Intra- and Extra-Cellular Events Related to Altered Glycosylation of MUC1 Promote Chronic Inflammation, Tumor Progression, Invasion, and Metastasis
Abstract
:1. Introduction
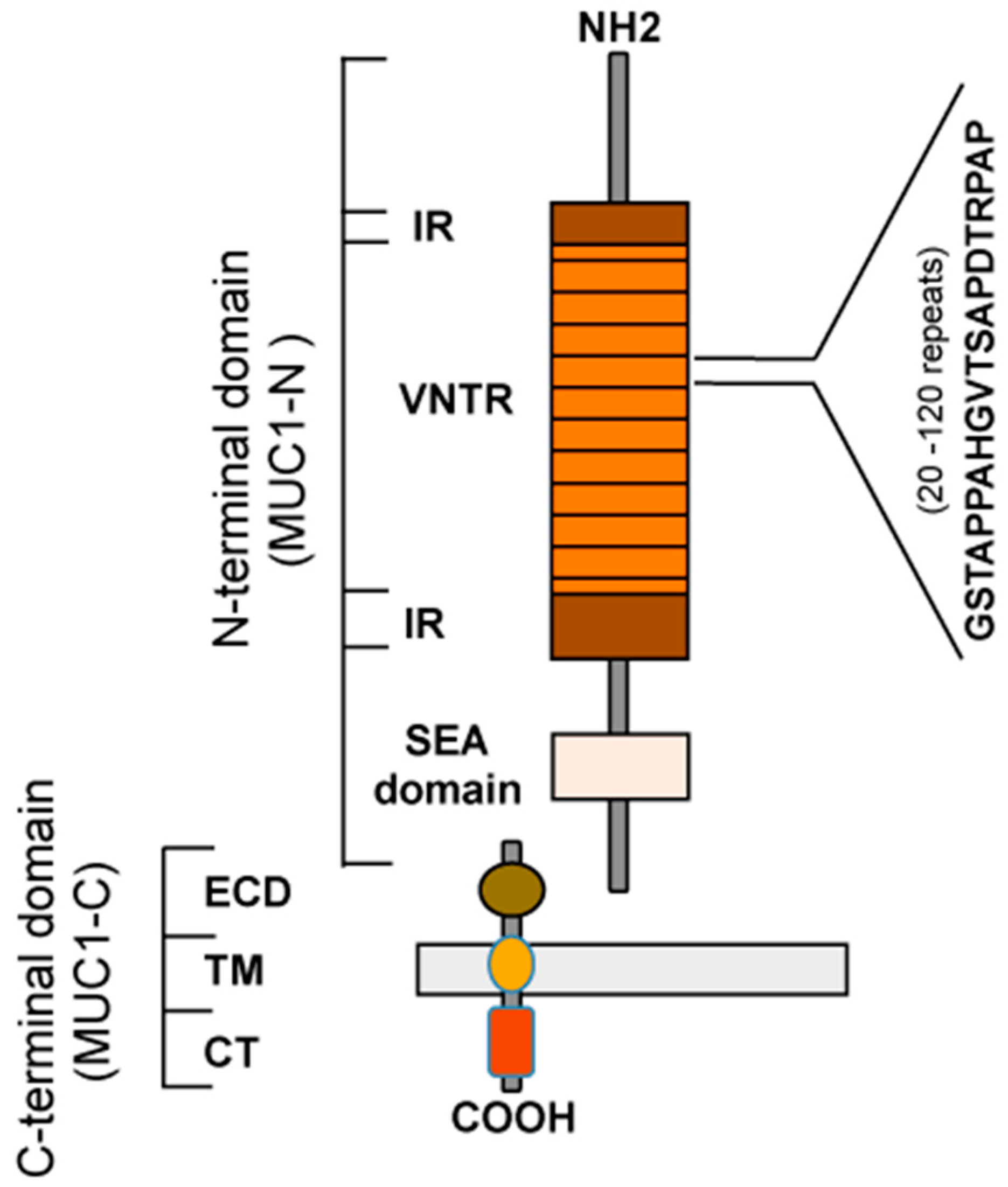
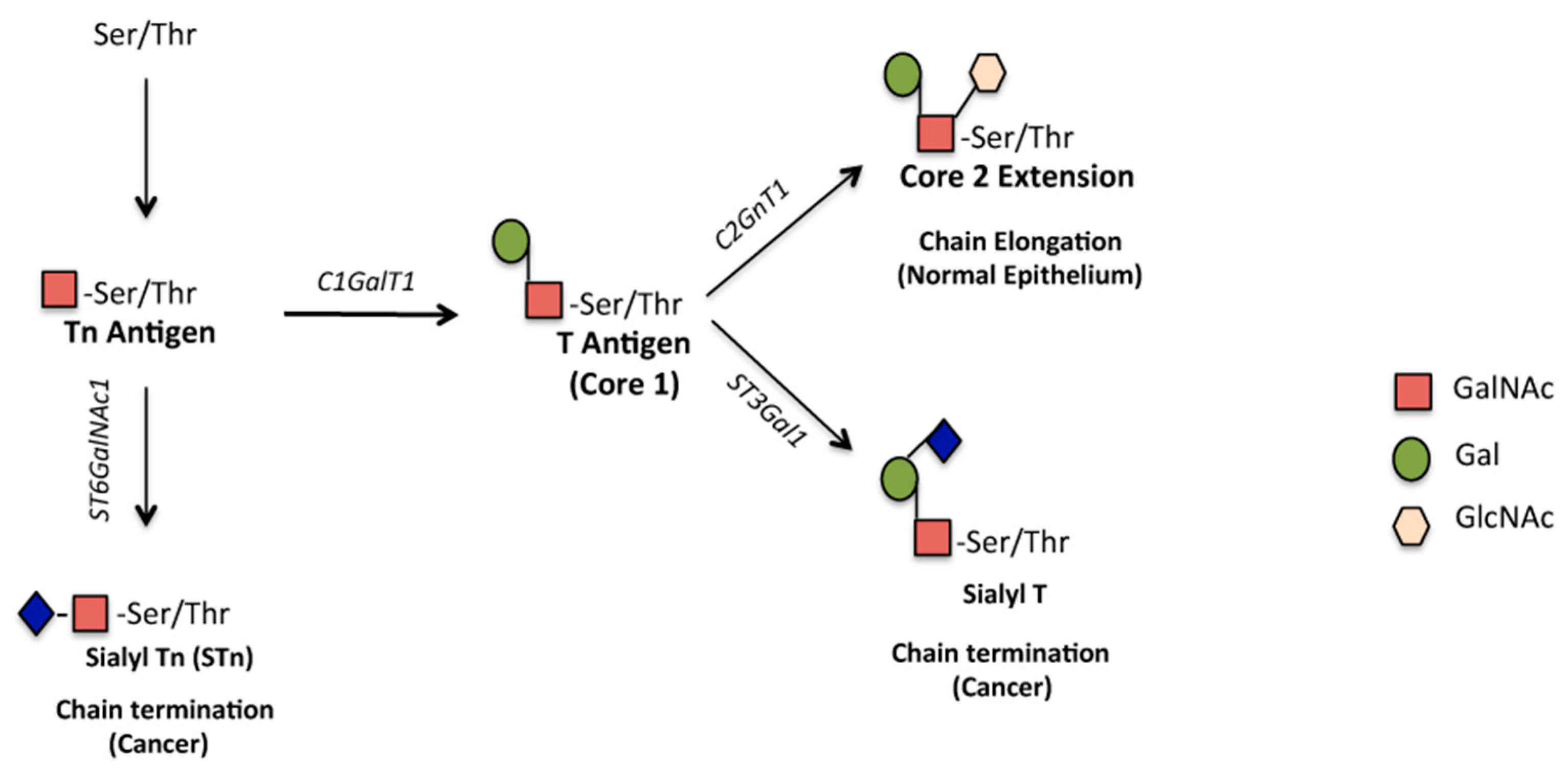
2. Aberrant Glycosylation of MUC1 Is Associated with Cancer
2.1. O-glycosylation
2.2. N-glycosylation
3. Aberrant Glycosylation of MUC1 Facilitates New Interactions with Cellular Proteins and Affects Intracellular Signaling in Cancer Cells
4. MUC1 Glycosylation in Cancer Progression and Metastasis
5. MUC1 Hypoglycosylation in Inflammation
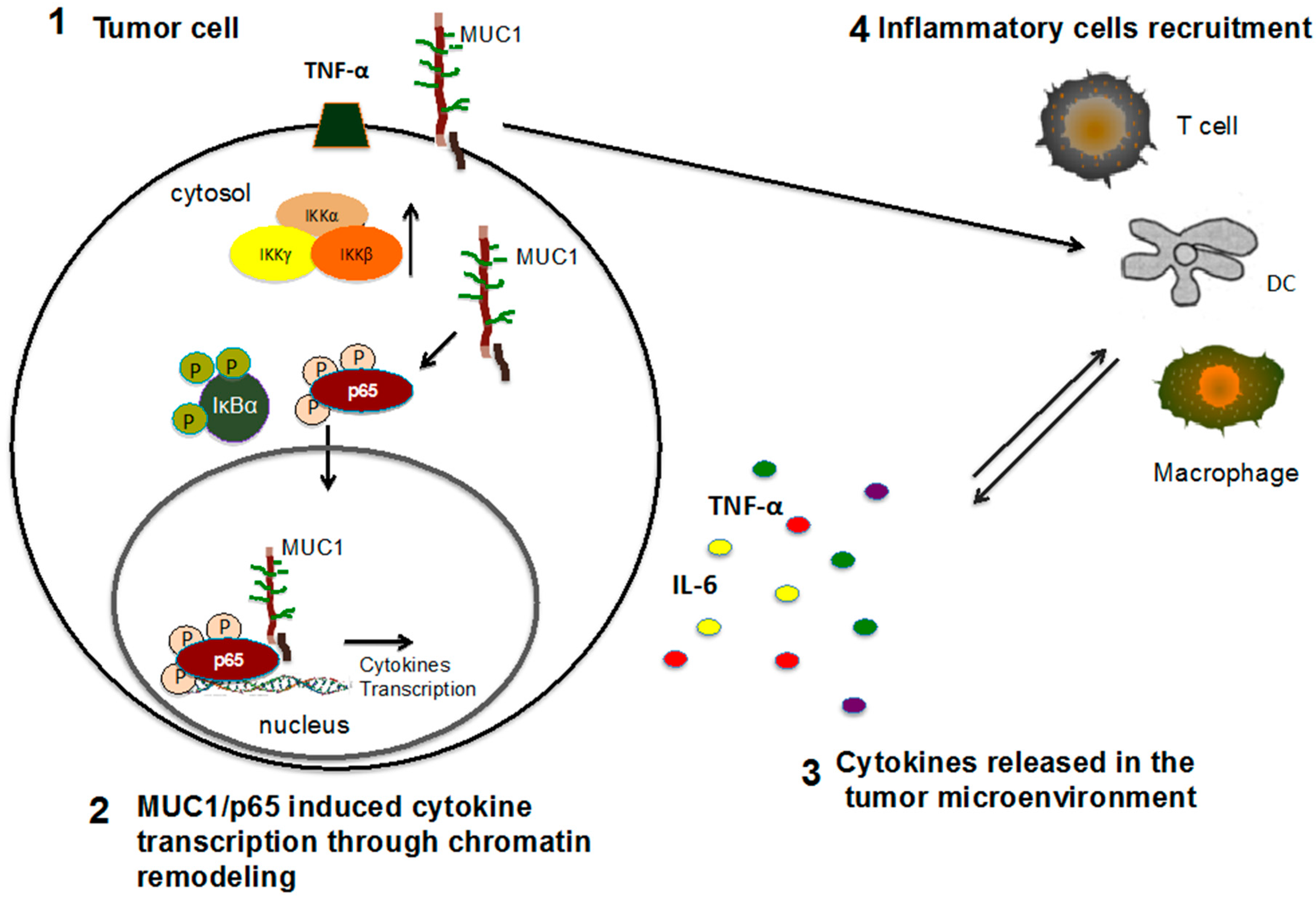
6. Abnormal MUC1 in the Tumor Microenvironment
7. Conclusions
Acknowledgments
Conflicts of interest
References
- Kinlough, C.L.; Poland, P.A.; Gendler, S.J.; Mattila, P.E.; Mo, D.; Weisz, O.A.; Hughey, R.P. Core-glycosylated mucin-like repeats from MUC1 are an apical targeting signal. J. Biol. Chem. 2011, 286, 39072–39081. [Google Scholar] [CrossRef] [PubMed]
- Cadron, I.; Van Gorp, T.; Mihalyi, A.; Luyten, C.; Drijkoningen, K.; Amant, F.; Leunen, K.; Vergote, I. The impact of enzastaurin (LY317615.HCL) on CA125 biosynthesis and shedding in ovarian cancer cells. Gynecol. Oncol. 2010, 118, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Hollingsworth, M.A.; Swanson, B.J. Mucins in cancer: Protection and control of the cell surface. Nat. Rev. Cancer 2004, 4, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, P.; Chakraborty, S.; Ponnusamy, M.P.; Lakshmanan, I.; Jain, M.; Batra, S.K. Mucins in the pathogenesis of breast cancer: Implications in diagnosis, prognosis and therapy. Biochim. Biophys. Acta 2011, 1815, 224–240. [Google Scholar] [CrossRef] [PubMed]
- Schlondorff, J.; Blobel, C.P. Metalloprotease-disintegrins: Modular proteins capable of promoting cell-cell interactions and triggering signals by protein-ectodomain shedding. J. Cell Sci. 1999, 112, 3603–3617. [Google Scholar] [PubMed]
- Blobel, C.P. Adams: Key components in EGFR signalling and development. Nat. Rev. Mol. Cell Biol. 2005, 6, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Thathiah, A.; Blobel, C.P.; Carson, D.D. Tumor necrosis factor-α converting enzyme/ADAM 17 mediates MUC1 shedding. J. Biol. Chem. 2003, 278, 3386–3394. [Google Scholar] [CrossRef] [PubMed]
- Goth, C.K.; Halim, A.; Khetarpal, S.A.; Rader, D.J.; Clausen, H.; Schjoldager, K.T. A systematic study of modulation of ADAM-mediated ectodomain shedding by site-specific O-glycosylation. Proc. Natl. Acad. Sci. USA 2015, 112, 14623–14628. [Google Scholar] [CrossRef] [PubMed]
- Thathiah, A.; Brayman, M.; Dharmaraj, N.; Julian, J.J.; Lagow, E.L.; Carson, D.D. Tumor necrosis factor α stimulates MUC1 synthesis and ectodomain release in a human uterine epithelial cell line. Endocrinology 2004, 145, 4192–4203. [Google Scholar] [CrossRef] [PubMed]
- Varki, A. Biological roles of oligosaccharides: All of the theories are correct. Glycobiology 1993, 3, 97–130. [Google Scholar] [CrossRef] [PubMed]
- Nath, S.; Mukherjee, P. MUC1: A multifaceted oncoprotein with a key role in cancer progression. Trends Mol. Med. 2014, 20, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Kharbanda, S.; Kufe, D. Association of the DF3/MUC1 breast cancer antigen with GRB2 and the SOS/RAS exchange protein. Cancer Res. 1995, 55, 4000–4003. [Google Scholar] [PubMed]
