Specification and Patterning of Drosophila Appendages
Abstract
1. Introduction
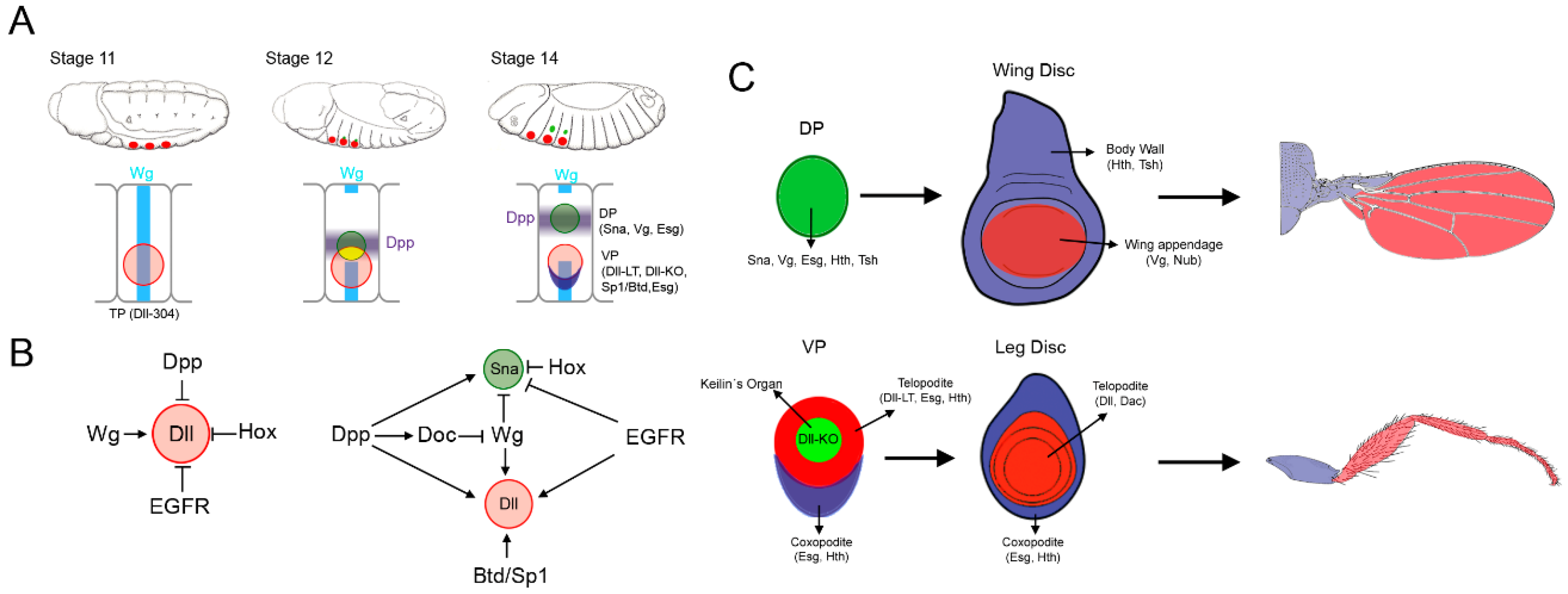
2. Allocation of the Thoracic Appendage Primordia
2.1. Specification of the Ventral and Dorsal Primordia
2.2. Dual Developmental Origin of the Drosophila Wing and Its Evolutionary Implications
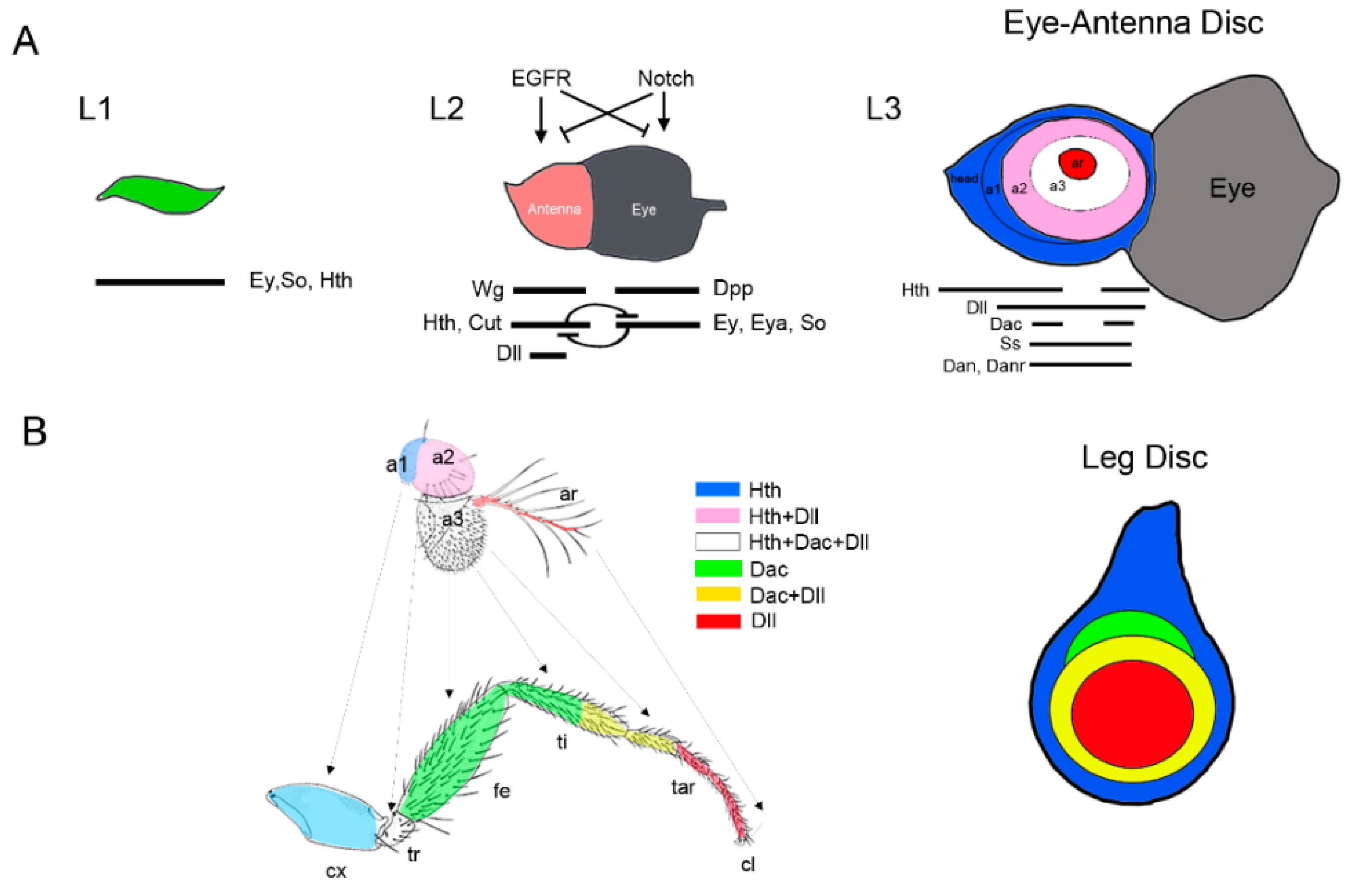
2.3. Proximo-Distal Subdivision of the Ventral Primordia
3. Specification of the Appendage Domain and Patterning of the Leg, Antenna and Wing Imaginal Discs
3.1. Patterning of the Leg Imaginal Disc
3.2. Patterning of the Wing Imaginal Disc
3.3. Patterning of the Eye-Antennal Disc
4. Perspectives
Funding
Acknowledgments
Conflicts of Interest
References
- Engel, M.S. Insect evolution. Curr. Biol. 2015, 25, R868–R872. [Google Scholar] [CrossRef] [PubMed]
- Angelini, D.R.; Kaufman, T.C. Insect appendages and comparative ontogenetics. Dev. Biol. 2005, 286, 57–77. [Google Scholar] [CrossRef] [PubMed]
- Tabin, C.; Carroll, S.; Panganiban, G. Out on a limb: Parallels in vertebrate and invertebrate limb patterning and the origin of appendages. Am. Zool. 1999, 39, 650–663. [Google Scholar] [CrossRef]
- Pueyo, J.I.; Couso, J.P. Parallels between the proximal-distal development of vertebrate and arthropod appendages: Homology without an ancestor? Curr. Opin. Genet. Dev. 2005, 15, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Panganiban, G.; Irvine, S.M.; Lowe, C.; Roehl, H.; Corley, L.S.; Sherbon, B.; Grenier, J.K.; Fallon, J.F.; Kimble, J.; Walker, M.; et al. The origin and evolution of animal appendages. Proc. Natl. Acad. Sci. USA 1997, 94, 5162–5166. [Google Scholar] [CrossRef] [PubMed]
- Shubin, N.; Tabin, C.; Carroll, S. Fossils, genes and the evolution of animal limbs. Nature 1997, 388, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Shubin, N.; Tabin, C.; Carroll, S. Deep homology and the origins of evolutionary novelty. Nature 2009, 457, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Cohen, B.; Simcox, A.A.; Cohen, S.M. Allocation of the thoracic imaginal primordia in the Drosophila embryo. Development 1993, 117, 597–608. [Google Scholar] [PubMed]
- Bate, M.; Arias, A.M. The embryonic origin of imaginal discs in Drosophila. Development 1991, 112, 755–761. [Google Scholar] [PubMed]
- Estrada, B.; Casares, F.; Sanchez-Herrero, E. Development of the genitalia in Drosophila melanogaster. Differentiation 2003, 71, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Younossi-Hartenstein, A.; Tepass, U.; Hartenstein, V. Embryonic origin of the imaginal discs of the head of Drosophila melanogaster. Roux. Arch. Dev. Biol. 1993, 203, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Haynie, J.L.; Bryant, P.J. Development of the eye-antenna imaginal disc and morphogenesis of the adult head in Drosophila melanogaster. J. Exp. Zool. 1986, 237, 293–308. [Google Scholar] [CrossRef] [PubMed]
- Requena, D.; Alvarez, J.A.; Gabilondo, H.; Loker, R.; Mann, R.S.; Estella, C. Origins and specification of the Drosophila wing. Curr. Biol. 2017, 27, 3826–3836. [Google Scholar] [CrossRef] [PubMed]
- Estella, C.; McKay, D.J.; Mann, R.S. Molecular integration of wingless, decapentaplegic, and autoregulatory inputs into Distalless during Drosophila leg development. Dev. Cell 2008, 14, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Galindo, M.I.; Fernandez-Garza, D.; Phillips, R.; Couso, J.P. Control of distal-less expression in the Drosophila appendages by functional 3′ enhancers. Dev. Biol. 2011, 353, 396–410. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.J.; Estella, C.; Mann, R.S. The origins of the Drosophila leg revealed by the cis-regulatory architecture of the Distalless gene. Development 2009, 136, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Bieli, D.; Kanca, O.; Requena, D.; Hamaratoglu, F.; Gohl, D.; Schedl, P.; Affolter, M.; Slattery, M.; Müller, M.; Estella, C. Establishment of a developmental compartment requires interactions between three synergistic cis-regulatory modules. PLoS Genet. 2015, 11, e1005376. [Google Scholar] [CrossRef] [PubMed]
