Plant Functional Genomics in A Few Days: Laser-Assisted Delivery of Double-Stranded RNA to Higher Plants
Abstract
1. Introduction
2. Results and Discussion
2.1. Laser Generated Perforations
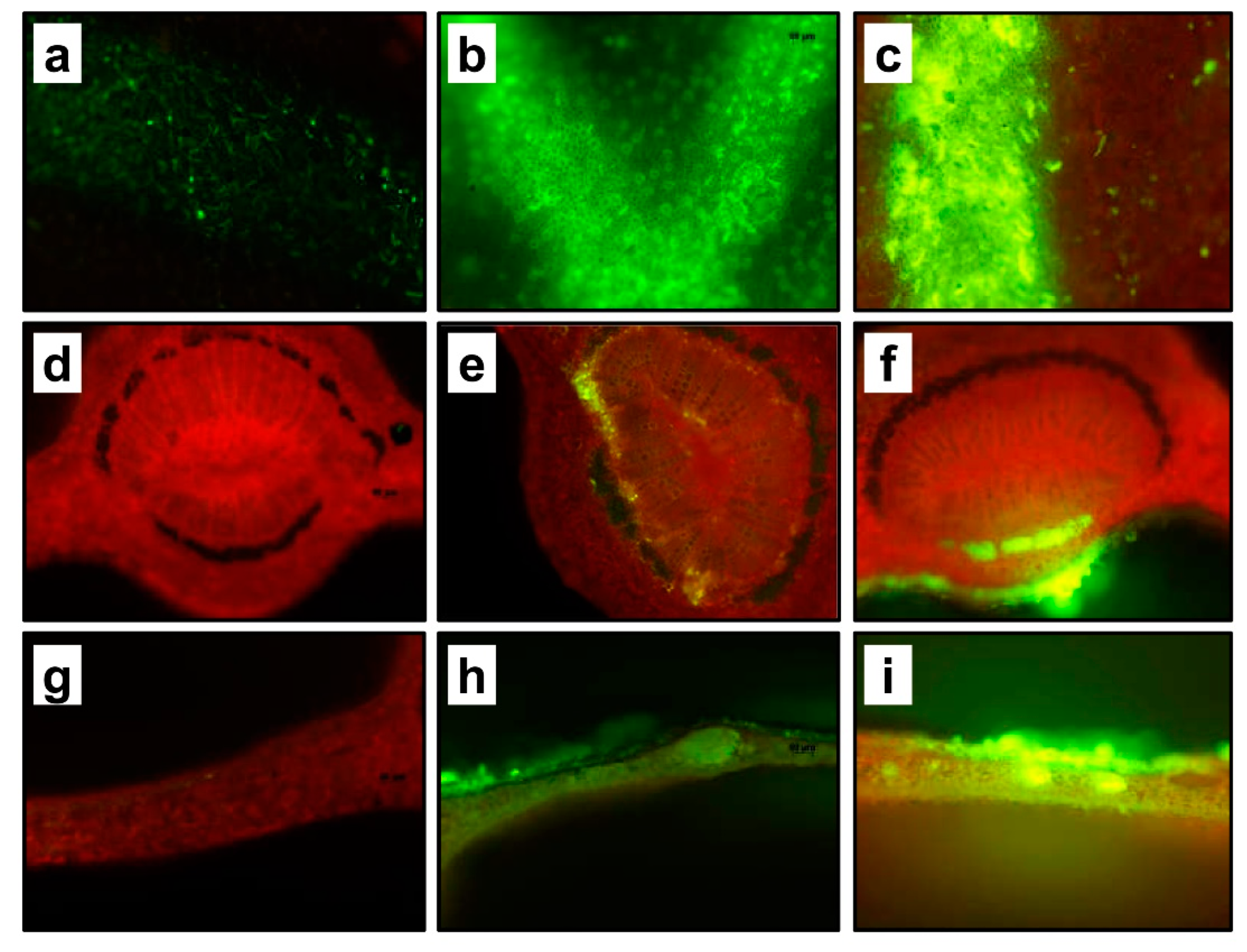
2.2. Leaf Penetration and Mobility of Experimental Solutions
2.3. Phenotype Changes and Gene Expression
3. Material and Methods
3.1. Plant Material
3.2. Synthesizing of the dsRNA
3.3. Labeling of the dsRNA
3.4. Laser Treatment
3.5. Experimental Solutions
3.6. Microscopy
3.7. RNA Extraction and Gene Expression
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CLas | Candidatus Liberibacter asiaticus |
| dsRNA | Double stranded RNA |
| GFP | Green fluorescent protein |
| HLB | Huanglongbing |
| mRNA | Messenger RNA |
| 2-NBDG | 2-[ N -(7-nitrobenz2-oxa-1,3-diazol-4-yl)amino]-2-deoxyglucose |
| PDS | Phytoene desaturase |
| RNAi | RNA interference |
| siRNAs | Small interfering RNAs |
| VIGS | Virus-induced gene silencing |
References
- Price, D.R.G.; Gatehouse, J.A. RNAi-mediated crop protection against insects. Trends Biotechnol. 2008, 26, 393–400. [Google Scholar] [CrossRef]
- Koch, A.; Kogel, K.H. New wind in the sails: Improving the agronomic value of crop plants through RNAi-mediated gene silencing. Plant Biotechnol. J. 2014, 12, 821–831. [Google Scholar] [CrossRef]
