Effect of Crude Oil on Growth, Oxidative Stress and Response of Antioxidative System of Two Rye (Secale cereale L.) Varieties
Abstract
1. Introduction
2. Results
2.1. Effect of Toxic Concentrations of Crude Oil on Plant Growth and Biomass of Rye Varieties
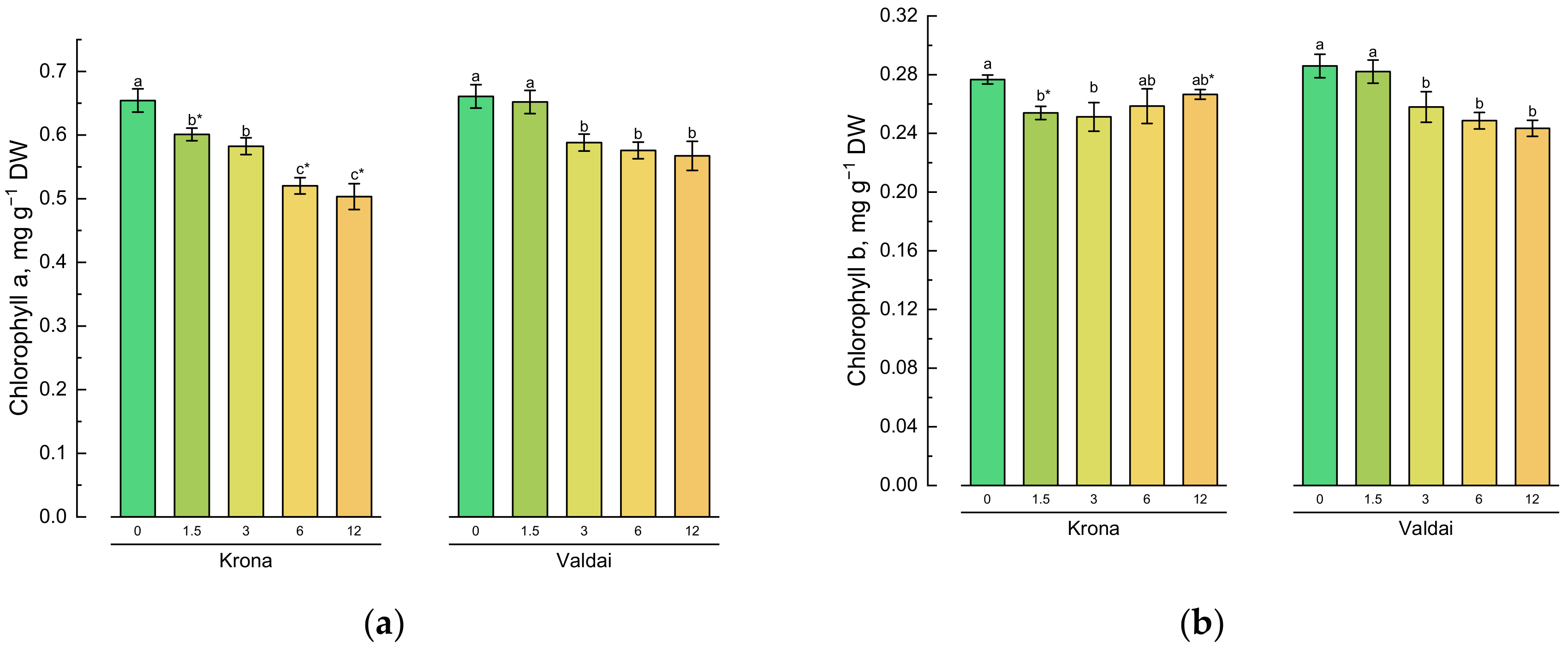
2.2. Effect of Toxic Concentrations of Crude Oil on Chlorophylls and Carotenoids of Rye Varieties
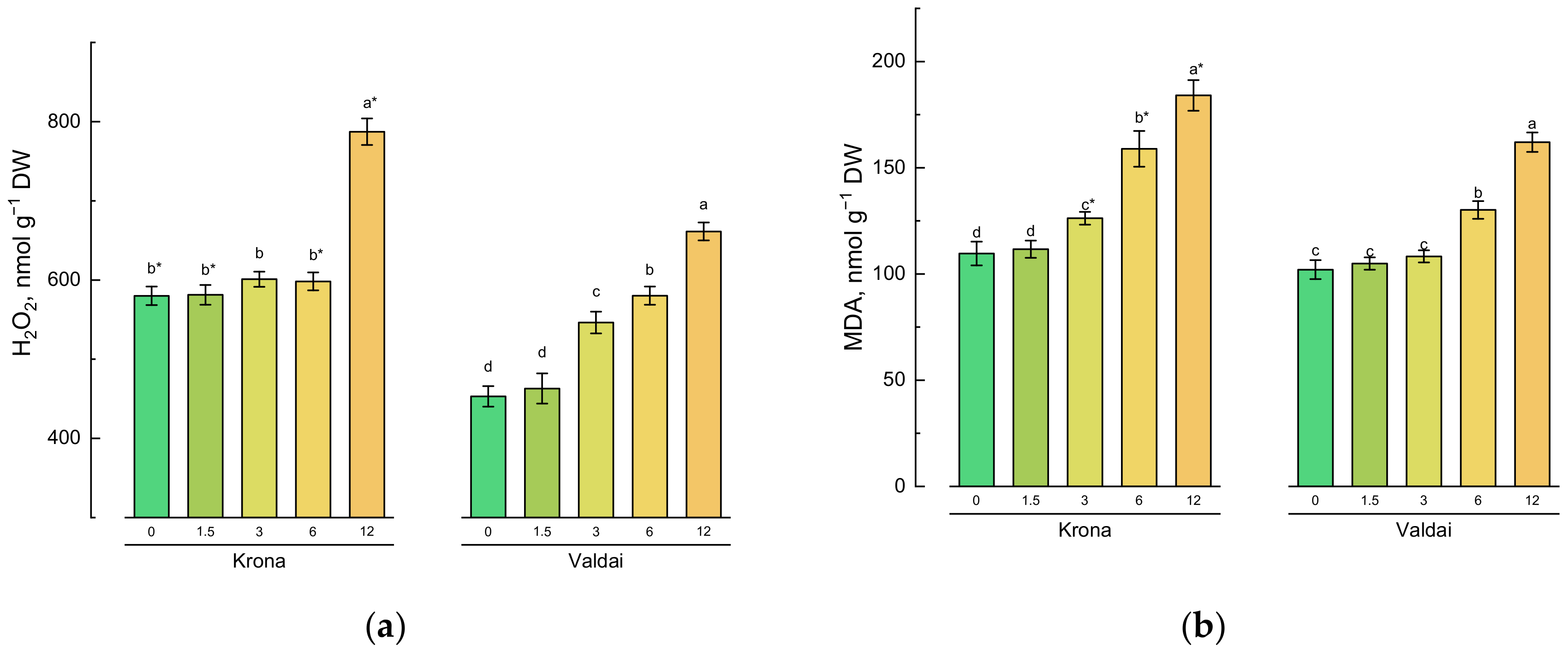
2.3. Effect of Toxic Concentrations of Crude Oil on Oxidative Stress Parameters in Rye Varieties
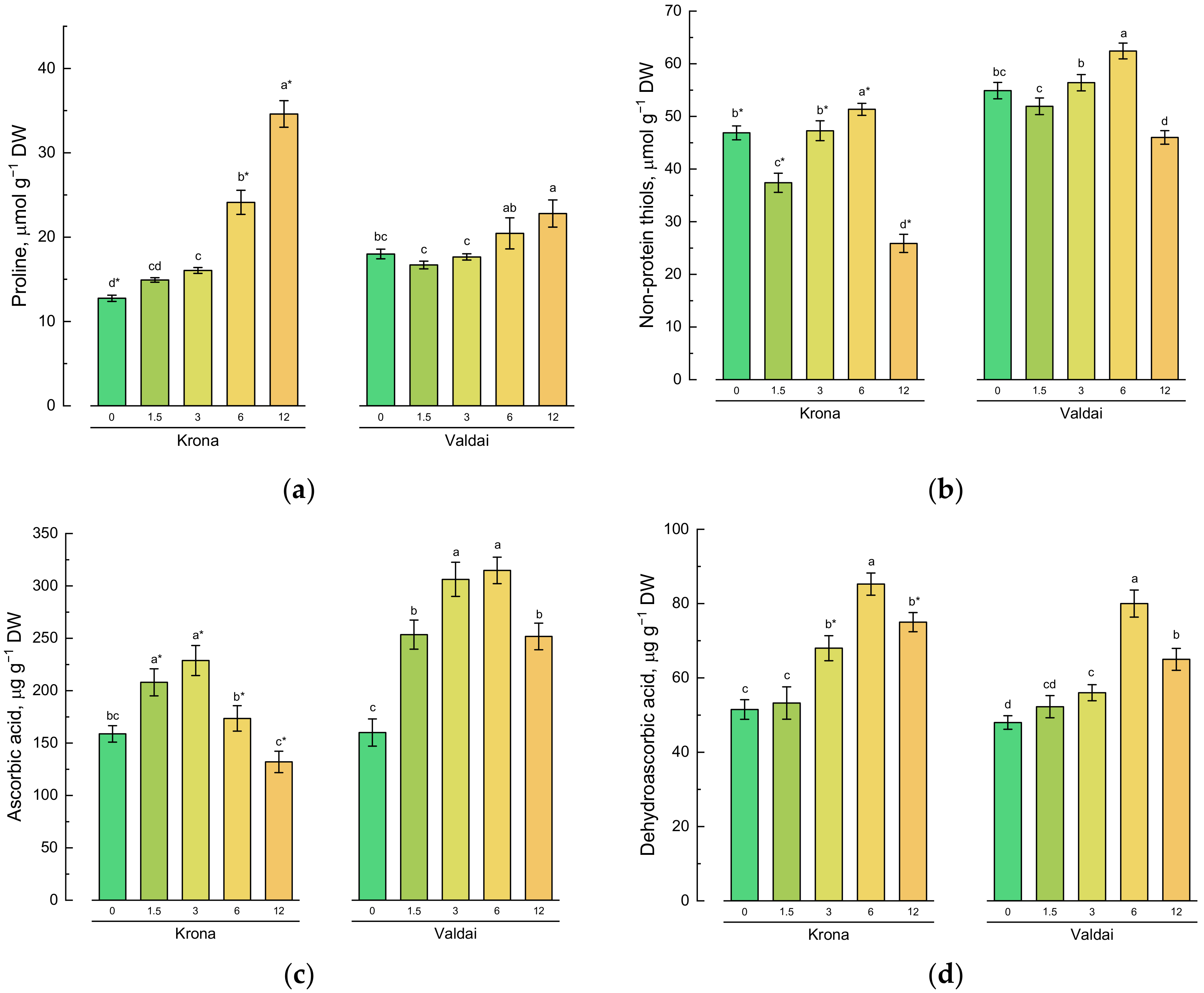
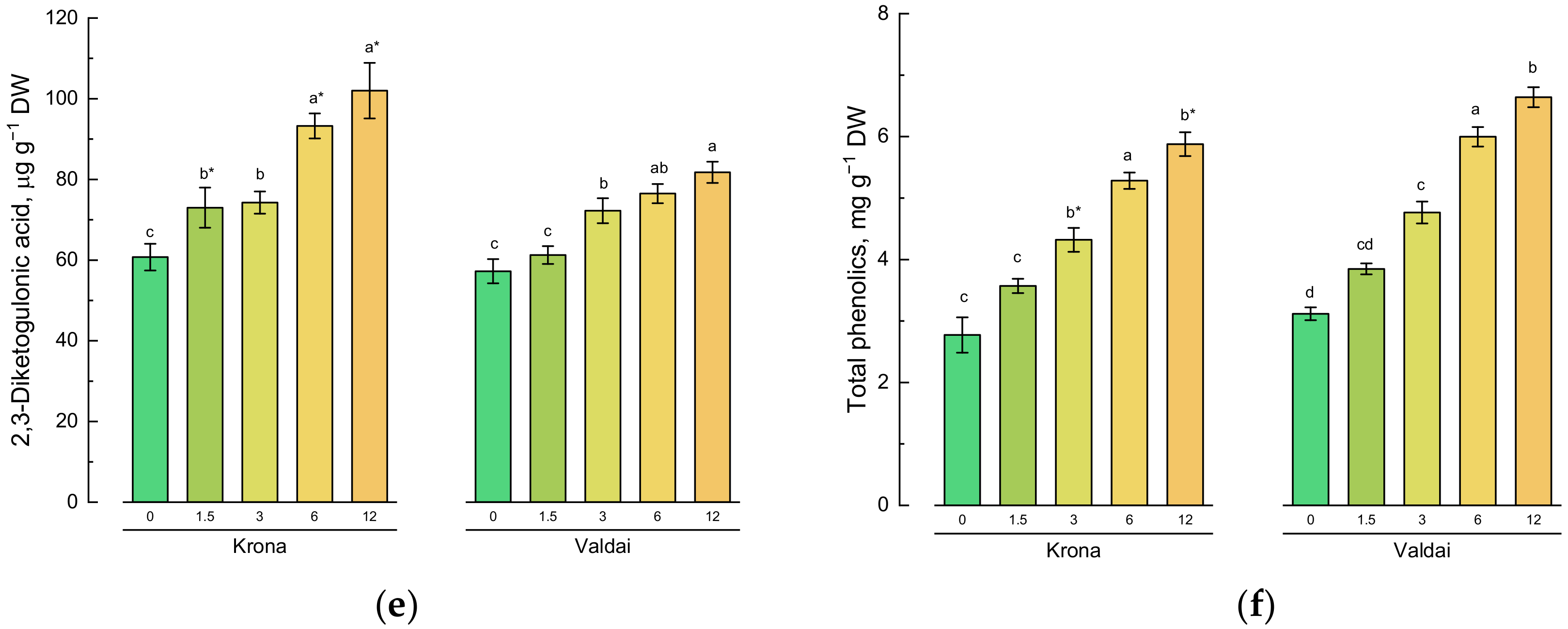
2.4. Effect of Toxic Concentrations of Crude Oil on Non-Enzymatic Antioxidants of Rye Varieties
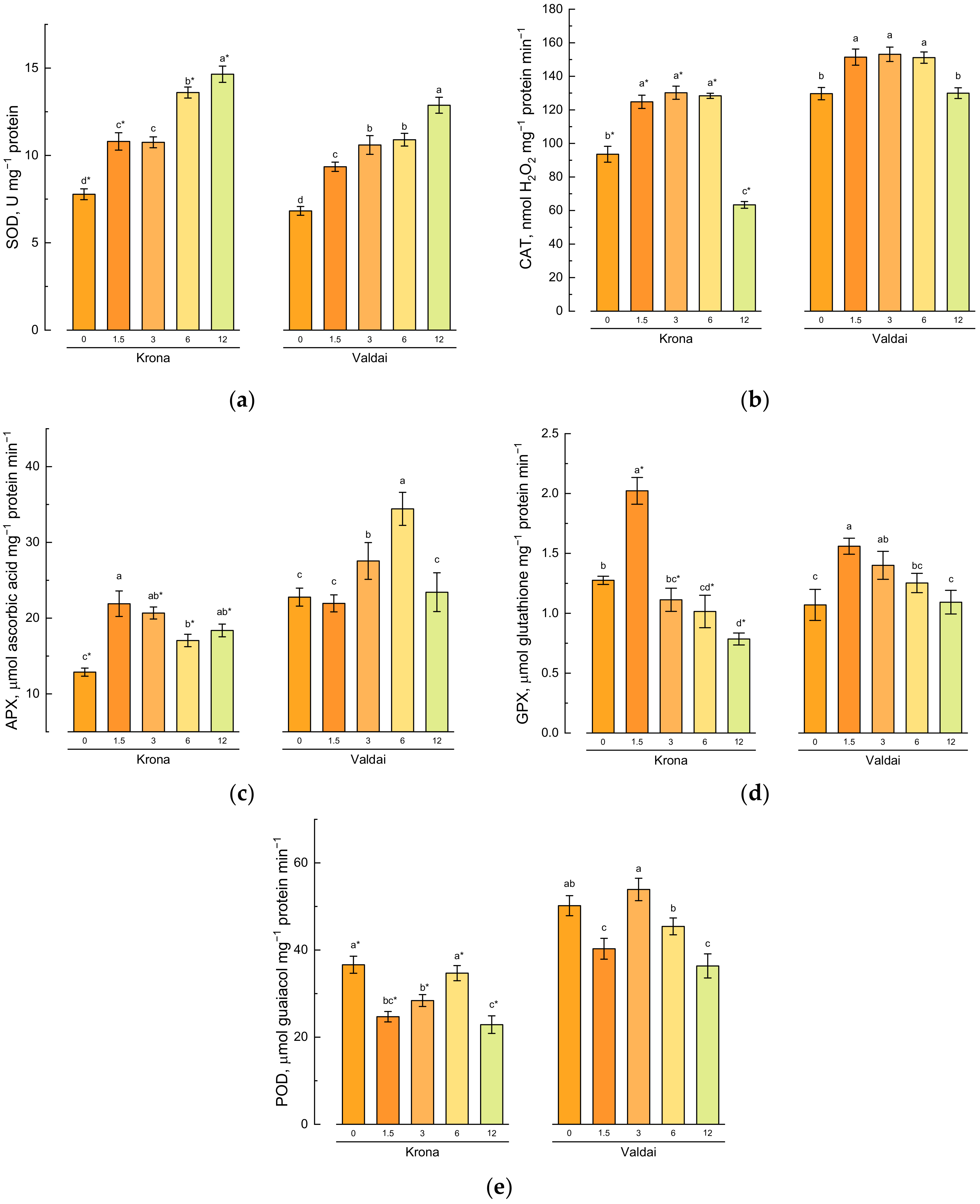
2.5. Effect of Toxic Concentrations of Crude Oil on Activities of Antioxidative Enzymes in Rye Varieties
2.6. Relationship between Growth Parameters, Oxidative Stress and Antioxidative Response of Rye Varieties
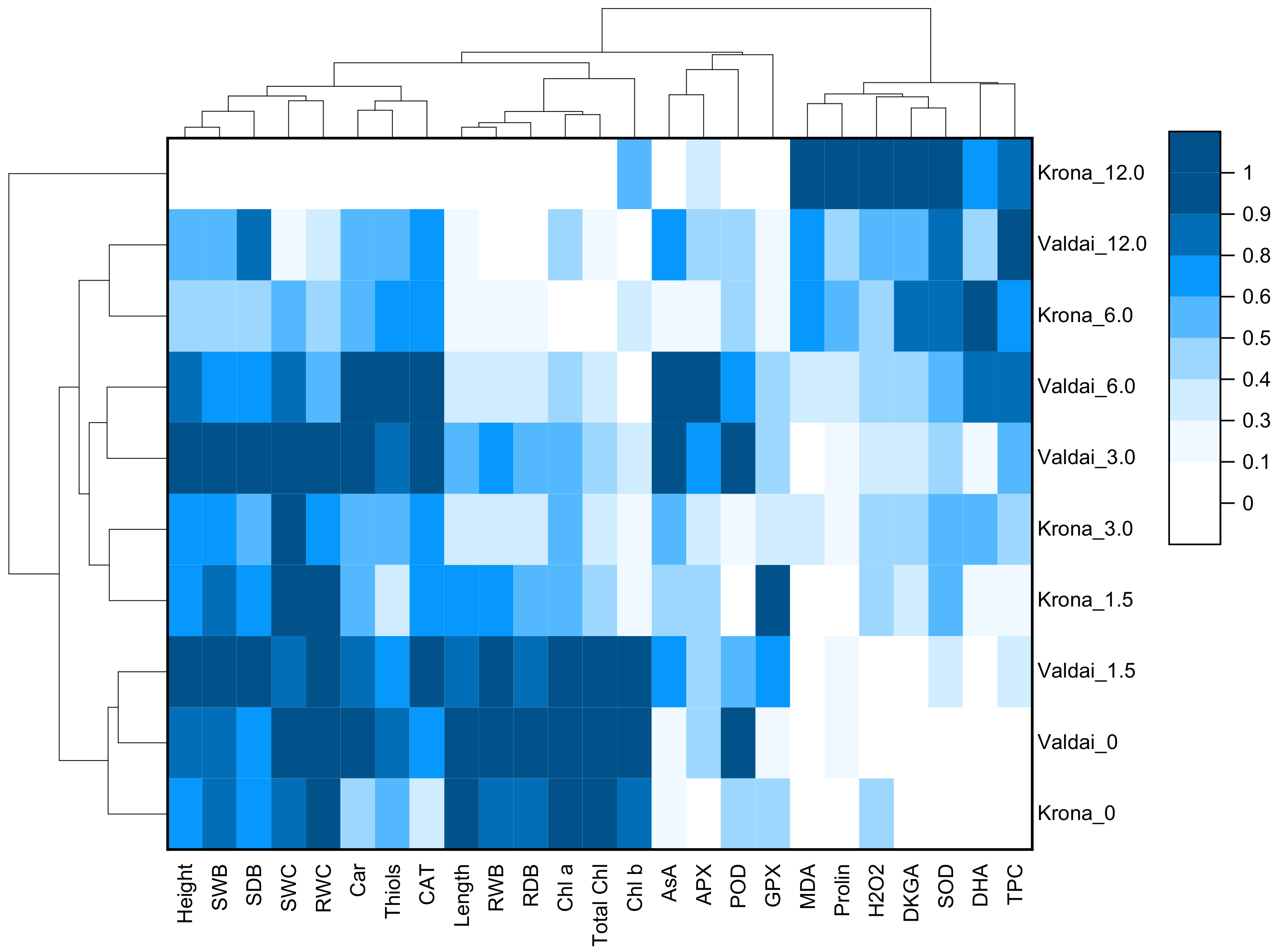
2.7. Heat Map and Cluster Analysis of Growth Parameters, Oxidative Stress and Antioxidative Response of Rye Varieties at Different Concentration of Crude Oil in Soil
3. Discussion
3.1. Difference in Growth Response of Two Rye Varieties to Oil Pollution
3.2. Changes in Biochemical Parameters of Rye under Crude Oil-Induced Stress
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Plant Materials and Growth Conditions
4.3. Plant Harvesting and Sample Preparation
4.4. Analysis of the Biochemical Parameters of the Plants
4.4.1. Photosynthetic Pigments
4.4.2. Malondialdehyde
4.4.3. Hydrogen Peroxide
4.4.4. Proline
4.4.5. Non-Protein Thiols
4.4.6. Ascorbic, Dehydroascorbic and 2,3-Diketogulonic Acids
4.4.7. Total Phenolic Compounds
4.4.8. Antioxidative Enzymes Activity
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Main Effects 1 | Factors | Chl a | Chl b | Total Chl | Car | MDA | H2O2 |
|---|---|---|---|---|---|---|---|
| Oil concentration | 0 | 0.658a | 0.281a | 0.939a | 0.192a | 105.9d | 516.5c |
| (O) | 1.5 | 0.626b | 0.268b | 0.894b | 0.192a | 108.3d | 522.1c |
| 3.0 | 0.585c | 0.255c | 0.840c | 0.197a | 117.3c | 573.6b | |
| 6.0 | 0.548d | 0.254c | 0.802d | 0.195a | 144.5b | 589.3b | |
| 12.0 | 0.535d | 0.255c | 0.790d | 0.171b | 173.0a | 724.3a | |
| Rye variety | Krona | 0.572b | 0.261a | 0.834b | 0.178b | 138.1a | 629.6a |
| (V) | Valdai | 0.609a | 0.264a | 0.872a | 0.201a | 121.5b | 540.8b |
| Significance | O | * | * | * | * | * | * |
| V | * | ns | * | * | * | * | |
| O*V | * | * | * | ns | * | * |
| Main Effects 1 | Factors | Proline | -SH | AsA | DHA | DKGA | TPC | SOD | CAT | APX | GPX | POD |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oil concentration | 0 | 15.38c | 50.89b | 159d | 49.8d | 59.0e | 2.95e | 7.30a | 111.6b | 17.8d | 1.17b | 43.4a |
