(3α,5α)3-Hydroxypregnan-20-one (3α,5α-THP) Regulation of the HPA Axis in the Context of Different Stressors and Sex
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. 3α,5α-THP Administration
2.3. Stress Paradigms
2.3.1. Restraint Stress
2.3.2. Forced Swim Stress
2.4. Immunoblotting
2.5. Antibodies
2.6. Corticosterone Assay
2.7. Statistical Analysis
3. Results
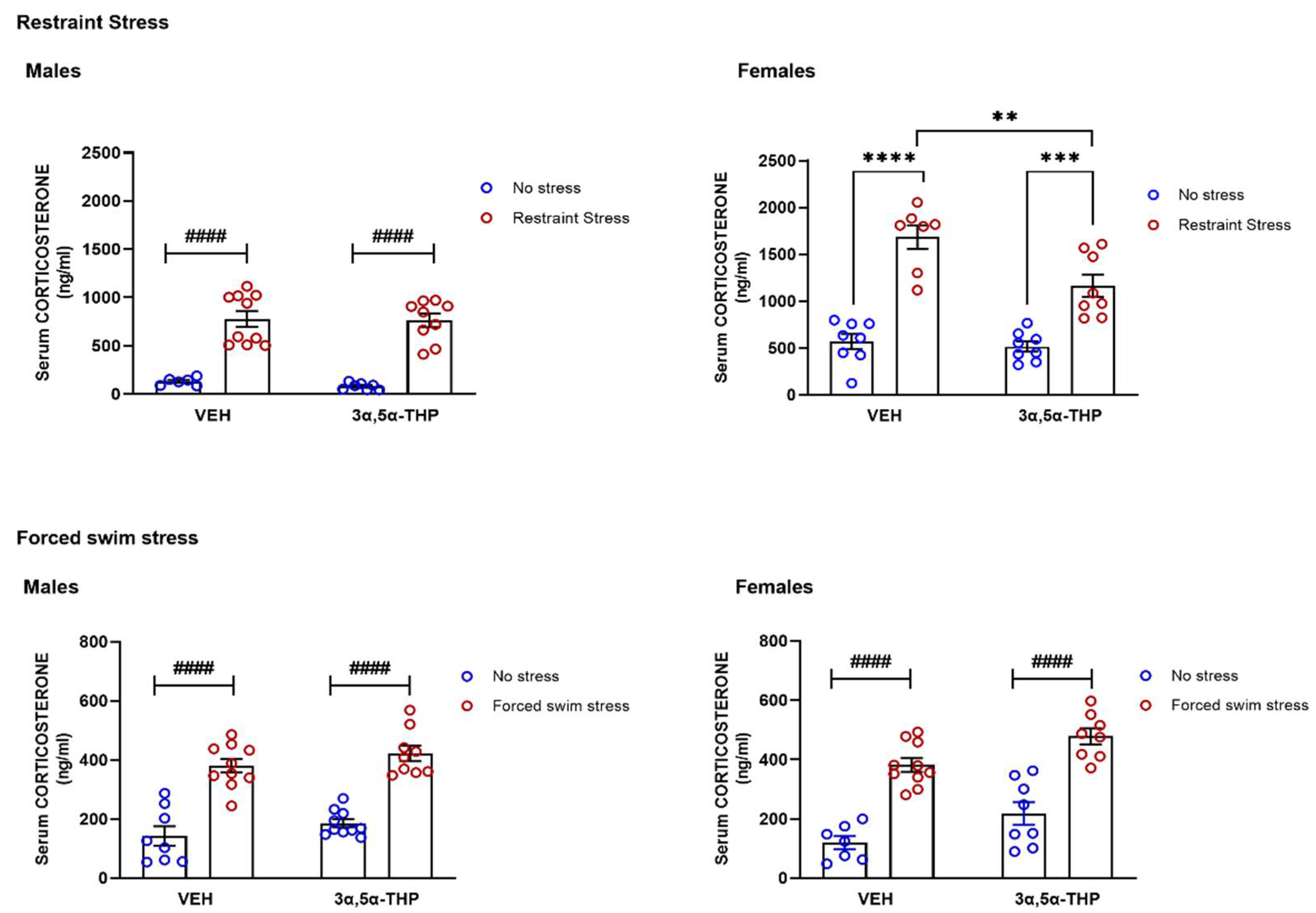
3.1. 3α,5α-THP Partially Prevents the Corticosterone Stress Response after Restraint Stress in Female, but Not in Male Rats
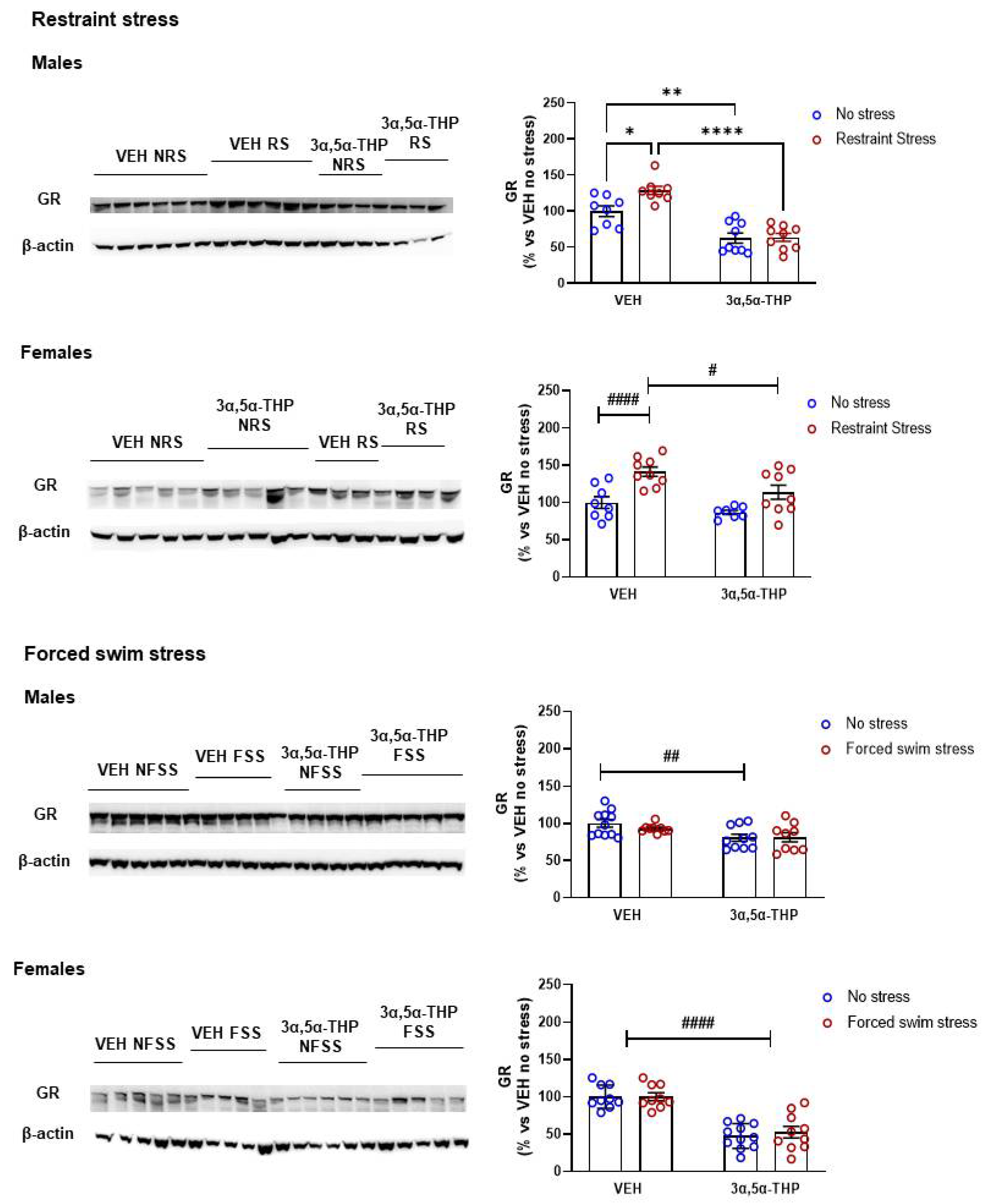
3.2. Glucocorticoid Receptor (GR) Regulation by 3α,5α-THP Differs Based on Type of Stressor, but Not Sex
3.3. 3α,5α-THP Diminished CRF Stress-Induced Response after Forced Swim Stress, but Only in Female Rats after Restraint Stress
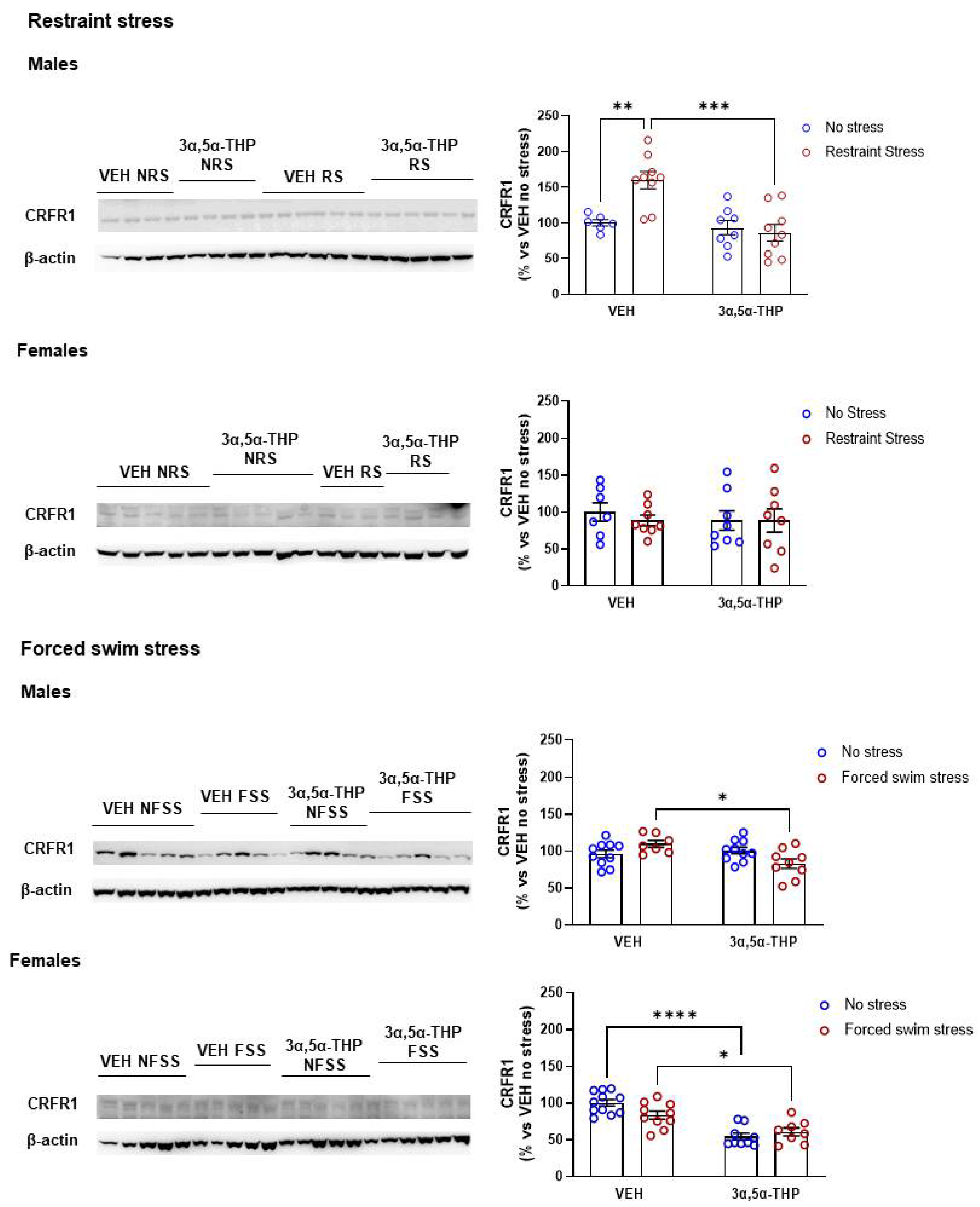
3.4. 3α,5α-THP Regulation of CRFR1 Is Both Stressor and Sex-Dependent
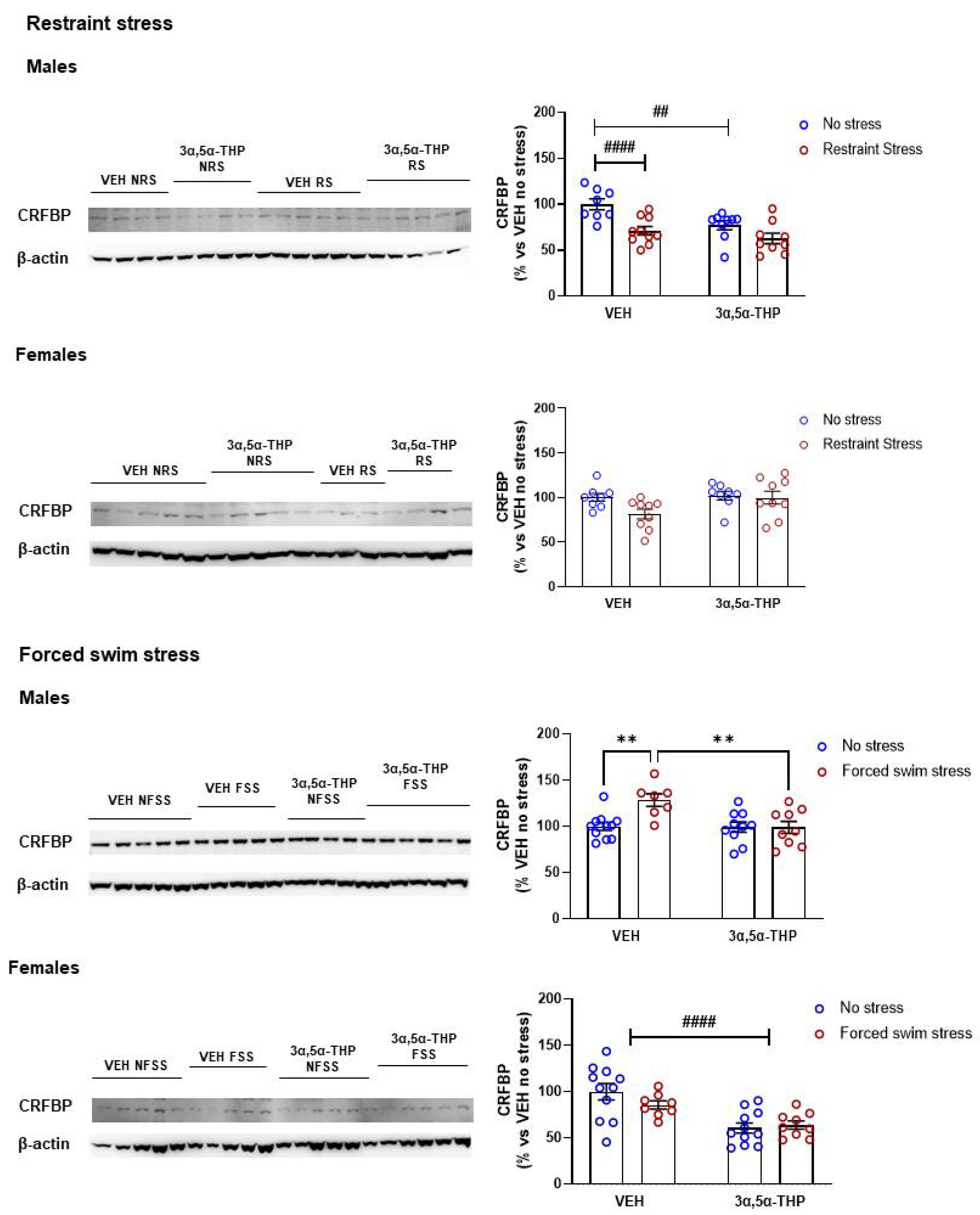
3.5. 3α,5α-THP Effect on CRF Binding Protein (CRFBP) Differs between Stressors and Sex
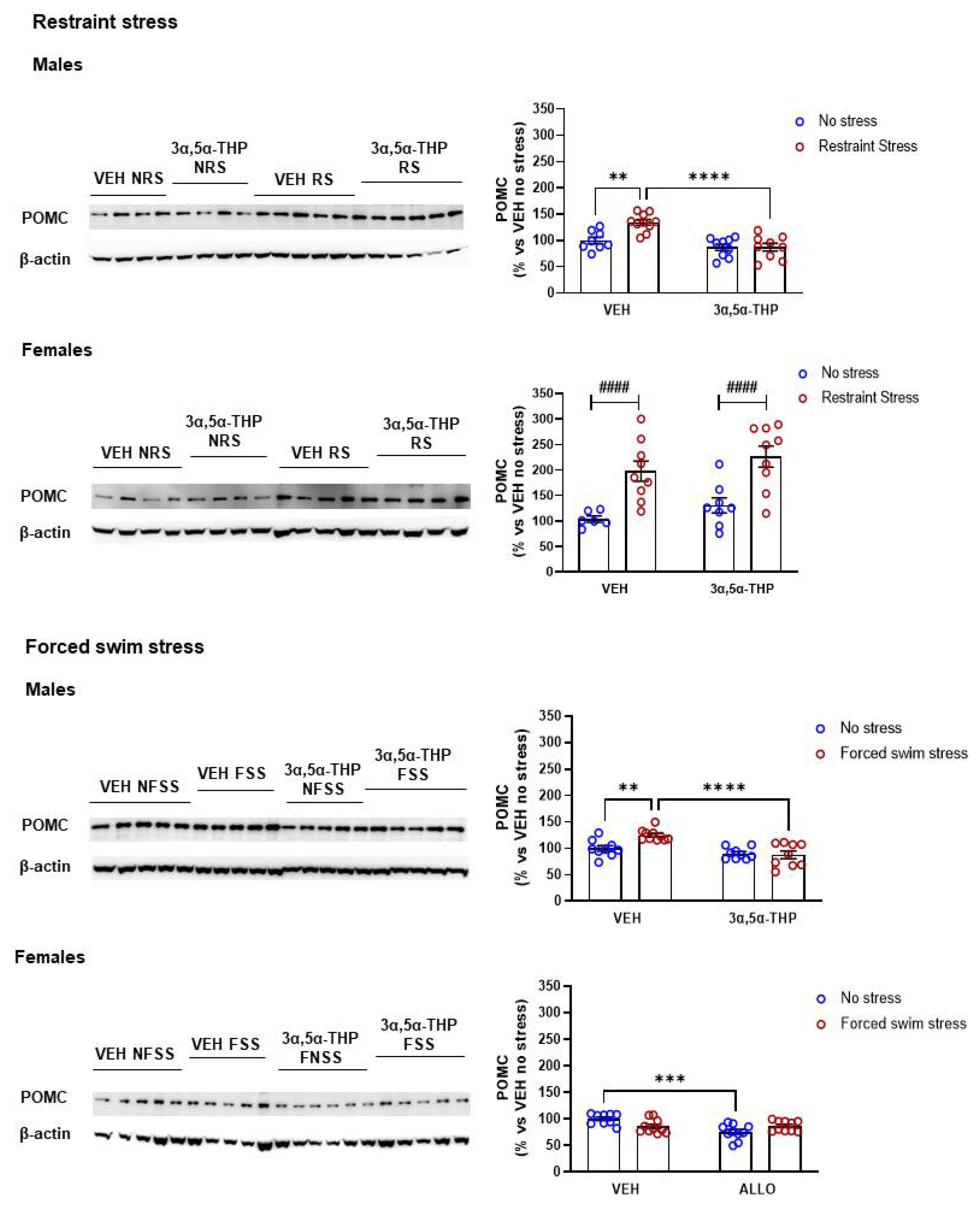
3.6. 3α,5α-THP Decreased Stress-Induced Hypothalamic POMC Levels in Male, but Not in Female Rats
4. Discussion
4.1. Circulating Corticosterone
4.2. Hypothalamic CRF
4.3. CRF Binding Protein (CRFBP)
4.4. CRF Receptor 1 (CRFR1)
4.5. Glucocorticoid Receptors (GRs)
4.6. Pro-Opiomelanocortin (POMC)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Heim, C.; Nemeroff, C.B. The role of childhood trauma in the neurobiology of mood and anxiety disorders: Preclinical and clinical studies. Biol. Psychiatry 2001, 49, 1023–1039. [Google Scholar] [CrossRef]
- Vale, W.; Spiess, J.; Rivier, C.; Rivier, J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 1981, 213, 1394–1397. [Google Scholar] [CrossRef]
- Eberwine, J.H.; Roberts, J.L. Glucocorticoid regulation of pro-opiomelanocortin gene transcription in the rat pituitary. J. Biol. Chem. 1984, 259, 2166–2170. [Google Scholar] [CrossRef]
- Tasker, J.G.; Herman, J.P. Mechanisms of rapid glucocorticoid feedback inhibition of the hypothalamic-pituitary-adrenal axis. Stress 2011, 14, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Owens, M.J.; Nemeroff, C.B. Physiology and pharmacology of corticotropin-releasing factor. Pharmacol. Rev. 1991, 43, 425–473. [Google Scholar]
- Potter, E.; Behan, D.P.; Fischer, W.H.; Linton, E.A.; Lowry, P.J.; Vale, W.W. Cloning and characterization of the cDNAs for human and rat corticotropin releasing factor-binding proteins. Nature 1991, 349, 423–426. [Google Scholar] [CrossRef] [PubMed]
