Assessing the NLRP3 Inflammasome Activating Potential of a Large Panel of Micro- and Nanoplastics in THP-1 Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Test Materials
2.2. Generation and Characterization of PET Particles
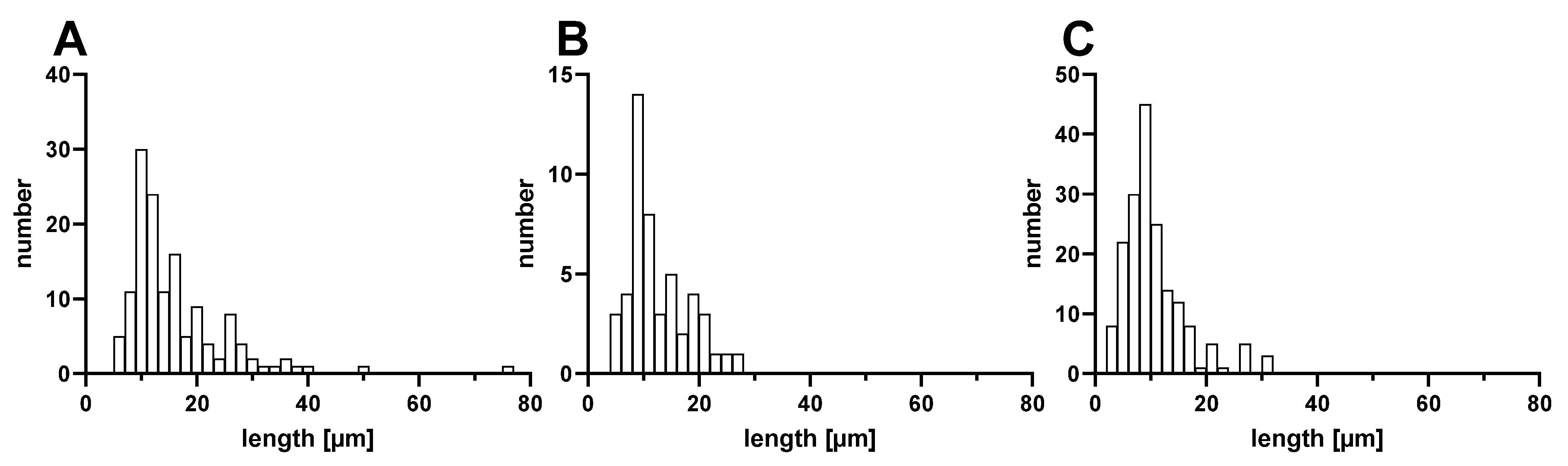
2.3. Production and Characterization of PES, PAN and PA6 Microfibers
2.4. Cell Culture
2.5. Generation of an NLRP3-Deficient THP-1 Cell Line Using the CRISPR/Cas9 System
2.6. Experimental Procedure
2.7. Cytokine Quantification by Enzyme-Linked Immune-Sorbent Assay (ELISA)
2.8. Assessment of Cytotoxicity via LDH Assay
2.9. Statistical Analysis
3. Results
3.1. PET Particles Generated by Laser Ablation
3.2. Characterization of PES, PAN, and PA6 Microparticles Produced via Cryotome
3.3. Interference of Materials with the ELISA and LDH Assay
3.4. Characterization of THP-1 Cells as a Screening Tool for NLRP3 Activation
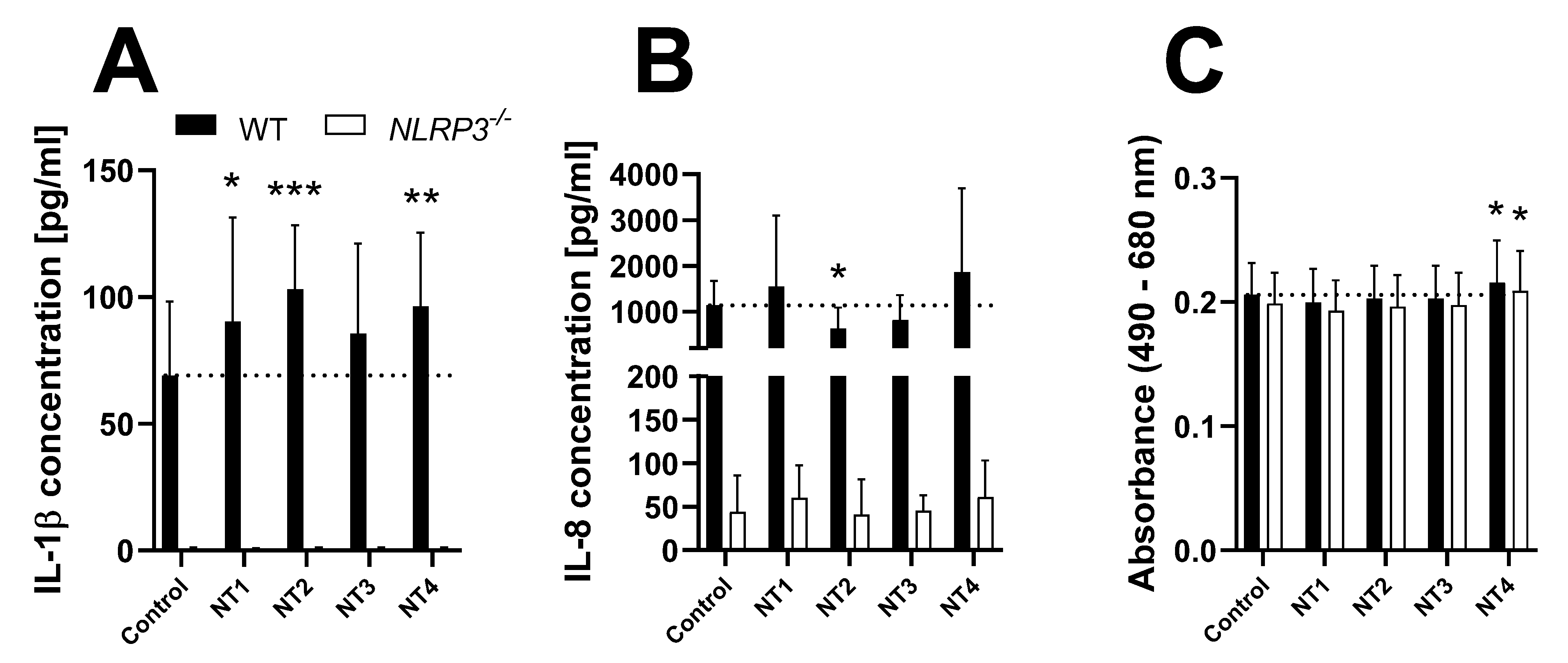
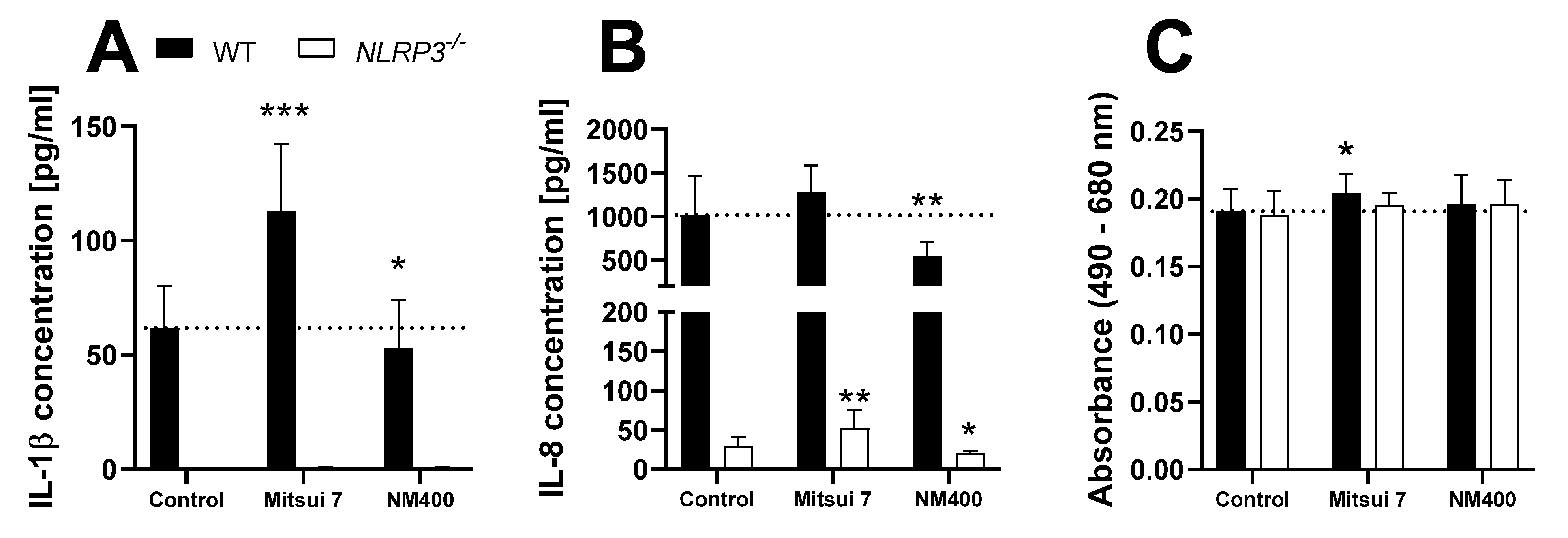
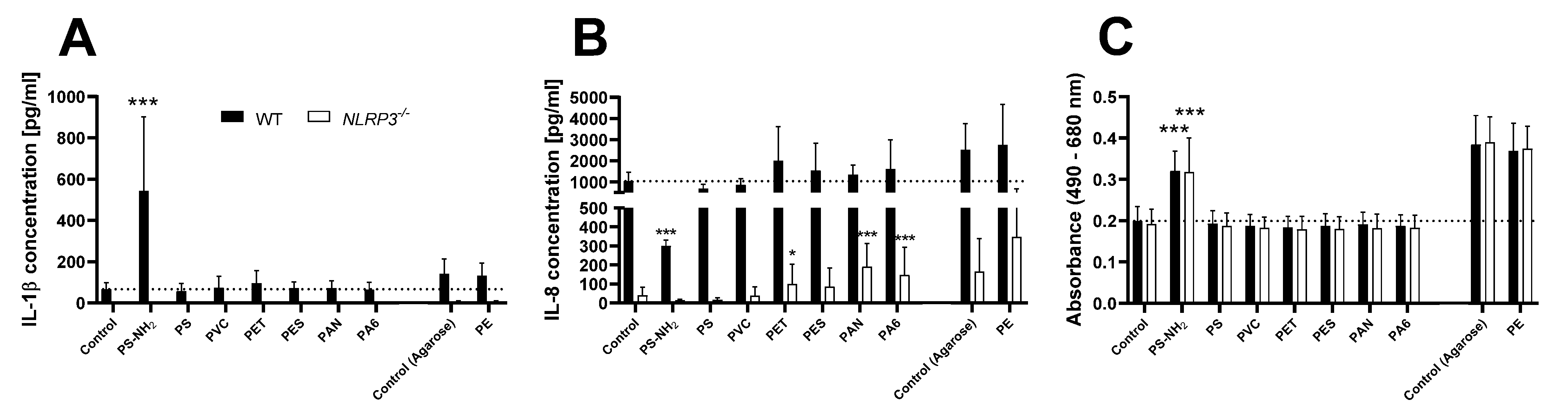
3.5. Screening of Eight Different MNPs for NLRP3 Activation in THP-1 Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Julienne, F.; Delorme, N.; Lagarde, F. From macroplastics to microplastics: Role of water in the fragmentation of polyethylene. Chemosphere 2019, 236, 124409. [Google Scholar] [CrossRef] [PubMed]
- Andrady, A.L. Biodegradation of Plastics: Monitoring What Happens. In Plastics Additives: An A–Z Reference; Brewis, D., Briggs, D., Pritchard, G., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 32–40. ISBN 978-94-010-6477-4. [Google Scholar]
- Guerranti, C.; Martellini, T.; Perra, G.; Scopetani, C.; Cincinelli, A. Microplastics in cosmetics: Environmental issues and needs for global bans. Environ. Toxicol. Pharmacol. 2019, 68, 75–79. [Google Scholar] [CrossRef] [PubMed]
- De Falco, F.; Di Pace, E.; Cocca, M.; Avella, M. The contribution of washing processes of synthetic clothes to microplastic pollution. Sci. Rep. 2019, 9, 6633. [Google Scholar] [CrossRef] [PubMed]
- Andrady, A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef]
- Nizzetto, L.; Futter, M.; Langaas, S. Are Agricultural Soils Dumps for Microplastics of Urban Origin? Environ. Sci. Technol. 2016, 50, 10777–10779. [Google Scholar] [CrossRef]
- Amato-Lourenço, L.F.; Dos Santos Galvão, L.; de Weger, L.A.; Hiemstra, P.S.; Vijver, M.G.; Mauad, T. An emerging class of air pollutants: Potential effects of microplastics to respiratory human health? Sci. Total Environ. 2020, 749, 141676. [Google Scholar] [CrossRef]