| (O) | 1.5 | 15.81c | 44.66c | 231b | 52.8d | 67.1d | 3.71d | 10.08b | 138.1a | 21.9bc | 1.79a | 32.5c |
| 3.0 | 16.85c | 51.84b | 268a | 62.0c | 73.3c | 4.54c | 10.68c | 141.7a | 24.1ab | 1.26b | 41.2ab | |
| 6.0 | 22.29b | 56.89a | 244b | 82.6a | 84.9b | 5.64b | 12.25d | 139.8a | 25.7a | 1.13b | 40.1b | |
| 12.0 | 28.70a | 35.94d | 191c | 70.1b | 91.8a | 6.26a | 13.76e | 96.7c | 20.9c | 0.94c | 29.6c | |
| Rye variety | Krona | 20.49a | 41.76b | 180a | 66.7a | 80.7a | 4.37b | 11.52a | 108.1b | 18.2b | 1.24a | 29.5b |
| (V) | Valdai | 19.12b | 54.33a | 257b | 60.3b | 69.8b | 4.87a | 10.11b | 143.1a | 26.0a | 1.28a | 45.2a |
| Significance | O | * | * | * | * | * | * | * | * | * | * | * |
| V | * | * | * | * | * | * | * | * | * | ns | * | |
| O*V | * | * | * | * | * | * | * | * | * | * | * |
References
- Abdollahzadeh, T.; Niazi, A.; Moghadam, A.; Heydarian, Z.; Ghasemi-Fasaei, R.; Kaviani, E.; Pourdad, N. Phytoremediation of petroleum-contaminated soil by Salicornia: From PSY activity to physiological and morphological communications. Environ. Technol. 2019, 40, 2789–2801. [Google Scholar] [CrossRef]
- Odukoya, J.; Lambert, R.; Sakrabani, R. Impact of crude oil on yield and phytochemical composition of selected green leafy vegetables. Int. J. Veg. Sci. 2019, 25, 554–570. [Google Scholar] [CrossRef]
- Gamage, S.S.W.; Masakorala, K.; Brown, M.T.; Gamage, S.M.K.W. Tolerance of Impatiens balsamina L., and Crotalaria retusa L. to grow on soil contaminated by used lubricating oil: A comparative study. Ecotoxicol. Environ. Saf. 2020, 188, 109911. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Wang, A.; Zhang, M.; Li, H.; Du, S.; Bai, L.; Zhong, M.; Chen, S. Compared the physiological response of two petroleum tolerant-contrasting plants to petroleum stress. Int. J. Phytoremediation 2018, 20, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Rusin, M.; Gospodarek, J.; Barczyk, G.; Nadgórska-Socha, A. Antioxidant responses of Triticum aestivum plants to petroleum-derived substances. Ecotoxicology 2018, 27, 1353–1367. [Google Scholar] [CrossRef] [PubMed]
- Peretiemo-Clarke, B.O.; Achuba, F.I. Phytochemical effect of petroleum on peanut (Arachis hypogea) seedlings. Plant Pathol. J. 2007, 6, 179–182. [Google Scholar] [CrossRef]
- Achuba, F.I. Petroleum products in soil mediated oxidative stress in cowpea (Vigna unguiculata) and maize (Zea mays) seedlings. Open J. Soil Sci. 2014, 4, 417–435. [Google Scholar] [CrossRef]
- Otitoju, O.; Udebuani, A.C.; Ebulue, M.M.; Onwurah, I.N. Enzyme-based assay for toxicological evaluation of soil ecosystem polluted with spent engine oil. Agric. Ecol. Res. Int. J. 2017, 11, 1–13. [Google Scholar] [CrossRef]
- Achuba, F.I.; Ja-anni, M.O. Effect of abattoir waste water on metabolic and antioxidant profiles of cowpea seedlings grown in crude oil contaminated soil. Int. J. Recycl. Org. Waste Agric. 2018, 7, 59–66. [Google Scholar] [CrossRef]
