Himantoglossum adriaticum H. Baumann × Himantoglossum robertianum (Loisel.) P. Delforge: A New Interspecific Hybrid Assessed by Barcoding Analysis
Abstract
1. Introduction
2. Results
2.1. Cross-Pollination and Capsule Development
2.2. In Vitro Sowing and Germination
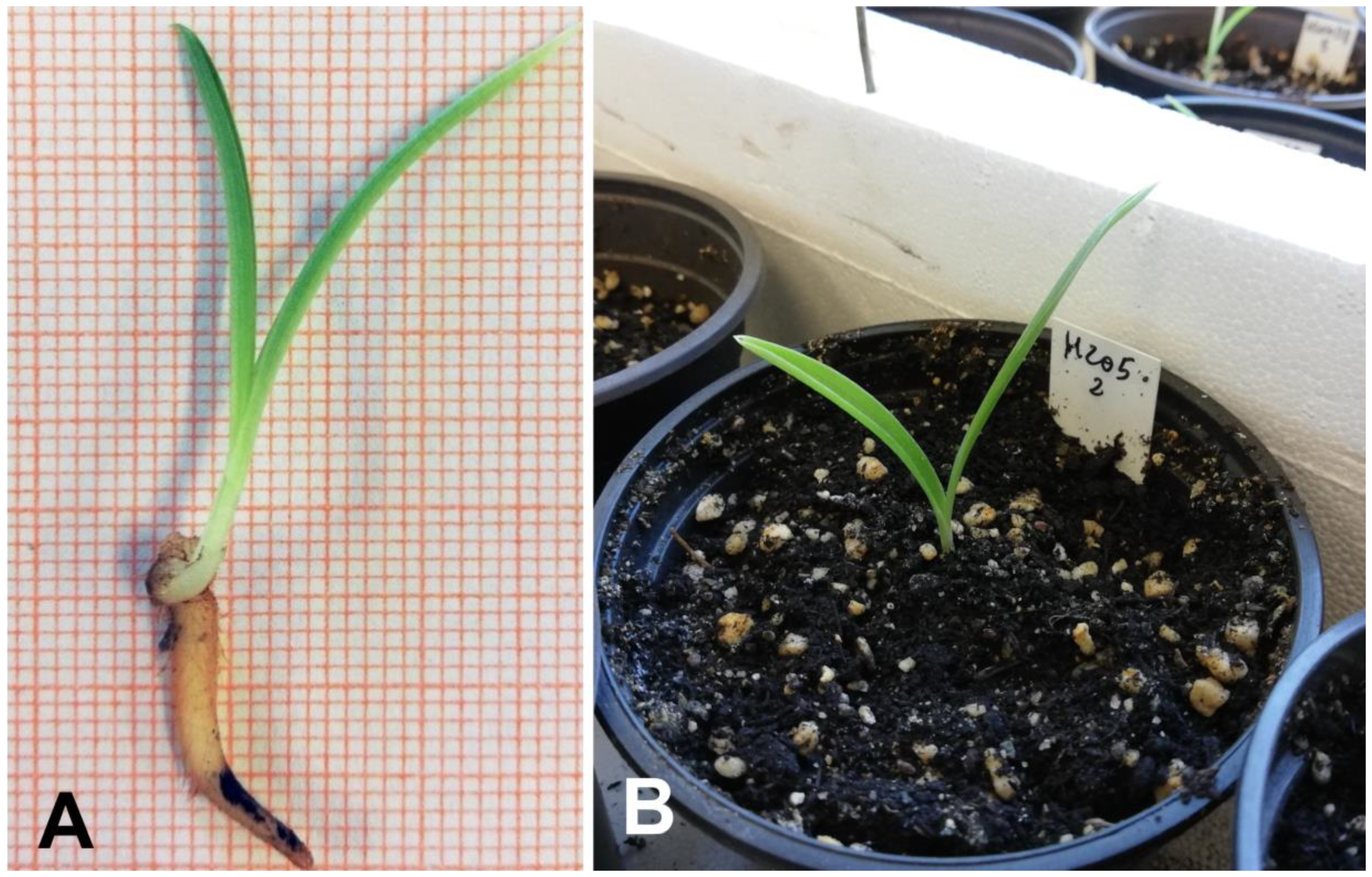
2.3. In Vitro Establishment and Cultivation
2.4. Acclimatization
2.5. Molecular Analysis
3. Discussion
4. Materials and Methods
4.1. Interspecific Hybridization
4.1.1. Anther Conservation
4.1.2. Cross-Pollination
4.1.3. In Vitro Sowing and Germination
4.1.4. In Vitro Establishment and Cultivation
4.1.5. Acclimatization
4.2. Molecular Analysis
4.2.1. Sample Collection and DNA Extraction
4.2.2. PCR Amplification, Cloning and Sequencing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moore, D.M.; Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Orchideaceae in Flora Europea; Cambridge University Press: Cambridge, UK, 1980. [Google Scholar]
- Arditti, J.; Ghani, A.K.A. Transley review, 110—numerical and physical properties of orchid seeds and their biological implication. New Phytol. 2000, 145, 367–421. [Google Scholar] [CrossRef]
- Tremblay, R.L.; Ackerman, J.D.; Zimmerman, J.K.; Calvo, R.N. Variation in sexual reproduction in orchids and its evolutionary consequences: A spasmodic journey to diversification. Biol. J. Linn. Soc. 2005, 84, 1–54. [Google Scholar] [CrossRef]
- Ackerman, J. On the Evidence for a Primitively Epiphytic Habit in Orchids. Am. Soc. Plant Taxon. (ASPT) 1983, 8, 474–477. [Google Scholar]
- De, L.C.; Rao1, A.N.; Rajeevan, P.K.; Pathak, P. Orchid Improvement—An Overview. J. Orchid. Soc. India 2014, 28, 35–45. [Google Scholar]
- Khuraijam, J.S.; Sharma, S.C.; Roy, R.K. Orchids: Potential Ornamental Crop in North India. Int. J. Hortic. Crop Sci. Res. 2017, 7, 1–8. [Google Scholar]
- AIPH. The International Statistics Flowers and Plants 2018 Yearbook; International Association of Horticultural Producers (AIPH) and Union Fleurs: Oxfordshire, UK, 2018; ISBN 978-1-9164807-0-4. [Google Scholar]
- IUCN. IUCN Guidelines for the Prevention of Biodiversity Loss due to Biological Invasion. Species 31–32; IUCN: Gland, Switzerland, 1999; pp. 28–42. [Google Scholar]
- Kreutz, C.A.J. Kompendium der Europaischen Orchideen, 1st ed.; Karel, C.A.J., Ed.; Kreutz Publishers: Landgraaf, The Netherlands, 2004; 240p, ISBN 90-806626-4-X. [Google Scholar]
- Leake, J.R. The biology of myco-heterotrophic (‘saprophytic’) plants. New Phytol. 1994, 127, 171–216. [Google Scholar] [CrossRef]
- Leake, J.R.; Cameron, D.D. Physiological ecology of mycoheterotrophy. New Phytol. 2010, 185, 601–605. [Google Scholar] [CrossRef]
- Read, D.J. Mycorrhizas in ecosystems. Experientia 1991, 47, 376–391. [Google Scholar] [CrossRef]