- McClennen, S.J.; Cortright, D.N.; Seasholtz, A.F. Regulation of pituitary corticotropin-releasing hormone-binding protein messenger ribonucleic acid levels by restraint stress and adrenalectomy. Endocrinology 1998, 139, 4435–4441. [Google Scholar] [CrossRef]
- Kishimoto, T.; Pearse, R.V., 2nd; Lin, C.R.; Rosenfeld, M.G. A sauvagine/corticotropin-releasing factor receptor expressed in heart and skeletal muscle. Proc. Natl. Acad. Sci. USA 1995, 92, 1108–1112. [Google Scholar] [CrossRef]
- Potter, E.; Sutton, S.; Donaldson, C.; Chen, R.; Perrin, M.; Lewis, K.; Sawchenko, P.E.; Vale, W. Distribution of corticotropin-releasing factor receptor mRNA expression in the rat brain and pituitary. Proc. Natl. Acad. Sci. USA 1994, 91, 8777–8781. [Google Scholar] [CrossRef]
- Vaughan, J.; Donaldson, C.; Bittencourt, J.; Perrin, M.H.; Lewis, K.; Sutton, S.; Chan, R.; Turnbull, A.V.; Lovejoy, D.; Rivier, C.; et al. Urocortin, a mammalian neuropeptide related to fish urotensin I and to corticotropin-releasing factor. Nature 1995, 378, 287–292. [Google Scholar] [CrossRef]
- Huising, M.O.; Metz, J.R.; van Schooten, C.; Taverne-Thiele, A.J.; Hermsen, T.; Verburg-van Kemenade, B.M.; Flik, G. Structural characterisation of a cyprinid (Cyprinus carpio L.) CRH, CRH-BP and CRH-R1, and the role of these proteins in the acute stress response. J. Mol. Endocrinol. 2004, 32, 627–648. [Google Scholar] [CrossRef] [PubMed]
- Cullinan, W.E.; Ziegler, D.R.; Herman, J.P. Functional role of local GABAergic influences on the HPA axis. Brain Struct. Funct. 2008, 213, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, J.; Wakefield, S.; MacKenzie, G.; Moss, S.J.; Maguire, J. Neurosteroidogenesis is required for the physiological response to stress: Role of neurosteroid-sensitive GABAA receptors. J. Neurosci. 2011, 31, 18198–18210. [Google Scholar] [CrossRef] [PubMed]
- Purdy, R.H.; Morrow, A.L.; Moore, P.H., Jr.; Paul, S.M. Stress-induced elevations of gamma-aminobutyric acid type A receptor-active steroids in the rat brain. Proc. Natl. Acad. Sci. USA 1991, 88, 4553–4557. [Google Scholar] [CrossRef]
- Barbaccia, M.L.; Concas, A.; Serra, M.; Biggio, G. Stress and neurosteroids in adult and aged rats. Exp. Gerontol. 1998, 33, 697–712. [Google Scholar] [CrossRef]
- Genazzani, A.R.; Petraglia, F.; Bernardi, F.; Casarosa, E.; Salvestroni, C.; Tonetti, A.; Nappi, R.E.; Luisi, S.; Palumbo, M.; Purdy, R.H.; et al. Circulating levels of allopregnanolone in humans: Gender, age, and endocrine influences. J. Clin. Endocr. Metab. 1998, 83, 2099–2103. [Google Scholar] [CrossRef]
