YKL-40, Soluble IL-2 Receptor, Angiotensin Converting Enzyme and C-Reactive Protein: Comparison of Markers of Sarcoidosis Activity
Abstract
1. Introduction
2. Materials and Methods
- In treatment: Patients who started corticosteroid therapy after diagnosis.
- Spontaneous Remission: Spontaneous clinical and/or radiological improvement.
- Treatment-related remission: Clinical/radiological improvement following cessation of corticosteroid therapy.
- Chronic disease: if the disease is not more than two years.
2.1. Sample Collection
2.2. Assay of Biochemical Parameters
2.3. Statistical Analysis
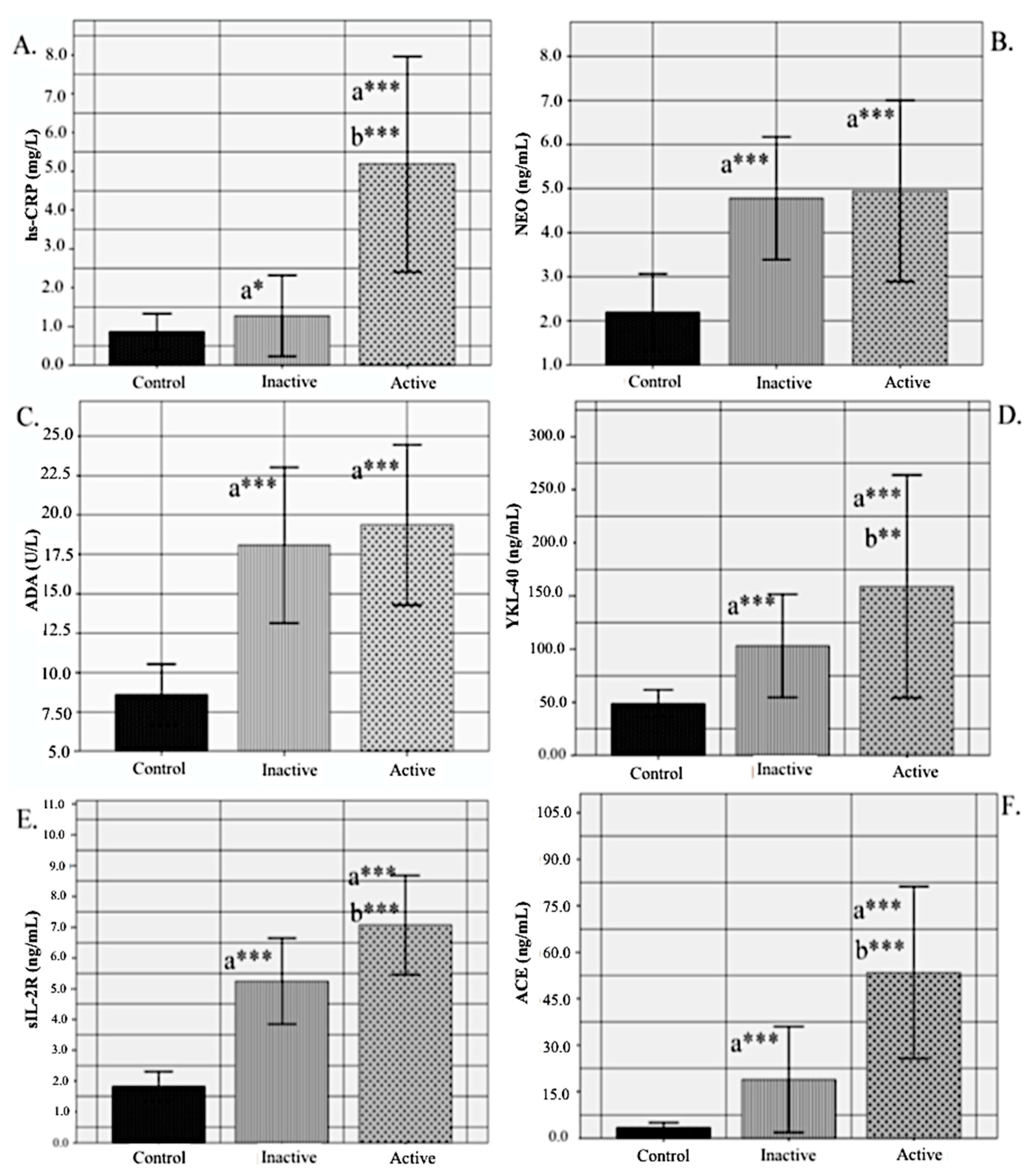
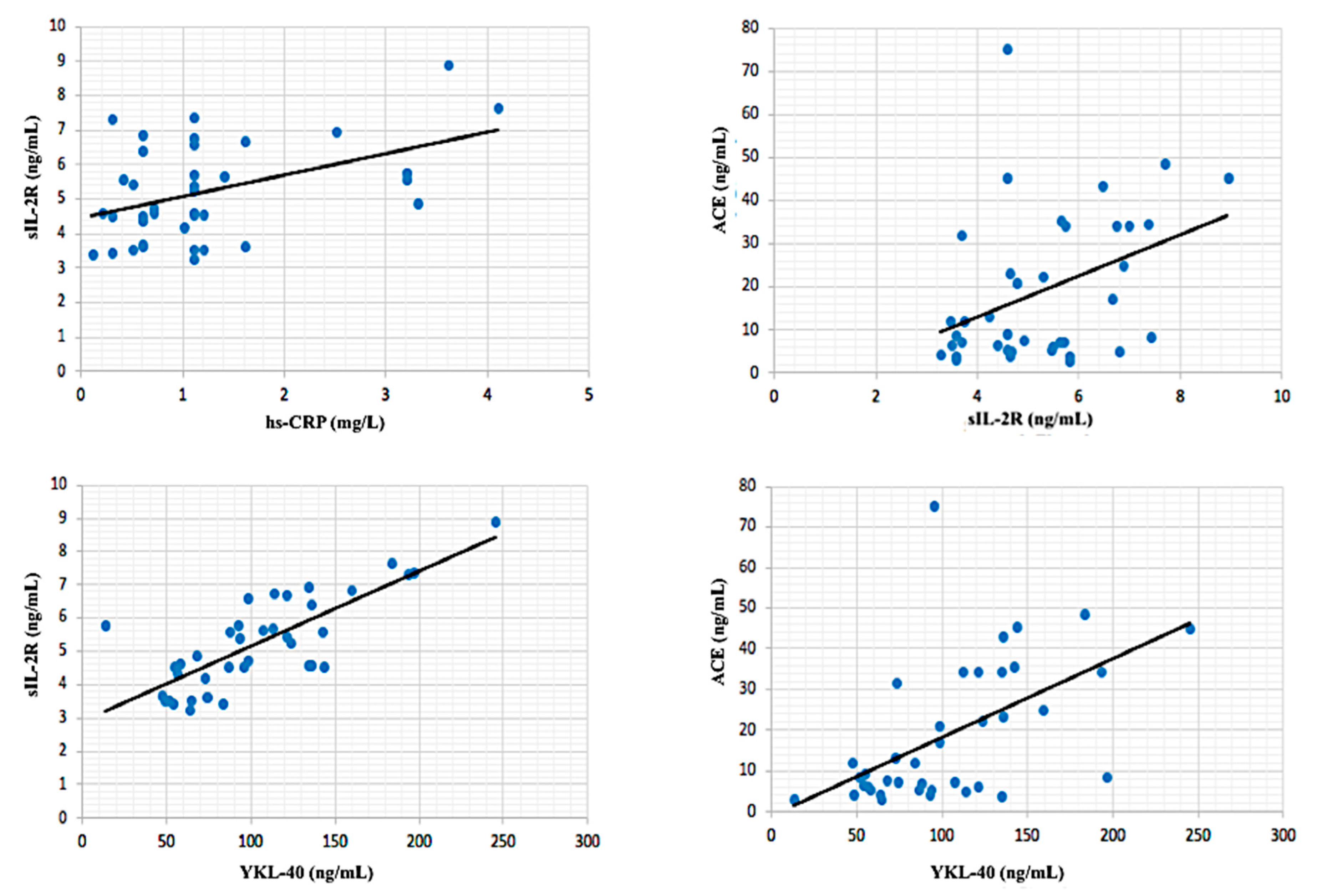
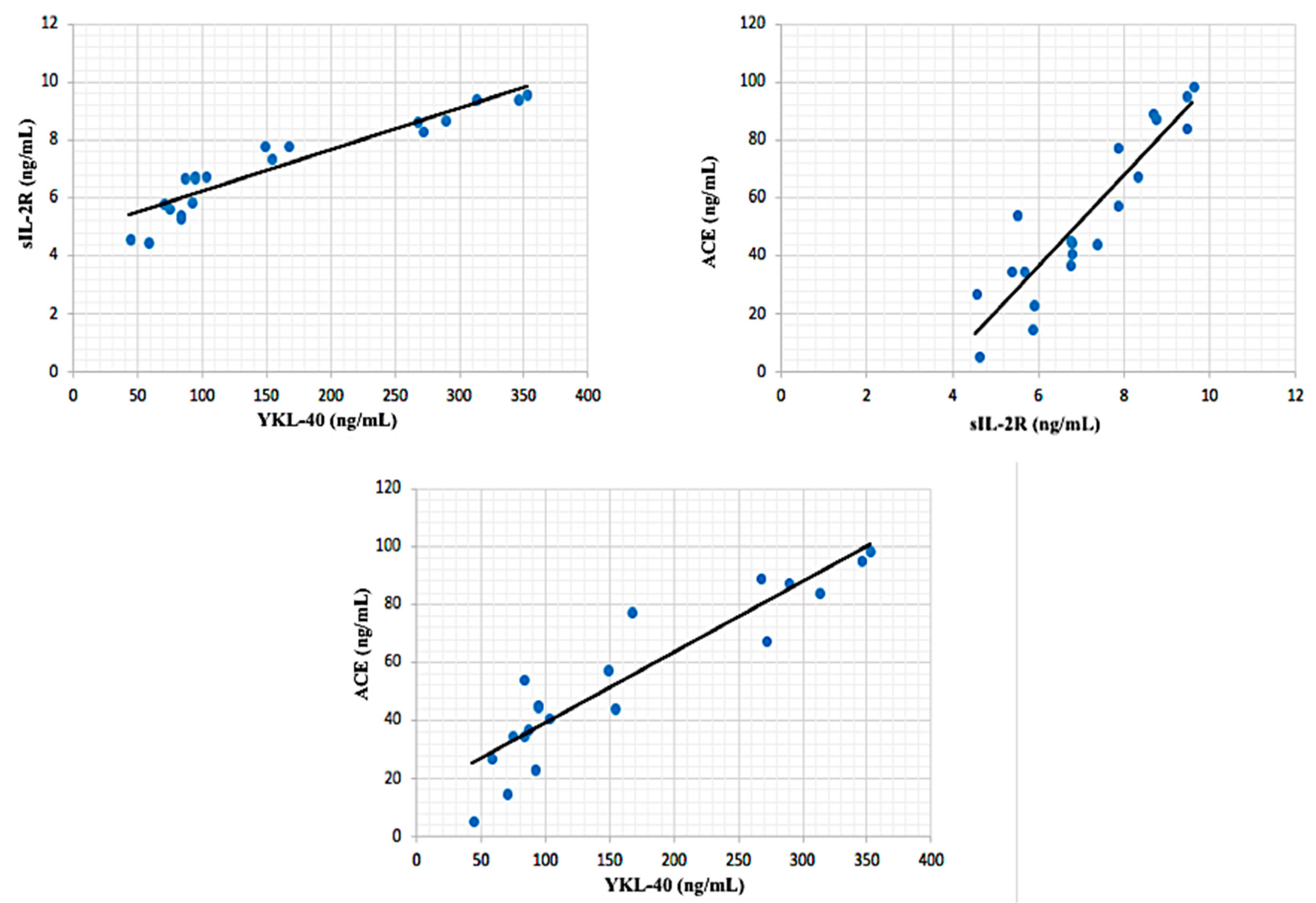
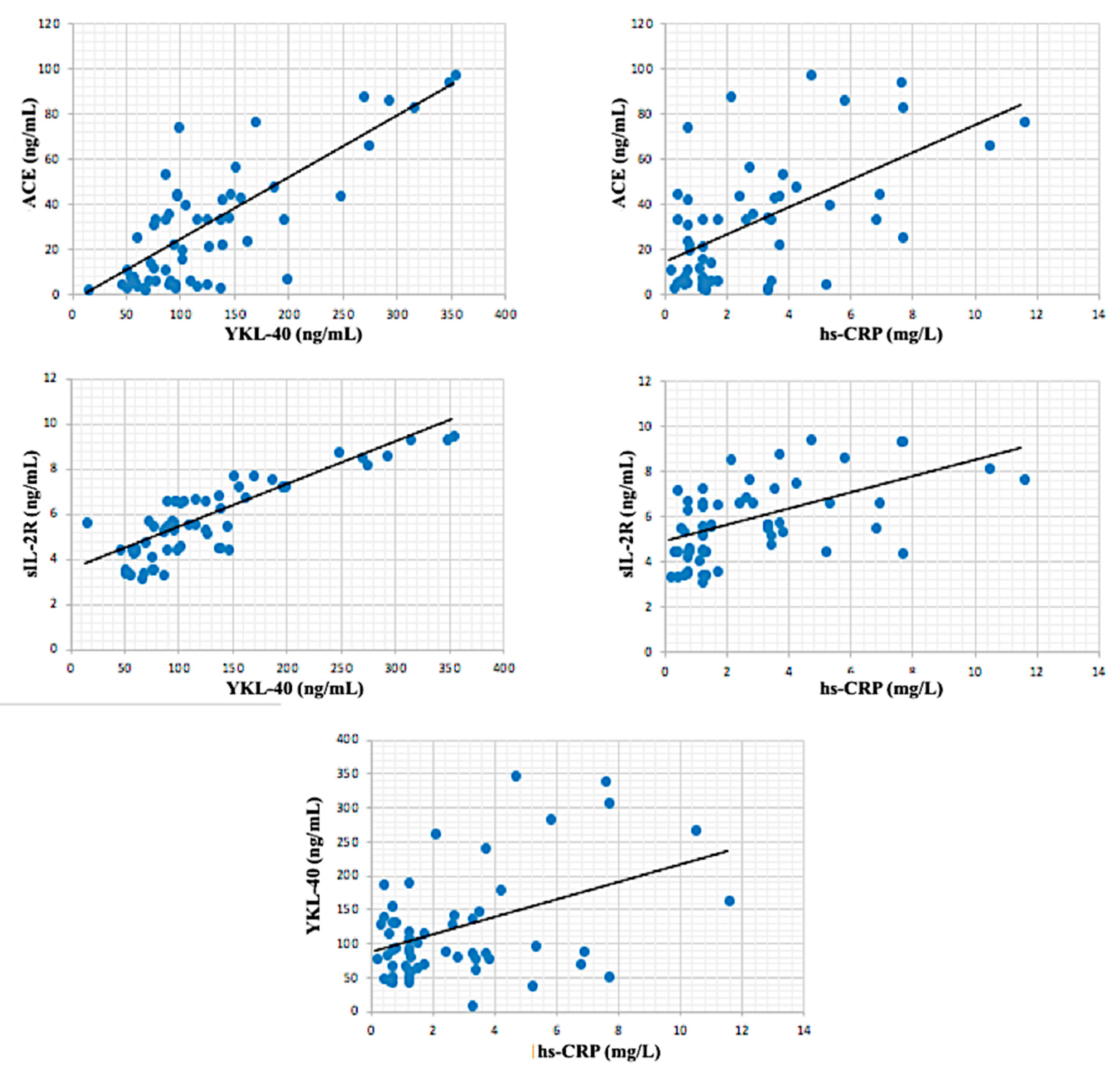
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Pierce, T.B.; Margolis, M.; Razzuk, M.A. Sarcoidosis: Still a mystery? Proc. (Bayl. Univ. Med. Cent.) 2001, 14, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Gundlach, E.; Hoffmann, M.M.; Prasse, A.; Heinzelmann, S.; Ness, T. Interleukin-2 Receptor and Angiotensin-Converting Enzyme as Markers for Ocular Sarcoidosis. PLoS ONE 2016, 11, e0147258. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A. Serum adenosine deaminase activity is increased in sarcoidosis. Clin. Chem. 1984, 30, 499. [Google Scholar] [PubMed]
- Ahmadzai, H.; Loke, W.S.J.; Huang, S.; Herbert, C.; Wakefield, D.; Thomas, P. Biomarkers in sarcoidosis: A review. Curr. Biomark. Find. 2014, 4, 93. [Google Scholar] [CrossRef]
- Rothkrantz-Kos, S.; van Dieijen-Visser, M.P.; Mulder, P.G.H.; Drent, M. Potential usefulness of inflammatory markers to monitor respiratory functional impairment in sarcoidosis. Clin. Chem. 2003, 49, 1510–1517. [Google Scholar] [CrossRef] [PubMed]