- Kalčíková, G. Aquatic vascular plants—A forgotten piece of nature in microplastic research. Environ. Pollut. 2020, 262, 114354. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Lopes, C.; Oliveira, P.; Bessa, F.; Otero, V.; Henriques, B.; Raimundo, J.; Caetano, M.; Vale, C.; Guilhermino, L. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Sci. Total Environ. 2019, 717, 134625. [Google Scholar] [CrossRef]
- Ibrahim, Y.S.; Tuan Anuar, S.; Azmi, A.A.; Wan Mohd Khalik, W.M.A.; Lehata, S.; Hamzah, S.R.; Ismail, D.; Ma, Z.F.; Dzulkarnaen, A.; Zakaria, Z.; et al. Detection of microplastics in human colectomy specimens. JGH Open 2021, 5, 116–121. [Google Scholar] [CrossRef]
- Destoumieux-Garzón, D.; Mavingui, P.; Boetsch, G.; Boissier, J.; Darriet, F.; Duboz, P.; Fritsch, C.; Giraudoux, P.; Le Roux, F.; Morand, S.; et al. The One Health Concept: 10 Years Old and a Long Road Ahead. Front. Vet. Sci. 2018, 5, 14. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Ding, Y.; Cheng, X.; Sheng, D.; Xu, Z.; Rong, Q.; Wu, Y.; Zhao, H.; Ji, X.; Zhang, Y. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 2019, 244, 125492. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.; Jeong, J.; Song, K.S.; Sung, J.H.; Oh, S.M.; Choi, J. Inhalation toxicity of polystyrene micro(nano)plastics using modified OECD TG 412. Chemosphere 2021, 262, 128330. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, E.; Campbell, M.; Doyle, S.L. Targeting the NLRP3 inflammasome in chronic inflammatory diseases: Current perspectives. J. Inflamm. Res. 2015, 8, 15–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Hara, H.; Núñez, G. Mechanism and Regulation of NLRP3 Inflammasome Activation. Trends Biochem. Sci. 2016, 41, 1012–1021. [Google Scholar] [CrossRef] [Green Version]
- Palomäki, J.; Välimäki, E.; Sund, J.; Vippola, M.; Clausen, P.A.; Jensen, K.A.; Savolainen, K.; Matikainen, S.; Alenius, H. Long, needle-like carbon nanotubes and asbestos activate the NLRP3 inflammasome through a similar mechanism. ACS Nano 2011, 5, 6861–6870. [Google Scholar] [CrossRef]
- Dostert, C.; Pétrilli, V.; van Bruggen, R.; Steele, C.; Mossman, B.T.; Tschopp, J. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 2008, 320, 674–677. [Google Scholar] [CrossRef] [Green Version]