- Owens, M.J.; Ritchie, J.C.; Nemeroff, C.B. 5 alpha-pregnane-3 alpha, 21-diol-20-one (THDOC) attenuates mild stress-induced increases in plasma corticosterone via a non-glucocorticoid mechanism: Comparison with alprazolam. Brain Res. 1992, 573, 353–355. [Google Scholar] [CrossRef]
- Patchev, V.K.; Hassan, A.H.S.; Holsboer, F.; Almeida, O.F.X. The neurosteroid tetrahydroprogesterone attenuates the endocrine response to stress and exerts glucocorticoid-like effects on vasopressin gene transcription in the rat hypothalamus. Neuropsychopharmacology 1996, 15, 533–540. [Google Scholar] [CrossRef]
- Patchev, V.K.; Shoaib, M.; Holsboer, F.; Almeida, O.F.X. The neurosteroid tetrahydroprogesterone counteracts corticotropin-releasing hormone-induced anxiety and alters the release and gene expression of corticotropin-releasing hormone in the rat hypothalamus. Neuroscience 1994, 62, 265–271. [Google Scholar] [CrossRef]
- Boero, G.; Tyler, R.E.; Todd, C.A.; O’Buckley, T.K.; Balan, I.; Besheer, J.; Morrow, A.L. (3alpha,5alpha)3-hydroxypregnan-20-one (3alpha,5alpha-THP) regulation of hypothalamic and extrahypothalamic corticotropin releasing factor (CRF): Sexual dimorphism and brain region specificity in Sprague Dawley rats. Neuropharmacology 2021, 186, 108463. [Google Scholar] [CrossRef]
- Servatius, R.J.; Salameh, G.; Coyle, K.M.; Paré, W.P. Restraint Stress. In Encyclopedia of Stress, 2nd ed.; Fink, G., Ed.; Elsevier: San Diego, CA, USA, 2007; pp. 389–390. [Google Scholar] [CrossRef]
- Nequin, L.G.; Alvarez, J.; Schwartz, N.B. Measurement of serum steroid and gonadotropin levels and uterine and ovarian variables throughout 4 day and 5 day estrous cycles in the rat. Biol. Reprod 1979, 20, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.E. Neuroendocrine Control of the Ovarian Cycle of the Rat. In Knobil and Neill’s Physiology of Reproduction, 3rd ed.; Neill, J.D., Ed.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 2, pp. 2327–2388. [Google Scholar] [CrossRef]
- Balan, I.; Beattie, M.C.; O’Buckley, T.K.; Aurelian, L.; Morrow, A.L. Endogenous Neurosteroid (3⍺,5⍺)3-Hydroxypregnan-20-one Inhibits Toll-like-4 Receptor Activation and Pro-inflammatory Signaling in Macrophages and Brain. Sci. Rep. 2019, 9, 1220. [Google Scholar] [CrossRef] [PubMed]
- Crawley, J.N.; Glowa, J.R.; Majewska, M.D.; Paul, S.M. Anxiolytic activity of an endogenous adrenal steroid. Brain Res. 1986, 398, 382–385. [Google Scholar] [CrossRef]
