Front. Plant Sci. 2016, 7, 1285. [Google Scholar] [CrossRef]
- Chaves-Barrantes, N.F.; Gutiérrez-Soto, M.V. Respuestas al estrés por calor en los cultivos. I. aspectos moleculares, bioquímicos y fisiológicos. Agron. Mesoam. 2017, 28, 237–253. [Google Scholar] [CrossRef]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M.R. Heat Tolerance in Plants: An Overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Wang, L.-C.; Tsai, M.-C.; Chang, K.-Y.; Fan, Y.-S.; Yeh, C.-H.; Wu, S.-J. Involvement of the Arabidopsis HIT1/AtVPS53 Tethering Protein Homologue in the Acclimation of the Plasma Membrane to Heat Stress. J. Exp. Bot. 2011, 62, 3609–3620. [Google Scholar] [CrossRef]
- Savchenko, G.E.; Klyuchareva, E.A.; Abramchik, L.M.; Serdyuchenko, E.V. Effect of Periodic Heat Shock on the Inner Membrane System of Etioplasts. Russ. J. Plant Physiol. 2002, 49, 349–359. [Google Scholar] [CrossRef]
- Los, D.A.; Murata, N. Membrane Fluidity and Its Roles in the Perception of Environmental Signals. Biochim. Biophys. Acta BBA-Biomembr. 2004, 1666, 142–157. [Google Scholar] [CrossRef]
- Porch, T.G.; Hall, A.E. Heat Tolerance. In Genomics and Breeding for Climate-Resilient Crops: Vol. 2 Target Traits; Kole, C., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 167–202. ISBN 978-3-642-37048-9. [Google Scholar]
- Almeselmani, M.; Deshmukh, P.S.; Sairam, R.K.; Kushwaha, S.R.; Singh, T.P. Protective Role of Antioxidant Enzymes under High Temperature Stress. Plant Sci. 2006, 171, 382–388. [Google Scholar] [CrossRef]
- Nagesh Babu, R.; Rangaiah, D.V. High Temperature and Salt Stress Response in French Bean (Phaseolus vulgaris). Aust. J. Crop Sci. 2008, 2, 40–48. [Google Scholar]
- Upchurch, R.G. Fatty Acid Unsaturation, Mobilization, and Regulation in the Response of Plants to Stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Hikosaka, K.; Ishikawa, K.; Borjigidai, A.; Muller, O.; Onoda, Y. Temperature Acclimation of Photosynthesis: Mechanisms Involved in the Changes in Temperature Dependence of Photosynthetic Rate. J. Exp. Bot. 2006, 57, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.M.; Roychowdhury, R.; Fujita, M. Physiological, Biochemical, and Molecular Mechanisms of Heat Stress Tolerance in Plants. Int. J. Mol. Sci. 2013, 14, 9643–9684. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive Oxygen Species and Antioxidant Machinery in Abiotic Stress Tolerance in Crop Plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Müller-Xing, R.; Xing, Q.; Goodrich, J. Footprints of the Sun: Memory of UV and Light Stress in Plants. Front. Plant Sci. 2014, 5, 474. [Google Scholar] [CrossRef]
- Jenkins, G.I. Signal Transduction in Responses to UV-B Radiation. Annu. Rev. Plant Biol. 2009, 60, 407–431. [Google Scholar] [CrossRef]
- Jenkins, G.I.; Fuglevand, G.; Christie, J.M. UV-B Perception and Signal Transduction. In Plants and UV-B: Responses to Environmental Change; Lumsden, P., Ed.; Society for Experimental Biology Seminar Series; Cambridge University Press: Cambridge, UK, 2010; pp. 135–156. ISBN 978-0-521-57222-4. [Google Scholar]