- Cassel, S.; Eisenbarth, S.; Iyer, S.; Sadler, J.; Colegio, O.; Tephly, L.; Carter, A.; Rothman, P.; Flavell, R.; Sutterwala, F. The Nalp3 inflammasome is essential for the development of silicosis. Proc. Natl. Acad. Sci. USA 2008, 26, 9035–9040. [Google Scholar] [CrossRef] [Green Version]
- Zheng, H.; Wang, J.; Wei, X.; Chang, L.; Liu, S. Proinflammatory properties and lipid disturbance of polystyrene microplastics in the livers of mice with acute colitis. Sci. Total Environ. 2021, 750, 143085. [Google Scholar] [CrossRef]
- Lomer, M.C.E.; Thompson, R.P.H.; Powell, J.J. Fine and ultrafine particles of the diet: Influence on the mucosal immune response and association with Crohn’s disease. Proc. Nutr. Soc. 2002, 61, 123–130. [Google Scholar] [CrossRef] [Green Version]
- Busch, M.; Bredeck, G.; Kämpfer, A.A.; Schins, R.P. Investigations of acute effects of polystyrene and polyvinyl chloride micro- and nanoplastics in an advanced in vitro triple culture model of the healthy and inflamed intestine. Environ. Res. 2021, 193, 110536. [Google Scholar] [CrossRef]
- Lehnert, B.E. Pulmonary and thoracic macrophage subpopulations and clearance of particles from the lung. Environ. Health Perspect. 1992, 97, 17–46. [Google Scholar] [CrossRef] [PubMed]
- LeFevre, M.E.; Warren, J.B.; Joel, D.D. Particles and macrophages in murine Peyer’s patches. Exp. Cell Biol. 1985, 53, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhao, W. NLRP3 Inflammasome-A Key Player in Antiviral Responses. Front. Immunol. 2020, 11, 211. [Google Scholar] [CrossRef] [Green Version]
- Siegmund, B. Interleukin-1β converting enzyme (caspase-1) in intestinal inflammation. Biochem. Pharmacol. 2002, 64, 1–8. [Google Scholar] [CrossRef]
- Parikh, A.A.; Salzman, A.L.; Kane, C.D.; Fischer, J.E.; Hasselgren, P. IL-6 Production in Human Intestinal Epithelial Cells Following Stimulation with IL-1β Is Associated with Activation of the Transcription Factor NF-kB. J. Surg. Res. 1997, 69, 139–144. [Google Scholar] [CrossRef]
- Vlahopoulos, S.; Boldogh, I.; Casola, A.; Brasier, A.R. Nuclear Factor-κB–Dependent Induction of Interleukin-8 Gene Expression by Tumor Necrosis Factor α: Evidence for an Antioxidant Sensitive Activating Pathway Distinct From Nuclear Translocation. Blood 1999, 94, 1878–1889. [Google Scholar] [CrossRef] [PubMed]
- Kunsch, C.; Rosen, C.A. NF-kappa B subunit-specific regulation of the interleukin-8 promoter. Mol. Cell. Biol. 1993, 13, 6137–6146. [Google Scholar] [CrossRef] [PubMed]