- Bayman, P.; Gonzàlez, E.J.; Fumero, J.J.; Tremblay, R.L. Are fungi necessary? How fungicides affect growth and survival of the orchid Lepanthes rupestris in the field. J. Ecol. 2002, 90, 1002–1008. [Google Scholar] [CrossRef]
- Ramsay, R.R.; Sivasithamparam, K.; Dixon, K.W. Patterns of infection and endophytes associated with Western Australian orchids. Lindteyana 1986, 1, 203–214. [Google Scholar]
- Arditti, J.; Ernst, R.; Wing Yam, T.; Glabe, C. The contribution of orchid mycorrhizal fungi to seed germination: A speculative review. Lindleyana 1990, 5, 249–255. [Google Scholar]
- Pecoraro, L.; Girlanda, M.; Kull, T.; Perini, C.; Perotto, S. Fungi from the roots of the terrestrial photosynthetic orchid Himantoglossum adriaticum. Plant Ecol. Evol. 2013, 146, 145–152. [Google Scholar] [CrossRef]
- Bateman, R.M.; Molnár, V.A.; Sramkó, G. In situ morphometric survey elucidates the evolutionary systematics of the Eurasian Himantoglossum clade (Orchidaceae: Orchidinae). PeerJ 2018, 5, e2893. [Google Scholar] [CrossRef]
- Delforge, P. Contribution taxonomique et nomenclaturale au genere Himantoglossum (Orchidaceae). Nat. Belg. 1999, 80, 387–408. [Google Scholar]
- Bateman, R.M.; Hollingsworth, P.M.; Preston, J.; Luo, Y.-B.; Pridgeon, A.M.; Chase, M.W. Molecular phylogenetics and evolution of Orchidinae and selected Habenariinae (Orchidaceae). Bot. J. Linn. Soc. 2003, 142, 1–40. [Google Scholar] [CrossRef]
- Bartolucci, F.; Peruzzi, L.; Galasso, G.; Albano, A.; Alessandrini, A.; Ardenghi, N.M.G.; Astuti, G.; Bacchetta, G.; Ballelli, S.; Banfi, E.; et al. An updated checklist of the vascular flora native to Italy. Plant Biosyst. 2018, 152, 179–303. [Google Scholar] [CrossRef]
- Mallet, J. Hybridization as an invasion of the genome. Trends Ecol. Evol. 2005, 20, 229–237. [Google Scholar] [CrossRef]
- Marques, I.; Draper, D.; Riofrío, L.; Naranjo, C. Multiple hybridization events, polyploidy and low postmating isolation entangle the evolution of neotropical species of Epidendrum (Orchidaceae). BMC Evol. Biol. 2014, 14, 20. [Google Scholar] [CrossRef]
- Bernardos, S.; Santos, M.A.; Tyteca, D.; Amich, F. Phylogenetic relationships of Mediterranean Neottieae and Orchideae (Orchidaceae) inferred from nuclear ribosomal ITS sequences. Acta Bot. Gall. 2006, 153, 153–165. [Google Scholar] [CrossRef][Green Version]
- Xu, S.; Schlüter, P.M.; Scopece, G.; Breitkopf, H.; Gross, K.; Cozzolino, S.; Schiestl, F.P. Floral isolation is the main reproductive barrier among closely related sexually deceptive orchids. Evolution 2012, 65, 2606–2620. [Google Scholar] [CrossRef]
- Leal, B.S.S.; Chaves, C.J.N.; Koehler, S.; Borba, E.L. When hybrids are not hybrids: A case study of a putative hybrid zone between Cattleya coccinea and C. brevipedunculata (Orchidaceae). Bot. J. Linn. Soc. 2016, 181, 621–639. [Google Scholar] [CrossRef]
