Hermes (Rbpms) is a Critical Component of RNP Complexes that Sequester Germline RNAs during Oogenesis
Abstract
:1. Introduction
2. Experimental Section
2.1. Oocytes and Microinjection
2.2. Myc-Hermes/Rbpms Immunoprecipitation
2.3. Mutagenesis, Cloning, and RNA Synthesis
2.4. In Vitro Translation
2.5. RNA-AADA Pull-Down
2.6. Yeast-Two-Hybrid
2.7. Immunofluorescence Microscopy and Antibodies
2.8. Ultrastructural Analysis
2.9. Ethical Statements
3. Results
3.1. Hermes/Rbpms Co-Localizes with Nanos RNA within Germinal Granules
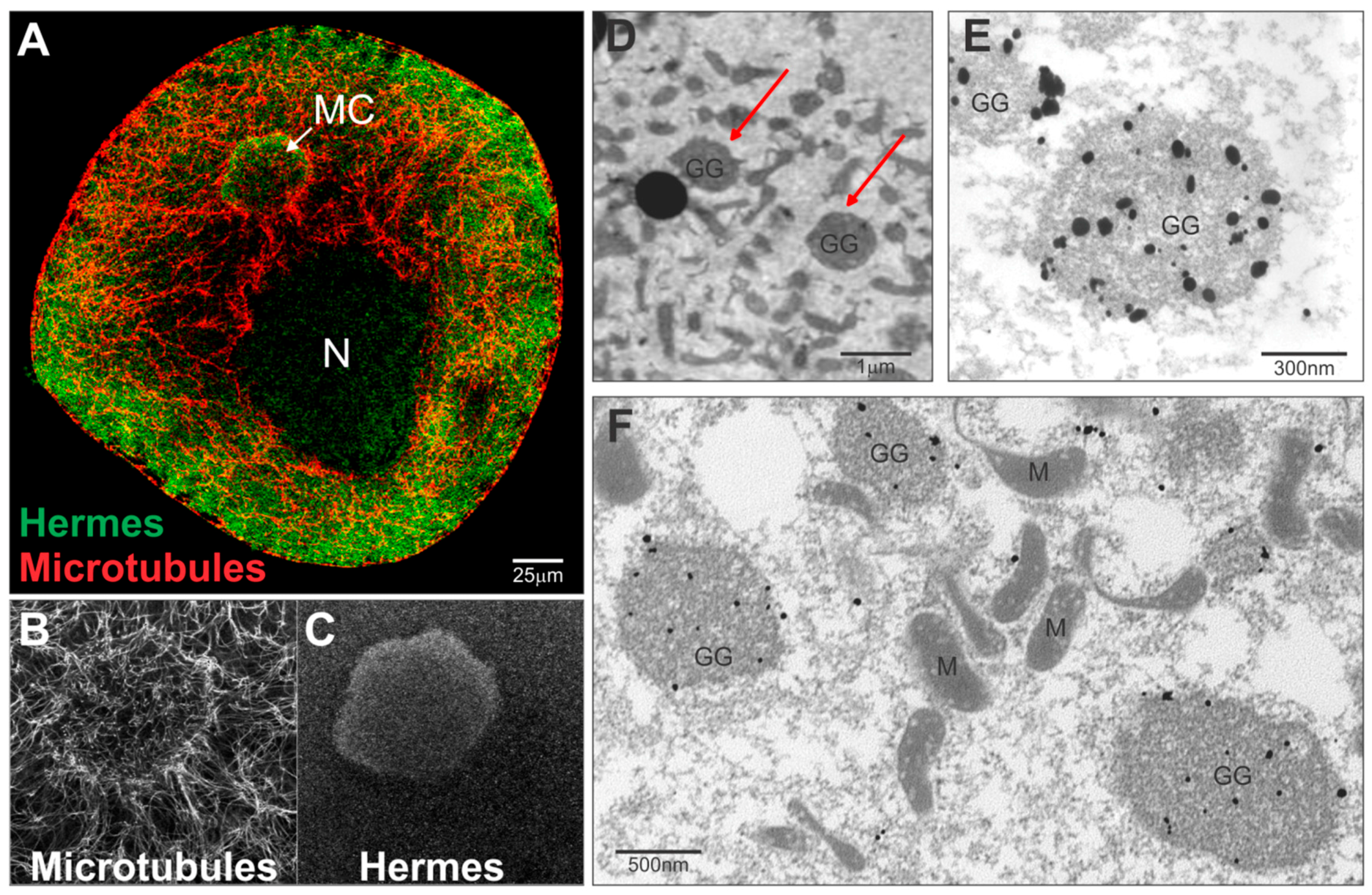
3.2. Hermes/Rbpms Protein Specifically Binds the Nanos Mitochondrial Cloud Localization Signal
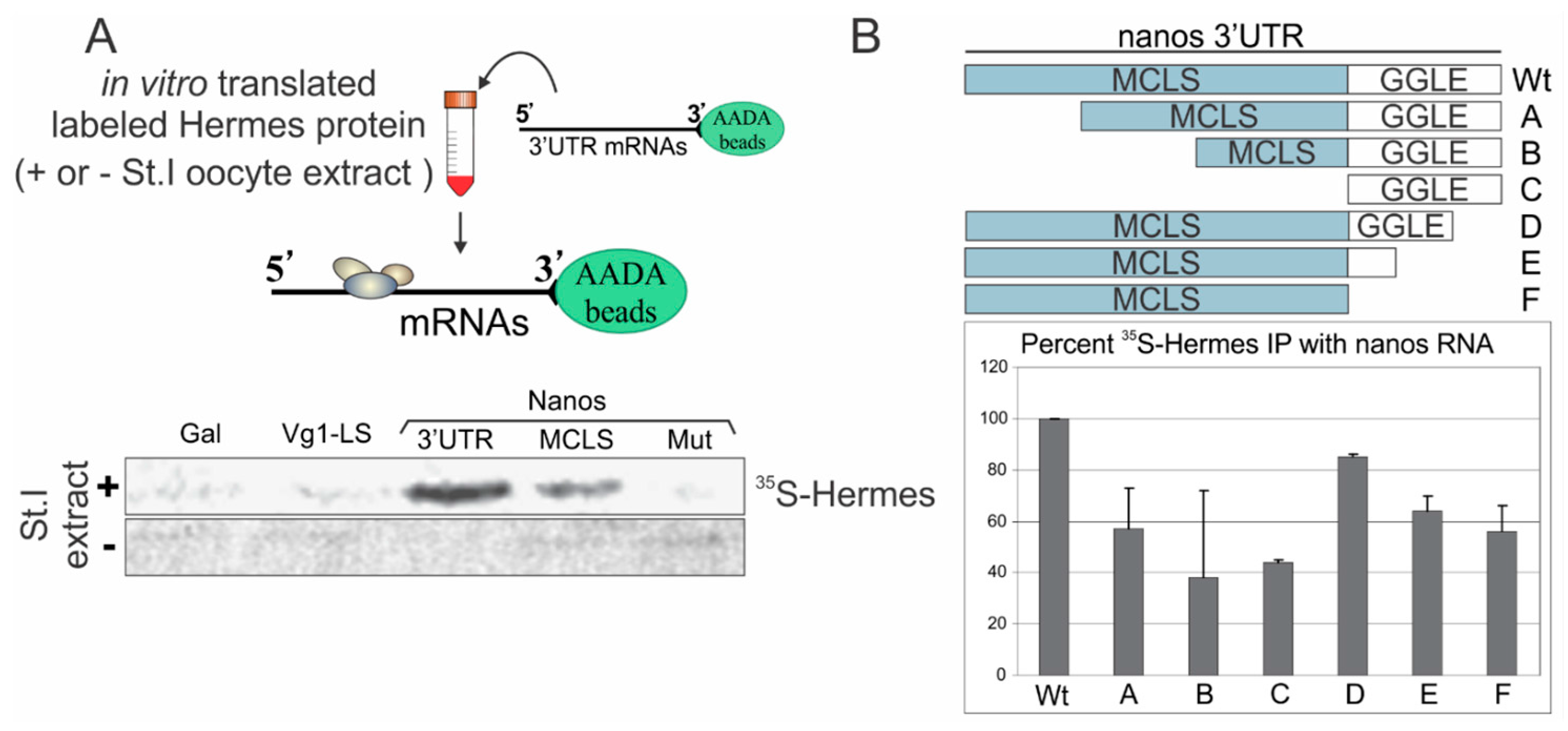
3.3. Mapping Hermes/Rbpms for Regions Required for Nanos-Hermes/Rbpms Interaction
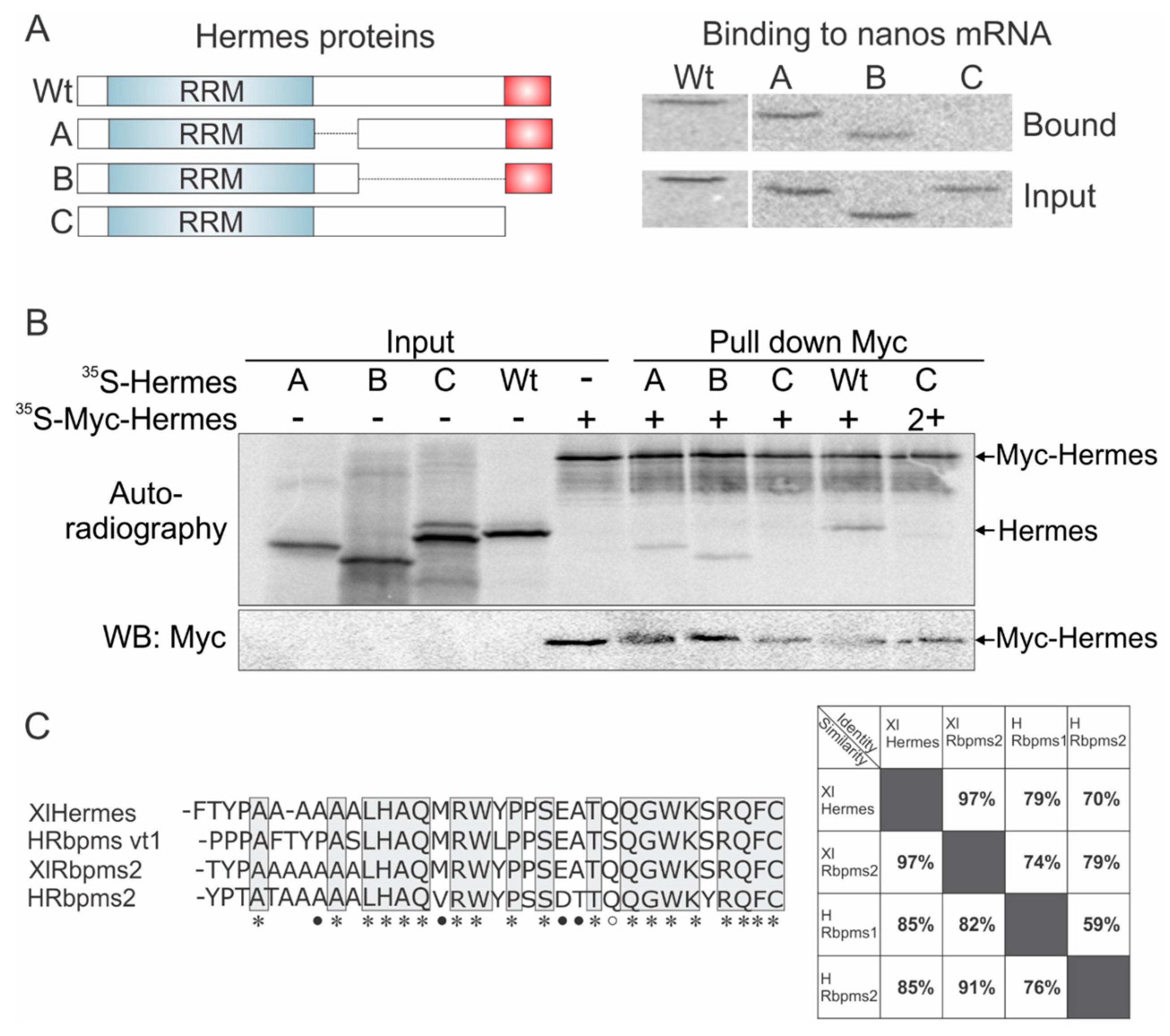
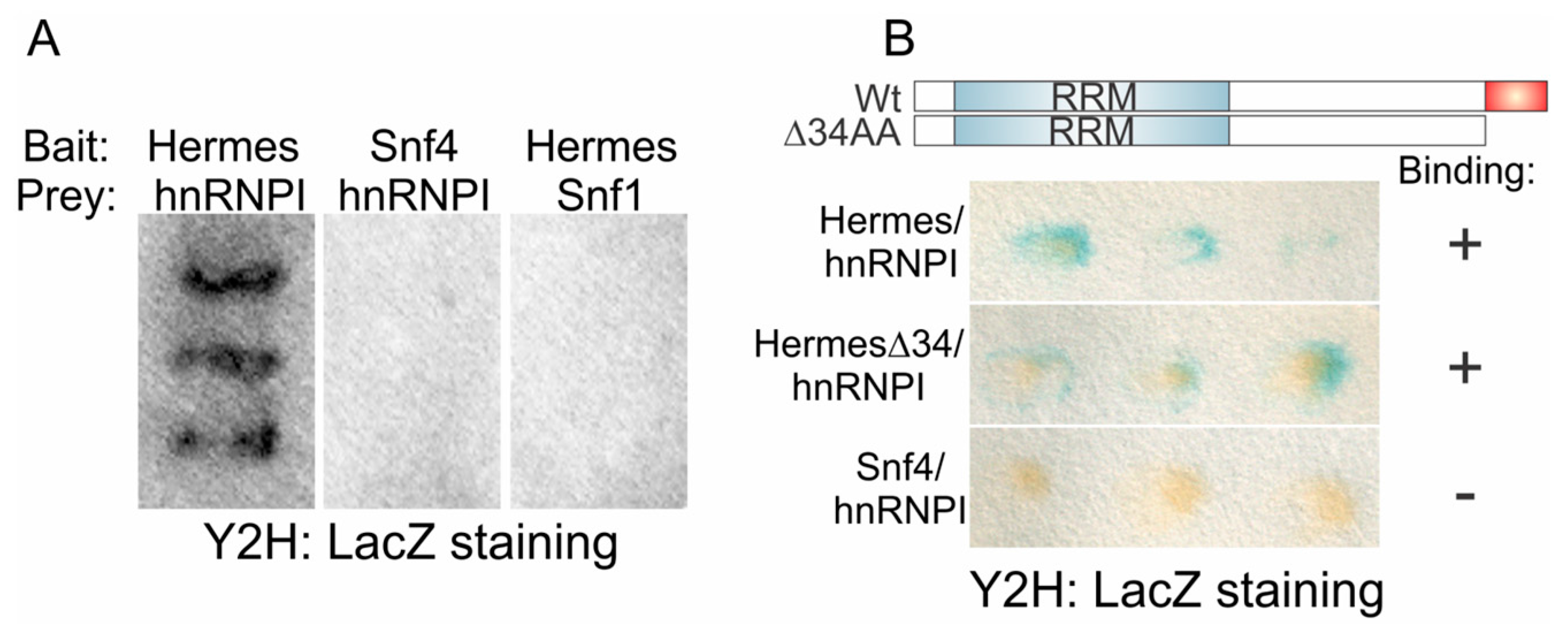
3.4. Hermes/Rbpms Interacts with hnRNP I
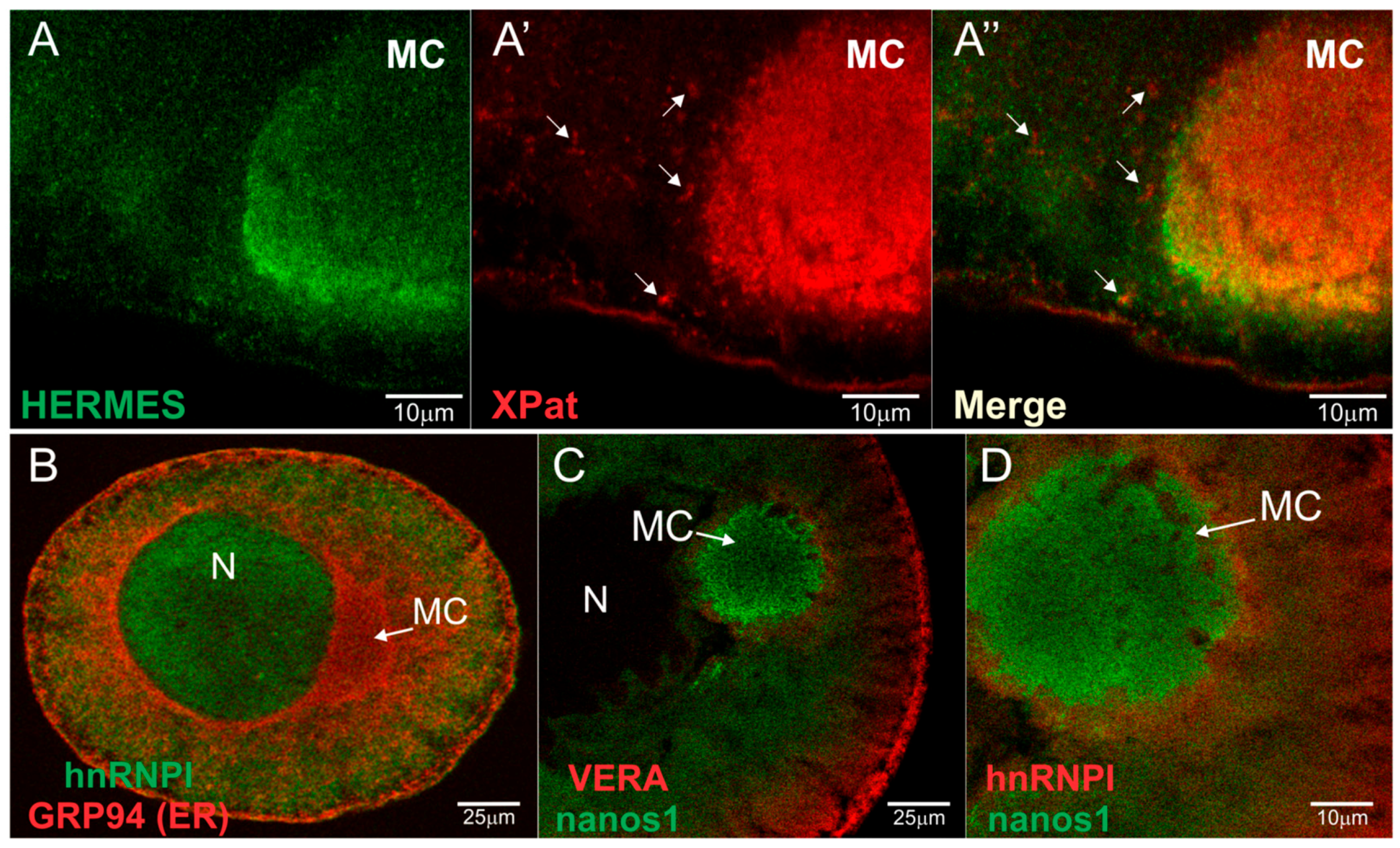
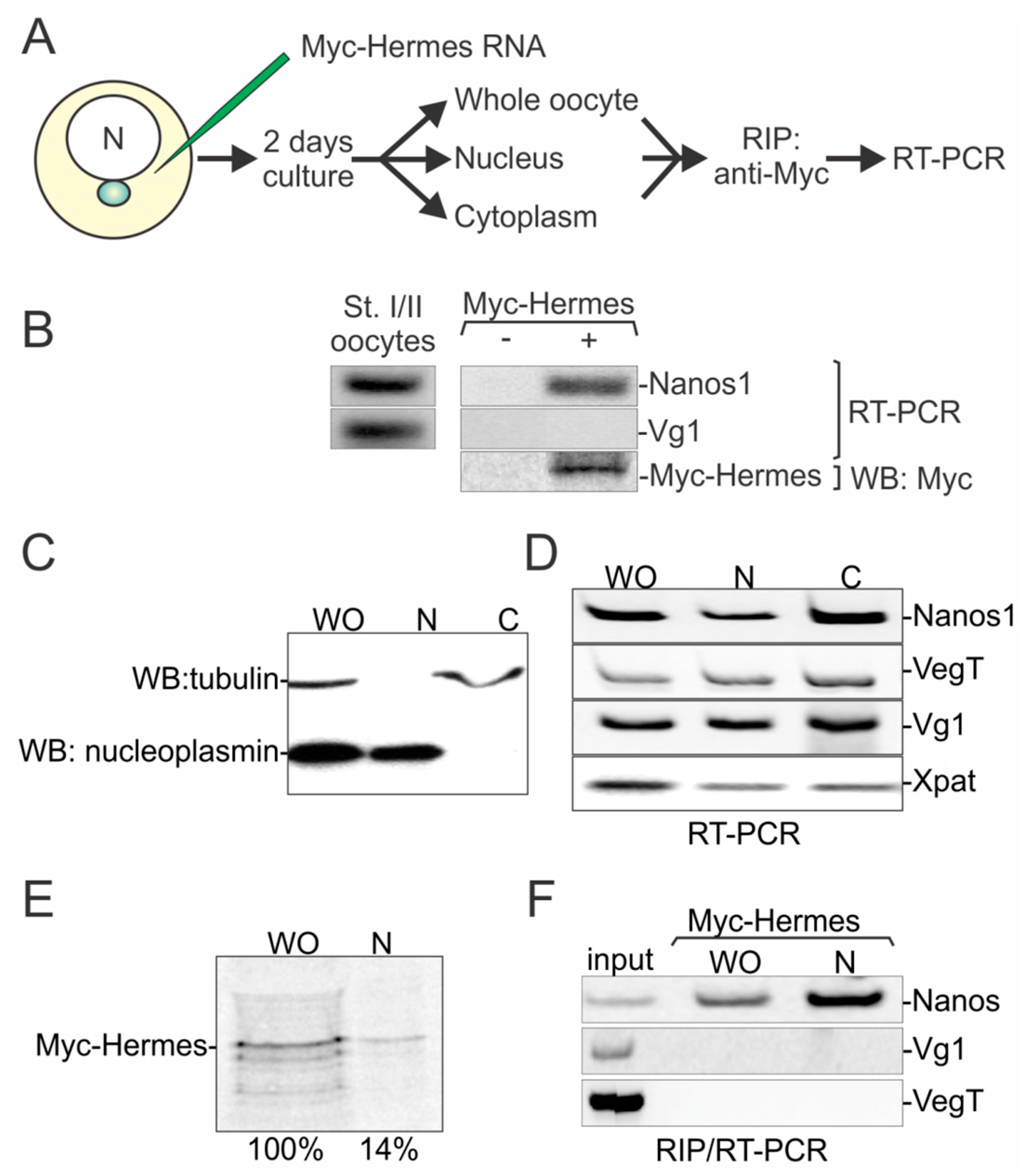
3.5. Hermes/Rbpms Associates with Nanos RNA within the Nucleus of Stage I Oocytes
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heasman, J.; Quarmby, J.; Wylie, C.C. The mitochondrial cloud of Xenopus oocytes: The source of germinal granule material. Dev. Biol. 1984, 105, 458–469. [Google Scholar] [CrossRef]
- Gard, D.L.; Cha, B.J.; King, E. The organization and animal-vegetal asymmetry of cytokeratin filaments in stage VI Xenopus oocytes is dependent upon F-actin and microtubules. Dev. Biol. 1997, 184, 95–114. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kloc, M.; Dougherty, M.T.; Bilinski, S.; Chan, A.P.; Brey, E.; King, M.L.; Patrick, C.W., Jr.; Etkin, L.D. Three-dimensional ultrastructural analysis of RNA distribution within germinal granules of Xenopus. Dev. Biol. 2002, 241, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; King, M.L. Localization of Xcat-2 RNA, a putative germ plasm component, to the mitochondrial cloud in Xenopus stage I oocytes. Development 1996, 122, 2947–2953. [Google Scholar] [PubMed]