- Brown, B.A.; Cloix, C.; Jiang, G.H.; Kaiserli, E.; Herzyk, P.; Kliebenstein, D.J.; Jenkins, G.I. A UV-B-Specific Signaling Component Orchestrates Plant UV Protection. Proc. Natl. Acad. Sci. USA 2005, 102, 18225–18230. [Google Scholar] [CrossRef]
- Christie, J.M.; Arvai, A.S.; Baxter, K.J.; Heilmann, M.; Pratt, A.J.; O’Hara, A.; Kelly, S.M.; Hothorn, M.; Smith, B.O.; Hitomi, K.; et al. Plant UVR8 Photoreceptor Senses UV-B by Tryptophan-Mediated Disruption of Cross-Dimer Salt Bridges. Science 2012, 335, 1492–1496. [Google Scholar] [CrossRef]
- Favory, J.-J.; Stec, A.; Gruber, H.; Rizzini, L.; Oravecz, A.; Funk, M.; Albert, A.; Cloix, C.; Jenkins, G.I.; Oakeley, E.J.; et al. Interaction of COP1 and UVR8 Regulates UV-B-Induced Photomorphogenesis and Stress Acclimation in Arabidopsis. EMBO J. 2009, 28, 591–601. [Google Scholar] [CrossRef]
- Kaiserli, E.; Jenkins, G.I. UV-B Promotes Rapid Nuclear Translocation of the Arabidopsis UV-B Specific Signaling Component UVR8 and Activates Its Function in the Nucleus. Plant Cell 2007, 19, 2662–2673. [Google Scholar] [CrossRef]
- Tilbrook, K.; Arongaus, A.B.; Binkert, M.; Heijde, M.; Yin, R.; Ulm, R. The UVR8 UV-B Photoreceptor: Perception, Signaling and Response. Arab. Book 2013, 11, e0164. [Google Scholar] [CrossRef]
- Verdaguer, D.; Jansen, M.A.K.; Llorens, L.; Morales, L.O.; Neugart, S. UV-A Radiation Effects on Higher Plants: Exploring the Known Unknown. Plant Sci. 2017, 255, 72–81. [Google Scholar] [CrossRef]
- Kuhlmann, F.; Müller, C. Independent Responses to Ultraviolet Radiation and Herbivore Attack in Broccoli. J. Exp. Bot. 2009, 60, 3467–3475. [Google Scholar] [CrossRef]
- Mewis, I.; Schreiner, M.; Nguyen, C.N.; Krumbein, A.; Ulrichs, C.; Lohse, M.; Zrenner, R. UV-B Irradiation Changes Specifically the Secondary Metabolite Profile in Broccoli Sprouts: Induced Signaling Overlaps with Defense Response to Biotic Stressors. Plant Cell Physiol. 2012, 53, 1546–1560. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Suresh Kumar, J.; Suprasanna, P. Seed ‘Primeomics’: Plants Memorize Their Germination under Stress. Biol. Rev. 2021, 96, 1723–1743. [Google Scholar] [CrossRef] [PubMed]
- Garfin, G.; Franco, G.; Blanco, H.; Comrie, A.; Gonzalez, P.; Piechota, T.; Smyth, R.; Waskom, R.; Melillo, J.M.; Richmond, T.; et al. Chapter 20: Southwest. In Climate Change Impacts in the United States: The Third National Climate Assessment; U.S. Global Change Research Program: Washington, DC, USA, 2014; pp. 418–440. [Google Scholar]
- Satheesh, N.; Workneh Fanta, S. Kale: Review on Nutritional Composition, Bio-Active Compounds, Anti-Nutritional Factors, Health Beneficial Properties and Value-Added Products. Cogent. Food Agric. 2020, 6, 1811048. [Google Scholar] [CrossRef]
- Jacobo-Velázquez, D.A.; Benavides, J. Non-Thermal Technologies as Tools to Increase the Content of Health-Promoting Compounds in Whole Fruits and Vegetables while Retaining Quality Attributes. Foods 2021, 10, 2904. [Google Scholar] [CrossRef]
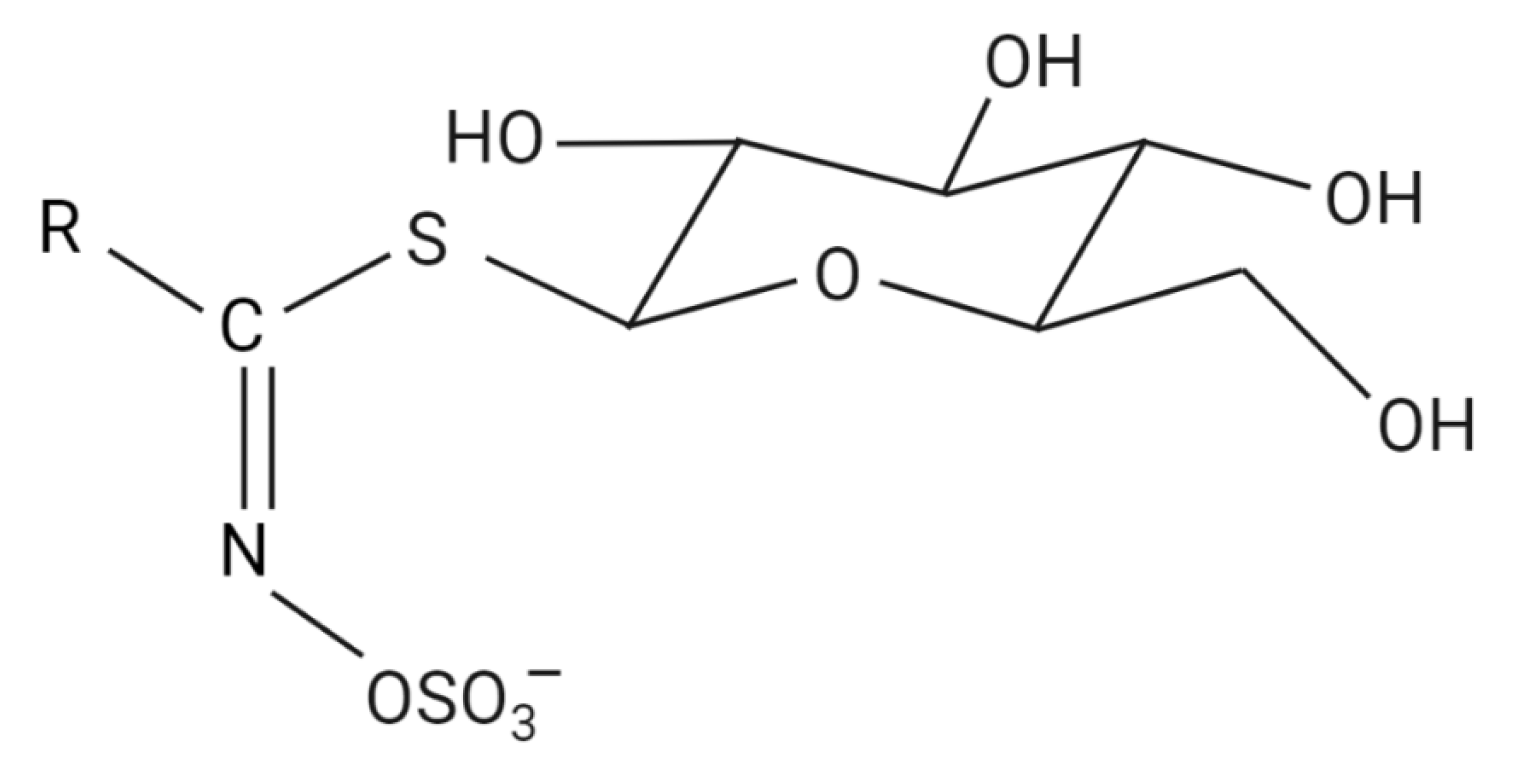

| Glucosinolates | ||