- Bellout, Y.; Khelif, L.; Guivarch, A.; Haouche, L.; Djebbar, R.; Carol, P.; Abrous Belbachir, O. Impact of edaphic hydrocarbon pollution on the morphology and physiology of pea roots (Pisum sativum L.). Appl. Ecol. Environ. Res. 2016, 14, 511–525. [Google Scholar] [CrossRef]
- Athar, H.-R.; Ambreen, S.; Javed, M.; Hina, M.; Rasul, S.; Zafar, Z.U.; Manzoor, H.; Ogbaga, C.C.; Afzal, M.; Al-Qurainy, F.; et al. Influence of sub-lethal crude oil concentration on growth, water relations and photosynthetic capacity of maize (Zea mays L.) plants. Environ. Sci. Pollut. Res. 2016, 23, 18320–18331. [Google Scholar] [CrossRef]
- Al-Hawas, G.H.S.; Shukry, W.M.; Azzoz, M.M.; Al-Moaik, R.M.S. The effect of sublethal concentrations of crude oil on the metabolism of Jojoba (Simmodsia chinensis) seedlings. Int. Res. J. Plant Sci. 2012, 3, 54–62. [Google Scholar]
- Poljšak, B.; Fink, R. The Protective Role of Antioxidants in the Defence against ROS/RNS-Mediated Environmental Pollution. Oxidative Med. Cell Longev. 2014, 2014, 671539. [Google Scholar] [CrossRef]
- Maslennikov, P.V.; Chupakhina, G.N.; Skrypnik, L.N.; Feduraev, P.V.; Melnik, A.S. Assessment of the Antioxidant Potential of Plants in Urban Ecosystems under Conditions of Anthropogenic Pollution of Soils. Russ. J. Ecol. 2018, 49, 384–394. [Google Scholar] [CrossRef]
- Zaid, A.; Wani, S.H. Reactive Oxygen Species Generation, Scavenging and Signaling in Plant Defense Responses. In Bioactive Molecules in Plant Defense; Jogaiah, S., Abdelrahman, M., Eds.; Springer: Cham, Switzerland, 2019; pp. 111–132. [Google Scholar] [CrossRef]
- Kohli, S.K.; Khanna, K.; Bhardwaj, R.; Abd_Allah, E.F.; Ahmad, P.; Corpas, F.J. Assessment of Subcellular ROS and NO Metabolism in Higher Plants: Multifunctional Signaling Molecules. Antioxidants 2019, 8, 641. [Google Scholar] [CrossRef]
- Rusin, M.; Gospodarek, J.; Nadgórska-Socha, A. Soil Pollution by Petroleum-Derived Substances and its Bioremediation: The Effect on Aphis fabae Scop. Infestation and Antioxidant Response in Vicia faba L. Agronomy 2020, 10, 147. [Google Scholar] [CrossRef]
- Chupakhina, G.N.; Maslennikov, P.V. Plant adaptation to oil stress. Russ. J. Ecol. 2004, 35, 290–295. [Google Scholar] [CrossRef]
- Noori, A.S.; Maivan, H.Z.; Alaie, E. Changes in total phenol and flavonoid contents in Chrysanthemum leucanthemum under crude oil contamination. Adv. Environ. Biol. 2012, 6, 3057–3064. [Google Scholar]
- Pihlava, J.M.; Hellström, J.; Kurtelius, T.; Mattila, P. Flavonoids, anthocyanins, phenolamides, benzoxazinoids, lignans and alkylresorcinols in rye (Secale cereale) and some rye products. J. Cereal Sci. 2018, 79, 183–192. [Google Scholar] [CrossRef]