- Kloc, M.; Bilinski, S.; Dougherty, M.T.; Brey, E.M.; Etkin, L.D. Formation, architecture and polarity of female germline cyst in Xenopus. Dev. Biol. 2004, 266, 43–61. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.; Torres, J.; Lewis, R.A.; Mowry, K.L.; Houliston, E.; King, M.L. Localization of RNAs to the mitochondrial cloud in Xenopus oocytes through entrapment and association with endoplasmic reticulum. Mol. Biol. Cell 2004, 15, 4669–4681. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, B.; Deshler, J.O. RNA localization to the Balbiani body in Xenopus oocytes is regulated by the energy state of the cell and is facilitated by kinesin II. RNA 2009, 15, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Wilk, K.; Bilinski, S.; Dougherty, M.T.; Kloc, M. Delivery of germinal granules and localized RNAs via the messenger transport organizer pathway to the vegetal cortex of Xenopus oocytes occurs through directional expansion of the mitochondrial cloud. Int. J. Dev. Biol. 2005, 49, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Tada, H.; Mochii, M.; Orii, H.; Watanabe, K. Ectopic formation of primordial germ cells by transplantation of the germ plasm: Direct evidence for germ cell determinant in Xenopus. Dev. Biol. 2012, 371, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Kloc, M.; Bilinski, S.; Chan, A.P.; Allen, L.H.; Zearfoss, N.R.; Etkin, L.D. RNA localization and germ cell determination in Xenopus. Int. Rev. Cytol. 2001, 203, 63–91. [Google Scholar] [PubMed]
- Houston, D.W.; King, M.L. Germ plasm and molecular determinants of germ cell fate. Curr. Top. Dev. Biol. 2000, 50, 155–181. [Google Scholar] [PubMed]
- Houston, D.W.; King, M.L. A critical role for Xdazl, a germ plasm-localized RNA, in the differentiation of primordial germ cells in Xenopus. Development 2000, 127, 447–456. [Google Scholar] [PubMed]
- Lai, F.; Zhou, Y.; Luo, X.; Fox, J.; King, M.L. Nanos1 functions as a translational repressor in the Xenopus germline. Mech. Dev. 2011, 128, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.; Singh, A.; King, M.L. Xenopus Nanos1 is required to prevent endoderm gene expression and apoptosis in primordial germ cells. Development 2012, 139, 1476–1486. [Google Scholar] [CrossRef] [PubMed]
