Introduction to the Study on Regeneration in Lizards as an Amniote Model of Organ Regeneration
1. The Lizard Model: Brief Historical Notes
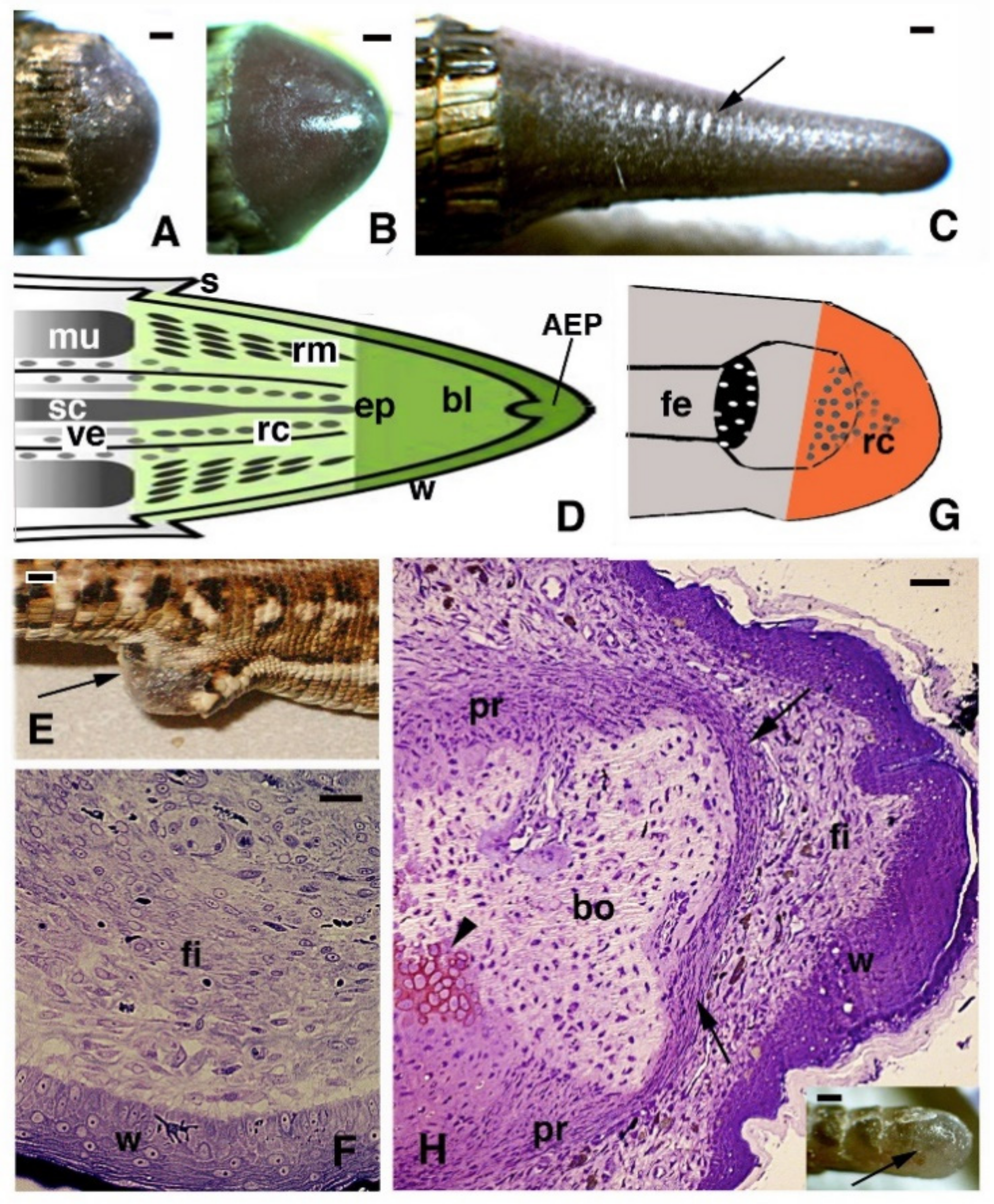
2. General Histological Aspects
3. Regeneration Evolved Only in Lizards, Providing Clues for Amniote Regeneration
Acknowledgments
Conflicts of Interest
References
- Dinsmore, C.H. Urodele limb and tail regeneration in early biological thought: An essay on scientific controversy and social change. Int. J. Dev. Biol. 1996, 40, 621–627. [Google Scholar] [PubMed]
- Fraisse, P. Die Regeneration von Geweben und Organen bei den Wirbeltieren Besonders bei Amphibien und Reptilien; Fischer: Berlin, Germany, 1885. [Google Scholar]
- Kudokotsev, V.P. Regeneration process of extremity in lizard, stimulated by the method of supplementary innervation. Dokl. Akad. Sci. SSSR 1960, 126, 233–236. (in Russian). [Google Scholar]
- Kudokotsev, V.P. Regeneration of the limbs in the dersert snake-eyed skink (Ablepharus deserti Strauch). Dokl. Akad. Sci. SSSR 1960, 126, 464–467. [Google Scholar]
- Boring, A.M.; Lan-Fen, C.; Wei-His, C. Autotomy and regeneration in the tail of lizards. Peking Natur. Hist. Bull. 1948–1949, 17, 85–108. [Google Scholar]
- Bellairs, A.D.A.; Bryant, S.V. Autotomy and regeneration in reptiles. In Biology of the Reptilia; Gans, C., Billet, F., Maderson, P.F.A., Eds.; Wiley: New York, NY, USA, 1985; Volume 15B, pp. 302–410. [Google Scholar]
- Alibardi, L. Morphological and cellular aspects of tail and limb regeneration in lizard: A model system with implications for tissue regeneration in mammals. Adv. Anat. Embryol. Cell Biol. 2010, 207, 1–112. [Google Scholar]
- Calori, L. Sullo scheletro della Lacerta viridis Linn., sulla riproduzione della coda nelle lucertole, e sulle ossa cutanee del teschio dei sauri. Mem. Atti. Acc. Sci. Bologna. 1858, 9, 46–50. [Google Scholar]
- Giuliani, M. Sulla struttura del midollo spinale. Sulla riproduzione della coda della Lacerta viridis. Ric. Lab. Anat. Norm. 1878, I–II, 137–150. [Google Scholar]
- Misuri, A. Ricerche sulla struttura della coda normale e rigenerata nella Lacerta muralis. Memor. Boll. Soc. Zool. Ital. 1910, 11, 103–135. [Google Scholar]
- Woodland, W.N.F. Some observations on caudal autotomy and regeneration in the gecko (Hemidactylus flaviviridis, Ruppel), with notes on the tails of Sphenodon and Pygopus. Quart. J. Microsc. Sci. 1920, 65, 63–100. [Google Scholar]
- White, C.P. Regeneration of the lizard’s tail. J. Path. Bact. 1925, 28, 63–68. [Google Scholar] [CrossRef]
- Quattrini, D. Piano di autotomia e rigenerazione della coda nei Sauri. Arch. Ital. Anat. Embr. 1954, 59, 225–282. [Google Scholar]
- Terni, T. Studio anatomico di una coda doppia. Arch. Ital. Anat. Embryol. 1915, 14, 290–314. [Google Scholar]
- Terni, T. Sulla correlazione fra amplezza del territorio di innervazioni e grandezza della cellule gangliare. 2. Richerche sui gangli spinali che innervano la coda rigenerata, dei Sauri (Gongylus ocellatus). Arch. Ital. Anat. Embriol. 1920, 17, 507–543. [Google Scholar]