- Emmons, R.B.; Duncan, D.; Duncan, I. Regulation of the Drosophila distal antennal determinant spineless. Dev. Biol. 2007, 302, 412–426. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.M. Specification of limb development in the Drosophila embryo by positional cues from segmentation genes. Nature 1990, 343, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Weaver, T.A.; White, R.A. Headcase, an imaginal specific gene required for adult morphogenesis in Drosophila melanogaster. Development 1995, 121, 4149–4160. [Google Scholar] [PubMed]
- Hayashi, S.; Hirose, S.; Metcalfe, T.; Shirras, A.D. Control of imaginal cell development by the escargot gene of Drosophila. Development 1993, 118, 105–115. [Google Scholar] [CrossRef]
- Carroll, S.B.; Weatherbee, S.D.; Langeland, J.A. Homeotic genes and the regulation and evolution of insect wing number. Nature 1995, 375, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Vachon, G.; Cohen, B.; Pfeifle, C.; McGuffin, M.E.; Botas, J.; Cohen, S.M. Homeotic genes of the Bithorax complex repress limb development in the abdomen of the Drosophila embryo through the target gene Distal-less. Cell 1992, 71, 437–450. [Google Scholar] [CrossRef]
- Castelli-Gair, J.; Akam, M. How the hox gene Ultrabithorax specifies two different segments: The significance of spatial and temporal regulation within metameres. Development 1995, 121, 2973–2982. [Google Scholar] [PubMed]
- Hughes, C.L.; Kaufman, T.C. Hox genes and the evolution of the arthropod body plan. Evol. Dev. 2002, 4, 459–499. [Google Scholar] [CrossRef] [PubMed]
- Whiteley, M.; Noguchi, P.D.; Sensabaugh, S.M.; Odenwald, W.F.; Kassis, J.A. The Drosophila gene escargot encodes a zinc finger motif found in snail-related genes. Mech. Dev. 1992, 36, 117–127. [Google Scholar] [CrossRef]
- Fuse, N.; Hirose, S.; Hayashi, S. Determination of wing cell fate by the escargot and snail genes in Drosophila. Development 1996, 122, 1059–1067. [Google Scholar] [PubMed]
- Williams, J.A.; Bell, J.B.; Carroll, S.B. Control of Drosophila wing and haltere development by the nuclear vestigial gene product. Genes Dev. 1991, 5, 2481–2495. [Google Scholar] [CrossRef] [PubMed]
- Schock, F.; Purnell, B.A.; Wimmer, E.A.; Jackle, H. Common and diverged functions of the Drosophila gene pair D-Sp1 and buttonhead. Mech. Dev. 1999, 89, 125–132. [Google Scholar] [CrossRef]
- Estella, C.; Rieckhof, G.; Calleja, M.; Morata, G. The role of buttonhead and Sp1 in the development of the ventral imaginal discs of Drosophila. Development 2003, 130, 5929–5941. [Google Scholar] [CrossRef] [PubMed]
- Panganiban, G.; Rubenstein, J.L. Developmental functions of the Distal-less/Dlx homeobox genes. Development 2002, 129, 4371–4386. [Google Scholar] [PubMed]
- Cohen, S.M.; Jurgens, G. Proximal-distal pattern formation in Drosophila: Cell autonomous requirement for Distal-less gene activity in limb development. EMBO J. 1989, 8, 2045–2055. [Google Scholar] [PubMed]
- Goto, S.; Hayashi, S. Specification of the embryonic limb primordium by graded activity of Decapentaplegic. Development 1997, 124, 125–132. [Google Scholar] [PubMed]
- Gebelein, B.; Culi, J.; Ryoo, H.D.; Zhang, W.; Mann, R.S. Specificity of Distalless repression and limb primordia development by abdominal Hox proteins. Dev. Cell 2002, 3, 487–498. [Google Scholar] [CrossRef]
- Gebelein, B.; McKay, D.J.; Mann, R.S. Direct integration of Hox and segmentation gene inputs during Drosophila development. Nature 2004, 431, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Uhl, J.D.; Zandvakili, A.; Gebelein, B. A Hox transcription factor collective binds a highly conserved distal-less cis-regulatory module to generate robust transcriptional outcomes. PLoS Genet. 2016, 12, e1005981. [Google Scholar] [CrossRef] [PubMed]
- Wieschaus, E.; Gehring, W. Clonal analysis of primordial disc cells in the early embryo of Drosophila melanogaster. Dev. Biol. 1976, 50, 249–263. [Google Scholar] [CrossRef]