- Claessens, J.; Kleynen, J. (Eds.) The Flower of the European Orchid: Form and Function; Jean Claessens & Jacques Kleynen: Geuelle, The Netherlands, 2011; p. 440. [Google Scholar]
- Carey, P.D. Changes in the distribution and abundance of Himantoglossum hircinum (L.) Sprengel (Orchidaceae) over the last 100 years. Watsonia 1999, 22, 353–364. [Google Scholar]
- Carey, P.D.; Farrell, L.; Stewart, N.F. The sudden increase in the abundance of Himantoglossum hircinum in England in the past decade and what has caused it. In Trends and Fluctuations and Underlying Mechanisms in Terrestrial Orchid Populations; Kindlmann, P., Willems, J., Whigham, D.F., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2002; pp. 187–208. [Google Scholar]
- Pfeifer, M.; Wiegand, K.; Heinrich, W.; Jetschke, G. Long-term demographic fluctuations in an orchid species driven by weather: Implications for conservation planning. J. Appl. Ecol. 2006, 43, 313–324. [Google Scholar] [CrossRef]
- Kreutz, C.A.J.; Steinfeld, P. Himantoglossum hircinum var. aestivalis, eine spätblühende und lockerblütige Sippe der Bocks-Riemenzunge. J. Eur. Orch. 2013, 45, 317–328. [Google Scholar]
- Biró, É.; Bódis, J.; Nagy, T.; Takács, A.; Tökölyi, J.; Molnár, V.A. Reproductive success of Himantoglossum species. In Proceedings of the International Conference on Temperate Orchids, Research and Conservation, Programme and Abstracts, Samos Island, Greece, 14–18 April 2015; p. 75. [Google Scholar]
- G.I.R.O.S. Gruppo Italiano Ricerca Orchidee Spontanee. 2012. Available online: http://www.giros.it/Genera/Barlia.htm (accessed on 2 October 2020).
- Rankou, H. Himantoglossum robertianum. The IUCN Red List of Threatened Species. 2011, p. e.T175926A7145415. Available online: https://www.iucnredlist.org/species/175926/7145415 (accessed on 9 October 2020).
- Del Fuoco, C. Genere Barlia. In Orchidee d’Italia. Guida Alle Orchidee Spontanee, 2nd ed.; GIROS, Il Castello Edizioni: Cornaredo, Italy, 2016; p. 166. ISBN 978-88-6520-801-4. [Google Scholar]
- Bódis, J.; Biró, É.; Nagy, T.; Takács, A.; Sramkó, G.; Bateman, R.M.; Gilián, L.; Illyés, Z.; Tökölyi, J.; Lukács, B.A.; et al. Biological flora of Central Europe Himantoglossum adriaticum H. Baumann. Perspect. Plant Ecol. Evol. Syst. 2019, 40, 125461. [Google Scholar] [CrossRef]
- Bódis, J.; Molnár, E. Long-term monitoring of Himantoglossum adriaticum H. Baumann population in Keszthely hills, Hungary. Nat. Som. 2009, 15, 27–40. [Google Scholar]
- Bernardo, L. Genere Himantoglossum. In Orchidee d’Italia. Guida Alle Orchidee Spontanee, 2nd ed.; GIROS, Il Castello Edizioni: Cornaredo, Italy, 2016; p. 164. ISBN 978-88-6520-801-4. [Google Scholar]
- Himantoglossum Adriaticum. 2020. Available online: https://www.iucnredlist.org/species/162219/5559772#geographic-range (accessed on 9 October 2020).