- Alibardi, L. Histochemical, Biochemical and Cell Biological aspects of tail regeneration in lizard, an amniote model for studies on tissue regeneration. Prog. Histochem. Cytoch. 2014, 48, 143–244. [Google Scholar] [CrossRef]
- Ramachandran, A.V. Biochemistry and metabolism of lizard tail regeneration. J. Anim. Morphol. Physiol. 2006, 43, 1–13. [Google Scholar]
- Hughes, A.; New, D. Tail regeneration in the geckonid lizard, Sphaerodactylus. J. Embryol. Exp. Morphol. 1959, 7, 281–302. [Google Scholar] [CrossRef]
- Werner, Y.L. Regeneration of the caudal axial skeleton in a gekkonid lizard (Hemidactylus) with particular reference to the “latent” period. Acta Zool. 1967, 48, 103–125. [Google Scholar] [CrossRef]
- Magon, D.K. Glucose metabolism in the regenerating tail of the scincid lizard, Mabuya striata: Glycogen, phosphorylase and aldolase activity. J. Nat. Hist. 1977, 11, 121–126. [Google Scholar] [CrossRef]
- Purvis, M.D. Early stages of tail regeneration in Lampropholis guichenoti. Aust. Zool. 1979, 20, 289–296. [Google Scholar]
- Byerly, T.C. Note on the partial regeneration of the caudal region of Sphenodon punctatus. Anat. Rec. 1925, 30, 61–66. [Google Scholar] [CrossRef]
- Ali, S.M. Studies on the comparative anatomy of the tail in sauria and rhynchocephalia Sphenodon punctatus Gray. Proc. Ind. Acad. Sci. 1941, B13, 171–192. [Google Scholar] [CrossRef]
- Alibardi, L.; Meyer-Rochow, V.B. Microscopical observations on the regenerating tail in the tuatara Sphenodon punctatus indicate a tendency to scarring, but also influence from somatic growth. J. Morph. 2019, 280, 411–422. [Google Scholar] [CrossRef]
- Alibardi, L.; Meyer-Rochow, V.B. Regeneration in Reptiles Generally and the New Zealand Tuatara in Particular as a Model to Analyze Organ Regrowth in Amniotes. J. Dev. Biol. 2021, 9, 36. [Google Scholar] [CrossRef]
- Marcucci, E. Gli arti e la coda della Lacerta muralis rigenerano nello stadio embrionale? Boll. Soc. Natural. 1915, 27, 98–101. [Google Scholar]
- Marcucci, E. Il potere rigenerativo degli arti nei Rettili. Ricerche sperimentali sopra alcune specie di Sauri. Arch. Zool. Ital. 1930, 14, 227–252. [Google Scholar]
- Marcucci, E. La rigenerazione nei rettili. Archivio Zool. Ital. 1930, 16, 455–458. [Google Scholar]
- Guyénot, E.; Matthey, R. Les processus régénératifs dans la patte posterieure du lezard. Wilhelm Roux’Archiv Entwick. Organ. 1928, 113, 520–529. [Google Scholar] [CrossRef]
- Barber, L.W. Correlations between wound healing and regeneration in fore-limbs and tails of lizards. Anat. Rec. 1944, 89, 441–453. [Google Scholar] [CrossRef]
- Bellairs, A.D.A.; Bryant, S.V. Effects of amputation on limbs and digits of lacertid lizards. Anat. Rec. 1968, 161, 489–496. [Google Scholar] [CrossRef]
- Alibardi, L. Tail regeneration in lepidosauria as an exception to the generalized lack of organ regeneration in amniotes. J. Exp. Zool. Part B Mol. Dev. Evol. 2021, 336, 145–164. [Google Scholar] [CrossRef]
- Simpson, S.B. Regeneration of the lizard tail. In Regeneration in Animals and Related Problems; Kiortsis, V., Trampusch, H.A.L., Eds.; North-Holland: Amsterdam, The Netherlands, 1965; pp. 431–443. [Google Scholar]