|---|---|---|
| Side Chain Name | Trivial Name | Isothiocyanate |
| Methylthiobutyl | Glucoerucin | Erucin |
| 3-Methylsulfinylpropyl | Glucoiberin | Iberin |
| 3-Methylsulfinylbutyl | Glucoraphanin | Sulforaphane |
| 2-Phenylethyl | Gluconasturtiin | Phenylethyl ITC |
| 3-Indolylmethyl | Glucobrassicin | indol-3-carbinol |
| 2-Hydroxyl-3-butenyl | Progoitrin | 2-hydroxyalkenyl |
| Compound | Metabolic Effect | Main Findings | Reference |
|---|---|---|---|
| Sulforaphane (SFN) | Nrf2 Mitigate insulin resistance | C57BL/6JSlc mice were supplemented with 0.3% glucoraphanin for 14 weeks. Results showed diminished weight gain, reduced hepatic steatosis, and improved insulin sensitivity. | [47] |
| C/EBPα Adipocyte differentiation | 3T3-L1 pre-adipocytes cell line treated with SFN inhibited the elevation of triglycerides in the adipocytes by activation of PPARγ and (C/EBP) α, and inhibition of (C/EBP) β. SFN arrested the cell cycle at the G0/G1 phase. | [48] | |
| C/EBPα | 3T3-L1 pre-adipocytes cell line treated with SFN (10 μM) showed significant inhibition of adipocyte differentiation and lipid accumulation by degradation of CCAAT/enhancer-binding protein (C/EBP) β. | [50] | |
| Benzyl isothiocyanate Phenethyl isothiocyanate | Adipocyte differentiation | C57BL/6J mice were fed with 1g/kg of benzyl isothiocyanate or phenethyl isothiocyanate for 18 weeks. Results showed that these isothiocyanates have the potential to prevent body weight gain. | [54] |
| Allyl isothiocyanate | Sirt1/AMPK NF-κB | AML-12 mouse hepatocyte cell line was treated with allyl isothiocyanate (20 μmol/L) for 24 h. Allyl isothiocyanate reduced lipid accumulation and inflammation in vitro through the Sirt1/AMPK and NF-κB signaling pathways. | [55] |
| Phenethyl isothiocyanate | Hormone regulation | C57BL/6J mice were fed with 25 mg/kg phenethyl isothiocyanate for 18 weeks. Results showed that phenethyl isothiocyanate has the potential to stimulate hypothalamic leptin signaling. | [60] |
| Indol-3-carbinol (I3C) | Liver enzymes | C57BL/6J mice were fed with 40 mg/kg I3C for 35 days. Treatment modulated glucose tolerance and insulin sensibility. Also, supplementation with I3C increased SOD, CAT, GPx levels. | [61] |
| Indol-3-carbinol (I3C) | Apoptosis | I3C inhibited the phosphorylation and following activation of enzyme Akt kinase in PC-3 cell line. Akt kinase is involved in apoptosis and cell cycle regulation. | [62] |
| Apoptosis | In vitro, I3C caused DNA strand breaks in three cervical cancer cell lines. (LD50) = 200 μmol/L I3C. | [63] | |
| Adipogenesis | Mice fed with a I3C-supplemented diet (1 g/kg diet) for 10 weeks showed significantly decreased expression levels of key adipogenic transcription factor PPARγ2, and its target genes, such as leptin and adipocyte protein 2. | [64] | |
| Anti-carcinogenic | MCF-7 cell line treated with I3C (10–125 μm/L) showed a significant inhibition of the ER-alpha signaling and the expression of the estrogen-responsive genes, pS2, and cathepsin-D. On the other hand, breast cancer susceptibility gene 1 (BRCA1) expression was upregulated. | [65] | |