- Johansen, J.S.; Milman, N.; Hansen, M.; Garbarsch, C.; Price, P.A.; Graudal, N. Increased serum YKL-40 in patients with pulmonary sarcoidosis—A potential marker of disease activity? Respir. Med. 2005, 99, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Ziegenhagen, M.W.; Rothe, M.E.; Schlaak, M.; Müller-Quernheim, J. Bronchoalveolar and serological parameters reflecting the severity of sarcoidosis. Eur. Respir. J. 2003, 21, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Hunninghake, G.W.; Costabel, U.; Ando, M.; Baughman, R.; Cordier, J.F.; du Bois, R.; Eklund, A.; Kitaichi, M.; Lynch, J.; Rizzato, G.; et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc. Diffus. Lung Dis. Off. J. WASOG 1999, 16, 149–173. [Google Scholar]
- Grutters, J.C.; Fellrath, J.-M.; Mulder, L.; Janssen, R.; van den Bosch, J.M.M.; van Velzen-Blad, H. Serum Soluble Interleukin-2 Receptor Measurement in Patients With Sarcoidosis: A Clinical Evaluation. Chest 2003, 124, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; Kazerooni, E.A.; Gay, S.E. Pulmonary sarcoidosis. Clin. Chest Med. 1997, 18, 755–785. [Google Scholar] [CrossRef]
- Rust, M.; Bergmann, L.; Kühn, T.; Tuengerthal, S.; Bartmann, K.; Mitrou, P.S.; Meier-Sydow, J. Prognostic Value of Chest Radiograph, Serum-Angiotensin-Converting Enzyme and T Helper Cell Count in Blood and in Bronchoalveolar Lavage of Patients with Pulmonary Sarcoidosis. Respiration 1985, 48, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Drent, M.; Wirnsberger, R.; de Vries, J.; van Dieijen-Visser, M.; Wouters, E.; Schols, A. Association of fatigue with an acute phase response in