- Simpson, S.B. Morphology of the regenerated spinal cord in the lizard Anolis carolinensis. J. Comp. Neur. 1968, 134, 193–210. [Google Scholar] [CrossRef] [PubMed]
- Zika, J.; Singer, M. The relation between nerve fiber number and limb regenerative capacity in the lizard, Anolis. Anat. Rec. 1965, 152, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.G. Some aspects of tail regeneration in the lizard, Anolis carolinensis. I. A description based on histology and autoradiography. J. Exp. Zool. 1969, 171B, 127–150. [Google Scholar] [CrossRef]
- Egar, M.; Simpson, S.B.; Singer, M. The growth and differentiation of the regenerating spinal cord after autotomy of the lizard Anolis carolinensis. J. Morph. 1970, 131, 131–152. [Google Scholar] [CrossRef]
- Simpson, S.B.; Bayne, E.K. In vivo and in vitro studies of regenerating muscle in the lizard Anolis. In Muscle Regeneration; Mauro, A., Ed.; Raven Press: New York, NY, USA, 1979; pp. 189–200. [Google Scholar]
- Kahn, E.B.; Simpson, S.B. Satellite cells in mature, uninjured skeletal muscle of the lizard tail. Dev. Biol. 1974, 37, 219–223. [Google Scholar] [CrossRef]
- Simpson, S.B.; Duffy, M.T. The lizard spinal cord: A model system for the study of spinal cord injury and repair. Progr. Brain Res. 1994, 103, 229–241. [Google Scholar]
- Chlebowsky, J.S.; Przybylski, R.J.; Cox, P.G. Ultrastructural studies of lizard (Anolis carolinensis) myogenesis in vitro. Dev. Biol. 1973, 33, 80–99. [Google Scholar] [CrossRef]
- Marusich, M.F.; Simpson, S.B. Changes in cell surface antigens during in vitro lizard myogenesis. Dev. Biol. 1983, 97, 313–328. [Google Scholar] [CrossRef]
- Dalla Valle, L.; Toffolo, V.; Belvedere, P.; Alibardi, L. Isolation of a mRNA encoding a glycine-proline-rich beta-keratin expressed in the regenerating epidermis of lizard. Dev. Dyn. 2005, 234, 934–947. [Google Scholar] [CrossRef]
- Alibardi, L.; Dalla Valle, L.; Nardi, A.; Toni, M. Evolution of hard proteins in the sauropsid integument in relation to the cornification of skin derivatives in amniotes. J. Anat. 2009, 214, 560–586. [Google Scholar] [CrossRef]
- Holthaus, K.B.; Eckhart, L.; Dalla Valle, L.; Alibardi, L. Evolution and diversification of corneous beta-proteins, the characteristic epidermal proteins of reptiles and birds. J. Exp. Zool. 2019, 330B, 438–453. [Google Scholar] [CrossRef]
- Holthaus, K.B.; Alibardi, L.; Tschachler, E.; Eckhart, L. Identification of epidermal differentiation genes of the tuatara provides insights into the early evolution of lepidosaurian skin. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, R.; Jang, S.; Zhou, W.; Liu, Y.; Gu, Q.; Gu, Y.; Dong, Y.; Liu, M.; Ding, F.; et al. Gecko CD59 is implicated in proximodistal identity during tail regeneration. PLoS ONE 2011, 6, e17878. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Xu, Q.; Li, D.; Zhao, L.; Wang, Y.; Liu, M.; Gu, X.; Liu, Y. Early neurogenesis during caudal spinal cord regeneration in adult Gekko japonicus. J. Mol. Histol. 2013, 44, 291–297. [Google Scholar] [CrossRef]