- Forbes, A.; Lehmann, R. Nanos and Pumilio have critical roles in the development and function of Drosophila germline stem cells. Development 1998, 125, 679–690. [Google Scholar] [PubMed]
- Kobayashi, S.; Yamada, M.; Asaoka, M.; Kitamura, T. Essential role of the posterior morphogen nanos for germline development in Drosophila. Nature 1996, 380, 708–711. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, M.; Sasaoka, Y.; Kiso, M.; Abe, K.; Haraguchi, S.; Kobayashi, S.; Saga, Y. Conserved role of nanos proteins in germ cell development. Science 2003, 301, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lin, H. Nanos maintains germline stem cell self-renewal by preventing differentiation. Science 2004, 303, 2016–2019. [Google Scholar] [CrossRef] [PubMed]
- Yisraeli, J.K.; Sokol, S.; Melton, D.A. A two-step model for the localization of maternal mRNA in Xenopus oocytes: Involvement of microtubules and microfilaments in the translocation and anchoring of Vg1 mRNA. Development 1990, 108, 289–298. [Google Scholar] [PubMed]
- Betley, J.N.; Heinrich, B.; Vernos, I.; Sardet, C.; Prodon, F.; Deshler, J.O. Kinesin II mediates Vg1 mRNA transport in Xenopus oocytes. Curr. Biol. 2004, 14, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.J.; Mowry, K.L. Xenopus Staufen is a component of a ribonucleoprotein complex containing Vg1 RNA and kinesin. Development 2004, 131, 3035–3045. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Houston, D.W.; King, M.L.; Payne, C.; Wylie, C.; Heasman, J. The role of maternal VegT in establishing the primary germ layers in Xenopus embryos. Cell 1998, 94, 515–524. [Google Scholar] [CrossRef]
- Joseph, E.M.; Melton, D.A. Mutant Vg1 ligands disrupt endoderm and mesoderm formation in Xenopus embryos. Development 1998, 125, 2677–2685. [Google Scholar] [PubMed]
- Chan, A.P.; Kloc, M.; Etkin, L.D. fatvg encodes a new localized RNA that uses a 25-nucleotide element (FVLE1) to localize to the vegetal cortex of Xenopus oocytes. Development 1999, 126, 4943–4953. [Google Scholar] [PubMed]