- Alonzi, A.; Ercole, S.; Piccini, C. La protezione delle specie della flora e della fauna selvatica: Quadro di riferimento legislativo regionale. APAT Rapp. 2006, 75, 258. [Google Scholar]
- Rossi, G.; Montagnani, C.; Gargano, D.; Peruzzi, L.; Abeli, T.; Ravera, S.; Cogoni, A.; Fenu, G.; Magrini, S.; Gennai, M.; et al. (Eds.) Lista Rossa della Flora Italiana. 1. Policy Species e Altre Specie Minacciate; Comitato Italiano IUCN e Minor ATTM: Roma, Italy, 2013. [Google Scholar]
- Rieseberg, L.H.; Ellstrand, N.C. What can molecular and morphological markers tell us about plant hybridization? Crit. Rev. Plant Sci. 1993, 12, 213–241. [Google Scholar]
- Nagayoshi, T.; Hatanaka, T.; Suzuki, T. Seed Propagation of Habenaria radiata. Morphological and Physiological Characteristics of Plants Derived from in vitro Cultured Seedlings. Nat. Hum. Act. 1996, 1, 67–81. [Google Scholar]
- Chen, J.; Moinard, M.; Xu, J.; Wang, S.; Foulongne-Oriol, M.; Zhao, R.; Hyde, K.D.; Callac, P. Genetic Analyses of the Internal Transcribed Spacer Sequences Suggest Introgression and Duplication in the Medicinal Mushroom Agaricus subrufescens. PLoS ONE 2016, 11, e0156250. [Google Scholar] [CrossRef]
- CBOL Plant Working Group. A DNA barcode for land plants. Proc. Natl. Acad. Sci. USA 2009, 106, 12794–12797. [Google Scholar] [CrossRef]
- Euromed PlantBase. 2020. Available online: http://euromed.luomus.fi/euromed_map.php?taxon=498930&size=medium (accessed on 13 October 2020).
- Euromed PlantBase. 2020. Available online: http://euromed.luomus.fi/euromed_map.php?taxon=531028&size=medium (accessed on 13 October 2020).
- G.I.R.O.S. Gruppo Italiano Ricerca Orchidee Spontanee. 2013. Available online: http://www.giros.it/forum/viewtopic.php?f=131&t=1485 (accessed on 2 October 2020).
- Sinumporn, P.; Narumi-Kawasaki, T.; Fukai, S. Development of interspecific hybrids between Habenaria radiate and Habenaria rhodocheila complex. Adv. Hort. Sci. 2020, 34, 3–10. [Google Scholar]
- Chase, M.W.; Knapp, S.; Cox, A.V.; Clarkson, J.J.; Butsko, I.Y.; Joseph, J.; Savolainen, V.; Parokonny, A.S. Molecular systematics, GISH and the origin of hybrid taxa in Nicotiana (Solanaceae). Ann. Bot. 2003, 92, 107–127. [Google Scholar] [CrossRef]
- Grassi, F.; Labra, M.; Minuto, L.; Casazza, G.; Sala, F. Natural hybridization in Saxifraga callosa Sm. Plant Biol. 2006, 8, 243–252. [Google Scholar] [CrossRef]
- Gilián, L.D.; Bódis, J.L.; Eszéki, E.; Illyés, Z.; Biró, É.; Nagy, J. Germination traits of adriatic lizard orchid (Himantoglossum Adriaticum) in Hungary. Appl. Ecol. Environ. Res. 2018, 16, 1155–1171. [Google Scholar] [CrossRef]
- Johnson, T.R.; Kane, M.E. Asymbiotic germination of ornamental Vanda: In vitro germination and development of three hybrids. Plant Cell Tissue Organ Cult. 2007, 91, 251–261. [Google Scholar] [CrossRef]