- Huang, L.; Chen, D.; Liu, D.; Yin, L.; Kharbanda, S.; Kufe, D. MUC1 oncoprotein blocks glycogen synthase kinase 3β-mediated phosphorylation and degradation of β-catenin. Cancer Res. 2005, 65, 10413–10422. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Xu, H.; Kufe, D. MUC1 oncoprotein stabilizes and activates estrogen receptor α. Mol. Cell 2006, 21, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Raina, D.; Joshi, M.D.; Kawano, T.; Ren, J.; Kharbanda, S.; Kufe, D. MUC1-C oncoprotein functions as a direct activator of the nuclear factor-κB p65 transcription factor. Cancer Res. 2009, 69, 7013–7021. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Raina, D.; Trivedi, V.; Ren, J.; Rajabi, H.; Kharbanda, S.; Kufe, D. MUC1 oncoprotein activates the IκB kinase β complex and constitutive NF-κB signalling. Nat. Cell Biol. 2007, 9, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Cascio, S.; Zhang, L.; Finn, O.J. MUC1 protein expression in tumor cells regulates transcription of proinflammatory cytokines by forming a complex with nuclear factor-κB p65 and binding to cytokine promoters: Importance of extracellular domain. J. Biol. Chem. 2011, 286, 42248–42256. [Google Scholar] [CrossRef] [PubMed]
- Brockhausen, I. Biosynthesis and functions of O-glycans and regulation of mucin antigen expression in cancer. Biochem. Soc. Trans. 1997, 25, 871–874. [Google Scholar] [CrossRef] [PubMed]
- Brockhausen, I.; Schachter, H.; Stanley, P. O-GalNAc glycans. In Essentials of Glycobiology, 2nd ed.; Varki, A., Cummings, R.D., Esko, J.D., Freeze, H.H., Stanley, P., Bertozzi, C.R., Hart, G.W., Etzler, M.E., Eds.; Cold Spring Harbor: New York, NY, USA, 2009. [Google Scholar]
- Sewell, R.; Backstrom, M.; Dalziel, M.; Gschmeissner, S.; Karlsson, H.; Noll, T.; Gatgens, J.; Clausen, H.; Hansson, G.C.; Burchell, J.; et al. The ST6GALNAc-I sialyltransferase localizes throughout the Golgi and is responsible for the synthesis of the tumor-associated sialyl-Tn O-glycan in human breast cancer. J. Biol. Chem. 2006, 281, 3586–3594. [Google Scholar] [CrossRef] [PubMed]
- Ju, T.; Cummings, R.D. A unique molecular chaperone Cosmc required for activity of the mammalian core 1 β3-galactosyltransferase. Proc. Natl. Acad. Sci. USA 2002, 99, 16613–16618. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ju, T.; Ding, X.; Xia, B.; Wang, W.; Xia, L.; He, M.; Cummings, R.D. Cosmc is an essential chaperone for correct protein O-glycosylation. Proc. Natl. Acad. Sci. USA 2010, 107, 9228–9233. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, P.; Dabelsteen, S.; Madsen, F.B.; Francavilla, C.; Kopp, K.L.; Steentoft, C.; Vakhrushev, S.Y.; Olsen, J.V.; Hansen, L.; Bennett, E.P.; et al. Immature truncated O-glycophenotype of cancer directly induces oncogenic features. Proc. Natl. Acad. Sci. USA 2014, 111, E4066–E4075. [Google Scholar] [CrossRef] [PubMed]
- Burchell, J.; Poulsom, R.; Hanby, A.; Whitehouse, C.; Cooper, L.; Clausen, H.; Miles, D.; Taylor-Papadimitriou, J. An α2,3 sialyltransferase (ST3Gal I) is elevated in primary breast carcinomas. Glycobiology 1999, 9, 1307–1311. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, C.; Burchell, J.; Gschmeissner, S.; Brockhausen, I.; Lloyd, K.O.; Taylor-Papadimitriou, J. A transfected sialyltransferase that is elevated in breast cancer and localizes to the medial/trans-Golgi apparatus inhibits the development of core-2-based O-glycans. J. Cell Biol. 1997, 137, 1229–1241. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in cancer: Mechanisms and clinical implications. Nat. Rev. Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Roy, L.D.; Sahraei, M.; Subramani, D.B.; Besmer, D.; Nath, S.; Tinder, T.L.; Bajaj, E.; Shanmugam, K.; Lee, Y.Y.; Hwang, S.I.; et al. MUC1 enhances invasiveness of pancreatic cancer cells by inducing epithelial to mesenchymal transition. Oncogene 2011, 30, 1449–1459. [Google Scholar] [CrossRef] [PubMed]