- He, B.; Song, H.; Wang, Y. Self-control of inflammation during tail regeneration of lizards. J. Dev. Biol. 2021, 9, 48, (this issue). [Google Scholar] [CrossRef]
- McLean, C.E.; Vickaryous, M.K. A novel amniote model of epimorphic regeneration: The leopard gecko, Eublepharis macularius. BMC Dev. Biol. 2011, 11, 50–74. [Google Scholar] [CrossRef] [Green Version]
- Gilbert, E.A.B.; Payne, S.L.; Vickaryous, M.K. The anatomy and histology of caudal autotomy and regeneration in lizards. Physiolog. Bioch. Zool. 2013, 86, 631–644. [Google Scholar] [CrossRef]
- Gilbert, E.A.B.; Delorme, S.L.; Vickaryous, M.K. The regeneration blastema of lizards: An amniote model for the study on appendage replacement. Regeneration 2015, 2, 45–53. [Google Scholar] [CrossRef]
- Gilbert, R.W.D.; Vickaryous, M.K.; Victoria-Petit, A.M. Signalling by transforming growth factor beta isoforms in wound healing and tissue regeneration. J. Dev. Biol. 2016, 6, 21. [Google Scholar] [CrossRef]
- Gilbert, E.A.B.; Vickaryous, M.K. Neural stem/progenitor cells are activated during tail regeneration in the leopard gecko (Eleublepharis macularius). J. Comp. Neurol. 2018, 526, 285–309. [Google Scholar] [CrossRef]
- Fisher, R.E.; Geiger, L.A.; Stroik, L.K.; Hutchins, E.D.; George, R.M.; DeNardo, D.F.; Kusumi, K.; Rawls, J.A.; Wilson-Rawls, J. A histological comparison of the original and regenerated tail in the green anole, Anolis carolinensis. Anat. Rec. 2012, 295, 1609–1619. [Google Scholar] [CrossRef] [Green Version]
- Lozito, T.P.; Tuan, R.S. Lizard tail regeneration: Regulation of two distinct cartilage regions by Indian hedgehog. Dev. Biol. 2015, 399, 249–262. [Google Scholar] [CrossRef] [Green Version]
- Lozito, T.P.; Tuan, S.R. Lizard tail regeneration as an instructive model of enhanced healing capabilities in an adult amniote. Connect. Tiss. Res. 2016, 58, 145–154. [Google Scholar] [CrossRef] [Green Version]
- Lozito, T.P.; Tuan, R.S. Lizard tail skeletal regeneration combines aspects of fracture healing and blastema-based regeneration. Development 2016, 143, 2946–2957. [Google Scholar] [CrossRef] [Green Version]
- Hutchins, E.D.; Wilson-Rawls, J.; Kusumi, K. Regeneration: Lessons from lizards. In Innovations in Molecular Mechanisms and Tissue Engineering, Stem Cell Biology and Regenerative Medicine; Wilson-Rawls, J., Kusumi, K., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 23–35. [Google Scholar]
- Londono, R.; Wenzhong, W.; Wang, B.; Tuan, R.S.; Lozito, T.P. Cartilage and muscle cell fate and origin during lizard tail regeneration. Front. Bioeng. Biotech. 2017, 5, 70. [Google Scholar] [CrossRef] [Green Version]
- Palade, J.; Djordjevic, D.; Hutchins, E.D.; George, R.M.; Cornelius, J.A.; Rawls, A.; Ho, J.W.K.; Kusumi, K.; Wilson-Rawls, J. Identification of satellite cells from anole lizard skeletal muscles and demonstration of expanded musculoskeletal potential. Dev. Biol. 2018, 433, 344–356. [Google Scholar] [CrossRef]