- Kolling, J.; Tigges, J.; Hellack, B.; Albrecht, C.; Schins, R.P.F. Evaluation of the NLRP3 Inflammasome Activating Effects of a Large Panel of TiO2 Nanomaterials in Macrophages. Nanomaterials 2020, 10, 1876. [Google Scholar] [CrossRef]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique, Special ed.; UFAW: Potters Bar, UK, 1992; ISBN 0900767782. [Google Scholar]
- Van Berlo, D.; Wilhelmi, V.; Boots, A.W.; Hullmann, M.; Kuhlbusch, T.A.J.; Bast, A.; Schins, R.P.F.; Albrecht, C. Apoptotic, inflammatory, and fibrogenic effects of two different types of multi-walled carbon nanotubes in mouse lung. Arch. Toxicol. 2014, 88, 1725–1737. [Google Scholar] [CrossRef]
- Gram, A.M.; Wright, J.A.; Pickering, R.J.; Lam, N.L.; Booty, L.M.; Webster, S.J.; Bryant, C.E. Salmonella Flagellin Activates NAIP/NLRC4 and Canonical NLRP3 Inflammasomes in Human Macrophages. J. Immunol. 2021, 206, 631–640. [Google Scholar] [CrossRef]
- Eisfeld, H.S.; Simonis, A.; Winter, S.; Chhen, J.; Ströh, L.J.; Krey, T.; Koch, M.; Theobald, S.J.; Rybniker, J. Viral Glycoproteins Induce NLRP3 Inflammasome Activation and Pyroptosis in Macrophages. Viruses 2021, 13, 2076. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; McLeland, C.B.; Potter, T.M.; Stern, S.T.; Adiseshaiah, P.P. Assessing NLRP3 Inflammasome Activation by Nanoparticles. In Characterization of Nanoparticles Intended for Drug Delivery, 2nd ed.; McNeil, S.E., Ed.; Humana Press: New York, NY, USA, 2018; pp. 135–147. ISBN 978-1-4939-7352-1. [Google Scholar]
- Stone, V.; Johnston, H.; Schins, R.P.F. Development of in vitro systems for nanotoxicology: Methodological considerations. Crit. Rev. Toxicol. 2009, 39, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Busch, M.; Kämpfer, A.A.; Schins, R.P. An inverted in vitro triple culture model of the healthy and inflamed intestine: Adverse effects of polyethylene particles. Chemosphere 2021, 284, 131345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Gökce, B.; Barcikowski, S. Laser Synthesis and Processing of Colloids: Fundamentals and Applications. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar] [CrossRef] [PubMed]
- Rehbock, C.; Jakobi, J.; Gamrad, L.; van der Meer, S.; Tiedemann, D.; Taylor, U.; Kues, W.; Rath, D.; Barcikowski, S. Current state of laser synthesis of metal and alloy nanoparticles as ligand-free reference materials for nano-toxicological assays. Beilstein J. Nanotechnol. 2014, 5, 1523–1541. [Google Scholar] [CrossRef] [PubMed]