- Julien, S.; Lagadec, C.; Krzewinski-Recchi, M.A.; Courtand, G.; Le Bourhis, X.; Delannoy, P. Stable expression of sialyl-Tn antigen in T47-D cells induces a decrease of cell adhesion and an increase of cell migration. Breast Cancer Res. Treat. 2005, 90, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Mungul, A.; Cooper, L.; Brockhausen, I.; Ryder, K.; Mandel, U.; Clausen, H.; Rughetti, A.; Miles, D.W.; Taylor-Papadimitriou, J.; Burchell, J.M. Sialylated core 1 based O-linked glycans enhance the growth rate of mammary carcinoma cells in MUC1 transgenic mice. Int. J. Oncol. 2004, 25, 937–943. [Google Scholar] [PubMed]
- Ozaki, H.; Matsuzaki, H.; Ando, H.; Kaji, H.; Nakanishi, H.; Ikehara, Y.; Narimatsu, H. Enhancement of metastatic ability by ectopic expression of ST6GalNAcI on a gastric cancer cell line in a mouse model. Clin. Exp. Metastasis 2012, 29, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Krishn, S.R.; Kaur, S.; Smith, L.M.; Johansson, S.L.; Jain, M.; Patel, A.; Gautam, S.K.; Hollingsworth, M.A.; Mandel, U.; Clausen, H.; et al. Mucins and associated glycan signatures in colon adenoma-carcinoma sequence: Prospective pathological implication(s) for early diagnosis of colon cancer. Cancer Lett. 2016, 374, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Itzkowitz, S.H.; Bloom, E.J.; Kokal, W.A.; Modin, G.; Hakomori, S.; Kim, Y.S. Sialosyl-Tn. A novel mucin antigen associated with prognosis in colorectal cancer patients. Cancer 1990, 66, 1960–1966. [Google Scholar] [CrossRef]
- Ogata, S.; Koganty, R.; Reddish, M.; Longenecker, B.M.; Chen, A.; Perez, C.; Itzkowitz, S.H. Different modes of sialyl-Tn expression during malignant transformation of human colonic mucosa. Glycoconj. J. 1998, 15, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Muller, S.; Hanisch, F.G. Recombinant MUC1 probe authentically reflects cell-specific O-glycosylation profiles of endogenous breast cancer mucin. High density and prevalent core 2-based glycosylation. J. Biol. Chem. 2002, 277, 26103–26112. [Google Scholar] [CrossRef] [PubMed]
- Premaratne, P.; Welen, K.; Damber, J.E.; Hansson, G.C.; Backstrom, M. O-glycosylation of MUC1 mucin in prostate cancer and the effects of its expression on tumor growth in a prostate cancer xenograft model. Tumour Biol. 2011, 32, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Backstrom, M.; Thomsson, K.A.; Karlsson, H.; Hansson, G.C. Sensitive liquid chromatography-electrospray mass spectrometry allows for the analysis of the O-glycosylation of immunoprecipitated proteins from cells or tissues: Application to MUC1 glycosylation in cancer. J. Proteome Res. 2009, 8, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Borzym-Kluczyk, M.; Radziejewska, I.; Cechowska-Pasko, M. Increased expression of MUC1 and sialyl Lewis antigens in different areas of clear renal cell carcinoma. Clin. Exp. Nephrol. 2015, 19, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Gulzar, Z.G.; St Hill, C.A.; Walcheck, B.; Brooks, J.D. Increased expression of GCNT1 is associated with altered O-glycosylation of PSA, PAP, and MUC1 in human prostate cancers. Prostate 2014, 74, 1059–1067. [Google Scholar] [CrossRef] [PubMed]