- Devaud, L.L.; Purdy, R.H.; Morrow, A.L. The neurosteroid, 3α-hydroxy-5α-pregnan-20-one, protects against bicuculline-induced seizures during ethanol withdrawal in rats. Alcohol. Clin. Exp. Res. 1995, 19, 350–355. [Google Scholar] [CrossRef]
- Mendelson, W.B.; Martin, J.V.; Perlis, M.; Wagner, R.; Majewska, M.D.; Paul, S.M. Sleep induction by an adrenal steroid in the rat. Psychopharmacology 1987, 93, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Timpl, P.; Spanagel, R.; Sillaber, I.; Kresse, A.; Reul, J.M.; Stalla, G.K.; Blanquet, V.; Steckler, T.; Holsboer, F.; Wurst, W. Impaired stress response and reduced anxiety in mice lacking a functional corticotropin-releasing hormone receptor 1. Nat. Genet. 1998, 19, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Heinrichs, S.C.; Lapsansky, J.; Lovenberg, T.W.; De Souza, E.B.; Chalmers, D.T. Corticotropin-releasing factor CRF1, but not CRF2, receptors mediate anxiogenic-like behavior. Regul. Pept 1997, 71, 15–21. [Google Scholar] [CrossRef]
- Seasholtz, A.F.; Valverde, R.A.; Denver, R.J. Corticotropin-releasing hormone-binding protein: Biochemistry and function from fishes to mammals. J. Endocrinol. 2002, 175, 89–97. [Google Scholar] [CrossRef]
- De Souza, F.S.; Santangelo, A.M.; Bumaschny, V.; Avale, M.E.; Smart, J.L.; Low, M.J.; Rubinstein, M. Identification of neuronal enhancers of the proopiomelanocortin gene by transgenic mouse analysis and phylogenetic footprinting. Mol. Cell Biol. 2005, 25, 3076–3086. [Google Scholar] [CrossRef]
- Morrow, A.L.; Porcu, P.; Boyd, K.N.; Grant, K.A. Hypothalamic-pituitary-adrenal axis modulation of GABAergic neuroactive steroids influences ethanol sensitivity and drinking behavior. Dialogues Clin. Neurosci. 2006, 8, 463–477. [Google Scholar] [CrossRef]
- Biggio, G.; Pisu, M.G.; Biggio, F.; Serra, M. Allopregnanolone modulation of HPA axis function in the adult rat. Psychopharmacology 2014, 231, 3437–3444. [Google Scholar] [CrossRef] [PubMed]
- Porcu, P.; Morrow, A.L. Divergent neuroactive steroid responses to stress and ethanol in rat and mouse strains: Relevance for human studies. Psychopharmacology 2014, 231, 3257–3272. [Google Scholar] [CrossRef] [PubMed]
- Guo, A.-L.; Petraglia, F.; Criscuolo, M.; Ficarra, G.; Nappi, R.E.; Palumbo, M.A.; Trentini, G.P.; Purdy, R.H.; Genazzani, A.R. Evidence for a role of neurosteroids in modulation of diurnal changes and acute stress-induced corticosterone secretion in rats. Gynecol. Endocrinol. 1995, 9, 1–7. [Google Scholar] [CrossRef]