- Kuzel, S.; Romanova, I.; Augspole, I.; Linina, A. Winter rye (Secale cereale L.) antioxidant capacity, total phenolic content and quality indices. Agron. Res. 2020, 18, 1751–1759. [Google Scholar] [CrossRef]
- Miedaner, T.; Laidig, F. Hybrid breeding in rye (Secale cereale L.). In Advances in Plant Breeding Strategies: Cereals; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer: Cham, Switzerland, 2019; Volume 5, pp. 343–372. [Google Scholar] [CrossRef]
- Choden, D.; Pokethitiyook, P.; Poolpak, T.; Kruatrachue, M. Phytoremediation of soil co-contaminated with zinc and crude oil using Ocimum gratissimum (L.) in association with Pseudomonas putida MU02. Int. J. Phytoremediation 2020. [Google Scholar] [CrossRef]
- Hatami, E.; Abbaspour, A.; Dorostkar, V. Phytoremediation of a petroleum-polluted soil by native plant species in Lorestan Province, Iran. Environ. Sci. Pollut. Res. Int. 2019, 26, 24323–24330. [Google Scholar] [CrossRef]
- Liao, C.; Xu, W.; Lu, G.; Deng, F.; Liang, X.; Guo, C.; Dang, Z. Biosurfactant-enhanced phytoremediation of soils contaminated by crude oil using maize (Zea mays L). Ecol. Eng. 2016, 92, 10–17. [Google Scholar] [CrossRef]
- Moubasher, H.A.; Hegazy, A.K.; Mohamed, N.H.; Moustafa, Y.M.; Kabiel, H.F.; Hamad, A.A. Phytoremediation of soils polluted with crude petroleum oil using Bassia scoparia and its associated rhizosphere microorganisms. Int. Biodeterior. Biodegrad. 2015, 98, 113–120. [Google Scholar] [CrossRef]
- Ayotamuno, J.M.; Kogbara, R.B. Determining the tolerance level of Zea mays (maize) to a crude oil polluted agricultural soil. Afr. J. Biotechnol. 2007, 6, 1332–1337. [Google Scholar]
- Gospodarek, J.; Rusin, M.; Nadgórska-Socha, A. Effect of Petroleum-Derived Substances and their Bioremediation on Triticum aestivum L. Growth and Chemical Composition. Pol. J. Environ. Stud. 2019, 28, 2131–2137. [Google Scholar] [CrossRef]
- Odiyi, B.O.; Giwa, G.O.; Abiya, S.E.; Babatunde, O.S. Effects of crude oil pollution on the morphology, growth and heavy metal content of maize (Zea mays Linn.). J. Environ. Manag. 2020, 24, 119–125. [Google Scholar] [CrossRef]
- Grifoni, M.; Rosellini, I.; Angelini, P.; Petruzzelli, G.; Pezzarossa, B. The effect of residual hydrocarbons in soil following oil spillages on the growth of Zea mays plants. Environ. Pollut. 2020, 265, 114950. [Google Scholar] [CrossRef]
- De Sousa, A.; AbdElgawad, H.; Fidalgo, F.; Teixeira, J.; Matos, M.; Hamed, B.A.; Selim, S.; Hozzein, W.N.; Beemster, G.T.S.; Asard, H. Al exposure increases proline levels by different pathways in an Al-sensitive and an Al-tolerant rye genotype. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Goncharenko, A.A.; Makarov, A.V.; Ermakov, S.A.; Semenova, T.V.; Tochilin, V.N.; Tsygankova, N.V.; Skatova, S.E.; Krakhmaleva, O.A. Ecological Stability of Short Stemmed Winter Rye Varieties. Russ. Agric. Sci. 2019, 45, 315–322. [Google Scholar] [CrossRef]