sarcoidosis. Eur. Respir. J. 1999, 13, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Sobic-Saranovic, D.; Grozdic, I.; Videnovi-Ivanov, J.; Vucinic-Mihailovic, V.; Artiko, V.; Saranovic, D.; Djuric-Stefanovic, A.; Masulovic, D.; Odalovic, S.; Ilic-Dudvarski, A.; et al. The Utility of 18F-FDG PET/CT for Diagnosis and Adjustment of Therapy in Patients with Active Chronic Sarcoidosis. J. Nucl. Med. 2012, 53, 1543–1549. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.D.; Hunninghake, G.W. Current concepts of the pathogenesis of sarcoidosis. Am. Rev. Respir. Dis. 1987, 135, 747–760. [Google Scholar] [CrossRef] [PubMed]
- Gungor, S.; Ozseker, F.; Yalcinsoy, M.; Akkaya, E.; Can, G.; Eroglu, H.; Genc, N.S. Conventional markers in determination of activity of sarcoidosis. Int. Immunopharmacol. 2015, 25, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Prasse, A.; Katic, C.; Germann, M.; Buchwald, A.; Zissel, G.; Müller-Quernheim, J. Phenotyping Sarcoidosis from a Pulmonary Perspective. Am. J. Respir. Crit. Care Med. 2008, 177, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Tuleta, I.; Skowasch, D.; Biener, L.; Pizarro, C.; Schueler, R.; Nickenig, G.; Schahab, N.; Schaefer, C.; Pingel, S. Impaired Vascular Function in Sarcoidosis Patients. Adv. Exp. Med. Biol. 2017, 980, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ando, M.; Miyazaki, E.; Hatano, Y.; Nishio, S.; Torigoe, C.; Yamasue, M.; Miyazaki, E.; Kadota, J. Subcutaneous sarcoidosis: A clinical analysis of nine patients. Clin. Rheumatol. 2016, 35, 2277–2281. [Google Scholar] [CrossRef] [PubMed]