- Zearfoss, N.R.; Chan, A.P.; Wu, C.F.; Kloc, M.; Etkin, L.D. Hermes is a localized factor regulating cleavage of vegetal blastomeres in Xenopus laevis. Dev. Biol. 2004, 267, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Claussen, M.; Horvay, K.; Pieler, T. Evidence for overlapping, but not identical, protein machineries operating in vegetal RNA localization along early and late pathways in Xenopus oocytes. Development 2004, 131, 4263–4273. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kloc, M.; Zearfoss, N.R.; Etkin, L.D. Mechanisms of subcellular mRNA localization. Cell 2002, 108, 533–544. [Google Scholar] [CrossRef]
- King, M.L.; Messitt, T.J.; Mowry, K.L. Putting RNAs in the right place at the right time: RNA localization in the frog oocyte. Biol. Cell 2005, 97, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Betley, J.N.; Frith, M.C.; Graber, J.H.; Choo, S.; Deshler, J.O. A ubiquitous and conserved signal for RNA localization in chordates. Curr. Biol. 2002, 12, 1756–1761. [Google Scholar] [CrossRef]
- Bubunenko, M.; Kress, T.L.; Vempati, U.D.; Mowry, K.L.; King, M.L. A consensus RNA signal that directs germ layer determinants to the vegetal cortex of Xenopus oocytes. Dev. Biol. 2002, 248, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.A.; Kress, T.L.; Cote, C.A.; Gautreau, D.; Rokop, M.E.; Mowry, K.L. Conserved and clustered RNA recognition sequences are a critical feature of signals directing RNA localization in Xenopus oocytes. Mech. Dev. 2004, 121, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Mowry, K.L. Complex formation between stage-specific oocyte factors and a Xenopus mRNA localization element. Proc. Natl. Acad. Sci. USA 1996, 93, 14608–14613. [Google Scholar] [CrossRef] [PubMed]
- Deshler, J.O.; Highett, M.I.; Schnapp, B.J. Localization of Xenopus Vg1 mRNA by Vera protein and the endoplasmic reticulum. Science 1997, 276, 1128–1131. [Google Scholar] [CrossRef] [PubMed]