- Wieschaus, E.; Gehring, W. Gynandromorph analysis of the thoracic disc primordia in Drosophila melanogaster. Wilhelm Roux. Arch. Dev. Biol. 1976, 180, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, P.A.; Morata, G. The early development of mesothoracic compartments in Drosophila. An analysis of cell lineage and fate mapping and an assessment of methods. Dev. Biol. 1977, 56, 40–51. [Google Scholar] [CrossRef]
- Kubota, K.; Goto, S.; Eto, K.; Hayashi, S. EGF receptor attenuates Dpp signaling and helps to distinguish the wing and leg cell fates in Drosophila. Development 2000, 127, 3769–3776. [Google Scholar] [PubMed]
- Kubota, K.; Goto, S.; Hayashi, S. The role of Wg signaling in the patterning of embryonic leg primordium in Drosophila. Dev. Biol. 2003, 257, 117–126. [Google Scholar] [CrossRef]
- Hamaguchi, T.; Yabe, S.; Uchiyama, H.; Murakami, R. Drosophila Tbx6-related gene, Dorsocross, mediates high levels of Dpp and Scw signal required for the development of amnioserosa and wing disc primordium. Dev. Biol. 2004, 265, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Reim, I.; Lee, H.H.; Frasch, M. The T-box-encoding Dorsocross genes function in amnioserosa development and the patterning of the dorsolateral germ band downstream of Dpp. Development 2003, 130, 3187–3204. [Google Scholar] [CrossRef] [PubMed]
- Niwa, N.; Akimoto-Kato, A.; Niimi, T.; Tojo, K.; Machida, R.; Hayashi, S. Evolutionary origin of the insect wing via integration of two developmental modules. Evol. Dev. 2010, 12, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Clark-Hachtel, C.M.; Tomoyasu, Y. Exploring the origin of insect wings from an evo-devo perspective. Curr. Opin. Insect Sci. 2016, 13, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Crampton, G. The phylogenetic origin and the nature of the wings of insects according to the paranotal theory. J. N. Y. Entomol. Soc. 1916, 24, 1–39. [Google Scholar]
- Kukalova-Peck, J. Origin and evolution of insect wings and their relation to metamorphosis, as documented by the fossil record. J. Morphol. 1978, 156, 53–125. [Google Scholar] [CrossRef]
- Kukalova-Peck, J. Origin of the insect wing and wing articulation from the arthropodan leg. Can. J. Zool. 1983, 61, 1618–1669. [Google Scholar] [CrossRef]
- Averof, M.; Cohen, S.M. Evolutionary origin of insect wings from ancestral gills. Nature 1997, 385, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Linz, D.M.; Tomoyasu, Y. Dual evolutionary origin of insect wings supported by an investigation of the abdominal wing serial homologs in Tribolium. Proc. Natl. Acad. Sci. USA 2018, 115, E658–E667. [Google Scholar] [CrossRef] [PubMed]
- Clark-Hachtel, C.M.; Linz, D.M.; Tomoyasu, Y. Insights into insect wing origin provided by functional analysis of vestigial in the red flour beetle, Tribolium castaneum. Proc. Natl. Acad. Sci. USA 2013, 110, 16951–16956. [Google Scholar] [CrossRef] [PubMed]
- Elias-Neto, M.; Belles, X. Tergal and pleural structures contribute to the formation of ectopic prothoracic wings in cockroaches. R. Soc. Open Sci. 2016, 3, 160347. [Google Scholar] [CrossRef] [PubMed]
- Medved, V.; Marden, J.H.; Fescemyer, H.W.; Der, J.P.; Liu, J.; Mahfooz, N.; Popadic, A. Origin and diversification of wings: Insights from a neopteran insect. Proc. Natl. Acad. Sci. USA 2015, 112, 15946–15951. [Google Scholar] [CrossRef] [PubMed]
- Estella, C.; Mann, R.S. Non-redundant selector and growth-promoting functions of two sister genes, buttonhead and Sp1, in Drosophila leg development. PLoS Genet. 2010, 6, e1001001. [Google Scholar] [CrossRef] [PubMed]