- Dohi, D.F.; Sutton, R.C.; Frazier, M.L.; Nakamori, S.; McIsaac, A.M.; Irimura, T. Regulation of sialomucin production in colon carcinoma cells. J. Biol. Chem. 1993, 268, 10133–10138. [Google Scholar] [PubMed]
- Nakamori, S.; Kameyama, M.; Imaoka, S.; Furukawa, H.; Ishikawa, O.; Sasaki, Y.; Kabuto, T.; Iwanaga, T.; Matsushita, Y.; Irimura, T. Increased expression of sialyl Lewisx antigen correlates with poor survival in patients with colorectal carcinoma: Clinicopathological and immunohistochemical study. Cancer Res. 1993, 53, 3632–3637. [Google Scholar] [PubMed]
- Ricardo, S.; Marcos-Silva, L.; Valente, C.; Coelho, R.; Gomes, R.; David, L. Mucins MUC16 and MUC1 are major carriers of SLea and SLex in borderline and malignant serous ovarian tumors. Virchows Arch. 2016, 468, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Hanski, C.; Klussmann, E.; Wang, J.; Bohm, C.; Ogorek, D.; Hanski, M.L.; Kruger-Krasagakes, S.; Eberle, J.; Schmitt-Graff, A.; Riecken, E.O. Fucosyltransferase III and sialyl-Lex expression correlate in cultured colon carcinoma cells but not in colon carcinoma tissue. Glycoconj. J. 1996, 13, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Hiraiwa, N.; Sawada-Kasugai, M.; Akamatsu, S.; Tachikawa, T.; Kasai, Y.; Akiyama, S.; Ito, K.; Takagi, H.; Kannagi, R. Altered mRNA expression of specific molecular species of fucosyl- and sialyl-transferases in human colorectal cancer tissues. Int. J. Cancer 1997, 71, 556–564. [Google Scholar] [CrossRef]
- Petretti, T.; Kemmner, W.; Schulze, B.; Schlag, P.M. Altered mRNA expression of glycosyltransferases in human colorectal carcinomas and liver metastases. Gut 2000, 46, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Hauselmann, I.; Borsig, L. Altered tumor-cell glycosylation promotes metastasis. Front. Oncol. 2014, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Kojima, N.; Handa, K.; Newman, W.; Hakomori, S. Inhibition of selectin-dependent tumor cell adhesion to endothelial cells and platelets by blocking O-glycosylation of these cells. Biochem. Biophys. Res. Commun. 1992, 182, 1288–1295. [Google Scholar] [CrossRef]
- Zhang, D.; Gao, J.; Zhu, L.; Hu, Z.; Hou, R.; Liu, S.; Tan, M.; Liu, J.; Lin, B. Chemoresistance is associated with MUC1 and Lewis y antigen expression in ovarian epithelial cancers. Int. J. Mol. Sci. 2013, 14, 11024–11033. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.S.; Batra, S.K.; Qi, W.N.; Metzgar, R.S.; Hollingsworth, M.A. Cloning and sequencing of a human pancreatic tumor mucin cDNA. J. Biol. Chem. 1990, 265, 15294–15299. [Google Scholar] [PubMed]
- Parry, S.; Hanisch, F.G.; Leir, S.H.; Sutton-Smith, M.; Morris, H.R.; Dell, A.; Harris, A. N-glycosylation of the MUC1 mucin in epithelial cells and secretions. Glycobiology 2006, 16, 623–634. [Google Scholar] [CrossRef] [PubMed]
- Shental-Bechor, D.; Levy, Y. Effect of glycosylation on protein folding: A close look at thermodynamic stabilization. Proc. Natl. Acad. Sci. USA 2008, 105, 8256–8261. [Google Scholar] [CrossRef] [PubMed]