- Verwoerd, A.; Vorselaars, A.D.M.; van Moorsel, C.H.M.; Bos, W.J.W.; van Velzen-Blad, H.; Grutters, J.C. Discrepant elevation of sIL-2R levels in sarcoidosis patients with renal insufficiency. Eur. Respir. J. 2015, 46, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Zurkova, M.; Kolek, V.; Tomankova, T.; Kriegova, E. Extrapulmonary involvement in patients with sarcoidosis and comparison of routine laboratory and clinical data to pulmonary involvement. Biomed. Pap. 2014, 158, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, S.; Hamada, H.; Kadowaki, T.; Hamaguchi, N.; Ito, R.; Irifune, K.; Higaki, J. Comparative Evaluation of Serum Markers in Pulmonary Sarcoidosis. Chest 2010, 137, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Keijsers, R.G.; Verzijlbergen, F.J.; Oyen, W.J.; van den Bosch, J.M.; Ruven, H.J.; van Velzen-Blad, H.; Grutters, J.C. 18F-FDG PET, genotype-corrected ACE and sIL-2R in newly diagnosed sarcoidosis. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Ina, Y.; Takada, K.; Sato, T.; Yamamoto, M.; Noda, M.; Morishita, M. Soluble Interleukin 2 Receptors in Patients With Sarcoidosis. Possible origin. Chest 1992, 102, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, E.C.; Brousseau, K.P.; Berger, M.B.; Kurman, C.C.; Marcon, L.; Nelson, D.L. Elevated Concentrations of Soluble Interleukin-2 Receptors in Serum Samples and Bronchoalveolar Lavage Fluids in Active Sarcoidosis. Am. Rev. Respir. Dis. 1988, 137, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Bargagli, E.; Bianchi, N.; Margollicci, M.; Olivieri, C.; Luddi, A.; Coviello, G.; Grosso, S.; Rottoli, P. Chitotriosidase and soluble IL-2 receptor: Comparison of two markers of sarcoidosis severity. Scand. J. Clin. Lab. Investig. 2008, 68, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Su, R.; Nguyen, M.