- Waag, F.; Streubel, R.; Gökce, B.; Barcikowski, S. Synthesis of gold, platinum, and gold-platinum alloy nanoparticle colloids with high-power megahertz-repetition-rate lasers: The importance of the beam guidance method. Appl. Nanosci. 2021, 11, 1303–1312. [Google Scholar] [CrossRef]
- Magrì, D.; Sánchez-Moreno, P.; Caputo, G.; Gatto, F.; Veronesi, M.; Bardi, G.; Catelani, T.; Guarnieri, D.; Athanassiou, A.; Pompa, P.P.; et al. Laser Ablation as a Versatile Tool to Mimic Polyethylene Terephthalate Nanoplastic Pollutants: Characterization and Toxicology Assessment. ACS Nano 2018, 12, 7690–7700. [Google Scholar] [CrossRef]
- Elaboudi, I.; Lazare, S.; Belin, C.; Talaga, D.; Labrugère, C. Underwater excimer laser ablation of polymers. Appl. Phys. 2008, 92, 743–748. [Google Scholar] [CrossRef]
- Nachev, P.; van ’T Zand, D.D.; Coger, V.; Wagener, P.; Reimers, K.; Vogt, P.M.; Barcikowski, S.; Pich, A. Synthesis of hybrid microgels by coupling of laser ablation and polymerization in aqueous medium. J. Laser Appl. 2012, 24, 42012. [Google Scholar] [CrossRef]
- Cole, M. A novel method for preparing microplastic fibers. Sci. Rep. 2016, 6, 34519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramachandran, H.; Martins, S.; Kontarakis, Z.; Krutmann, J.; Rossi, A. Fast but not furious: A streamlined selection method for genome-edited cells. Life Sci. Alliance 2021, 4, e202101051. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-A.; Ahn, B.-N.; Kong, C.-S.; Kim, S.-K. Anti-inflammatory action of sulfated glucosamine on cytokine regulation in LPS-activated PMA-differentiated THP-1 macrophages. Inflamm. Res. 2011, 60, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Genin, M.; Clement, F.; Fattaccioli, A.; Raes, M.; Michiels, C. M1 and M2 macrophages derived from THP-1 cells differentially modulate the response of cancer cells to etoposide. BMC Cancer 2015, 15, 577. [Google Scholar] [CrossRef] [Green Version]
- Susewind, J.; de Souza Carvalho-Wodarz, C.; Repnik, U.; Collnot, E.-M.; Schneider-Daum, N.; Griffiths, G.W.; Lehr, C.-M. A 3D co-culture of three human cell lines to model the inflamed intestinal mucosa for safety testing of nanomaterials. Nanotoxicology 2016, 10, 53–62. [Google Scholar] [CrossRef]