- Tokuyama, M.A.; Xu, C.; Fisher, R.E.; Wilson-Rawls, J.; Kusumi, K.; Newbern, J.M. Developmental and adult-specific processes contribute to de novo neuromuscular regeneration in the lizard tail. Dev. Biol. 2018, 433, 287–296. [Google Scholar] [CrossRef]
- Sharma, P.; Suresh, B. Influence of Cox-2-induced PGE2 on the initiation and progression of tail regeneration in Northern house gecko, Hemidactylus flaviviridis. Folia Biol. 2008, 54, 193–201. [Google Scholar]
- Nambiar, V.V.; Bhatt, I.Y.; Deshmukh, P.A.; Jape, D.D.J.; Jivani, P.N.; Kavale, H.R.; Prakashkar, S.S.; Ramachandran, A.V. Assessment of extracellular matrix remodeling during tail regeneration in the lizard Hemidactylus flaviviridis. J. Endocr. Reprod. 2008, 2, 67–72. [Google Scholar]
- Murawala, H.; Ranadive, I.; Patel, S.; Desai, I.; Balakrishnan, S. Protein expression pattern and analysis of differentially expressed peptides during various stages of tail regeneration in Hemidactylus flaviviridis. Mech. Dev. 2018, 150, 1–9. [Google Scholar] [CrossRef]
- Hutchins, E.D.; Markov, G.J.; Eckalbar, W.L.; Gorge, R.M.; King, J.M.; Tokuyama, M.A.; Geiger, L.A.; Emmert, N.; Ammar, M.J.; Allen, A.P.; et al. Transcriptomic analysis of tail regeneration in the lizard Anolis carolinensis reveals activation of conserved vertebrate developmental and repair mechanisms. PLoS ONE 2014, 9, e105004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchins, E.D.; Eckalbar, W.L.; Walter, J.M.; Mangone, M.; Kosumi, K. Differential expression of conserved and novel microRNAs during tail regeneration in the lizard Anolis carolinensis. BMC Genom. 2016, 17, 339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Zhou, Q.; Wang, Y.; Luo, L.; Yang, J.; Yang, L.; Liu, M.; Li, Y.; Qian, T.; Zheng, Y.; et al. Gekko japonicus genome reveal evolution of adhesive toe pads and tail regeneration. Nat. Commun. 2015, 6, 10033. [Google Scholar] [CrossRef] [PubMed]
- Vitulo, N.; Dalla Valle, L.; Skobo, T.; Valle, G.; Alibardi, L. Transcriptome analysis of the regenerating tail versus the scarring limb in lizard reveals pathways leading to successful versus unsuccessful organ regeneration in amniotes. Dev. Dyn. 2017, 246, 116–134. [Google Scholar] [CrossRef] [Green Version]
- Vitulo, N.; Dalla Valle, L.; Skobo, T.; Valle, G.; Alibardi, L. Down-regulation of lizard immuno-genes in the regenerating tail and myo-genes in the scarring limb suggests that tail regeneration occurs in an immuno-privileged organ. Protoplasma 2017, 254, 2127–2141. [Google Scholar] [CrossRef]
- Degan, M.; Dalla Valle, L.; Alibardi, L. Gene expression in regenerating and scarring tails of lizard evidences three main key genes (wnt2b, egfl6 and arhgap28) activated during the regulated process of tail regeneration. Protoplasma 2021, 258, 3–17. [Google Scholar] [CrossRef]
- Alibardi, L. Biological and molecular differences between tail regeneration and limb scarring in lizard, an inspiring model addressing limb regeneration in amniotes. J. Exp. Zool. B 2017, 328, 493–514. [Google Scholar] [CrossRef]
- Alibardi, L. Hyaluronic acid in the tail and limb of amphibians and lizards recreates permissive embryonic conditions for regeneration due to its hygroscopic and immuno-suppressive properties. J. Exp. Zool. B 2017, 328, 760–771. [Google Scholar] [CrossRef]