| Anti-carcinogenic | Rats treated intraperitoneally with I3C showed significant inhibition in the development and metastases of prostate cancer and overall survival advantage. | [66] |
| Compound | Metabolic Effect | Main Findings | Reference |
|---|---|---|---|
| Quercetin | Antioxidant activity Neuroprotective | Cortical cells treated with quercetin (100 μM) inhibited up to 61% of the neurotoxicity produced by adding NMDA (n-methyl-d-aspartate) and kainate. In addition, quercetin showed a significant decrease in free radicals in brain injury caused by exposure to chemical agents. | [77] |
| Vasodilator Normotensive, Normocholesteroleic | Healthy volunteers treated with quercetin (200–400 mg) showed an increase in brachial arterial diameter, demonstrating its vasodilator effects. That effect was correlated with an increase in plasma and urinary levels of glutathione. | [78] | |
| Kaempferol and quercetin | Anti-inflammatory | The capacity of flavanoids wa proved to effectively inhibit the lipopolysaccharide (LPS)-induced PGE2 production, LPS-induced COX-2 expression, and mPGES-1 expression in activated macrophages. | [79] |
| Antiproliferative | HepG2 cells exposed to a Ginkgo biloba leaf extract, kaempferol, and quercetin (50 and–100 μM) showed DNA damage and topoisomerase II inhibition. | [80] | |
| Kaempferol- glucoside | Synergistic effect with Lactobacillus paracasei A221 | The oral administration of kaempferol-rich kale extract to Sod1-deficient mice improved various pathologies, including skin thinning, fatty liver, and anemia. | [81] |
| Gallic, proto-catechuic,p-hydroxybenzoic, vanillic, salicylic,p-coumaric,caffeic, ferulic and sinapic acid | Antioxidant and anti-bacterial activities | Phenolic extracts of kale showed anti-bacterial effect on Gram-positive (S. aureus, E. faecalis, B. subtilis), Gram-negative (M. catarrhalis) bacteria, and two yeast-like fungi (C. tropicali and C. albicans). | [82] |
| Compound | Metabolic Effect | Main Findings | Reference |
|---|---|---|---|
| Lutein | Antiproliferative | Lutein induced a decrease in the proliferation of PC-3 cells (in vitro), modulating the expression of growth genes associated with prostate cancer cells. | [98] |
| Antioxidant activity | Healthy subjects supplemented with 12 mg/d of lutein for 12 weeks showed improved visual function, particularly in contrast sensitivity. | [99] | |
| Neuroprotective | Rats supplemented with lutein (30, 15, and 7.5 mg/kg) significantly elevated the activities of superoxide dismutase, glutathione peroxidase, and catalase in brain and decreased the neurological deficit scores. | [100] | |
| Neuroprotective | Rats supplemented with lutein (50–100 mg/kg) for 14 days showed a fat loss, reduced neurobehavioral alterations, and reduced oxidative stress. | [101] | |
| Antidiabetic and obesity control | Rats supplemented with lutein significantly improved body weight, hepatic levels of lipid accumulation, and insulin sensitivity. | [102] | |