-L.T.; Agarwal, M.R.; Kirby, C.; Nguyen, C.P.; Ramstein, J.; Darnell, E.P.; Gomez, A.D.; Ho, M.; Woodruff, P.G.; et al. Interferon-inducible chemokines reflect severity and progression in sarcoidosis. Respir. Res. 2013, 14, 121. [Google Scholar] [CrossRef] [PubMed]
- Groen-Hakan, F.; Eurelings, L.; ten Berge, J.C.; van Laar, J.; Ramakers, C.R.B.; Dik, W.A.; Rothova, A. Diagnostic Value of Serum-Soluble Interleukin 2 Receptor Levels vs. Angiotensin-Converting Enzyme in Patients With Sarcoidosis-Associated Uveitis. JAMA Ophthalmol. 2017, 135, 1352. [Google Scholar] [CrossRef] [PubMed]
- Thi Hong Nguyen, C.; Kambe, N.; Kishimoto, I.; Ueda-Hayakawa, I.; Okamoto, H. Serum soluble interleukin-2 receptor level is more sensitive than angiotensin-converting enzyme or lysozyme for diagnosis of sarcoidosis and may be a marker of multiple organ involvement. J. Dermatol. 2017, 44, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Tanimura, H.; Mizuno, K.; Okamoto, H. Serum levels of soluble CD163 as a specific marker of macrophage/monocyte activity in sarcoidosis patients. Sarcoidosis Vasc. Diffus. Lung Dis. Off. J. WASOG 2015, 32, 99–105. [Google Scholar]
- Niederer, R.L.; Al-Janabi, A.; Lightman, S.L.; Tomkins-Netzer, O. Serum angiotensin converting enzyme (ACE) has a high negative predictive value in the investigation for systemic sarcoidosis. Am. J. Ophthalmol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sejdic, A.; Graudal, N.; Baslund, B. Clinical and biochemical presentation of sarcoidosis with high and normal serum angiotensin-converting enzyme. Scand. J. Rheumatol. 