- Karmanenko, N.M. Response to low temperature, soil acidification and aluminium in the varieties of cereal crops. Agric. Biol. 2014, 5, 66–77. [Google Scholar] [CrossRef]
- Odukoya, J.; Lambert, R.; Sakrabani, R. Understanding the Impacts of Crude Oil and its Induced Abiotic Stresses on Agrifood Production: A Review. Horticulturae 2019, 5, 47. [Google Scholar] [CrossRef]
- Langer, I.; Syafruddin, S.; Steinkellner, S.; Puschenreiter, M.; Wenzel, W.W. Plant growth and root morphology of Phaseolus vulgaris L. grown in a split-root system is affected by heterogeneity of crude oil pollution and mycorrhizal colonization. Plant Soil 2010, 332, 339–355. [Google Scholar] [CrossRef]
- Basumatary, B.; Bordoloi, S.; Sarma, H.P. Crude Oil-Contaminated Soil Phytoremediation by Using Cyperus brevifolius (Rottb.) Hassk. Water Air Soil Pollut. 2012, 223, 3373–3383. [Google Scholar] [CrossRef]
- Merkl, N.; Schultze-Kraft, R.; Infante, C. Assessment of tropical grasses and legumes for phytoremediation of petroleum-contaminated soils. Water Air Soil Pollut. 2005, 165, 195–209. [Google Scholar] [CrossRef]
- García-Segura, D.; Castillo-Murrieta, I.M.; Martínez-Rabelo, F.; Gomez-Anaya, A.; Rodríguez-Campos, J.; Hernández-Castellanos, B.; Contreras-Ramos, S.M.; Barois, I. Macrofauna and mesofauna from soil contaminated by oil extraction. Geoderma 2018, 332, 180–189. [Google Scholar] [CrossRef]
- Cevher-Keskin, B.; Selçukcan-Erol, Ç.; Yüksel, B.; Ertekin, Ö.; Yıldızhan, Y.; Onarıcı, S.; Kulen, O.; Memon, A.R. Comparative transcriptome analysis of Zea mays in response to petroleum hydrocarbon stress. Environ. Sci. Pollut. Res. 2018, 25, 32660–32674. [Google Scholar] [CrossRef]
- Hajihashemi, S.; Brestic, M.; Kalaji, H.M.; Skalicky, M.; Noedoost, F. Environmental pollution is reflected in the activity of the photosynthetic apparatus. Photosynthetica 2020, 58, 529–539. [Google Scholar] [CrossRef]
- Kreslavski, V.D.; Brestic, M.; Zharmukhamedov, S.K.; Lyubimov, V.Y.; Lankin, A.V.; Jajoo, A.; bAllakhverdiev, S.I. Mechanisms of inhibitory effects of polycyclic aromatic hydrocarbons in photosynthetic primary processes in pea leaves and thylakoid preparations. Plant Biol. 2017, 19, 683–688. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.B.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants 2020, 9, 681. [Google Scholar] [CrossRef]
- Zhang, C.G.; Leung, K.K.; Wong, Y.S.; Tam, N.F.Y. Germination, growth and physiological responses of mangrove plant (Bruguiera gymnorrhiza) to lubricating oil pollution. Environ. Exp. Bot. 2007, 60, 127–136. [Google Scholar] [CrossRef]
- Maslennikov, P.; Golovina, E.; Artemenko, A. Ecological and Geochemical Conditions for the Accumulation of Antioxidants in the Leaves of Lathyrus maritimus (L.) Bigel. Plants 2020, 9, 746. [Google Scholar] [CrossRef]