- Alibardi, L. Appendage regeneration in anamniotes utilizes genes active during larval-metamorphic stages that have been lost or altered in amniotes: The case for studying lizard tail regeneration. J. Morphol. 2020, 281, 1358–1381. [Google Scholar] [CrossRef]
- Alibardi, L. Regeneration of the tail in lizards appears regulated by a balanced expression of oncogenes and tumor suppressors likely controlling epidermal mesenchymal transition and apical tail growth. Ann. Anat. 2021, 239, 151824. [Google Scholar] [CrossRef]
- Jacyniak, K.; McDonald, R.P.; Vickaryous, M.K. Tail regeneration and other phenomena of wound healing and tissue restoration in lizards. J. Exp. Biol. 2017, 220, 2858–2869. [Google Scholar] [CrossRef] [Green Version]
- Alibardi, L. Spinal ganglia and peripheral nerves innervating the regenerating tail and muscles of lizards. J. Morphol. 2021. [Google Scholar] [CrossRef]
- Pough, H.F.; Janis, C.M.; Heiser, J.B. Vertebrate Life; Pearson Benjamin Cummings: San Francisco, CA, USA, 2009. [Google Scholar]
- Arnold, N. The throwaway tail. New Sci. 1990, 125, 42–45. [Google Scholar]
- Ananieva, N.B.; Gordeev, D.A.; Korost, D.V. The review of the autotomy of agamid lizards with considerations about the types of autotomy and regeneration. J. Dev. Biol. 2021, 9, 32. [Google Scholar] [CrossRef]
- Bateman, J.J.; Boisvert, C.A.; Bateman, P.W. At what cost? Trade-off and influences on energetic investment to tail regeneration in lizards following autotomy. J. Dev. Biol. 2021, in press. [Google Scholar]
- Bryant, S.V.; Bellairs, A.D.A. Amnio-allantoic constriction bands in lizard embryos and their effects on tail regeneration. J. Zool. 1967, 152, 155–161. [Google Scholar] [CrossRef]
- Alibardi, L. Immunolocalization indicates that both original and regenerated lizard tail tissues contain populations of long retaining cells, putative stem/progenitor cells. Micr. Res. Techn. 2015, 78, 1032–1045. [Google Scholar] [CrossRef]
- Holthaus, K.B.; Spisni, E.; Alibardi, L. Microbicide activity of two reptilian antimicrobial peptides on Gram positive and Gram negative bacteria. J. Immunobiol. 2016, 1, 10–4172. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alibardi, L. Introduction to the Study on Regeneration in Lizards as an Amniote Model of Organ Regeneration. J. Dev. Biol. 2021, 9, 51. https://doi.org/10.3390/jdb9040051
Alibardi L. Introduction to the Study on Regeneration in Lizards as an Amniote Model of Organ Regeneration. Journal of Developmental Biology. 2021; 9(4):51. https://doi.org/10.3390/jdb9040051
Chicago/Turabian StyleAlibardi, Lorenzo. 2021. "Introduction to the Study on Regeneration in Lizards as an Amniote Model of Organ Regeneration" Journal of Developmental Biology 9, no. 4: 51. https://doi.org/10.3390/jdb9040051
APA StyleAlibardi, L. (2021). Introduction to the Study on Regeneration in Lizards as an Amniote Model of Organ Regeneration. Journal of Developmental Biology, 9(4), 51. https://doi.org/10.3390/jdb9040051