- Gautreau, D.; Cote, C.A.; Mowry, K.L. Two copies of a subelement from the Vg1 RNA localization sequence are sufficient to direct vegetal localization in Xenopus oocytes. Development 1997, 124, 5013–5020. [Google Scholar] [PubMed]
- Deshler, J.O.; Highett, M.I.; Abramson, T.; Schnapp, B.J. A highly conserved RNA-binding protein for cytoplasmic mRNA localization in vertebrates. Curr. Biol. 1998, 8, 489–496. [Google Scholar] [CrossRef]
- Schwartz, S.P.; Aisenthal, L.; Elisha, Z.; Oberman, F.; Yisraeli, J.K. A 69-kDa RNA-binding protein from Xenopus oocytes recognizes a common motif in two vegetally localized maternal mRNAs. Proc. Natl. Acad. Sci. USA 1992, 89, 11895–11899. [Google Scholar] [CrossRef] [PubMed]
- Elisha, Z.; Havin, L.; Ringel, I.; Yisraeli, J.K. Vg1 RNA binding protein mediates the association of Vg1 RNA with microtubules in Xenopus oocytes. EMBO J. 1995, 14, 5109–5114. [Google Scholar] [PubMed]
- Kwon, S.; Abramson, T.; Munro, T.P.; John, C.M.; Kohrmann, M.; Schnapp, B.J. UUCAC- and vera-dependent localization of VegT RNA in Xenopus oocytes. Curr. Biol. 2002, 12, 558–564. [Google Scholar] [CrossRef]
- Cote, C.A.; Gautreau, D.; Denegre, J.M.; Kress, T.L.; Terry, N.A.; Mowry, K.L. A Xenopus protein related to hnRNP I has a role in cytoplasmic RNA localization. Mol. Cell 1999, 4, 431–437. [Google Scholar] [CrossRef]
- Wilhelm, J.E.; Hilton, M.; Amos, Q.; Henzel, W.J. Cup is an eIF4E binding protein required for both the translational repression of oskar and the recruitment of Barentsz. J. Cell Biol. 2003, 163, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.M.; Jiang, C.; Kroll, T.T.; Huber, P.W. A proline-rich protein binds to the localization element of Xenopus Vg1 mRNA and to ligands involved in actin polymerization. EMBO J. 2001, 20, 2315–2325. [Google Scholar] [CrossRef] [PubMed]
- Kress, T.L.; Yoon, Y.J.; Mowry, K.L. Nuclear RNP complex assembly initiates cytoplasmic RNA localization. J. Cell Biol. 2004, 165, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Sato, K.; Hanyu-Nakamura, K. Drosophila cup is an eIF4E binding protein that associates with Bruno and regulates oskar mRNA translation in oogenesis. Dev. Cell 2004, 6, 69–78. [Google Scholar] [CrossRef]