| β-carotene | Apoptotic | β-carotene (1 μM) induced apoptosis in MCF cells by caspase-3 activity and inhibited the expression of the anti-apoptotic proteins, Bcl-2 and PARP. | [103] |
| Antioxidant activity | Patients supplemented with 60 mg/d of β-carotene for 3 weeks showed a reduction in LDL susceptibility to oxidation. | [104] | |
| Antioxidant activity | The content of β-carotene in plasma was significantly and positively correlated with telomerase activity of Alzheimer disease patients, independent of gender. | [105] |
| Abiotic Stress | Treatment | Main Findings on the Biosynthesis of Phytochemicals | References |
|---|---|---|---|
| Saline stress | 6-week-old kale seedlings were supplemented with sulphur (S) solution (0.0, 0.5, 1.0-, and 2.0-mM) for 28 days. | Maximum levels of total GLSs and glucobrassicin were found in the leaves supplemented with 2 mM S. Aliphatic glucosinolates, and total glucosinolates increased by 67% and 35%, respectively. Glucobrassicin was the main glucosinolate accumulated. | [121] |
| 2-week-old kale sprouts (Winterbor, Redbor, and Toscano) were treated with sulphur (S) solution (4, 8, 16, 32, and 64 mg/L) for 45 days. | There was a significant increase in the glucoiberin, glucobrassicin, neoglucobrassicin and 4-hydroxygluco-brassicin content in the leaves supplemented with 32 and 64 mg of S/L. Glucobrassicin was the most abundant glucosinolate. There was no significant change in carotenoid accumulation by S treatment. | [122] | |
| 6-week-old kale seedlings (Brassica oleracea var. sabellica) were exposed to Na2SeO3 (2 mg/L), NaCl (80 mM), or a combination for 14 days. | After seven days of treatment, gluconasturtiin concentration increased by 15%, 19%, and 27% with NaCl, Na2SeO3, or both, respectively. ITC concentration increased 30% with the combination of both treatments after 14 days. | [123] | |
| Five-day-old kale sprouts (Brassica oleracea var. alboglabra) were treated with NaCl solution (160 mM) for two days. | Total glucosinolate increased (129%) by NaCl treatment, mainly aliphatic glucosinolates (142%). Glucobrassicin, glucoiberin, glucoraphanin, glucoerucin, gluconapin, and progoitrin increased by 233%, 190%, 144%, 166%, 175%, and 10%, respectively. Ascorbic acid and total carotenoids were 83% and 53% higher, respectively, in treated sprouts. | [124] | |
| Kale seedlings (Brassica oleracea var. acephala) were grown in 1% agar plates containing NaCl (50–200 mM) | There was no change in chlorophyll pigments in kale in the salt conditions. There was a decrease in salicylic acid (27.5%), caffeic acid (31.1%), and 4-coumaric acid (108.1%) in kale under severe stress (200 mM NaCl). | [125] | |
| Phytohormone Application | Thirty-day-old chinese kale seeds (Brassica oleracea var. alboglabra) were sprayed with a MeJA solution (100 μM) 6 days before sampling. | Glucobrassicin (520%), neoglucobrassicin (1420%), and total indole glucosinolates (230%) showed a significant increase after 1 d of treatment. MeJA treatment did not exert a significant effect on the content of vitamin C, carotenoids or phenolics. | [126] |