- Kauth, P.J.; Kane, M.E.; Vendrame, W.A.; Reinhardt-Adams, C. Asymbiotic germination response to photoperiod and nutritional media in six populations of Calopogon tuberosus var. tuberosus (Orchidaceae): Evidence for ecotypic differentiation. Ann. Bot. 2008, 102, 783–793. [Google Scholar] [CrossRef]
- Dowling, A.B.; Manfred Jusaitis, A.B. Asymbiotic in vitro germination and seed quality assessment of Australian terrestrial orchids. Nicole Aust. J. Bot. 2012, 60, 592–601. [Google Scholar] [CrossRef]
- Wood, C.B.; Pritchard, H.W. Determination of intra-specific variation in orchid seed viability using fluorescein diacetate. Seed Sci. Technol. 2004, 32, 629–635. [Google Scholar] [CrossRef]
- Johnson, T.R.; Kane, M.E.; Perez, H.E. Examining the interaction of light, nutrients and carbohydrates on seed germination and early seedling development of Bletia purpurea (Orchidaceae). Plant Growth Regul. 2011, 63, 89–99. [Google Scholar] [CrossRef]
- Aybeke, M. Morphological and histochemical investigations on Himantoglossum robertianum (Loisel.) P. Delforge (Orchidaceae) seeds. Plant Syst. Evol. 2014, 300, 91–97. [Google Scholar] [CrossRef]
- Antonetti, M.; Burchi, G.; Prisa, D.; Calevo, J.; Giovannini, A. Asymbiotic propagation of Italian orchids. Acta Hortic. 2019, 1262, 179–186. [Google Scholar] [CrossRef]
- Pierce, S.; Ceriani, R.M. Conservazione in situ e rafforzamento di popolazioni di orchidee spontanee. In Esperienze di Conservazione delle Orchidee; Magrini, S., Fonck, M., Zucconi, L., Eds.; Orto Botanica della Tuscia: Viterbo, Italy, 2014; pp. 33–38. [Google Scholar]
- Jacquemyn, H.; Honnay, O.; Cammue, B.P.A.; Brys, R.; Lievens, B. Low specificity and nested subset structure characterise mycorrhizal associations in five closely related species of the genus Orchis. Mol. Ecol. 2010, 19, 4086–4095. [Google Scholar] [CrossRef]
- Davenport, T.R.B.; Ndangalasi, H.J. An escalating trade in orchid tubers across Tanzania’s Southern Highlands: Assessment, dynamics and conservation implications. Oryx 2003, 37, 55–61. [Google Scholar] [CrossRef]
- Veldman, S.; Otieno, J.N.; van Andel, T.; Gravendeel, B.; de Boer, H.J. Efforts urged to tackle thriving illegal orchid trade in Tanzania and Zambia for chikanda production. Traffic Bull. 2014, 26, 47–50. [Google Scholar]
- Sezik, E. Turkish orchids and Salep. Acta Pharm. Turc. 2002, 44, 151–157. [Google Scholar]
- Kreziou, A.; De Boer, H.; Gravendeel, B. Harvesting of salep orchids in north-western Greece continues to threaten natural populations. Oryx 2015, 50, 393–396. [Google Scholar] [CrossRef]
- Giordani, E.; Ferri, A.; Trentacoste, E.; Radice, S. Viability and In Vitro Germinability of Pollen Grains of Olive Cultivars Grown in Different Environments. Acta Hort. 2014, 1057, 65–71. [Google Scholar] [CrossRef]
- Dutra, D.; Johnson, T.R.; Kauth, P.J.; Stewart, S.L.; Kane, M.E.; Richardson, L. Asymbiotic seed germination, in vitro seedling development, and greenhouse acclimatization of the threatened terrestrial orchid Bletia purpurea. Plant Cell Tiss. Organ Cult. 2008, 94, 11–21. [Google Scholar] [CrossRef]
