What Is Lost in the Weismann Barrier?
Abstract
1. Introduction
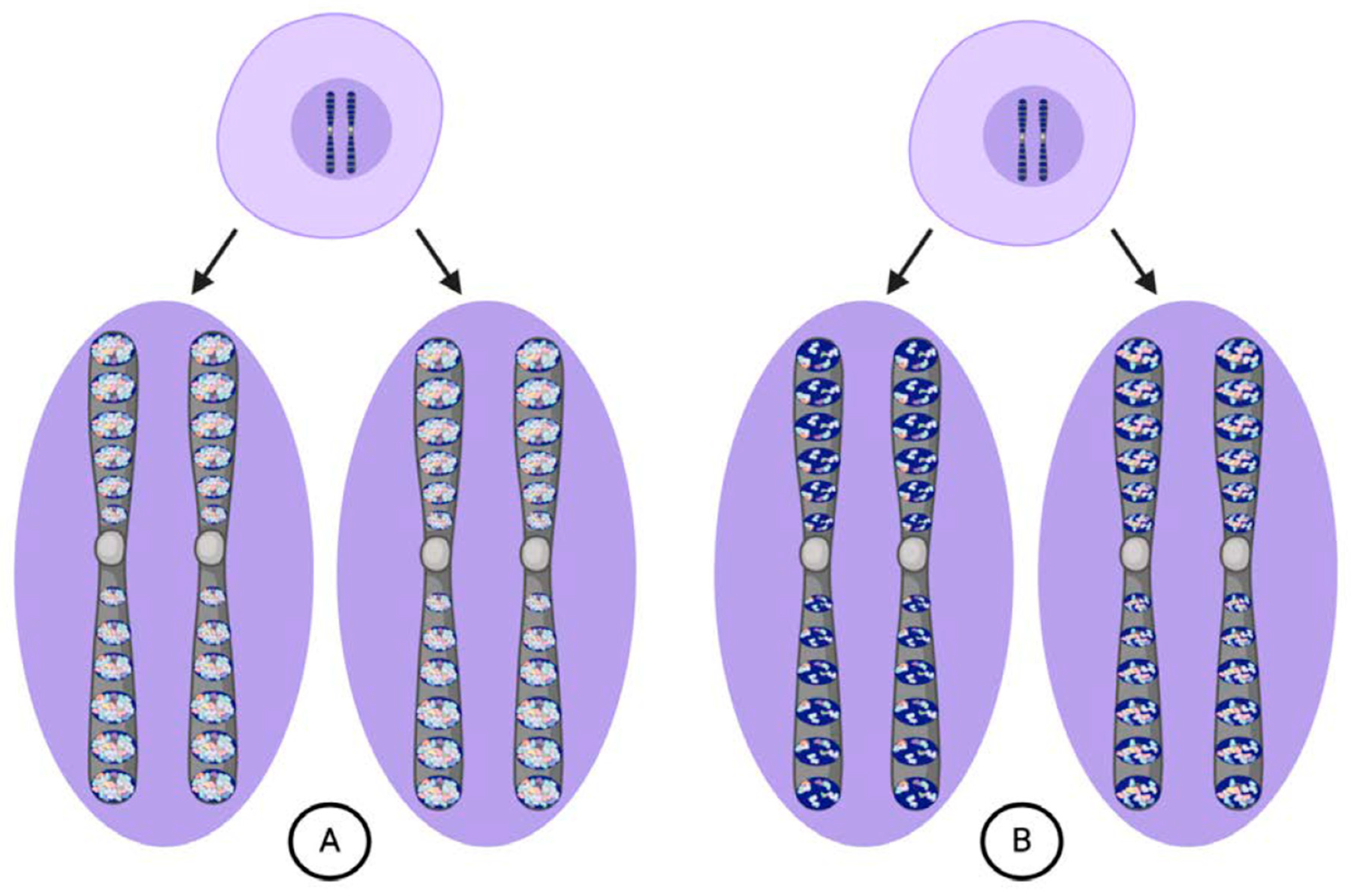
2. Weismann’s Model of Inheritance
3. Origins of the Weismann Barrier
4. What Is Lost in the Weismann Barrier?
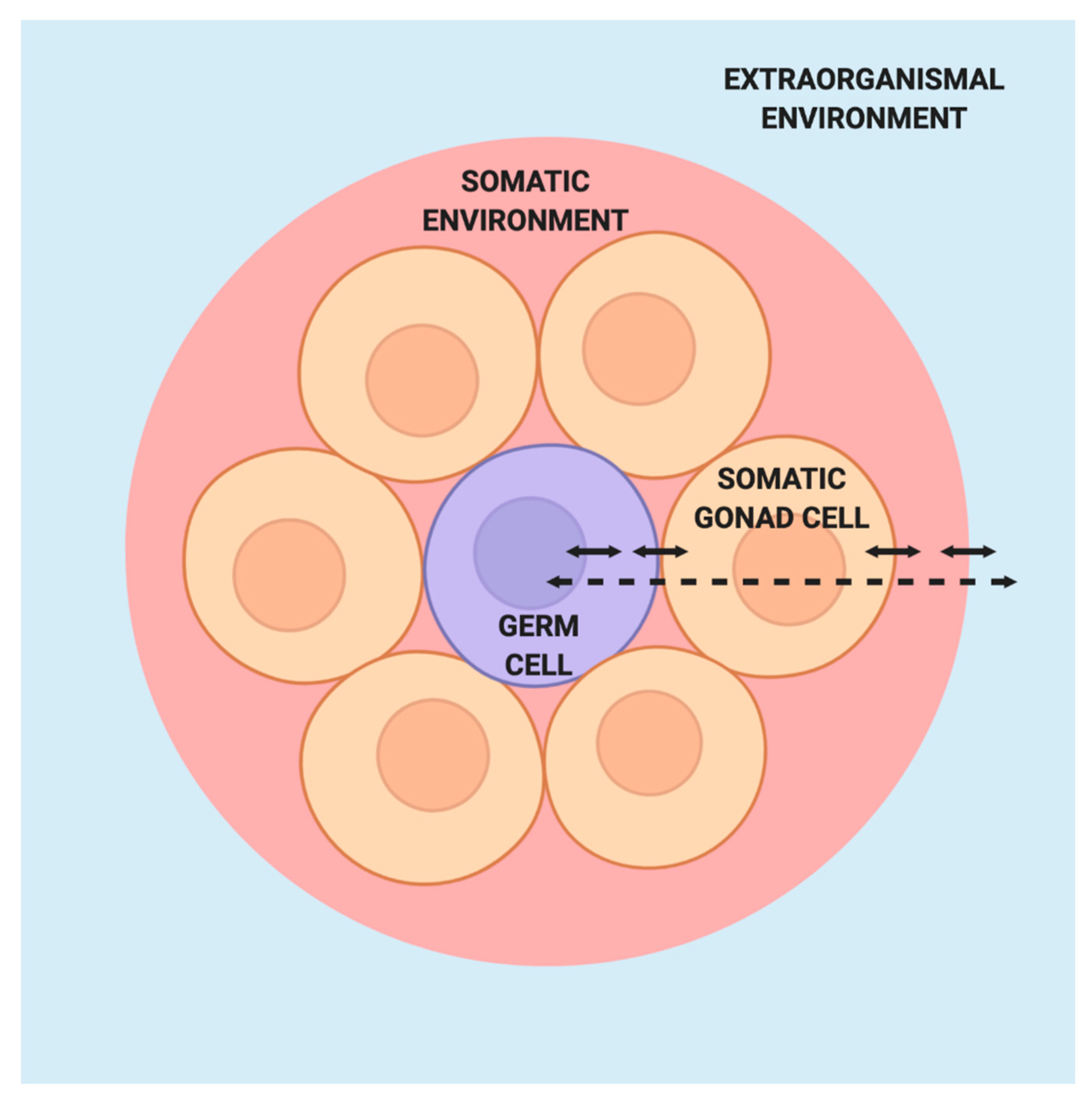
4.1. Emergent Germ Cell Fate and Function through Interaction with Somatic Cells
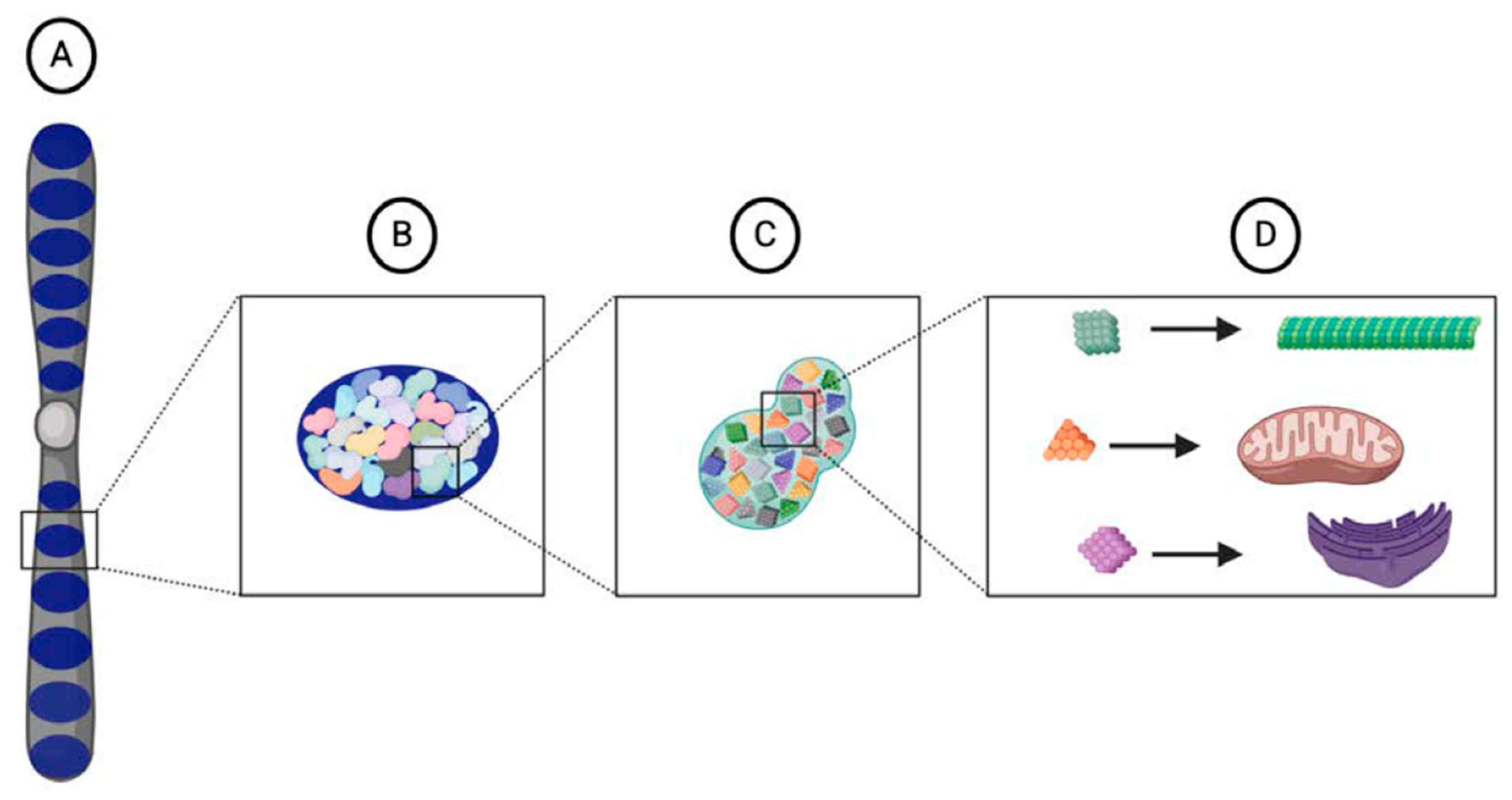
4.2. A Broader View of Heritable Information
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Sciamanna, I.; Serafino, A.; Shapiro, J.A.; Spadafora, C. The active role of spermatozoa in transgenerational inheritance. Proc. R. Soc. B Biol. Sci. 2019, 286, 20191263. [Google Scholar] [CrossRef] [PubMed]
- Deichmann, U. The social construction of the social epigenome and the larger biological context. Epigenet. Chromat. 2020, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Churchill, F.B. August Weismann, and a break from tradition. J. Hist. Biol. 1968, 1, 91–112. [Google Scholar] [CrossRef]
- Chen, D.; Sun, N.; Hou, L.; Kim, R.; Faith, J.; Aslanyan, M.; Tao, Y.; Zheng, Y.; Fu, J.; Liu, W.; et al. Human Primordial Germ Cells Are Specified from Lineage-Primed Progenitors. Cell Rep. 2019, 29, 4568–4582. [Google Scholar] [CrossRef] [PubMed]
- Weismann, A. The Germplasm: A Theory of Heredity; Charles Scribner’s Sons: New York, NY, USA, 1893. [Google Scholar]
- Aucamp, J.; Bronkhorst, A.J.; Badenhorst, C.P.S.; Pretorius, P.J. A historical and evolutionary perspective on the biological significance of circulating DNA and extracellular vesicles. Cell. Mol. Life Sci. 2016, 73, 4355–4381. [Google Scholar] [CrossRef] [PubMed]
- Darwin, C. On the Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life; John Murray: London, UK, 1859. [Google Scholar]
- Mayr, E. Weismann and evolution. J. Hist. Biol. 1985, 18, 295–329. [Google Scholar] [CrossRef] [PubMed]
- Churchill, F.B. August Weismann: Development, Heredity, and Evolution; Harvard University Press: Cambridge, MA, USA, 2015; ISBN 978-0-674-73689-4. [Google Scholar]