- Ahmad, P.; Jaleel, C.A.; Salem, M.A.; Nabi, G.; Sharma, S. Roles of enzymatic and nonenzymatic antioxidants in plants during abiotic stress. Crit. Rev. Biotechnol. 2010, 30, 161–175. [Google Scholar] [CrossRef]
- Maslennikov, P.V.; Chupakhina, G.N.; Skrypnik, L.N.; Feduraev, P.V.; Melnik, A.S. The contribution of polyphenols to plant resistance to Pb soil pollution. Int. J. Environ. Stud. 2018, 75, 719–731. [Google Scholar] [CrossRef]
- Radyukina, N.L.; Mikheeva, L.E.; Karbysheva, E.A. Low molecular weight antioxidants in cyanobacteria and plant cells. Biol. Bull. Rev. 2019, 9, 520–531. [Google Scholar] [CrossRef]
- Gong, Z.; Chen, W.; Bao, G.; Sun, J.; Ding, X.; Fan, C. Physiological response of Secale cereale L. seedlings under freezing-thawing and alkaline salt stress. Environ. Sci. Pollut. Res. 2020, 27, 1499–1507. [Google Scholar] [CrossRef]
- Siddique, A.; Kandpal, G.; Kumar, P. Proline accumulation and its defensive role under diverse stress condition in plants: An Overview. J. Pure Appl. Microbiol. 2018, 12, 1655–1659. [Google Scholar] [CrossRef]
- Ullah, R.; Hadi, F.; Ahmad, S.; Jan, A.U.; Rongliang, Q. Phytoremediation of lead and chromium contaminated soil improves with the endogenous phenolics and proline production in Parthenium, Cannabis, Euphorbia, and Rumex species. Water Air Soil Pollut. 2019, 230, 40. [Google Scholar] [CrossRef]
- Nadgórska-Socha, A.; Ptasiński, B.; Kita, A. Heavy metal bioaccumulation and antioxidative responses in Cardaminopsis arenosa and Plantago lanceolata leaves from metalliferous and non-metalliferous sites: A field study. Ecotoxicology 2013, 22, 1422–1434. [Google Scholar] [CrossRef]
- John, R.; Ahmad, P.; Gadgil, K.; Sharma, S. Heavy metal toxicity: Effect on plant growth, biochemical parameters and metal accumulation by Brassica juncea L. Int. J. Plant Prod. 2009, 3, 65–75. [Google Scholar]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef]
- Bhaduri, A.M.; Fulekar, M.H. Antioxidant enzyme responses of plants to heavy metal stress. Rev. Environ. Sci. Bio/Technol. 2012, 11, 55–69. [Google Scholar] [CrossRef]
- Rocha, A.C.S.; Cavenati, S.; Vasconcelos, M.T.S.; Basto, M.C.P.; Almeida, C.M.R. Metal Accumulation in Estuarine Plants. In Plants Under Metal and Metalloid Stress; Hasanuzzaman, M., Nahar, K., Fujita, M., Eds.; Springer: Singapore, 2018; pp. 185–205. [Google Scholar] [CrossRef]
- Pal, R.; Kaur, R.; Rajwar, D.; Narayan Rai, J.P. Induction of non-protein thiols and phytochelatins by cadmium in Eichhornia crassipes. Int. J. Phytoremediation 2019, 21, 790–798. [Google Scholar] [CrossRef]
- Kumar, D.; Singh, V.P.; Tripathi, D.K.; Prasad, S.M.; Chauhan, D.K. Effect of Arsenic on Growth, Arsenic Uptake, Distribution of Nutrient Elements and Thiols in Seedlings of Wrightia arborea (Dennst.) Mabb. Int. J. Phytoremediation 2015, 17, 128–134. [Google Scholar] [CrossRef]