- Prokop, J.; Pecharova, M.; Nel, A.; Hornschemeyer, T.; Krzeminska, E.; Krzeminski, W.; Engel, M.S. Paleozoic nymphal wing pads support dual model of insect wing origins. Curr. Biol. 2017, 27, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Couso, J.P.; Bate, M.; Martinez-Arias, A. A wingless-dependent polar coordinate system in Drosophila imaginal discs. Science 1993, 259, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.A.; Paddock, S.W.; Carroll, S.B. Pattern formation in a secondary field: A hierarchy of regulatory genes subdivides the developing Drosophila wing disc into discrete subregions. Development 1993, 117, 571–584. [Google Scholar] [PubMed]
- Ng, M.; Diaz-Benjumea, F.J.; Vincent, J.P.; Wu, J.; Cohen, S.M. Specification of the wing by localized expression of wingless protein. Nature 1996, 381, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Haro, E.; Delgado, I.; Junco, M.; Yamada, Y.; Mansouri, A.; Oberg, K.C.; Ros, M.A. Sp6 and sp8 transcription factors control AER formation and dorsal-ventral patterning in limb development. PLoS Genet. 2014, 10, e1004468. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, Y.; Esteban, C.R.; Matsui, T.; Rodriguez-Leon, J.; Kato, S.; Izpisua Belmonte, J.C. Sp8 and Sp9, two closely related buttonhead-like transcription factors, regulate Fgf8 expression and limb outgrowth in vertebrate embryos. Development 2004, 131, 4763–4774. [Google Scholar] [CrossRef] [PubMed]
- Bell, S.M.; Schreiner, C.M.; Waclaw, R.R.; Campbell, K.; Potter, S.S.; Scott, W.J. Sp8 is crucial for limb outgrowth and neuropore closure. Proc. Natl. Acad. Sci. USA 2003, 100, 12195–12200. [Google Scholar] [CrossRef] [PubMed]
- Beermann, A.; Aranda, M.; Schroder, R. The Sp8 zinc-finger transcription factor is involved in allometric growth of the limbs in the beetle Tribolium castaneum. Development 2004, 131, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Setton, E.V.W.; Sharma, P.P. Cooption of an appendage-patterning gene cassette in the head segmentation of arachnids. Proc. Natl. Acad. Sci. USA 2018, 115, E3491–E3500. [Google Scholar] [CrossRef] [PubMed]
- Schaeper, N.D.; Prpic, N.M.; Wimmer, E.A. A clustered set of three Sp-family genes is ancestral in the Metazoa: Evidence from sequence analysis, protein domain structure, developmental expression patterns and chromosomal location. BMC Evol. Biol. 2010, 10, 88. [Google Scholar] [CrossRef] [PubMed]
- Galindo, M.I.; Bishop, S.A.; Couso, J.P. Dynamic EGFR-Ras signalling in Drosophila leg development. Dev. Dyn. 2005, 233, 1496–1508. [Google Scholar] [CrossRef] [PubMed]
- Snodgrass, R. Principles of Insect Morphology; McGraw-Hill: New York, NY, USA, 1935; pp. 83–99. [Google Scholar]
- Bolinger, R.A.; Boekhoff-Falk, G. Distal-less functions in subdividing the Drosophila thoracic limb primordium. Dev. Dyn. 2005, 232, 801–816. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G.; Tomlinson, A. The roles of the homeobox genes Aristaless and Distal-less in patterning the legs and wings of Drosophila. Development 1998, 125, 4483–4493. [Google Scholar] [PubMed]
- Mandaravally Madhavan, M.; Schneiderman, H.A. Histological analysis of the dynamics of growth of imaginal discs and histoblast nests during the larval development of Drosophila melanogaster. Wilhelm Roux. Arch. Dev. Biol. 1977, 183, 269–305. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Crespo, S.; Abu-Shaar, M.; Torres, M.; Martinez, A.C.; Mann, R.S.; Morata, G. Antagonism between extradenticle function and Hedgehog signalling in the developing limb. Nature 1998, 394, 196–200. [Google Scholar] [CrossRef] [PubMed]
- González-Crespo, S.; Morata, G. Genetic evidence for the subdivision of the arthropod limb into coxopodite and telopodite. Development 1996, 122, 3921–3928. [Google Scholar] [PubMed]
- Irvine, K.D.; Rauskolb, C. Boundaries in development: Formation and function. Annu. Rev. Cell Dev. Biol. 2001, 17, 189–214. [Google Scholar] [CrossRef] [PubMed]
- Struhl, G. Genes controlling segmental specification in the Drosophila thorax. Proc. Natl. Acad. Sci. USA 1982, 79, 7380–7384. [Google Scholar] [CrossRef] [PubMed]