- Kitay, J.I. Sex differences in adrenal cortical secretion in the rat. Endocrinology 1961, 68, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Sze, Y.; Brunton, P.J. Effects of prenatal stress on neuroactive steroid responses to acute stress in adult male and female rats. J. Neuroendocr. 2021, 33, e12916. [Google Scholar] [CrossRef]
- Atkinson, H.C.; Waddell, B.J. Circadian variation in basal plasma corticosterone and adrenocorticotropin in the rat: Sexual dimorphism and changes across the estrous cycle. Endocrinology 1997, 138, 3842–3848. [Google Scholar] [CrossRef]
- Shoji, H.; Miyakawa, T. Differential effects of stress exposure via two types of restraint apparatuses on behavior and plasma corticosterone level in inbred male BALB/cAJcl mice. Neuropsychopharmacol. Rep. 2020, 40, 73–84. [Google Scholar] [CrossRef]
- Koob, G.F.; Zorrilla, E.P. Neurobiological mechanisms of addiction: Focus on corticotropin-releasing factor. Curr. Opin. Investig. Drugs 2010, 11, 63–71. [Google Scholar]
- Jutkiewicz, E.M.; Wood, S.K.; Houshyar, H.; Hsin, L.W.; Rice, K.C.; Woods, J.H. The effects of CRF antagonists, antalarmin, CP154,526, LWH234, and R121919, in the forced swim test and on swim-induced increases in adrenocorticotropin in rats. Psychopharmacology 2005, 180, 215–223. [Google Scholar] [CrossRef]
- Nielsen, D.M.; Carey, G.J.; Gold, L.H. Antidepressant-like activity of corticotropin-releasing factor type-1 receptor antagonists in mice. Eur. J. Pharmacol. 2004, 499, 135–146. [Google Scholar] [CrossRef]
- Dunlop, B.W.; Binder, E.B.; Iosifescu, D.; Mathew, S.J.; Neylan, T.C.; Pape, J.C.; Carrillo-Roa, T.; Green, C.; Kinkead, B.; Grigoriadis, D.; et al. Corticotropin-Releasing Factor Receptor 1 Antagonism Is Ineffective for Women With Posttraumatic Stress Disorder. Biol. Psychiatry 2017, 82, 866–874. [Google Scholar] [CrossRef]
- Tasker, J.G.; Di, S.; Malcher-Lopes, R. Minireview: Rapid glucocorticoid signaling via membrane-associated receptors. Endocrinology 2006, 147, 5549–5556. [Google Scholar] [CrossRef] [PubMed]
- Rupprecht, R.; Berning, B.; Hauser, C.A.E.; Holsboer, F.; Reul, J.M.H.M. Steroid receptor-mediated effects of neuroactive steroids: Characterization of structure-activity relationship. Eur. J. Pharmacol. 1996, 303, 227–234. [Google Scholar] [CrossRef]