- Johannsen, W. The Genotype Conception of Heredity. Am. Natural. 1911, 531, 129–159. [Google Scholar] [CrossRef]
- Wilson, E.B. The Cell in Development and Inheritance; The Macmillan Company: New York, NY, USA, 1896. [Google Scholar]
- Griesemer, J.R.; Wimsatt, W.C. Picturing Weismannism: A Case Study of Conceptual Evolution. In Popper and the Human Sciences; Currie, J., Musgrave, A., Eds.; Springer: Berlin/Heidelberg, Germany, 1989; pp. 75–137. [Google Scholar]
- Simpson, G.G.; Pittendrigh, C.S.; Tiffany, L.H. Life: An Introduction to Biology; Harcourt, Brace & World, Inc.: New York, NY, USA, 1957. [Google Scholar]
- Morgan, T.H. The Rise of Genetics. Science 1932, 76, 261–267. [Google Scholar] [CrossRef]
- Weiss, S.F. The Nazi Symbiosis: Human Genetics and Politics in the Third Reich; The University of Chicago Press: Chicago, IL, USA, 2010; ISBN 9780226891767. [Google Scholar]
- Kutschera, U.; Niklas, K.J. The modern theory of biological evolution: An expanded synthesis. Naturwissenschaften 2004, 91, 255–276. [Google Scholar] [CrossRef]
- Lickliter, R. The Origins of Variation. In Advances in Child Development and Behavior; Elsevier BV: Amsterdam, The Netherlands, 2013; Volume 44, pp. 193–223. [Google Scholar]
- Solana, J. Closing the circle of germline and stem cells: The Primordial Stem Cell hypothesis. EvoDevo 2013, 4, 2. [Google Scholar] [CrossRef]
- Dawkins, R. Extended Phenotype—But Not Too Extended. A Reply to Laland, Turner and Jablonka. Biol. Philos. 2004, 19, 377–396. [Google Scholar] [CrossRef]
- Anava, S.; Posner, R.; Rechavi, O. The soft genome. Worm 2014, 3, e989798. [Google Scholar] [CrossRef] [PubMed]
- Günesdogan, U.; Magnúsdóttir, E.; Surani, M.A. Correction to ‘Primoridal germ cell specification: A context-dependent cellular differentiation event’. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20140314. [Google Scholar] [CrossRef]