- Basler, K.; Struhl, G. Compartment boundaries and the control of Drosophila limb pattern by hedgehog protein. Nature 1994, 368, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Brook, W.J.; Cohen, S.M. Antagonistic interactions between wingless and decapentaplegic responsible for dorsal-ventral pattern in the Drosophila leg. Science 1996, 273, 1373–1377. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Struhl, G. Complementary and mutually exclusive activities of decapentaplegic and wingless organize axial patterning during Drosophila leg development. Cell 1996, 86, 401–409. [Google Scholar] [CrossRef]
- Johnston, L.A.; Schubiger, G. Ectopic expression of wingless in imaginal discs interferes with decapentaplegic expression and alters cell determination. Development 1996, 122, 3519–3529. [Google Scholar] [PubMed]
- Morimura, S.; Maves, L.; Chen, Y.; Hoffmann, F.M. Decapentaplegic overexpression affects Drosophila wing and leg imaginal disc development and wingless expression. Dev. Biol. 1996, 177, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Penton, A.; Hoffmann, F.M. Decapentaplegic restricts the domain of wingless during Drosophila limb patterning. Nature 1996, 382, 162–164. [Google Scholar] [CrossRef] [PubMed]
- Theisen, H.; Haerry, T.E.; O’Connor, M.B.; Marsh, J.L. Developmental territories created by mutual antagonism between wingless and decapentaplegic. Development 1996, 122, 3939–3948. [Google Scholar] [PubMed]
- Svendsen, P.C.; Formaz-Preston, A.; Leal, S.M.; Brook, W.J. The Tbx20 homologs midline and H15 specify ventral fate in the Drosophila melanogaster leg. Development 2009, 136, 2689–2693. [Google Scholar] [CrossRef] [PubMed]
- Held, L.I., Jr.; Heup, M.A. Genetic mosaic analysis of decapentaplegic and wingless gene function in the Drosophila leg. Dev. Genes Evol. 1996, 206, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G.; Weaver, T.; Tomlinson, A. Axis specification in the developing Drosophila appendage: The role of wingless, decapentaplegic, and the homeobox gene aristaless. Cell 1993, 74, 1113–1123. [Google Scholar] [CrossRef]
- Diaz-Benjumea, F.J.; Cohen, B.; Cohen, S.M. Cell interaction between compartments establishes the proximal-distal axis of Drosophila legs. Nature 1994, 372, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Lecuit, T.; Cohen, S.M. Proximal-distal axis formation in the Drosophila leg. Nature 1997, 388, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Estella, C.; Mann, R.S. Logic of wg and dpp induction of distal and medial fates in the Drosophila leg. Development 2008, 135, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Giorgianni, M.W.; Mann, R.S. Establishment of medial fates along the proximodistal axis of the Drosophila leg through direct activation of dachshund by Distalless. Dev. Cell 2011, 20, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Cohen, S.M. Proximodistal axis formation in the Drosophila leg: Subdivision into proximal and distal domains by homothorax and distal-less. Development 1999, 126, 109–117. [Google Scholar] [PubMed]
- Abu-Shaar, M.; Mann, R.S. Generation of multiple antagonistic domains along the proximodistal axis during Drosophila leg development. Development 1998, 125, 3821–3830. [Google Scholar] [PubMed]
- Weihe, U.; Dorfman, R.; Wernet, M.F.; Cohen, S.M.; Milan, M. Proximodistal subdivision of Drosophila legs and wings: The elbow-no ocelli gene complex. Development 2004, 131, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.D.; Chu, J.; Panganiban, G. Coexpression of the homeobox genes distal-less and homothorax determines Drosophila antennal identity. Development 2000, 127, 209–216. [Google Scholar] [PubMed]
- Casares, F.; Mann, R.S. A dual role for homothorax in inhibiting wing blade development and specifying proximal wing identities in Drosophila. Development 2000, 127, 1499–1508. [Google Scholar] [PubMed]
- Casares, F.; Mann, R.S. The ground state of the ventral appendage in Drosophila. Science 2001, 293, 1477–1480. [Google Scholar] [CrossRef] [PubMed]