| Kale cultivars Red Winter and Dwarf Blue Curled Vates were sprayed with a MeJA solution (250 μM) 4 days before harvest at commercial maturity. | MeJA treatments significantly increased total phenolics in Dwarf Blue Curled (2298 mg GAE/100 g) and Red Winter (2070 mg GAE/100 g) cultivar by 24% and 41%, respectively. In addition, antioxidant activity also increased by 31% in both kale cultivars. | [127] | |
| Phytohormone Application | Four-month-old kale plants were treated with a MeJA solution (250 μM) 4 days before sample collection. | Glucoraphanin (735%), glucobrassicin (1708%), and neoglucobrassicin (1800%) increase significantly. Higher expression of ST5a (Bol026200), CYP81F1 (Bol028913, Bol028914), and CYP81F4 genes were associated with this accumulation. | [128] |
| Drought | Kale ‘Winterbor’ were greenhouse growth at three different levels of volumetric water content (VWC): 0.15 m3, 0.25 m3, 0.35 m3. | Neoxanthin (16.6%) and antheraxanthin (18.2%) increased mainly with 0.35 m3 VWC treatment. Total phenolic content was higher with 0.25 and 0.35 m3 VWC treatments. Likewise, a higher concentration for glucoiberin (53%) and progoitrin (60%) was observed at 0.25 m3. | [129] |
| Five-week-old kale plants (Brassica oleracea var. acephala) were stressed by removing the nutrient solutions 7 days before harvest. | Total phenolic and flavonoid contents and antioxidant activities were significantly increased by 35%, 48%, and 34%, respectively, in treated samples at 3–4 days. | [130] | |
| Temperature | Five-week-old kale (Manchoo Collard) were subjected to 4 °C for 3 days. | Treated kale ‘Manchoo Collard’ exhibited a 15% higher total phenolic concentration and 17% higher antioxidant capacity than control. Individual phenolic compounds, such as caffeic acid, ferulic acid, and kaempferol, exhibited a similar trend to the total phenolic concentration. | [131] |
| Winterbor and Redbor kale were harvested in three periods: before frost (>0 °C), after medium (−5.0 °C), and heavy frost (−15.0 °C). | Ascorbic acid (27% and 14%), phenolics (60% and 90%), and antioxidant activity (340% and 80%) were higher after heavy frost in Winterbor and Redbor cultivars, respectively. Anthocyanins also increased significantly for ‘Redbor’ cultivar by 511% and 400%, repectively, with medium and heavy frost. | [132] | |
| Fifty-day-old kale sprouts (Brassica oleracea var. acephala) were subjected to low temperature for 4 days: 25 °C/20 °C, 20 °C/15 °C, 15 °C/10 °C | The total carotenoid content was higher at 25 °C/20 °C treatment, but lower at 15 °C/10 °C treatment. The lutein and β-carotene contents decreased 18.3% and 81.6% respectively, by 15℃/10℃ treatment; while zeaxanthin content increased 364% and α-carotene was not affected by cold stress. | [133] | |
| Temperature | Four-day-old kale sprouts were soaked at 50 °C in water for 10, 20, 30, 45, or 60 s. | Heat shock at 50 °C for 20 s induced 150% and 120% higher total phenolic concentration and antioxidants capacity, respectively, than control. Accumulation of total glucosinolates significantly increased by 33% with the same treatment. | [134] |