- Tang, W.W.C.; Kobayashi, T.; Irie, N.; Dietmann, S.; Surani, M.A. Specification and epigenetic programming of the human germ line. Nat. Rev. Genet. 2016, 17, 585–600. [Google Scholar] [CrossRef] [PubMed]
- Irie, N.; Weinberger, L.; Tang, W.W.; Kobayashi, T.; Viukov, S.; Manor, Y.S.; Dietmann, S.; Hanna, J.H.; Surani, M.A. SOX17 Is a Critical Specifier of Human Primordial Germ Cell Fate. Cell 2015, 160, 253–268. [Google Scholar] [CrossRef]
- Ohta, H.; Kurimoto, K.; Okamoto, I.; Nakamura, T.; Yabuta, Y.; Miyauchi, H.; Yamamoto, T.; Okuno, Y.; Hagiwara, M.; Shirane, K.; et al. In vitro expansion of mouse primordial germ cell-like cells recapitulates an epigenetic blank slate. EMBO J. 2017, 36, 1888–1907. [Google Scholar] [CrossRef]
- Gell, J.J.; Liu, W.; Sosa, E.; Chialastri, A.; Hancock, G.; Tao, Y.; Wamaitha, S.E.; Bower, G.; Dey, S.S.; Clark, A. An Extended Culture System that Supports Human Primordial Germ Cell-like Cell Survival and Initiation of DNA Methylation Erasure. Stem Cell Rep. 2020, 14, 433–446. [Google Scholar] [CrossRef]
- Yamashiro, C.; Sasaki, K.; Yokobayashi, S.; Kojima, Y.; Saitou, M. Generation of human oogonia from induced pluripotent stem cells in culture. Nat. Protoc. 2020, 15, 1560–1583. [Google Scholar] [CrossRef]
- Kimble, J. Molecular Regulation of the Mitosis/Meiosis Decision in Multicellular Organisms. Cold Spring Harb. Perspect. Biol. 2011, 3, a002683. [Google Scholar] [CrossRef]
- Feng, C.-W.; Bowles, J.; Koopman, P. Control of mammalian germ cell entry into meiosis. Mol. Cell. Endocrinol. 2014, 382, 488–497. [Google Scholar] [CrossRef]
- Carabatsos, M.J.; Sellitto, C.; Goodenough, D.A.; Albertini, D.F. Oocyte–Granulosa Cell Heterologous Gap Junctions Are Required for the Coordination of Nuclear and Cytoplasmic Meiotic Competence. Dev. Biol. 2000, 226, 167–179. [Google Scholar] [CrossRef] [PubMed]