- Basta-Kaim, A.; Budziszewska, B.; Jaworska-Feil, L.; Leskiewicz, M.; Tetich, M.; Otczyk, M.; Kubera, M.; Lason, W. Effects of neurosteroids on glucocorticoid receptor-mediated gene transcription in LMCAT cells—A possible interaction with psychotropic drugs. Eur. Neuropsychopharmacol. 2007, 17, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Basta-Kaim, A.; Budziszewska, B.; Jaworska-Feil, L.; Tetich, M.; Kubera, M.; Leskiewicz, M.; Lason, W. Mood stabilizers inhibit glucocorticoid receptor function in LMCAT cells. Eur. J. Pharmacol. 2004, 495, 103–110. [Google Scholar] [CrossRef]
- Basta-Kaim, A.; Budziszewska, B.; Jaworska-Feil, L.; Tetich, M.; Leskiewicz, M.; Kubera, M.; Lason, W. Chlorpromazine inhibits the glucocorticoid receptor-mediated gene transcription in a calcium-dependent manner. Neuropharmacology 2002, 43, 1035–1043. [Google Scholar] [CrossRef]
- Uphouse, L.; Adams, S.; Miryala, C.S.; Hassell, J.; Hiegel, C. RU486 blocks effects of allopregnanolone on the response to restraint stress. Pharm. Biochem. Behav. 2013, 103, 568–572. [Google Scholar] [CrossRef][Green Version]
- Rupprecht, R. The neuropsychopharmacological potential of neuroactive steroids. J. Psychiatr. Res 1997, 31, 297–314. [Google Scholar] [CrossRef]
- Kliewer, S.A.; Goodwin, B.; Willson, T.M. The nuclear pregnane X receptor: A key regulator of xenobiotic metabolism. Endocr. Rev. 2002, 23, 687–702. [Google Scholar] [CrossRef]
- Frye, C.A.; Koonce, C.J.; Walf, A.A. Role of pregnane xenobiotic receptor in the midbrain ventral tegmental area for estradiol- and 3alpha,5alpha-THP-facilitated lordosis of female rats. Psychopharmacology 2014, 231, 3365–3374. [Google Scholar] [CrossRef]
- Frye, C.A.; Koonce, C.J.; Walf, A.A. Novel receptor targets for production and action of allopregnanolone in the central nervous system: A focus on pregnane xenobiotic receptor. Front. Cell Neurosci. 2014, 8, 106. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Pai, H.V.; Zhou, J.; Amico, J.A.; Vollmer, R.R.; Xie, W. Activation of pregnane X receptor disrupts glucocorticoid and mineralocorticoid homeostasis. Mol. Endocrinol. 2007, 21, 138–147. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Non-genomic and genomic effects of steroids on neural activity. Trends. Pharmacol. Sci. 1991, 12, 141–147. [Google Scholar] [CrossRef]
- Busillo, J.M.; Azzam, K.M.; Cidlowski, J.A. Glucocorticoids sensitize the innate immune system through regulation of the NLRP3 inflammasome. J. Biol. Chem. 2011, 286, 38703–38713. [Google Scholar] [CrossRef]
- Sorrells, S.F.; Caso, J.R.; Munhoz, C.D.; Sapolsky, R.M. The stressed CNS: When glucocorticoids aggravate inflammation. Neuron 2009, 64, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Balan, I.; Aurelian, L.; Schleicher, R.; Boero, G.; O’Buckley, T.; Morrow, A.L. Neurosteroid allopregnanolone (3alpha,5alpha-THP) inhibits inflammatory signals induced by activated MyD88-dependent toll-like receptors. Transl. Psychiatry 2021, 11, 145. [Google Scholar] [CrossRef]