- Emerald, B.S.; Cohen, S.M. Spatial and temporal regulation of the homeotic selector gene antennapedia is required for the establishment of leg identity in Drosophila. Dev. Biol. 2004, 267, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Galindo, M.I.; Bishop, S.A.; Greig, S.; Couso, J.P. Leg patterning driven by proximal-distal interactions and EGFR signaling. Science 2002, 297, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G. Distalization of the Drosophila leg by graded EGF-receptor activity. Nature 2002, 418, 781–785. [Google Scholar] [CrossRef] [PubMed]
- Kojima, T. The mechanism of Drosophila leg development along the proximodistal axis. Dev. Growth Differ. 2004, 46, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Kojima, T. Developmental mechanism of the tarsus in insect legs. Curr. Opin. Insect Sci. 2017, 19, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Rauskolb, C. The establishment of segmentation in the Drosophila leg. Development 2001, 128, 4511–4521. [Google Scholar] [PubMed]
- Cordoba, S.; Requena, D.; Jory, A.; Saiz, A.; Estella, C. The evolutionarily conserved transcription factor Sp1 controls appendage growth through notch signaling. Development 2016, 143, 3623–3631. [Google Scholar] [CrossRef] [PubMed]
- De Celis, J.F.; Tyler, D.M.; de Celis, J.; Bray, S.J. Notch signalling mediates segmentation of the Drosophila leg. Development 1998, 125, 4617–4626. [Google Scholar] [PubMed]
- Rauskolb, C.; Irvine, K.D. Notch-mediated segmentation and growth control of the Drosophila leg. Dev. Biol. 1999, 210, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Kerber, B.; Monge, I.; Mueller, M.; Mitchell, P.J.; Cohen, S.M. The AP-2 transcription factor is required for joint formation and cell survival in Drosophila leg development. Development 2001, 128, 1231–1238. [Google Scholar] [PubMed]
- Mirth, C.; Akam, M. Joint development in the Drosophila leg: Cell movements and cell populations. Dev. Biol. 2002, 246, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Tajiri, R.; Misaki, K.; Yonemura, S.; Hayashi, S. Joint morphology in the insect leg: Evolutionary history inferred from Notch loss-of-function phenotypes in Drosophila. Development 2011, 138, 4621–4626. [Google Scholar] [CrossRef] [PubMed]
- Hao, I.; Green, R.B.; Dunaevsky, O.; Lengyel, J.A.; Rauskolb, C. The odd-skipped family of zinc finger genes promotes Drosophila leg segmentation. Dev. Biol. 2003, 263, 282–295. [Google Scholar] [CrossRef] [PubMed]
- Cordoba, S.; Estella, C. The bHLH-PAS transcription factor dysfusion regulates tarsal joint formation in response to notch activity during Drosophila leg development. PLoS Genet. 2014, 10, e1004621. [Google Scholar] [CrossRef] [PubMed]
- Suzanne, M. Molecular and cellular mechanisms involved in leg joint morphogenesis. Semin. Cell Dev. Biol. 2016, 55, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Paul, L.; Wang, S.H.; Manivannan, S.N.; Bonanno, L.; Lewis, S.; Austin, C.L.; Simcox, A. Dpp-induced Egfr signaling triggers postembryonic wing development in Drosophila. Proc. Natl. Acad. Sci. USA 2013, 110, 5058–5063. [Google Scholar] [CrossRef] [PubMed]
- Rafel, N.; Milan, M. Notch signalling coordinates tissue growth and wing fate specification in Drosophila. Development 2008, 135, 3995–4001. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.H.; Simcox, A.; Campbell, G. Dual role for Drosophila epidermal growth factor receptor signaling in early wing disc development. Genes Dev. 2000, 14, 2271–2276. [Google Scholar] [CrossRef] [PubMed]
- Baonza, A.; Roch, F.; Martin-Blanco, E. Der signaling restricts the boundaries of the wing field during Drosophila development. Proc. Natl. Acad. Sci. USA 2000, 97, 7331–7335. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Sebring, A.; Esch, J.J.; Kraus, M.E.; Vorwerk, K.; Magee, J.; Carroll, S.B. Integration of positional signals and regulation of wing formation and identity by Drosophila vestigial gene. Nature 1996, 382, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Azpiazu, N.; Morata, G. Function and regulation of homothorax in the wing imaginal disc of Drosophila. Development 2000, 127, 2685–2693. [Google Scholar] [PubMed]