- Gogoi, A.; Sarmah, N.; Kaldis, A.; Perdikis, D.; Voloudakis, A. Plant insects and mites uptake double-stranded RNA upon its exogenous application on tomato leaves. Planta 2017, 246, 1233–1241. [Google Scholar] [CrossRef]
- Grafton-Cardwell, E.E.; Stelinski, L.L.; Stansly, P.A. Biology and Management of Asian Citrus Psyllid, Vector of the Huanglongbing Pathogens. Annu. Rev. Entomol. 2013, 58, 413–432. [Google Scholar] [CrossRef]
- Hall, D.G.; Richardson, M.L.; Ammar, E.-D.; Halbert, S.E. Asian citrus psyllid, Diaphorina citri, vector of citrus huanglongbing disease. Entomol. Exp. Appl. 2013, 146, 207–223. [Google Scholar] [CrossRef]
- Folimonova, S.Y.; Robertson, C.J.; Garnsey, S.M.; Gowda, S.; Dawson, W.O. Examination of the Responses of Different Genotypes of Citrus to Huanglongbing (Citrus Greening) Under Different Conditions. Phytopathology 2009, 99, 1346–1354. [Google Scholar] [CrossRef]
- Bove, J.M. Huanglongbing: A destructive, newly-emerging, century-old disease of citrus. J. Plant Pathol. 2006, 88, 7–37. [Google Scholar]
- Singerman, A.; Useche, P. Impact of citrus greening on citrus operations in Florida. UF/IFAS Ext. 2016, #FE983. Available online: https://edis.ifas.ufl.edu/fe983 (accessed on 8 October 2020).