- Miyoshi, K.; Mii, M. Enhancement of seed germination and protocorm formation in Calanthe discolor (Orchidaceae) by NaOCl and polyphenol absorbent treatments. Plant Tissue Cult. Lett. 1995, 12, 267–272. [Google Scholar] [CrossRef]
- Yamazaki, J.; Miyoshi, K. In vitro asymbiotic germination of immature seed and formation of protocorm by Cephalanthera falcata (Orchidaceae). Ann. Bot. 2006, 98, 1197. [Google Scholar] [CrossRef] [PubMed]
- Magrini, S.; Bronzo, F.; Onofri, S.; Scoppola, A. Germinazione asimbiotica in vitro di semi immaturi di Orchis palustris Jacq. Stud. Trent. Sci. Nat. 2012, 90, 159–164. [Google Scholar]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Corpet, F. Multiple sequence alignment with hierarchical clustering. Nucl. Acids Res. 1988, 16, 10881–10890. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Manjunath, K. DNA Barcoding of Endemic and Endangered Orchids of India: A Molecular Method of Species Identification. Pharm. Mag. 2020, 16, 290–299. [Google Scholar] [CrossRef]
- Sang, T.; Crawford, D.J.; Stuessy, T.F. Documentation of reticulate evolution in peonies (Paeonia) using internal transcribed spacer sequences of nuclear ribosomal DNA: Implications for biogeography and concerted evolution. Proc. Natl. Acad. Sci. USA 1995, 92, 6813–6817. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, T.L.; Guo, Y.H.; Liu, X. Identification of Hybrids in Potamogeton: Incongruence between Plastid and ITS Regions Solved by a Novel Barcoding Marker PHYB. PLoS ONE 2016, 11, e0166177. [Google Scholar] [CrossRef]
- Sramkó, G.; Molnár, V.A.; Hawkins, J.; Bateman, R.M. Molecular phylogeny and evolutionary history of the Eurasiatic orchid genus Himantoglossum s.l. (Orchidaceae). Ann. Bot. 2014, 114, 1609–1626. [Google Scholar] [CrossRef]



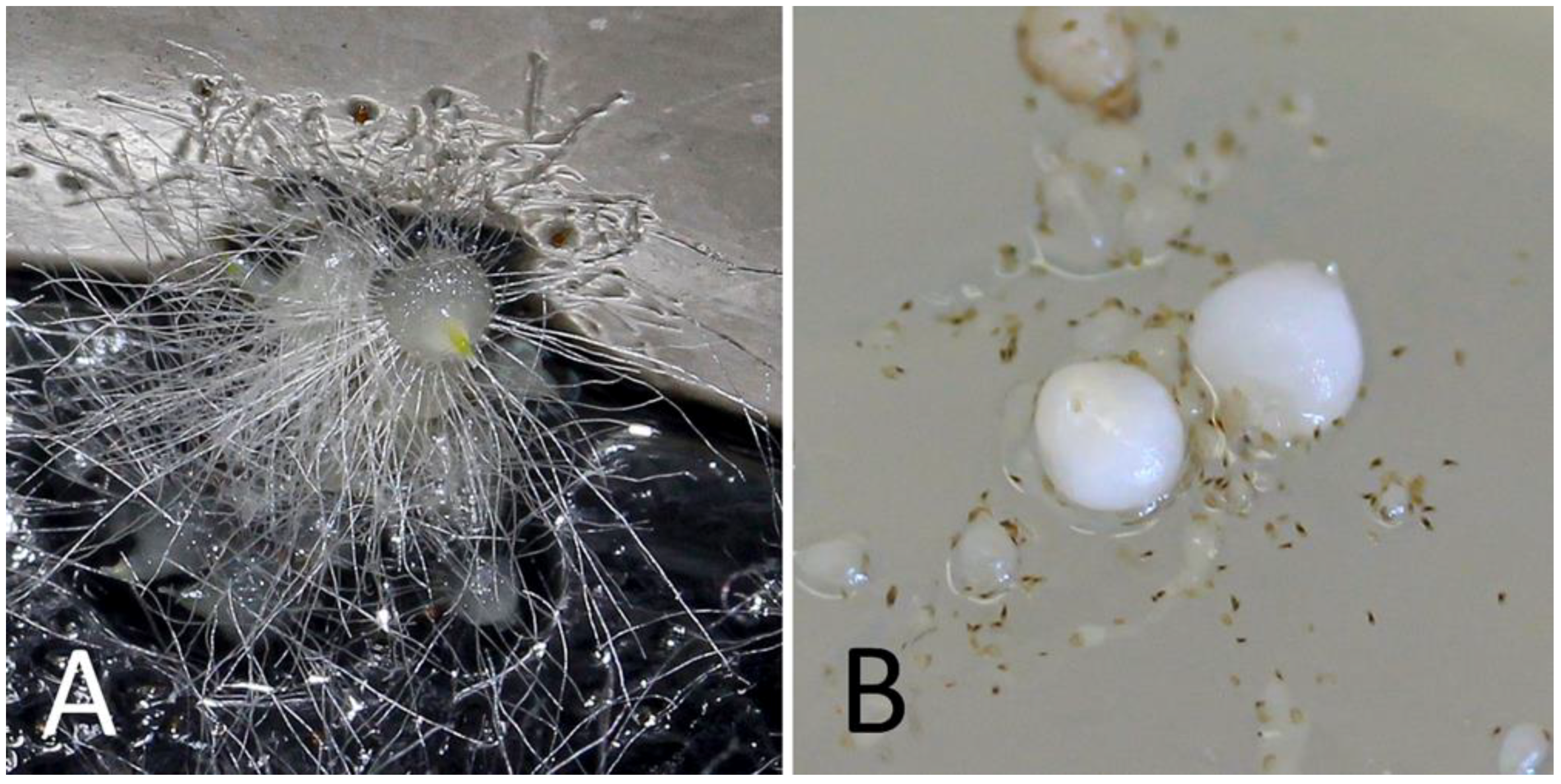

| Capsule N. | Sowing Date | Germinated Protocorms (N.) 1 | |
|---|---|---|---|
| BM1 | M551 | ||
| C1 | 21 June 2017 | 179 | 21 |
| C2 | 27 June 2017 | 150 | 132 |
| C3 | 27 June 2017 | 236 | 67 |
| C4 | 21 June 2017 | 149 | 25 |
| C5 | 26 June 2017 | 448 | 46 |
| C6 | 26 June 2017 | 443 | Contam. 2 |
| C7 | 27 June 2017 | 170 | 157 |
| C9 | 23 June 2017 | 311 | 69 |
| C10 | 28 June 2017 | 391 | 928 |
| C11 | 27 June 2017 | 145 | 247 |
| C13 | 26 June 2017 | 133 | 74 |
| C15 | 26 June 2017 | 211 | 94 |
| C20 | 28 June 2017 | 160 | 878 |
| C26 | 28 June 2017 | 398 | 1075 |
| C59 | 28 June 2017 | Contam. 2 | 1886 |
| C62 | 28 June 2017 | 370 | Contam. 2 |
| Total | 3894 | 5699 | |
| Position | 42 | 57 | 63 | 93 | 100 | 113 | 114 | 147 | 151 | 178 | 186 | 189 | 191 | 192 | 200 | 211 | 219 | 228 | 229 | 230 |
| H. adriaticum | T | T | C | G | T | T | T | C | T | T | A | T | C | A | T | C | T | C | T | T |
| colony 4 | T | T | C | G | T | T | T | C | T | T | A | T | C | A | T | C | T | C | T | T |