- Farina, K.L.; Singer, R.H. The nuclear connection in RNA transport and localization. Trends Cell Biol. 2002, 12, 466–472. [Google Scholar] [CrossRef]
- Kloc, M.; Bilinski, S.; Pui-Yee Chan, A.; Etkin, L.D. The targeting of Xcat2 mRNA to the germinal granules depends on a cis-acting germinal granule localization element within the 3′UTR. Dev. Biol. 2000, 217, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; King, M.L. RNA transport to the vegetal cortex of Xenopus oocytes. Dev. Biol. 1996, 179, 173–183. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gerber, W.V.; Yatskievych, T.A.; Antin, P.B.; Correia, K.M.; Conlon, R.A.; Krieg, P.A. The RNA-binding protein gene, hermes, is expressed at high levels in the developing heart. Mech. Dev. 1999, 80, 77–86. [Google Scholar] [CrossRef]
- Gerber, W.V.; Vokes, S.A.; Zearfoss, N.R.; Krieg, P.A. A role for the RNA-binding protein, hermes, in the regulation of heart development. Dev. Biol. 2002, 247, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Song, H.W.; Cauffman, K.; Chan, A.P.; Zhou, Y.; King, M.L.; Etkin, L.D.; Kloc, M. Hermes RNA-binding protein targets RNAs-encoding proteins involved in meiotic maturation, early cleavage, and germline development. Differentiation 2007, 75, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Gietz, R.D.; Schiestl, R.H. High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat. Protoc. 2007, 2, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Beckhelling, C.; Chang, P.; Chevalier, S.; Ford, C.; Houliston, E. Pre-M phase-promoting factor associates with annulate lamellae in Xenopus oocytes and egg extracts. Mol. Biol. Cell 2003, 14, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- Farazi, T.A.; Leonhardt, C.S.; Mukherjee, N.; Mihailovic, A.; Li, S.; Max, K.E.; Meyer, C.; Yamaji, M.; Cekan, P.; Jacobs, N.C.; et al. Identification of the RNA recognition element of the RBPMS family of RNA-binding proteins and their transcriptome-wide mRNA targets. RNA 2014, 20, 1090–1102. [Google Scholar] [CrossRef] [PubMed]