2018, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Mihailovic-Vucinic, V.; Popevic, L.; Popevic, S.; Stjepanovic, M.; Aleksic, A.; Stanojevic-Paovic, A. Utility of angiotensin-converting enzyme activity in aqueous humor in the diagnosis of ocular sarcoidosis. Indian J. Ophthalmol. 2017, 65, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Tetikkurt, C.; Yanardag, H.; Pehlivan, M.; Bilir, M. Clinical features and prognostic significance of splenic involvement in sarcoidosis. Monaldi Arch. Chest Dis. 2017, 87, 893. [Google Scholar] [CrossRef] [PubMed]
- Ivanišević, J.; Kotur-Stevuljević, J.; Stefanović, A.; Spasić, S.; Vučinić Mihailović, V.; Videnović Ivanov, J.; Jelić-Ivanović, Z. Association of serum amyloid A and oxidative stress with paraoxonase 1 in sarcoidosis patients. Eur. J. Clin. Investig. 2016, 46, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Yasar, Z.; Özgül, M.A.; Cetinkaya, E.; Kargi, A.; Gül, Ş.; Talay, F.; Tanriverdi, E.; Dincer, H.E. Angiotensin-converting Enzyme as a Predictor of Extrathoracic Involvement of Sarcoidosis. Sarcoidosis Vasc. Diffus. Lung Dis. Off. J. WASOG 2016, 32, 318–324. [Google Scholar]
- Kastrup, J. Can YKL-40 be a new inflammatory biomarker in cardiovascular disease? Immunobiology 2012, 217, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Kruit, A.; Grutters, J.C.; Ruven, H.J.T.; van Moorsel, C.C.M.; van den Bosch, J.M.M. A CHI3L1 gene polymorphism is associated with serum levels of YKL-40, a novel sarcoidosis marker. Respir. Med. 2007, 101, 1563–1571. [Google Scholar] [CrossRef] [PubMed]