- Cascio, S.; Farkas, A.M.; Hughey, R.P.; Finn, O.J. Altered glycosylation of MUC1 influences its association with CIN85: The role of this novel complex in cancer cell invasion and migration. Oncotarget 2013, 4, 1686–1697. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Akita, K.; Yashiro, M.; Sawada, T.; Hirakawa, K.; Murata, T.; Nakada, H. Binding of galectin-3, a β-galactoside-binding lectin, to MUC1 protein enhances phosphorylation of extracellular signal-regulated kinase 1/2 (ERK1/2) and Akt, promoting tumor cell malignancy. J. Biol. Chem. 2015, 290, 26125–26140. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, S.; Duraisamy, S.; Barbashov, S.; Kawano, T.; Kharbanda, S.; Kufe, D. The MUC1 and galectin-3 oncoproteins function in a microRNA-dependent regulatory loop. Mol. Cell 2007, 27, 992–1004. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.G.; Andrews, N.; Zhao, Q.; McKean, D.; Williams, J.F.; Connor, L.J.; Gerasimenko, O.V.; Hilkens, J.; Hirabayashi, J.; Kasai, K.; et al. Galectin-3 interaction with Thomsen-Friedenreich disaccharide on cancer-associated MUC1 causes increased cancer cell endothelial adhesion. J. Biol. Chem. 2007, 282, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Tanida, S.; Mori, Y.; Ishida, A.; Akita, K.; Nakada, H. Galectin-3 binds to MUC1-N-terminal domain and triggers recruitment of β-catenin in MUC1-expressing mouse 3T3 cells. Biochim. Biophys. Acta 2014, 1840, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Nishidate, T.; Kijima, K.; Ohashi, T.; Takegawa, K.; Fujikane, T.; Hirata, K.; Nakamura, Y.; Katagiri, T. Critical roles of mucin 1 glycosylation by transactivated polypeptide N-acetylgalactosaminyltransferase 6 in mammary carcinogenesis. Cancer Res. 2010, 70, 2759–2769. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.H.; Huang, M.J.; Chen, C.H.; Shyu, M.K.; Huang, J.; Hung, J.S.; Huang, C.S.; Huang, M.C. Up-regulation of C1GAlT1 promotes breast cancer cell growth through MUC1-C signaling pathway. Oncotarget 2015, 6, 6123–6135. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Gallup, M.; Zlock, L.; Chen, Y.T.; Finkbeiner, W.E.; McNamara, N.A. Pivotal role of MUC1 glycosylation by cigarette smoke in modulating disruption of airway adherens junctions in vitro. J. Pathol. 2014, 234, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Altschuler, Y.; Kinlough, C.L.; Poland, P.A.; Bruns, J.B.; Apodaca, G.; Weisz, O.A.; Hughey, R.P. Clathrin-mediated endocytosis of MUC1 is modulated by its glycosylation state. Mol. Biol. Cell 2000, 11, 819–831. [Google Scholar] [CrossRef] [PubMed]
- Duncan, T.J.; Watson, N.F.; Al-Attar, A.H.; Scholefield, J.H.; Durrant, L.G. The role of MUC1 and MUC3 in the biology and prognosis of colorectal cancer. World J. Surg. Oncol. 2007, 5, 31. [Google Scholar] [CrossRef] [PubMed]
- Inata, J.; Hattori, N.; Yokoyama, A.; Ohshimo, S.; Doi, M.; Ishikawa, N.; Hamada, H.; Kohno, N. Circulating KL-6/MUC1 mucin carrying sialyl Lewisa oligosaccharide is an independent prognostic factor in patients with lung adenocarcinoma. Int. J. Cancer 2007, 120, 2643–2649. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Shoda, J.; Kawamoto, T.; Shinozaki, E.; Miyahara, N.; Hotta, S.; Iizuka, Y.; Nakahara, A.; Tanaka, N.; Yanaka, A.; et al. Expression of MUC1 recognized by monoclonal antibody MY.1E12 is a useful biomarker for tumor aggressiveness of advanced colon carcinoma. Clin. Exp. Metastasis 2004, 21, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Havrylov, S.; Redowicz, M.J.; Buchman, V.L. Emerging roles of RUK/CIN85 in vesicle-mediated transport, adhesion, migration and malignancy. Traffic 