- El-Hayek, S.; Yang, Q.; Abbassi, L.; Fitzharris, G.; Clarke, H.J. Mammalian Oocytes Locally Remodel Follicular Architecture to Provide the Foundation for Germline-Soma Communication. Curr. Biol. 2018, 28, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Rode, K.; Weider, K.; Damm, O.S.; Wistuba, J.; Langeheine, M.; Brehm, R. Loss of connexin 43 in Sertoli cells provokes postnatal spermatogonial arrest, reduced germ cell numbers and impaired spermatogenesis. Reprod. Biol. 2018, 18, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Starich, T.A.; Hall, D.H.; Greenstein, D. Two Classes of Gap Junction Channels Mediate Soma-Germline Interactions Essential for Germline Proliferation and Gametogenesis in Caenorhabditis elegans. Genetics 2014, 198, 1127–1153. [Google Scholar] [CrossRef] [PubMed]
- Huelgas-Morales, G.; Greenstein, D. Control of oocyte meiotic maturation in C. elegans. Semin. Cell Dev. Biol. 2018, 84, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Mihalas, B.P.; Redgrove, K.A.; Bernstein, I.R.; Robertson, M.J.; McCluskey, A.; Nixon, B.; Holt, J.E.; McLaughlin, E.A.; Sutherland, J.M. Dynamin 2-dependent endocytosis is essential for mouse oocyte development and fertility. FASEB J. 2020, 34, 5162–5177. [Google Scholar] [CrossRef]
- Machtinger, R.; Laurent, L.C.; Baccarelli, A.A. Extracellular vesicles: Roles in gamete maturation, fertilization and embryo implantation. Hum. Reprod. Updat. 2015, 22, 182–193. [Google Scholar] [CrossRef]
- McCarter, J.; Bartlett, B.; Dang, T.; Schedl, T. Soma-Germ Cell Interactions in Caenorhabditis elegans: Multiple Events of Hermaphrodite Germline Development Require the Somatic Sheath and Spermathecal Lineages. Dev. Biol. 1997, 181, 121–143. [Google Scholar] [CrossRef]
- Conine, C.C.; Sun, F.; Song, L.; Rivera-Pérez, J.A.; Rando, O.J. Small RNAs Gained during Epididymal Transit of Sperm Are Essential for Embryonic Development in Mice. Dev. Cell 2018, 46, 470–480. [Google Scholar] [CrossRef]