| colony 7 | T | T | C | G | T | T | T | C | T | T | A | T | C | A | T | C | T | C | T | T |
| colony 10 | T | T | C | G | T | T | T | C | T | T | A | T | C | A | T | C | T | C | T | T |
| colony 11 | T | T | C | G | T | T | T | C | T | T | A | T | C | A | T | C | T | C | T | T |
| H. robertianum | A | C | T | A | C | G | C | T | C | C | G | C | A | G | A | A | A | A | A | A |
| colony 5 | A | C | T | A | C | G | C | T | C | C | G | C | A | G | A | A | A | A | A | A |
| colony 6 | A | C | T | A | C | G | C | T | C | C | G | C | A | G | A | A | A | A | A | A |
| colony 13 | A | C | T | A | C | G | C | T | C | C | G | C | A | G | A | A | A | A | A | A |
| colony 16 | A | C | T | A | C | G | C | T | C | C | G | C | A | G | A | A | A | A | A | A |
| Position | 233 | 374 | 414 | 422 | 426 | 429 | 431 | 444 | 445 | 470 | 476 | 481 | 483 | 497 | 498 | 513 | 514 | 519 | 551 | 553 |
| H. adriaticum | C | G | A | T | A | T | A | T | T | G | T | T | C | G | C | T | T | C | A | T |
| colony 4 | C | G | A | T | A | T | A | T | T | G | T | T | C | G | C | T | T | C | A | T |
| colony 7 | C | G | A | T | A | T | A | T | T | G | T | T | C | G | C | T | T | C | A | T |
| colony 10 | C | G | A | T | A | T | A | T | T | G | T | T | C | G | C | T | T | C | A | T |
| colony 11 | C | G | A | T | A | T | A | T | T | G | T | T | C | G | C | T | T | C | A | T |
| H. robertianum | G | A | C | C | G | C | G | A | A | A | C | C | T | A | T | A | G | A | G | C |
| colony 5 | G | A | C | C | G | C | G | A | A | A | C | C | T | A | T | A | G | A | G | C |
| colony 6 | G | A | C | C | G | C | G | A | A | A | C | C | T | A | T | A | G | A | G | C |
| colony 13 | G | A | C | C | G | C | G | A | A | A | C | C | T | A | T | A | G | A | G | C |
| colony 16 | G | A | C | C | G | C | G | A | A | A | C | C | T | A | T | A | G | A | G | C |
| Position | 587 | 593 | 597 | 601 | 624 | 625 | 626 | 627 | 634 | |||||||||||
| H. adriaticum | C | G | T | C | A | A | T | A | A | |||||||||||
| colony 4 | C | G | T | C | A | A | T | A | A | |||||||||||
| colony 7 | C | G | T | C | A | A | T | A | A | |||||||||||
| colony 10 | C | G | T | C | A | A | T | A | A | |||||||||||
| colony 11 | C | G | T | C | A | A | T | A | A | |||||||||||
| H. robertianum | A | T | A | G | G | T | C | G | G | |||||||||||
| colony 5 | A | T | A | G | G | T | C | G | G | |||||||||||
| colony 6 | A | T | A | G | G | T | C | G | G | |||||||||||
| colony 13 | A | T | A | G | G | T | C | G | G | |||||||||||
| colony 16 | A | T | A | G | G | T | C | G | G |
| matK 11/830 bp | rbcL 2/610 bp | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Position | 18 | 78 | 208 | 300 | 350 | 616 | 678 | 696 | 718 | 773 | 812 | 251 | 259 | |||||
| H. robertianum | A | A | T | C | G | G | C | G | C | G | A | G | A | |||||
| H. adriaticum | G | G | C | T | A | T | T | A | T | A | G | A | T | |||||
| Hybrid 6 | G | G | C | T | A | T | T | A | T | A | G | A | T | |||||