- Kämpfer, A.A.M.; Urbán, P.; Gioria, S.; Kanase, N.; Stone, V.; Kinsner-Ovaskainen, A. Development of an in vitro co-culture model to mimic the human intestine in healthy and diseased state. Toxicol. In Vitro 2017, 45, 31–43. [Google Scholar] [CrossRef]
- Diabaté, S.; Mülhopt, S.; Paur, H.-R.; Krug, H.F. The response of a co-culture lung model to fine and ultrafine particles of incinerator fly ash at the air-liquid interface. Altern. Lab. Anim. 2008, 36, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Cappellini, F.; Di Bucchianico, S.; Karri, V.; Latvala, S.; Malmlöf, M.; Kippler, M.; Elihn, K.; Hedberg, J.; Odnevall Wallinder, I.; Gerde, P.; et al. Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface-Dosimetry Considerations and Comparison to Submerged Exposure. Nanomaterials 2020, 10, 618. [Google Scholar] [CrossRef] [Green Version]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [Green Version]
- Lunov, O.; Syrovets, T.; Loos, C.; Nienhaus, G.U.; Mailänder, V.; Landfester, K.; Rouis, M.; Simmet, T. Amino-functionalized polystyrene nanoparticles activate the NLRP3 inflammasome in human macrophages. ACS Nano 2011, 5, 9648–9657. [Google Scholar] [CrossRef]
- Ruiz, P.A.; Morón, B.; Becker, H.M.; Lang, S.; Atrott, K.; Spalinger, M.R.; Scharl, M.; Wojtal, K.A.; Fischbeck-Terhalle, A.; Frey-Wagner, I.; et al. Titanium dioxide nanoparticles exacerbate DSS-induced colitis: Role of the NLRP3 inflammasome. Gut 2017, 66, 1216–1224. [Google Scholar] [CrossRef] [Green Version]
- Di Cristo, L.; Oomen, A.G.; Dekkers, S.; Moore, C.; Rocchia, W.; Murphy, F.; Johnston, H.J.; Janer, G.; Haase, A.; Stone, V.; et al. Grouping Hypotheses and an Integrated Approach to Testing and Assessment of Nanomaterials Following Oral Ingestion. Nanomaterials 2021, 11, 2623. [Google Scholar] [CrossRef]
- Kroll, A.; Pillukat, M.H.; Hahn, D.; Schnekenburger, J. Interference of engineered nanoparticles with in vitro toxicity assays. Arch. Toxicol. 2012, 86, 1123–1136. [Google Scholar] [CrossRef] [PubMed]
- Guadagnini, R.; Halamoda Kenzaoui, B.; Walker, L.; Pojana, G.; Magdolenova, Z.; Bilanicova, D.; Saunders, M.; Juillerat-Jeanneret, L.; Marcomini, A.; Huk, A.; et al. Toxicity screenings of nanomaterials: Challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology 2015, 9, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Forte, M.; Iachetta, G.; Tussellino, M.; Carotenuto, R.; Prisco, M.; de Falco, M.; Laforgia, V.; Valiante, S. Polystyrene nanoparticles internalization in human gastric adenocarcinoma cells. Toxicol. In Vitro 2016, 31, 126–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, D.M.; Wilson, M.R.; MacNee, W.; Stone, V.; Donaldson, K. Size-dependent proinflammatory effects of ultrafine polystyrene particles: A role for surface area and oxidative stress in the enhanced activity of ultrafines. Toxicol. Appl. Pharmacol. 