- Bilska, K.; Wojciechowska, N.; Alipour, S.; Kalemba, E.M. Ascorbic Acid—The Little-Known Antioxidant in Woody Plants. Antioxidants 2019, 8, 645. [Google Scholar] [CrossRef]
- Sumanta, N.; Haque, C.I.; Nishika, J.; Suprakash, R. Spectrophotometric analysis of chlorophylls and carotenoids from commonly grown fern species by using various extracting solvents. Res. J. Chem. Sci. 2014, 4, 63–69. [Google Scholar]
- Abbas, T.; Rizwan, M.; Ali, S.; Zia-ur-Rehman, M.; Qayyum, M.F.; Abbas, F.; Hannan, F.; Rinklebe, J.; Ok, Y.S. Effect of biochar on cadmium bioavailability and uptake in wheat (Triticum aestivum L.) grown in a soil with aged contamination. Ecotoxicol. Environ. Saf. 2017, 140, 37–47. [Google Scholar] [CrossRef]
- Sharma, S.; Anand, G.; Singh, N.; Kapoor, R. Arbuscular mycorrhiza augments arsenic tolerance in wheat (Triticum aestivum L.) by strengthening antioxidant defense system and thiol metabolism. Front. Plant Sci. 2017, 8, 906. [Google Scholar] [CrossRef]
- Khan, M.I.R.; Nazir, F.; Asgher, M.; Per, T.S.; Khan, N.A. Selenium and sulfur influence ethylene formation and alleviate cadmium-induced oxidative stress by improving proline and glutathione production in wheat. J. Plant Physiol. 2015, 173, 9–18. [Google Scholar] [CrossRef]
- Gajewska, E.; SkŁodowska, M. Differential effect of equal copper, cadmium and nickel concentration on biochemical reactions in wheat seedlings. Ecotoxicol. Environ. Saf. 2010, 73, 996–1003. [Google Scholar] [CrossRef]
- Sairam, R.K.; Rao, K.V.; Srivastava, G.C. Differential response of wheat genotypes to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci. 2002, 163, 1037–1046. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Li, J.; Gu, R.; Yue, L.; Wang, H.; Zhan, X.; Xing, B. Carotenoid and superoxide dismutase are the most effective antioxidants participating in ROS scavenging in phenanthrene accumulated wheat leaf. Chemosphere 2018, 197, 513–525. [Google Scholar] [CrossRef] [PubMed]
- Hartikainen, H.; Xue, T.; Piironen, V. Selenium as an anti-oxidant and pro-oxidant in ryegrass. Plant Soil 2000, 225, 193–200. [Google Scholar] [CrossRef]
- Dong, C.; Shao, L.; Liu, G.; Wang, M.; Liu, H.; Xie, B.; Li, B.; Fu, Y.; Liu, H. Photosynthetic characteristics, antioxidant capacity and biomass yield of wheat exposed to intermittent light irradiation with millisecond-scale periods. J. Plant Physiol. 2015, 184, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M. A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]


| Variety | Oil Concentration | Plant Height, cm | Shoot Fresh Weight, g Plant−1 | Shoot Dry Weight, g Plant−1 | Water Content in Shoots, % | Root Length, cm | Root Fresh Weight, g Plant−1 | Root Dry Weight, g Plant−1 | Water Content in Roots, % |