- Zhou, W.; Stanger, S.J.; Anderson, A.L.; Bernstein, I.R.; de Iuliis, G.; McCluskey, A.; McLaughlin, E.A.; Dun, M.D.; Nixon, B. Mechanisms of tethering and cargo transfer during epididymosome-sperm interactions. BMC Biol. 2019, 17, 35. [Google Scholar] [CrossRef]
- Sharma, U. Paternal Contributions to Offspring Health: Role of Sperm Small RNAs in Intergenerational Transmission of Epigenetic Information. Front. Cell Dev. Biol. 2019, 7, 215. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yan, M.; Cao, Z.; Li, X.; Zhang, Y.; Shi, J.; Feng, G.-H.; Peng, H.; Zhang, X.; Qian, J.; et al. Sperm tsRNAs contribute to intergenerational inheritance of an acquired metabolic disorder. Science 2016, 351, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Gapp, K.; van Steenwyk, G.; Germain, P.-L.; Matsushima, W.; Rudolph, K.L.M.; Manuella, F.; Roszkowski, M.; Vernaz, G.; Ghosh, T.; Pelczar, P.; et al. Alterations in sperm long RNA contribute to the epigenetic inheritance of the effects of postnatal trauma. Mol. Psychiatry 2020, 25, 2162–2174. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, A.B.; Morgan, C.P.; Bronson, S.L.; Revello, S.; Bale, T.L. Paternal Stress Exposure Alters Sperm MicroRNA Content and Reprograms Offspring HPA Stress Axis Regulation. J. Neurosci. 2013, 33, 9003–9012. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zheng, Y.; Lei, A.; Zhang, H.; Niu, H.; Li, X.; Zhang, P.; Liao, M.; Lv, Y.; Zhu, Z.; et al. Early cleavage of preimplantation embryos is regulated by tRNAGln-TTG–derived small RNAs present in mature spermatozoa. J. Biol. Chem. 2020, 295, 10885–10900. [Google Scholar] [CrossRef] [PubMed]
- Bošković, A.; Rando, O.J. Transgenerational Epigenetic Inheritance. Annu. Rev. Genet. 2018, 52, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Posner, R.; Toker, I.A.; Antonova, O.; Star, E.; Anava, S.; Azmon, E.; Hendricks, M.; Bracha, S.; Gingold, H.; Rechavi, O. Neuronal Small RNAs Control Behavior Transgenerationally. Cell 2019, 177, 1814–1826. [Google Scholar] [CrossRef]