2010, 11, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Julien, S.; Adriaenssens, E.; Ottenberg, K.; Furlan, A.; Courtand, G.; Vercoutter-Edouart, A.S.; Hanisch, F.G.; Delannoy, P.; Le Bourhis, X. ST6GalNAc I expression in MDA-MB-231 breast cancer cells greatly modifies their O-glycosylation pattern and enhances their tumourigenicity. Glycobiology 2006, 16, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Paszek, M.J.; DuFort, C.C.; Rossier, O.; Bainer, R.; Mouw, J.K.; Godula, K.; Hudak, J.E.; Lakins, J.N.; Wijekoon, A.C.; Cassereau, L.; et al. The cancer glycocalyx mechanically primes integrin-mediated growth and survival. Nature 2014, 511, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Yeh, K.; Takatani, T.; King, M.R. Three to tango: MUC1 as a ligand for both E-selectin and ICAM-1 in the breast cancer metastatic cascade. Front. Oncol. 2012, 2, 76. [Google Scholar] [CrossRef] [PubMed]
- Rahn, J.J.; Chow, J.W.; Horne, G.J.; Mah, B.K.; Emerman, J.T.; Hoffman, P.; Hugh, J.C. MUC1 mediates transendothelial migration in vitro by ligating endothelial cell ICAM-1. Clin. Exp. Metastasis 2005, 22, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Rahn, J.J.; Zhang, J.; Gunasekera, N.; Sun, X.; Shaw, A.R.; Hendzel, M.J.; Hoffman, P.; Bernier, A.; Hugh, J.C. MUC1 initiates SRC-CRKL-RAC1/CDC42-mediated actin cytoskeletal protrusive motility after ligating intercellular adhesion molecule-1. Mol. Cancer Res. 2008, 6, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Iurisci, I.; Tinari, N.; Natoli, C.; Angelucci, D.; Cianchetti, E.; Iacobelli, S. Concentrations of galectin-3 in the sera of normal controls and cancer patients. Clin. Cancer Res. 2000, 6, 1389–1393. [Google Scholar] [PubMed]
- Zhao, Q.; Guo, X.; Nash, G.B.; Stone, P.C.; Hilkens, J.; Rhodes, J.M.; Yu, L.G. Circulating galectin-3 promotes metastasis by modifying MUC1 localization on cancer cell surface. Cancer Res. 2009, 69, 6799–6806. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Bachvarova, M.; Morin, C.; Plante, M.; Gregoire, J.; Renaud, M.C.; Sebastianelli, A.; Bachvarov, D. Role of the polypeptide N-acetylgalactosaminyltransferase 3 in ovarian cancer progression: Possible implications in abnormal mucin O-glycosylation. Oncotarget 2014, 5, 544–560. [Google Scholar] [CrossRef] [PubMed]
- Maverakis, E.; Kim, K.; Shimoda, M.; Gershwin, M.E.; Patel, F.; Wilken, R.; Raychaudhuri, S.; Ruhaak, L.R.; Lebrilla, C.B. Glycans in the immune system and the altered glycan theory of autoimmunity: A critical review. J. Autoimmun. 2015, 57, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.M.; Nowack, D.D.; Omenn, G.S.; Haab, B.B. Mucin glycosylation is altered by pro-inflammatory signaling in pancreatic-cancer cells. J. Proteome Res. 2009, 8, 1876–1886. [Google Scholar] [CrossRef] [PubMed]
- Lagow, E.L.; Carson, D.D. Synergistic stimulation of MUC1 expression in normal breast epithelia and breast cancer cells by interferon-γ and tumor necrosis factor-α. J. Cell. Biochem. 2002, 86, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Gaemers, I.C.; Vos, H.L.; Volders, H.H.; van der Valk, S.W.; Hilkens, J. A stat-responsive element in the promoter of the episialin/MUC1 gene is involved in its overexpression in carcinoma cells. J. Biol. Chem. 2001, 276, 6191–6199. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; McConnell, M.J.; Yu, B.; Li, J.; Balko, J.M.; Black, E.P.; Johnson, J.O.; Lloyd, M.C.; Altiok, S.; Haura, E.B. MUC1 is a downstream target of STAT3 and regulates lung cancer cell survival and invasion. Int. J. Oncol. 2009, 35, 337–345. [Google Scholar] [PubMed]