| Radiation | Kale sprouts (Half Tall and Black Magic) were radiated with medium light (130 μmol photons/m2 s) or in high light (800 μmol photons/m2 s). | Total indole glucosinolates decreased in Black Magic cultivar (40%) treated with high light. However, glucoraphanin (150%), glucoerucin (350%), and total aliphatic glucosinolates (175%) content increased. Similarly, Half Tall cultivar showed an increase in glucoberverin (2%), glucoraphanin (400%), glucoerucin (100%), and total aliphatic glucosinolates (66%) with high light treatment. | [135] |
| Nine-week-old kale plants (Brassica oleracea var. acephala) were exposed to UV-B (0–3, 3–6 and 6–9 W/m2) for 4 h per day. | The increase in the concentration of total phenolics and flavonoids by UVB treatments was not significantly different. | [136] | |
| 10-day-old kale sprouts (Brassica oleracea var. sabellica) were light simulated 2 h each day with UV-B (0.0189 W/m2) and UV-A (69.502 W/m2). | Antioxidant activity increased 2–3-fold in UV-B-treated kale. Kaempferol glycosides decreased in favor of increasing quercetin glycosides. Quercetin-3,7,4 ′-O-d-triglucoside, quercetin-3-O-caffeoyl-sophoroside-7-O-glucoside and quercetin-3-O-sin-apoyl-sophoroside-7-O-d-glucoside significantly increased 26%, 30% and 33%, respectively, compared to control. | [137] | |
| 5-week-old kale seedlings (Brassica oleracea var. acephala) were radiated with UV-A LEDs (370 and 385 nm, 30 W/m2) continuously for 5 days. | Total phenolic content increased 25% and 42% in kale treated with UV-A LEDs 370 nm and at 385 nm, respectively, at 5 days of treatment. Caffeic acid (200% and 180%) and kaempferol (146% and 168%) concentrations were significantly increased by 370 and 385 nm UV-A radiation, respectively. | [138] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortega-Hernández, E.; Antunes-Ricardo, M.; Jacobo-Velázquez, D.A. Improving the Health-Benefits of Kales (Brassica oleracea L. var. acephala DC) through the Application of Controlled Abiotic Stresses: A Review. Plants 2021, 10, 2629. https://doi.org/10.3390/plants10122629
Ortega-Hernández E, Antunes-Ricardo M, Jacobo-Velázquez DA. Improving the Health-Benefits of Kales (Brassica oleracea L. var. acephala DC) through the Application of Controlled Abiotic Stresses: A Review. Plants. 2021; 10(12):2629. https://doi.org/10.3390/plants10122629
Chicago/Turabian StyleOrtega-Hernández, Erika, Marilena Antunes-Ricardo, and Daniel A. Jacobo-Velázquez. 2021. "Improving the Health-Benefits of Kales (Brassica oleracea L. var. acephala DC) through the Application of Controlled Abiotic Stresses: A Review" Plants 10, no. 12: 2629. https://doi.org/10.3390/plants10122629
APA StyleOrtega-Hernández, E., Antunes-Ricardo, M., & Jacobo-Velázquez, D. A. (2021). Improving the Health-Benefits of Kales (Brassica oleracea L. var. acephala DC) through the Application of Controlled Abiotic Stresses: A Review. Plants, 10(12), 2629. https://doi.org/10.3390/plants10122629