- Zirin, J.D.; Mann, R.S. Differing strategies for the establishment and maintenance of teashirt and homothorax repression in the Drosophila wing. Development 2004, 131, 5683–5693. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Cohen, S.M. Repression of teashirt marks the initiation of wing development. Development 2002, 129, 2411–2418. [Google Scholar] [PubMed]
- Blair, S.S.; Brower, D.L.; Thomas, J.B.; Zavortink, M. The role of apterous in the control of dorsoventral compartmentalization and ps integrin gene expression in the developing wing of Drosophila. Development 1994, 120, 1805–1815. [Google Scholar] [PubMed]
- Diaz-Benjumea, F.J.; Cohen, S.M. Interaction between dorsal and ventral cells in the imaginal disc directs wing development in Drosophila. Cell 1993, 75, 741–752. [Google Scholar] [CrossRef]
- Cohen, B.; McGuffin, M.E.; Pfeifle, C.; Segal, D.; Cohen, S.M. Apterous, a gene required for imaginal disc development in Drosophila encodes a member of the LIM family of developmental regulatory proteins. Genes Dev. 1992, 6, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Couso, J.P.; Knust, E.; Martinez Arias, A. Serrate and wingless cooperate to induce vestigial gene expression and wing formation in Drosophila. Curr. Biol. 1995, 5, 1437–1448. [Google Scholar] [CrossRef]
- Klein, T.; Arias, A.M. Different spatial and temporal interactions between notch, wingless, and vestigial specify proximal and distal pattern elements of the wing in Drosophila. Dev. Biol. 1998, 194, 196–212. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.; Arias, A.M. Interactions among delta, serrate and fringe modulate notch activity during Drosophila wing development. Development 1998, 125, 2951–2962. [Google Scholar] [PubMed]
- Letizia, A.; Barrio, R.; Campuzano, S. Antagonistic and cooperative actions of the EGFR and Dpp pathways on the iroquois genes regulate Drosophila mesothorax specification and patterning. Development 2007, 134, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Zecca, M.; Struhl, G. Subdivision of the Drosophila wing imaginal disc by EGFR-mediated signaling. Development 2002, 129, 1357–1368. [Google Scholar] [PubMed]
- Zecca, M.; Struhl, G. Control of growth and patterning of the Drosophila wing imaginal disc by EGFR-mediated signaling. Development 2002, 129, 1369–1376. [Google Scholar] [PubMed]
- Cavodeassi, F.; Rodriguez, I.; Modolell, J. Dpp signalling is a key effector of the wing-body wall subdivision of the Drosophila mesothorax. Development 2002, 129, 3815–3823. [Google Scholar] [PubMed]
- Calleja, M.; Herranz, H.; Estella, C.; Casal, J.; Lawrence, P.; Simpson, P.; Morata, G. Generation of medial and lateral dorsal body domains by the pannier gene of Drosophila. Development 2000, 127, 3971–3980. [Google Scholar] [PubMed]
- Minami, M.; Kinoshita, N.; Kamoshida, Y.; Tanimoto, H.; Tabata, T. Brinker is a target of dpp in Drosophila that negatively regulates Dpp-dependent genes. Nature 1999, 398, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Jazwinska, A.; Kirov, N.; Wieschaus, E.; Roth, S.; Rushlow, C. The Drosophila gene brinker reveals a novel mechanism of Dpp target gene regulation. Cell 1999, 96, 563–573. [Google Scholar] [CrossRef]
- Campbell, G.; Tomlinson, A. Transducing the Dpp morphogen gradient in the wing of Drosophila: Regulation of Dpp targets by brinker. Cell 1999, 96, 553–562. [Google Scholar] [CrossRef]
- Blair, S.S. Wing vein patterning in Drosophila and the analysis of intercellular signaling. Annu. Rev. Cell Dev. Biol. 2007, 23, 293–319. [Google Scholar] [CrossRef] [PubMed]
- De Celis, J.F. Pattern formation in the Drosophila wing: The development of the veins. BioEssays 2003, 25, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Schneuwly, S.; Klemenz, R.; Gehring, W.J. Redesigning the body plan of Drosophila by ectopic expression of the homoeotic gene antennapedia. Nature 1987, 325, 816–818. [Google Scholar] [CrossRef] [PubMed]