- Nijjar, S.; Woodland, H.R. Protein interactions in Xenopus germ plasm RNP particles. PLoS ONE 2013, 8, e80077. [Google Scholar] [CrossRef] [PubMed]
- Voeltz, G.K.; Ongkasuwan, J.; Standart, N.; Steitz, J.A. A novel embryonic poly (A) binding protein, ePAB, regulates mRNA deadenylation in Xenopus egg extracts. Genes Dev. 2001, 15, 774–788. [Google Scholar] [CrossRef] [PubMed]
- Caputi, M.; Mayeda, A.; Krainer, A.R.; Zahler, A.M. hnRNP A/B proteins are required for inhibition of HIV-1 pre-mRNA splicing. EMBO J. 1999, 18, 4060–4067. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; King, M.L. Sending RNAs into the future: RNA localization and germ cell fate. IUBMB Life 2004, 56, 19–27. [Google Scholar] [PubMed]
- Sonoda, J.; Wharton, R.P. Recruitment of Nanos to hunchback mRNA by Pumilio. Genes Dev. 1999, 13, 2704–2712. [Google Scholar] [CrossRef] [PubMed]
- Machado, R.J.; Moore, W.; Hames, R.; Houliston, E.; Chang, P.; King, M.L.; Woodland, H.R. Xenopus Xpat protein is a major component of germ plasm and may function in its organisation and positioning. Dev. Biol. 2005, 287, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Nijjar, S.; Woodland, H.R. Localisation of RNAs into the germ plasm of vitellogenic Xenopus oocytes. PLoS ONE 2013, 8, e61847. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.A.; Gagnon, J.A.; Mowry, K.L. PTB/hnRNP I is required for RNP remodeling during RNA localization in Xenopus oocytes. Mol. Cell. Biol. 2008, 28, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Bontems, F.; Stein, A.; Marlow, F.; Lyautey, J.; Gupta, T.; Mullins, M.C.; Dosch, R. Bucky ball organizes germ plasm assembly in zebrafish. Curr. Biol. 2009, 19, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Hornberg, H.; van Wollerton Horck, F.; Maurus, D.; Zwart, M.; Svoboda, H.; Harris, W.A.; Holt, C.E. RNA-binding protein Hermes/RBPMS inversely affects synapse density and axon arbor formation in retinal ganglion cells in vivo. J. Neurosci. 