- Shih, J.D.; Hunter, C.P. SID-1 is a dsRNA-selective dsRNA-gated channel. RNA 2011, 17, 1057–1065. [Google Scholar] [CrossRef]
- Diaz-Castillo, C.; Chamorro-Garcia, R.; Shioda, T.; Blumberg, B. Transgenerational Self-Reconstruction of Disrupted Chromatin Organization After Exposure to An Environmental Stressor in Mice. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef]
- Nuebler, J.; Fudenberg, G.; Imakaev, M.; Abdennur, N.; Mirny, L.A. Chromatin organization by an interplay of loop extrusion and compartmental segregation. Proc. Natl. Acad. Sci. USA 2018, 115, E6697–E6706. [Google Scholar] [CrossRef]
- Strom, A.R.; Emelyanov, A.V.; Mir, M.; Fyodorov, D.V.; Darzacq, X.; Karpen, G.H. Phase separation drives heterochromatin domain formation. Nat. Cell Biol. 2017, 547, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kutateladze, T.G. Liquid-Liquid Phase Separation is an Intrinsic Physiochemical Property of Chromatin. Nat. Struct. Mol. Biol. 2019, 26, 1085–1086. [Google Scholar] [CrossRef] [PubMed]
- Janssen, A.; Colmenares, S.U.; Karpen, G.H. Heterochromatin: Guardian of the Genome. Annu. Rev. Cell Dev. Biol. 2018, 34, 265–288. [Google Scholar] [CrossRef] [PubMed]
- Weismann, A. Essays upon Heredity and Kindred Biological Problems; Clarendon Press: Oxford, UK, 1889. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bline, A.P.; Le Goff, A.; Allard, P. What Is Lost in the Weismann Barrier? J. Dev. Biol. 2020, 8, 35. https://doi.org/10.3390/jdb8040035
Bline AP, Le Goff A, Allard P. What Is Lost in the Weismann Barrier? Journal of Developmental Biology. 2020; 8(4):35. https://doi.org/10.3390/jdb8040035
Chicago/Turabian StyleBline, Abigail P., Anne Le Goff, and Patrick Allard. 2020. "What Is Lost in the Weismann Barrier?" Journal of Developmental Biology 8, no. 4: 35. https://doi.org/10.3390/jdb8040035
APA StyleBline, A. P., Le Goff, A., & Allard, P. (2020). What Is Lost in the Weismann Barrier? Journal of Developmental Biology, 8(4), 35. https://doi.org/10.3390/jdb8040035