|---|---|---|---|---|---|---|---|---|---|
| Krona | 0 | 17.20a 1 | 0.738a | 0.073a | 90.1a | 8.22a | 0.338a | 0.026a | 90.1a |
| 1.5 | 16.91a | 0.770a | 0.074a | 90.4a | 6.62b | 0.283b | 0.022b | 90.4a | |
| 3.0 | 16.82a | 0.708a | 0.068b | 90.4a | 4.59c | 0.174c | 0.018c | 90.4a | |
| 6.0 | 14.08b | 0.564b | 0.062c | 88.9a | 3.58d | 0.133d | 0.016c | 88.9a | |
| 12.0 | 9.58c | 0.319c | 0.044d | 86.1b | 2.35e | 0.082e | 0.012d | 86.1b | |
| Valdai | 0 | 18.17b | 0.797ab | 0.073b | 90.8a | 8.38a | 0.379a * | 0.031a * | 90.8a |
| 1.5 | 19.93a * | 0.857a * | 0.087a * | 89.9a | 7.62b * | 0.349b * | 0.028a * | 89.9a | |
| 3.0 | 19.82a * | 0.874a * | 0.084a * | 90.4a | 6.03c * | 0.270c * | 0.023b * | 90.2a | |
| 6.0 | 18.02b * | 0.715b * | 0.072b * | 89.9a | 4.50d * | 0.170d * | 0.018c * | 89.9a | |
| 12.0 | 14.95c * | 0.597c * | 0.077b * | 87.1b | 3.18e * | 0.114e * | 0.014d | 87.1b | |
| Main effects 2 | |||||||||
| Oil concentration | 0 | 17.70bc | 0.767a | 0.0731b | 90.4a | 8.30a | 0.358a | 0.0285a | 92.0a |
| (O) | 1.5 | 18.41a | 0.814a | 0.0801a | 90.1a | 7.12b | 0.316b | 0.0252b | 92.1a |
| 3.0 | 18.30ab | 0.791a | 0.0759b | 90.3a | 5.31c | 0.222c | 0.0203c | 90.7b | |
| 6.0 | 16.04c | 0.639b | 0.0671c | 89.4a | 4.04d | 0.151d | 0.0169d | 88.8c | |
| 12.0 | 12.26d | 0.458c | 0.0605d | 86.6b | 2.77e | 0.098e | 0.0125e | 87.0d | |
| Rye variety | Krona | 14.91b | 0.620b | 0.0641b | 89.2a | 5.07b | 0.202b | 0.0186b | 89.7b |
| (V) | Valdai | 18.17a | 0.768a | 0.0786a | 89.6a | 5.94a | 0.256a | 0.0227a | 90.5a |
| Significance | O | * | * | * | * | * | * | * | * |
| V | * | * | * | ns | * | * | * | ns | |
| O*V | * | * | * | ns | * | * | ns | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skrypnik, L.; Maslennikov, P.; Novikova, A.; Kozhikin, M. Effect of Crude Oil on Growth, Oxidative Stress and Response of Antioxidative System of Two Rye (Secale cereale L.) Varieties. Plants 2021, 10, 157. https://doi.org/10.3390/plants10010157
Skrypnik L, Maslennikov P, Novikova A, Kozhikin M. Effect of Crude Oil on Growth, Oxidative Stress and Response of Antioxidative System of Two Rye (Secale cereale L.) Varieties. Plants. 2021; 10(1):157. https://doi.org/10.3390/plants10010157
Chicago/Turabian StyleSkrypnik, Liubov, Pavel Maslennikov, Anastasia Novikova, and Mikhail Kozhikin. 2021. "Effect of Crude Oil on Growth, Oxidative Stress and Response of Antioxidative System of Two Rye (Secale cereale L.) Varieties" Plants 10, no. 1: 157. https://doi.org/10.3390/plants10010157
APA StyleSkrypnik, L., Maslennikov, P., Novikova, A., & Kozhikin, M. (2021). Effect of Crude Oil on Growth, Oxidative Stress and Response of Antioxidative System of Two Rye (Secale cereale L.) Varieties. Plants, 10(1), 157. https://doi.org/10.3390/plants10010157