2013, 33, 10384–10395. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, M.T.; Sakamoto, H.; Inoue, K. Interaction and colocalization of HERMES/RBPMS with NonO, PSF, and G3BP1 in neuronal cytoplasmic RNP granules in mouse retinal line cells. Genes Cells 2015, 20, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Cheng, L.; Wang, Y.; Yuan, P.; Xu, X.; Ding, L.; Zhang, H.; Jiang, K.; Song, H.; Chen, Z.; et al. The RNA-binding protein RBPMS1 represses AP-1 signaling and regulates breast cancer cell proliferation and migration. Biochim. Biophys. Acta 2015, 1853, 1–13. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, H.; Bubunenko, M.; Houston, D.W.; King, M.L. Xcat2 RNA is a translationally sequestered germ plasm component in Xenopus. Mech. Dev. 1999, 84, 75–88. [Google Scholar] [CrossRef]
- Luo, X.; Nerlick, S.; An, W.; King, M.L. Xenopus germline nanos1 is translationally repressed by a novel structure-based mechanism. Development 2011, 138, 589–598. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguero, T.; Zhou, Y.; Kloc, M.; Chang, P.; Houliston, E.; King, M.L. Hermes (Rbpms) is a Critical Component of RNP Complexes that Sequester Germline RNAs during Oogenesis. J. Dev. Biol. 2016, 4, 2. https://doi.org/10.3390/jdb4010002
Aguero T, Zhou Y, Kloc M, Chang P, Houliston E, King ML. Hermes (Rbpms) is a Critical Component of RNP Complexes that Sequester Germline RNAs during Oogenesis. Journal of Developmental Biology. 2016; 4(1):2. https://doi.org/10.3390/jdb4010002
Chicago/Turabian StyleAguero, Tristan, Yi Zhou, Malgorzata Kloc, Patrick Chang, Evelyn Houliston, and Mary Lou King. 2016. "Hermes (Rbpms) is a Critical Component of RNP Complexes that Sequester Germline RNAs during Oogenesis" Journal of Developmental Biology 4, no. 1: 2. https://doi.org/10.3390/jdb4010002
APA StyleAguero, T., Zhou, Y., Kloc, M., Chang, P., Houliston, E., & King, M. L. (2016). Hermes (Rbpms) is a Critical Component of RNP Complexes that Sequester Germline RNAs during Oogenesis. Journal of Developmental Biology, 4(1), 2. https://doi.org/10.3390/jdb4010002