- Korthagen, N.M.; van Moorsel, C.H.M.; Zanen, P.; Ruven, H.J.; Grutters, J.C. Evaluation of Circulating YKL-40 Levels in Idiopathic Interstitial Pneumonias. Lung 2014, 192, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, K.; Suda, T.; Nakamura, Y.; Inui, N.; Hashimoto, D.; Miwa, S.; Hayakawa, H.; Kusagaya, H.; Nakano, Y.; Nakamura, H.; et al. Increased expression of YKL-40, a chitinase-like protein, in serum and lung of patients with idiopathic pulmonary fibrosis. Respir. Med. 2010, 104, 1204–1210. [Google Scholar] [CrossRef] [PubMed]
- Long, X.; Costabel, U.; He, X.; Guzman, J.; Theegarten, D.; Bonella, F. Serum YKL-40 as a candidate biomarker for hypersensitivity pneumonitis. Pneumologie 2015, 69, V269. [Google Scholar] [CrossRef]

| Variables | Control (n = 25) | Inactive Sarcoidosis (n = 39) | Active Sarcoidosis (n = 20) |
|---|---|---|---|
| Age, mean ± SD | 46.12 ± 8.27 | 42.44 ± 10.25 | 45.15 ± 11.26 |
| Sex (M/F) | 12/13 | 15/24 | 4/16 a,* |
| Smoking (Y/N) | 0/20 | 7/32 a,* | 5/15 a,** |
| BMI | 26 ± 6 | 27 ± 5 | 25 ± 4 |
| Duration of disease | - | 7 (2–3) | 6 (4–22) |
| Disease stage (I/II/III/IV) | - | 8/20/8/3 | 4/12/3/1 * |
| Lymphocyte count (/mm3) | - | 1308 ± 406 | 1608 ± 106 * |
| AUC | 95% Confidence Interval | p | Sensitivity | Specificity | Cut-off | |
|---|---|---|---|---|---|---|
| hs-CRP (mg/L) | 0.721 | 0.614–0.828 | 0.001 | 0.508 | 0.92 | 1.35 |
| NEO (ng/mL) | 0.942 | 0.897–0.987 | 0.000 | 0.881 | 0.76 | 2.85 |
| YKL-40 (ng/mL) | 0.917 | 0.859–0.976 | 0.000 | 0.831 | 0.920 | 62.88 |
| ADA (U/L) | 0.983 | 0.962–1.00 | 0.000 | 0.949 | 0.92 | 11.35 |
| sIL-2R (ng/mL) | 1.000 | 1.00–1.00 | 0.000 | 1.00 | 1.00 | 2.97 |
| ACE (ng/mL) | 0.956 | 0.918–0.994 | 0.000 | 0.881 | 0.92 | 5.37 |
| AUC | 95% Confidence Interval | p | Sensitivity | Specificity | Cut-off | |
|---|---|---|---|---|---|---|
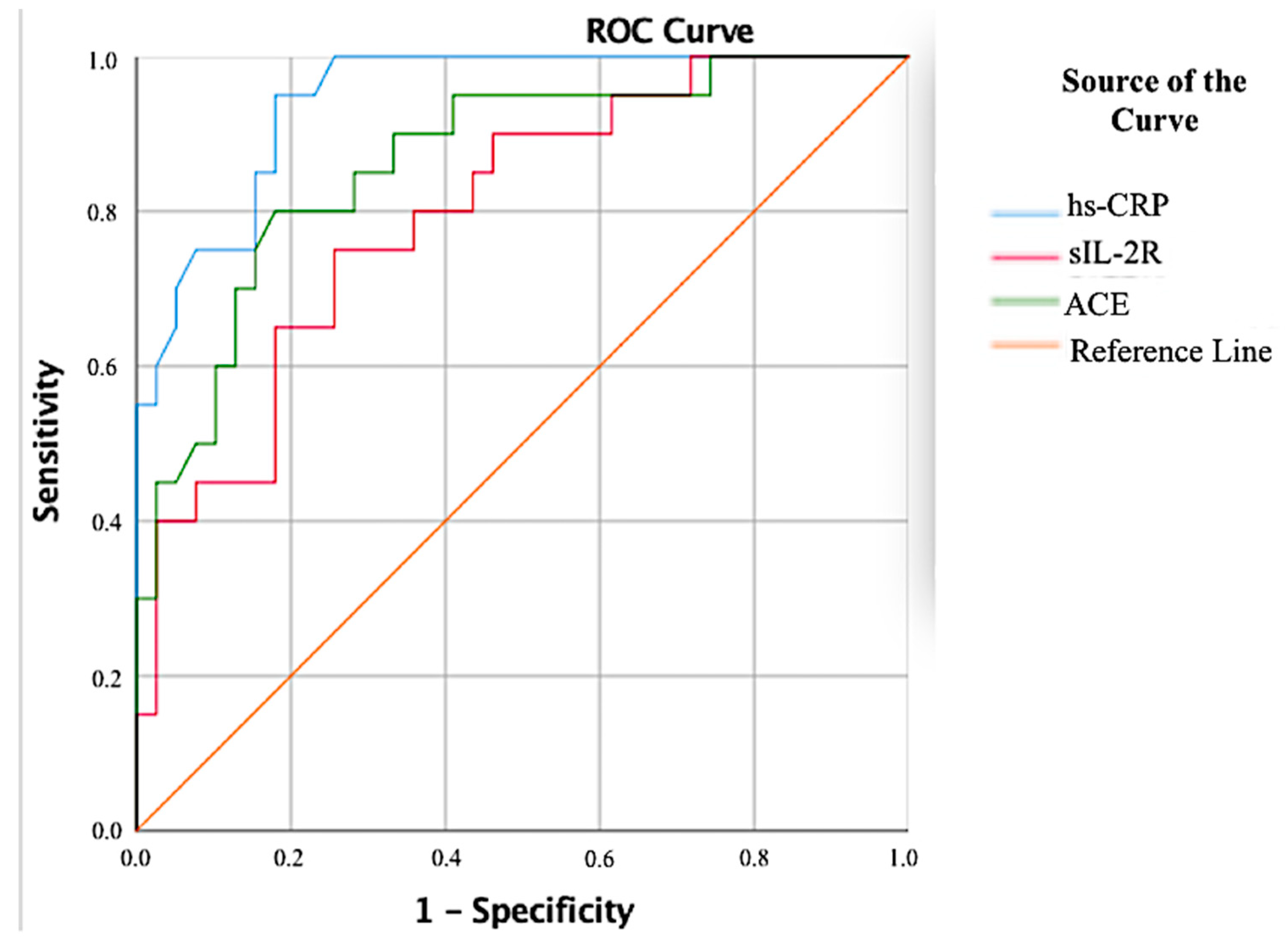
| hs-CRP (mg/L) | 0.946 | 0.894–0.997 | 0.000 | 0.950 | 0.821 | 1.8 |