| Hybrid 11 | G | G | C | T | A | T | T | A | T | A | G | A | T | |||||
| psbA-trnH36/813bp | ||||||||||||||||||
| Position | 58 | 59 | 60 | 61 | 62 | 63 | 91 | 92 | 93 | 94 | 95 | 96 | 141 | 142 | 143 | 144 | 145 | 146 |
| H. robertianum | - | - | - | - | - | - | - | - | - | - | - | - | T | T | T | C | T | C |
| H. adriaticum | T | C | C | C | C | A | G | G | G | A | T | G | - | - | - | - | - | - |
| Hybrid 6 | T | C | C | C | C | A | G | G | G | A | T | G | - | - | - | - | - | - |
| Hybrid 11 | T | C | C | C | C | A | G | G | G | A | T | G | - | - | - | - | - | - |
| psbA-trnH36/813 bp | ||||||||||||||||||
| Position | 147 | 148 | 149 | 242 | 243 | 244 | 245 | 246 | 247 | 248 | 249 | 250 | 172 | 196 | 260 | 274 | 336 | 720 |
| H. robertianum | C | G | A | A | G | A | A | A | A | G | T | C | G | G | C | G | T | A |
| H. adriaticum | - | - | - | - | - | - | - | - | - | - | - | - | T | T | G | T | A | C |
| Hybrid 6 | - | - | - | - | - | - | - | - | - | - | - | - | T | T | G | T | A | C |
| Hybrid 11 | - | - | - | - | - | - | - | - | - | - | - | - | T | T | G | T | A | C |
| Locus | Primer Name | Sequence (5’ → 3’) | Annealing T |
|---|---|---|---|
| rbcL | F1 | ATGTCACCACAAACAGAGACTAAAGC | 58 °C |
| R634 | GAAACGGTCTCTCCAACGCAT | ||
| matK | 3F | CGTACAGTACTTTTGTGTTTACGAG | 52 °C |
| 1R | ACCCAGTCCATCTGGAAATCTTGGTTC | ||
| psbA-trnH | Fwd | CGCGCATGGTGGATTCACAATCC | 55 °C |
| Rev | GTTATGCATGAACGTAATGCTC | ||
| ITS | ITS5 F | GGAAGTAAAAGTCGTAACAAGG | 50 °C |
| ITS4 R | TCCTCCGCTTATTGATATGC |
| ITS | matK | rbcl | psbA-trnH Spacer | |
|---|---|---|---|---|
| Himantoglossum adriaticum | FR750401 | MW367975 * | MW367974 * | MW316730 * |
| Himantoglossum robertianum | KJ596109 | AY368382 | AY368337 | MW316729 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antonetti, M.; Nin, S.; Burchi, G.; Biricolti, S.; Gori, M. Himantoglossum adriaticum H. Baumann × Himantoglossum robertianum (Loisel.) P. Delforge: A New Interspecific Hybrid Assessed by Barcoding Analysis. Plants 2021, 10, 107. https://doi.org/10.3390/plants10010107
Antonetti M, Nin S, Burchi G, Biricolti S, Gori M. Himantoglossum adriaticum H. Baumann × Himantoglossum robertianum (Loisel.) P. Delforge: A New Interspecific Hybrid Assessed by Barcoding Analysis. Plants. 2021; 10(1):107. https://doi.org/10.3390/plants10010107
Chicago/Turabian StyleAntonetti, Maurizio, Stefania Nin, Gianluca Burchi, Stefano Biricolti, and Massimo Gori. 2021. "Himantoglossum adriaticum H. Baumann × Himantoglossum robertianum (Loisel.) P. Delforge: A New Interspecific Hybrid Assessed by Barcoding Analysis" Plants 10, no. 1: 107. https://doi.org/10.3390/plants10010107
APA StyleAntonetti, M., Nin, S., Burchi, G., Biricolti, S., & Gori, M. (2021). Himantoglossum adriaticum H. Baumann × Himantoglossum robertianum (Loisel.) P. Delforge: A New Interspecific Hybrid Assessed by Barcoding Analysis. Plants, 10(1), 107. https://doi.org/10.3390/plants10010107