- Clark, S.; McGuckin, M.A.; Hurst, T.; Ward, B.G. Effect of interferon-γ and TNF-α on MUC1 mucin expression in ovarian carcinoma cell lines. Dis. Markers 1994, 12, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Albertsmeyer, A.C.; Kakkassery, V.; Spurr-Michaud, S.; Beeks, O.; Gipson, I.K. Effect of pro-inflammatory mediators on membrane-associated mucins expressed by human ocular surface epithelial cells. Exp. Eye Res. 2010, 90, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.I.; Chang, J.Y.; Kim, Y.Y.; Lee, G.; Kho, H.S. MUC1 expression in the oral mucosal epithelial cells of the elderly. Arch. Oral Biol. 2011, 56, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Dharmaraj, N.; Wang, P.; Carson, D.D. Cytokine and progesterone receptor interplay in the regulation of MUC1 gene expression. Mol. Endocrinol. 2010, 24, 2253–2266. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, L.; Nunes, D.P.; Troxler, R.F.; Offner, G.D. Pro-inflammatory cytokines up-regulate MUC1 gene expression in oral epithelial cells. J. Dent. Res. 2003, 82, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, Y.; Imai, S.; Inouye, Y.; Okano, T.; Taniguchi, A. Effects of carbocisteine on sialyl-Lewis X expression in an airway carcinoma cell line stimulated with tumor necrosis factor-α. Eur. J. Pharmacol. 2006, 530, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Rowse, G.J.; Tempero, R.M.; VanLith, M.L.; Hollingsworth, M.A.; Gendler, S.J. Tolerance and immunity to MUC1 in a human MUC1 transgenic murine model. Cancer Res. 1998, 58, 315–321. [Google Scholar] [PubMed]
- Beatty, P.; Ranganathan, S.; Finn, O.J. Prevention of colitis-associated colon cancer using a vaccine to target abnormal expression of the MUC1 tumor antigen. Oncoimmunology 2012, 1, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Cascio, S.; Finn, O.J. Complex of MUC1, CIN85 and CBL in colon cancer progression and metastasis. Cancers 2015, 7, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Beatty, P.L.; Plevy, S.E.; Sepulveda, A.R.; Finn, O.J. Cutting edge: Transgenic expression of human MUC1 in IL-10-/-mice accelerates inflammatory bowel disease and progression to colon cancer. J. Immunol. 2007, 179, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Crocker, P.R.; Varki, A. Siglecs in the immune system. Immunology 2001, 103, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Varki, A.; Angata, T. Siglecs—The major subfamily of I-type lectins. Glycobiology 2006, 16, 1R–27R. [Google Scholar] [CrossRef] [PubMed]
- Takamiya, R.; Ohtsubo, K.; Takamatsu, S.; Taniguchi, N.; Angata, T. The interaction between Siglec-15 and tumor-associated sialyl-Tn antigen enhances TGF-β secretion from monocytes/macrophages through the DAP12-Syk pathway. Glycobiology 2013, 23, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Nath, D.; Hartnell, A.; Happerfield, L.; Miles, D.W.; Burchell, J.; Taylor-Papadimitriou, J.; Crocker, P.R. Macrophage-tumour cell interactions: Identification of MUC1 on breast cancer cells as a potential counter-receptor for the macrophage-restricted receptor, sialoadhesin. Immunology 1999, 98, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Carlos, C.A.; Dong, H.F.; Howard, O.M.; Oppenheim, J.J.; Hanisch, F.G.; Finn, O.J. Human tumor antigen MUC1 is chemotactic for immature dendritic cells and elicits maturation but does not promote Th1 type immunity. J. Immunol. 