| sIL-2R (ng/mL) | 0.799 | 0.682–0.916 | 0.000 | 0.750 | 0.744 | 5.810 |
| ACE (ng/mL) | 0.865 | 0.767–0.963 | 0.000 | 0.85 | 0.718 | 26.23 |
| OR | p | |
|---|---|---|
| hs-CRP (mg/L) | 5.162 | 0.002 |
| sIL-2R (ng/mL) | 0.786 | 0.617 |
| ACE (ng/mL) | 1.085 | 0.029 |
| OR | p | |
|---|---|---|
| hs-CRP (mg/L) | 47.225 | 0.001 |
| sIL-2R (ng/mL) | 4.029 | 0.158 |
| ACE (ng/mL) | 4.445 | 0.201 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uysal, P.; Durmus, S.; Sozer, V.; Gelisgen, R.; Seyhan, E.C.; Erdenen, F.; Simsek, G.; Uzun, H. YKL-40, Soluble IL-2 Receptor, Angiotensin Converting Enzyme and C-Reactive Protein: Comparison of Markers of Sarcoidosis Activity. Biomolecules 2018, 8, 84. https://doi.org/10.3390/biom8030084
Uysal P, Durmus S, Sozer V, Gelisgen R, Seyhan EC, Erdenen F, Simsek G, Uzun H. YKL-40, Soluble IL-2 Receptor, Angiotensin Converting Enzyme and C-Reactive Protein: Comparison of Markers of Sarcoidosis Activity. Biomolecules. 2018; 8(3):84. https://doi.org/10.3390/biom8030084
Chicago/Turabian StyleUysal, Pelin, Sinem Durmus, Volkan Sozer, Remise Gelisgen, Ekrem Cengiz Seyhan, Fusun Erdenen, Gonul Simsek, and Hafize Uzun. 2018. "YKL-40, Soluble IL-2 Receptor, Angiotensin Converting Enzyme and C-Reactive Protein: Comparison of Markers of Sarcoidosis Activity" Biomolecules 8, no. 3: 84. https://doi.org/10.3390/biom8030084
APA StyleUysal, P., Durmus, S., Sozer, V., Gelisgen, R., Seyhan, E. C., Erdenen, F., Simsek, G., & Uzun, H. (2018). YKL-40, Soluble IL-2 Receptor, Angiotensin Converting Enzyme and C-Reactive Protein: Comparison of Markers of Sarcoidosis Activity. Biomolecules, 8(3), 84. https://doi.org/10.3390/biom8030084