- Calcia, M.A.; Bonsall, D.R.; Bloomfield, P.S.; Selvaraj, S.; Barichello, T.; Howes, O.D. Stress and neuroinflammation: A systematic review of the effects of stress on microglia and the implications for mental illness. Psychopharmacology 2016, 233, 1637–1650. [Google Scholar] [CrossRef]
- Weber, M.D.; Frank, M.G.; Tracey, K.J.; Watkins, L.R.; Maier, S.F. Stress induces the danger-associated molecular pattern HMGB-1 in the hippocampus of male Sprague Dawley rats: A priming stimulus of microglia and the NLRP3 inflammasome. J. Neurosci. 2015, 35, 316–324. [Google Scholar] [CrossRef]
- Torres-Platas, S.G.; Cruceanu, C.; Chen, G.G.; Turecki, G.; Mechawar, N. Evidence for increased microglial priming and macrophage recruitment in the dorsal anterior cingulate white matter of depressed suicides. Brain Behav. Immun. 2014, 42, 50–59. [Google Scholar] [CrossRef]
- Frick, L.R.; Williams, K.; Pittenger, C. Microglial dysregulation in psychiatric disease. Clin. Dev. Immunol. 2013, 2013, 608654. [Google Scholar] [CrossRef]
- Van Berckel, B.N.; Bossong, M.G.; Boellaard, R.; Kloet, R.; Schuitemaker, A.; Caspers, E.; Luurtsema, G.; Windhorst, A.D.; Cahn, W.; Lammertsma, A.A.; et al. Microglia activation in recent-onset schizophrenia: A quantitative (R)-[11C]PK11195 positron emission tomography study. Biol. Psychiatry 2008, 64, 820–822. [Google Scholar] [CrossRef] [PubMed]
- Reus, G.Z.; Fries, G.R.; Stertz, L.; Badawy, M.; Passos, I.C.; Barichello, T.; Kapczinski, F.; Quevedo, J. The role of inflammation and microglial activation in the pathophysiology of psychiatric disorders. Neuroscience 2015, 300, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Giatti, S.; Diviccaro, S.; Serafini, M.M.; Caruso, D.; Garcia-Segura, L.M.; Viviani, B.; Melcangi, R.C. Sex differences in steroid levels and steroidogenesis in the nervous system: Physiopathological role. Front. Neuroendocr. 2020, 56, 100804. [Google Scholar] [CrossRef] [PubMed]
- Meltzer-Brody, S.; Colquhoun, H.; Riesenberg, R.; Epperson, C.N.; Deligiannidis, K.M.; Rubinow, D.R.; Li, H.; Sankoh, A.J.; Clemson, C.; Schacterle, A.; et al. Brexanolone injection in post-partum depression: Two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet 2018, 392, 1058–1070. [Google Scholar] [CrossRef]