- Hajeri, S.; Killiny, N.; El-Mohtar, C.; Dawson, W.O.; Gowda, S. Citrus tristeza virus-based RNAi in citrus plants induces gene silencing in Diaphorina citri, a phloem-sap sucking insect vector of citrus greening disease (Huanglongbing). J. Biotechnol. 2014, 176, 42–49. [Google Scholar] [CrossRef]
- Ruiz, M.T.; Voinnet, O.; Baulcombe, D.C. Initiation and maintenance of virus-induced gene silencing. Plant Cell 1998, 10, 937–946. [Google Scholar]
- Anandalakshmi, R.; Pruss, G.J.; Ge, X.; Marathe, R.; Mallory, A.C.; Smith, T.H.; Vance, V.B. A viral suppressor of gene silencing in plants. Proc. Natl. Acad. Sci. USA 1998, 95, 13079–13084. [Google Scholar] [CrossRef]
- Ratcliff, F.; Martin-Hernandez, A.M.; Baulcombe, D.C. Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J. 2001, 25, 237–245. [Google Scholar] [CrossRef]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. Available online: http://www.nature.com/nature/journal/v391/n6669/suppinfo/391806a0_S1.html (accessed on 15 September 2020). [CrossRef]
- Killiny, N.; Hajeri, S.; Tiwari, S.; Gowda, S.; Stelinski, L.L. Double-Stranded RNA Uptake through Topical Application, Mediates Silencing of Five CYP4 Genes and Suppresses Insecticide Resistance in Diaphorina citri. PLoS ONE 2014, 9, e110536. [Google Scholar] [CrossRef]
- Badr, Y.A.; Kereim, M.A.; Yehia, M.A.; Fouad, O.O.; Bahieldin, A. Production of fertile transgenic wheat plants by laser micropuncture. Photochem. Photobiol. Sci. 2005, 4, 803–807. [Google Scholar] [CrossRef]
- Etxeberria, E.; Gonzalez, P.; Fanton Borges, A.; Brodersen, C. The use of laser light to enhance the uptake of foliar-applied substances into citrus (Citrus sinensis) leaves. Appl. Plant Sci. 2016, 4. [Google Scholar] [CrossRef]
- Etxeberria, E.; Gonzalez, P.; Tomlinson, P.; Pozueta-Romero, J. Existence of two parallel mechanisms for glucose uptake in heterotrophic plant cells. J. Exp. Bot. 2005, 56, 1905–1912. [Google Scholar] [CrossRef]
- Dubrovina, A.S.; Kiselev, K.V. Exogenous RNAs for gene regulation and plant resistance. Int. J. Mol. Sci. 2019, 20, 2282. [Google Scholar] [CrossRef]
- Uslu, V.V.; Wassenegger, M. Critical view on RNA silencing-mediated virus resistance using exogenously applied RNA. Curr. Opin. Virol. 2020, 42, 18–24. [Google Scholar] [CrossRef]
- Worrall, E.A.; Bravo-Cazar, A.; Nilon, A.T.; Fletcher, S.J.; Robinson, K.; Carr, J.P.; Mitter, N. Exogenous application of RNAi-Inducing double-stranded RNA inhibits aphid-mediated transmission of a plant virus. Front. Plant Sci. 2019, 10, 265. [Google Scholar] [CrossRef]
- Sood, P.; Exteberria, E.; Narciso, J.; Ference, C. Laser labeling and its effect on the storage quality of citrus fruits. In Proceedings of the American Society of Horticulture Science, St. Louis, MO, USA, 24–28 July 2009; p. 1004. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Killiny, N.; Gonzalez-Blanco, P.; Gowda, S.; Martini, X.; Etxeberria, E. Plant Functional Genomics in A Few Days: Laser-Assisted Delivery of Double-Stranded RNA to Higher Plants. Plants 2021, 10, 93. https://doi.org/10.3390/plants10010093
Killiny N, Gonzalez-Blanco P, Gowda S, Martini X, Etxeberria E. Plant Functional Genomics in A Few Days: Laser-Assisted Delivery of Double-Stranded RNA to Higher Plants. Plants. 2021; 10(1):93. https://doi.org/10.3390/plants10010093
Chicago/Turabian StyleKilliny, Nabil, Pedro Gonzalez-Blanco, Siddarame Gowda, Xavier Martini, and Ed Etxeberria. 2021. "Plant Functional Genomics in A Few Days: Laser-Assisted Delivery of Double-Stranded RNA to Higher Plants" Plants 10, no. 1: 93. https://doi.org/10.3390/plants10010093
APA StyleKilliny, N., Gonzalez-Blanco, P., Gowda, S., Martini, X., & Etxeberria, E. (2021). Plant Functional Genomics in A Few Days: Laser-Assisted Delivery of Double-Stranded RNA to Higher Plants. Plants, 10(1), 93. https://doi.org/10.3390/plants10010093