- Postlethwait, J.H.; Schneiderman, H.A. Pattern formation and determination in the antenna of the homoeotic mutant antennapedia of Drosophila melanogaster. Dev. Biol. 1971, 25, 606–640. [Google Scholar] [CrossRef]
- Kumar, J.P.; Moses, K. EGF receptor and notch signaling act upstream of Eyeless/Pax6 to control eye specification. Cell 2001, 104, 687–697. [Google Scholar] [CrossRef]
- Kenyon, K.L.; Ranade, S.S.; Curtiss, J.; Mlodzik, M.; Pignoni, F. Coordinating proliferation and tissue specification to promote regional identity in the Drosophila head. Dev. Cell 2003, 5, 403–414. [Google Scholar] [CrossRef]
- Wang, C.W.; Sun, Y.H. Segregation of eye and antenna fates maintained by mutual antagonism in Drosophila. Development 2012, 139, 3413–3421. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.P.; Moses, K. Eye specification in Drosophila: Perspectives and implications. Semin. Cell Dev. Biol. 2001, 12, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Bryant, P.J. Determination and pattern formation in the imaginal discs of Drosophila. Curr. Top. Dev. Biol. 1974, 8, 41–80. [Google Scholar] [PubMed]
- Morata, G.; Lawrence, P.A. Development of the eye-antenna imaginal disc of Drosophila. Dev. Biol. 1979, 70, 355–371. [Google Scholar] [CrossRef]
- Cho, K.O.; Chern, J.; Izaddoost, S.; Choi, K.W. Novel signaling from the peripodial membrane is essential for eye disc patterning in Drosophila. Cell 2000, 103, 331–342. [Google Scholar] [CrossRef]
- Baonza, A.; Freeman, M. Control of Drosophila eye specification by wingless signalling. Development 2002, 129, 5313–5322. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, M.; Casares, F. Organ specification-growth control connection: New in-sights from the Drosophila eye-antennal disc. Dev. Dyn. 2005, 232, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.D.; Chu, J.; Panganiban, G. Proximodistal domain specification and interactions in developing Drosophila appendages. Development 2001, 128, 2365–2372. [Google Scholar] [PubMed]
- Dong, P.D.; Dicks, J.S.; Panganiban, G. Distal-less and homothorax regulate multiple targets to pattern the Drosophila antenna. Development 2002, 129, 1967–1974. [Google Scholar] [PubMed]
- Casares, F.; Mann, R.S. Control of antennal versus leg development in Drosophila. Nature 1998, 392, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Duncan, D.; Kiefel, P.; Duncan, I. Control of the spineless antennal enhancer: Direct repression of antennal target genes by antennapedia. Dev. Biol. 2010, 347, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Suzanne, M.; Estella, C.; Calleja, M.; Sanchez-Herrero, E. The hernandez and fernandez genes of Drosophila specify eye and antenna. Dev. Biol. 2003, 260, 465–483. [Google Scholar] [CrossRef]
- Emerald, B.S.; Curtiss, J.; Mlodzik, M.; Cohen, S.M. Distal antenna and distal antenna related encode nuclear proteins containing pipsqueak motifs involved in antenna development in Drosophila. Development 2003, 130, 1171–1180. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Losada, M.; Blom-Dahl, D.; Córdoba, S.; Estella, C. Specification and Patterning of Drosophila Appendages. J. Dev. Biol. 2018, 6, 17. https://doi.org/10.3390/jdb6030017
Ruiz-Losada M, Blom-Dahl D, Córdoba S, Estella C. Specification and Patterning of Drosophila Appendages. Journal of Developmental Biology. 2018; 6(3):17. https://doi.org/10.3390/jdb6030017
Chicago/Turabian StyleRuiz-Losada, Mireya, David Blom-Dahl, Sergio Córdoba, and Carlos Estella. 2018. "Specification and Patterning of Drosophila Appendages" Journal of Developmental Biology 6, no. 3: 17. https://doi.org/10.3390/jdb6030017
APA StyleRuiz-Losada, M., Blom-Dahl, D., Córdoba, S., & Estella, C. (2018). Specification and Patterning of Drosophila Appendages. Journal of Developmental Biology, 6(3), 17. https://doi.org/10.3390/jdb6030017