- Pinna, G.; Almeida, F.B.; Davis, J.M. Allopregnanolone in Postpartum Depression. Front. Glob Womens Health 2022, 3, 823616. [Google Scholar] [CrossRef] [PubMed]

| (a) | ||||
| Males ♂ | ||||
| Restraint Stress | Forced Swim Stress | |||
| Marker | Stress | 3α,5α-THP | Stress | 3α,5α-THP |
| CORT | +595% **** vs. VNRS | No effect | +266% **** vs. VNRS | No effect |
| CRF | +76 *** vs. VNRS | +150% **** vs. VNRS | +55% vs. VNFSS | −73% **** vs. VFSS |
| GRs | +29% * vs. VNRS |
−36% ** vs. VNRS; −37% **** vs. VRS | No effect | −19% * vs. VNFSS |
| CRFR1 | +60% ** vs. VNRS | −67% *** vs. VRS | No effect | −17% * vs. VFSS |
| CRFBP | −29% ** vs. VNRS | −27% * vs. VNRS | +29% ** vs. VNFSS | −30% ** vs. VFSS |
| POMC | +35% ** vs. VNRS | −48% **** vs. VRS | +25% ** vs. VNFSS | −37% **** vs. VFSS |
| (b) | ||||
| Females ♀ | ||||
| Restraint Stress | Forced Swim Stress | |||
| Marker | Stress | 3α,5α-THP | Stress | 3α,5α-THP |
| CORT | +295 **** vs. VNRS | −69% ** vs. VRS | +321% **** | No effect |
| CRF | +144% **** vs. VNRS | −90% **** vs. VRS | +132% **** vs. NFSS | −176% **** vs. VFSS |
| GRs | +41% ** vs. VNRS | −13% vs. VRS | No effect |
−52% **** vs. VFSS; −47% **** vs. VNFSS |
| CRFR1 | No effect | No effect | No effect |
−45% **** vs. VNFSS; −39% * vs. VFSS |
| CRFBP | No effect | No effect | No effect |
−39% **** vs. NFSS; −46% * vs. VFSS |
| POMC | +98% ** vs. VNRS | +126% ** vs. VNRS | No effect | −25% *** vs. NFSS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boero, G.; Tyler, R.E.; O’Buckley, T.K.; Balan, I.; Besheer, J.; Morrow, A.L. (3α,5α)3-Hydroxypregnan-20-one (3α,5α-THP) Regulation of the HPA Axis in the Context of Different Stressors and Sex. Biomolecules 2022, 12, 1134. https://doi.org/10.3390/biom12081134
Boero G, Tyler RE, O’Buckley TK, Balan I, Besheer J, Morrow AL. (3α,5α)3-Hydroxypregnan-20-one (3α,5α-THP) Regulation of the HPA Axis in the Context of Different Stressors and Sex. Biomolecules. 2022; 12(8):1134. https://doi.org/10.3390/biom12081134
Chicago/Turabian StyleBoero, Giorgia, Ryan E. Tyler, Todd K. O’Buckley, Irina Balan, Joyce Besheer, and A. Leslie Morrow. 2022. "(3α,5α)3-Hydroxypregnan-20-one (3α,5α-THP) Regulation of the HPA Axis in the Context of Different Stressors and Sex" Biomolecules 12, no. 8: 1134. https://doi.org/10.3390/biom12081134
APA StyleBoero, G., Tyler, R. E., O’Buckley, T. K., Balan, I., Besheer, J., & Morrow, A. L. (2022). (3α,5α)3-Hydroxypregnan-20-one (3α,5α-THP) Regulation of the HPA Axis in the Context of Different Stressors and Sex. Biomolecules, 12(8), 1134. https://doi.org/10.3390/biom12081134