2001, 175, 191–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, R.; Sheng, C.; Lu, Y.; Zhang, Y.; Ren, H.; Lemos, B. Microplastics induce intestinal inflammation, oxidative stress, and disorders of metabolome and microbiome in zebrafish. Sci. Total Environ. 2019, 662, 246–253. [Google Scholar] [CrossRef]
- Wei, J.; Wang, X.; Liu, Q.; Zhou, N.; Zhu, S.; Li, Z.; Li, X.; Yao, J.; Zhang, L. The impact of polystyrene microplastics on cardiomyocytes pyroptosis through NLRP3/Caspase-1 signaling pathway and oxidative stress in Wistar rats. Environ. Toxicol. 2021, 36, 935–944. [Google Scholar] [CrossRef]
- Hou, J.; Lei, Z.; Cui, L.; Hou, Y.; Yang, L.; An, R.; Wang, Q.; Li, S.; Zhang, H.; Zhang, L. Polystyrene microplastics lead to pyroptosis and apoptosis of ovarian granulosa cells via NLRP3/Caspase-1 signaling pathway in rats. Ecotoxicol. Environ. Saf. 2021, 212, 112012. [Google Scholar] [CrossRef]
- Netea, M.G.; Nold-Petry, C.A.; Nold, M.F.; Joosten, L.A.B.; Opitz, B.; van der Meer, J.H.M.; van de Veerdonk, F.L.; Ferwerda, G.; Heinhuis, B.; Devesa, I.; et al. Differential requirement for the activation of the inflammasome for processing and release of IL-1β in monocytes and macrophages. Blood 2009, 113, 2324–2335. [Google Scholar] [CrossRef] [Green Version]
- Beijer, N.R.M.; Dehaut, A.; Carlier, M.P.; Wolter, H.; Versteegen, R.M.; Pennings, J.L.A.; de La Fonteyne, L.; Niemann, H.; Janssen, H.M.; Timmermans, B.G.; et al. Relationship between Particle Properties and Immunotoxicological Effects of Environmentally-Sourced Microplastics. Front. Water 2022, 4, 866732. [Google Scholar] [CrossRef]


| Material | Abbreviation | Size | Source | Characterization/ Previous Use | |
|---|---|---|---|---|---|
| Titanium dioxides | TiO2 (P25) | NT1 | 12–18 nm | Evonik, Essen, Germany | [29] |
| TiO2 (PC105) | NT2 | 10 nm | Cristal, Jeddah, Saudi Arabia | [29] | |
| TiO2 (SX001) | NT3 | 12–15 nm | Solaronix, Aubonne, Switzerland | [29] | |
| TiO2 (UT001) | NT4 | 16–17 nm | UNITO, Turin, Italy | [29] | |