2005, 175, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, L.A.; Van Vliet, S.J. A bitter sweet symphony: Immune responses to altered O-glycan epitopes in cancer. Biomolecules 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Napoletano, C.; Rughetti, A.; Agervig Tarp, M.P.; Coleman, J.; Bennett, E.P.; Picco, G.; Sale, P.; Denda-Nagai, K.; Irimura, T.; Mandel, U.; et al. Tumor-associated Tn-MUC1 glycoform is internalized through the macrophage galactose-type C-type lectin and delivered to the HLA class I and II compartments in dendritic cells. Cancer Res. 2007, 67, 8358–8367. [Google Scholar] [CrossRef] [PubMed]
- Monti, P.; Leone, B.E.; Zerbi, A.; Balzano, G.; Cainarca, S.; Sordi, V.; Pontillo, M.; Mercalli, A.; Di Carlo, V.; Allavena, P.; et al. Tumor-derived MUC1 mucins interact with differentiating monocytes and induce IL-10highIL-12low regulatory dendritic cell. J. Immunol. 2004, 172, 7341–7349. [Google Scholar] [CrossRef] [PubMed]
- Hiltbold, E.M.; Vlad, A.M.; Ciborowski, P.; Watkins, S.C.; Finn, O.J. The mechanism of unresponsiveness to circulating tumor antigen MUC1 is a block in intracellular sorting and processing by dendritic cells. J. Immunol. 2000, 165, 3730–3741. [Google Scholar] [CrossRef] [PubMed]
- Carrascal, M.A.; Severino, P.F.; Guadalupe Cabral, M.; Silva, M.; Ferreira, J.A.; Calais, F.; Quinto, H.; Pen, C.; Ligeiro, D.; Santos, L.L.; et al. Sialyl Tn-expressing bladder cancer cells induce a tolerogenic phenotype in innate and adaptive immune cells. Mol. Oncol. 2014, 8, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Waldhauer, I.; Steinle, A. NK cells and cancer immunosurveillance. Oncogene 2008, 27, 5932–5943. [Google Scholar] [CrossRef] [PubMed]
- Hudak, J.E.; Canham, S.M.; Bertozzi, C.R. Glycocalyx engineering reveals a siglec-based mechanism for NK cell immunoevasion. Nat. Chem. Biol. 2014, 10, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Jandus, C.; Boligan, K.F.; Chijioke, O.; Liu, H.; Dahlhaus, M.; Demoulins, T.; Schneider, C.; Wehrli, M.; Hunger, R.E.; Baerlocher, G.M.; et al. Interactions between Siglec-7/9 receptors and ligands influence NK cell-dependent tumor immunosurveillance. J. Clin. Investig. 2014, 124, 1810–1820. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cascio, S.; Finn, O.J. Intra- and Extra-Cellular Events Related to Altered Glycosylation of MUC1 Promote Chronic Inflammation, Tumor Progression, Invasion, and Metastasis. Biomolecules 2016, 6, 39. https://doi.org/10.3390/biom6040039
Cascio S, Finn OJ. Intra- and Extra-Cellular Events Related to Altered Glycosylation of MUC1 Promote Chronic Inflammation, Tumor Progression, Invasion, and Metastasis. Biomolecules. 2016; 6(4):39. https://doi.org/10.3390/biom6040039
Chicago/Turabian StyleCascio, Sandra, and Olivera J. Finn. 2016. "Intra- and Extra-Cellular Events Related to Altered Glycosylation of MUC1 Promote Chronic Inflammation, Tumor Progression, Invasion, and Metastasis" Biomolecules 6, no. 4: 39. https://doi.org/10.3390/biom6040039
APA StyleCascio, S., & Finn, O. J. (2016). Intra- and Extra-Cellular Events Related to Altered Glycosylation of MUC1 Promote Chronic Inflammation, Tumor Progression, Invasion, and Metastasis. Biomolecules, 6(4), 39. https://doi.org/10.3390/biom6040039