| Carbon nanotubes | Multi-walled carbon nanotubes 1 “Mitsui” | Mitsui 7 | 40–100 nm (diameter); 13 µm (length) | Mitsui & Co., Tokyo, Japan | [31] |
| Multi-walled carbon nanotubes 2 “NM400” | NM400 | 30 nm (diameter); 5 µm (length) | JRC repository, Ispra, Italy | [31] | |
| Micro/nanoplastics | Amine-modified polystyrene spheres | PS-NH2 | 50 nm | Sigma Aldrich, Schnelldorf, Germany (L0780) | [21] |
| Polystyrene spheres | PS | 50 nm | Polysciences Inc., Warrington, PA, USA (08691–10) | [21] | |
| Polyvinyl chloride powder | PVC | 235 nm (mode) | Werth–Metall, Erfurt, Germany | [21] | |
| Polyethylene spheres | PE | 611 nm (mode) | Cospheric LLC, Santa Barbara, CA, USA | [36] | |
| Polyethylene terephthalate fragments | PET | 16 nm (nanofraction); 5.7 µm (microfraction) | Produced via laser ablation | ||
| Polyester fibers | PES | 17.5 µm (diameter); 10 µm (length) | Produced via cryotome | ||
| Polyacrylonitrile fibers | PAN | 18.5 µm (diameter); 10 µm (length) | Produced via cryotome | ||
| Polyamide 6 (nylon) fibers | PA6 | 27.5 µm (diameter); 10 µm (length) | Produced via cryotome |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Busch, M.; Bredeck, G.; Waag, F.; Rahimi, K.; Ramachandran, H.; Bessel, T.; Barcikowski, S.; Herrmann, A.; Rossi, A.; Schins, R.P.F. Assessing the NLRP3 Inflammasome Activating Potential of a Large Panel of Micro- and Nanoplastics in THP-1 Cells. Biomolecules 2022, 12, 1095. https://doi.org/10.3390/biom12081095
Busch M, Bredeck G, Waag F, Rahimi K, Ramachandran H, Bessel T, Barcikowski S, Herrmann A, Rossi A, Schins RPF. Assessing the NLRP3 Inflammasome Activating Potential of a Large Panel of Micro- and Nanoplastics in THP-1 Cells. Biomolecules. 2022; 12(8):1095. https://doi.org/10.3390/biom12081095
Chicago/Turabian StyleBusch, Mathias, Gerrit Bredeck, Friedrich Waag, Khosrow Rahimi, Haribaskar Ramachandran, Tobias Bessel, Stephan Barcikowski, Andreas Herrmann, Andrea Rossi, and Roel P. F. Schins. 2022. "Assessing the NLRP3 Inflammasome Activating Potential of a Large Panel of Micro- and Nanoplastics in THP-1 Cells" Biomolecules 12, no. 8: 1095. https://doi.org/10.3390/biom12081095
APA StyleBusch, M., Bredeck, G., Waag, F., Rahimi, K., Ramachandran, H., Bessel, T., Barcikowski, S., Herrmann, A., Rossi, A., & Schins, R. P. F. (2022). Assessing the NLRP3 Inflammasome Activating Potential of a Large Panel of Micro- and Nanoplastics in THP-1 Cells. Biomolecules, 12(8), 1095. https://doi.org/10.3390/biom12081095







