Bioimprinting as a Receptor for Detection of Kwakhurin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
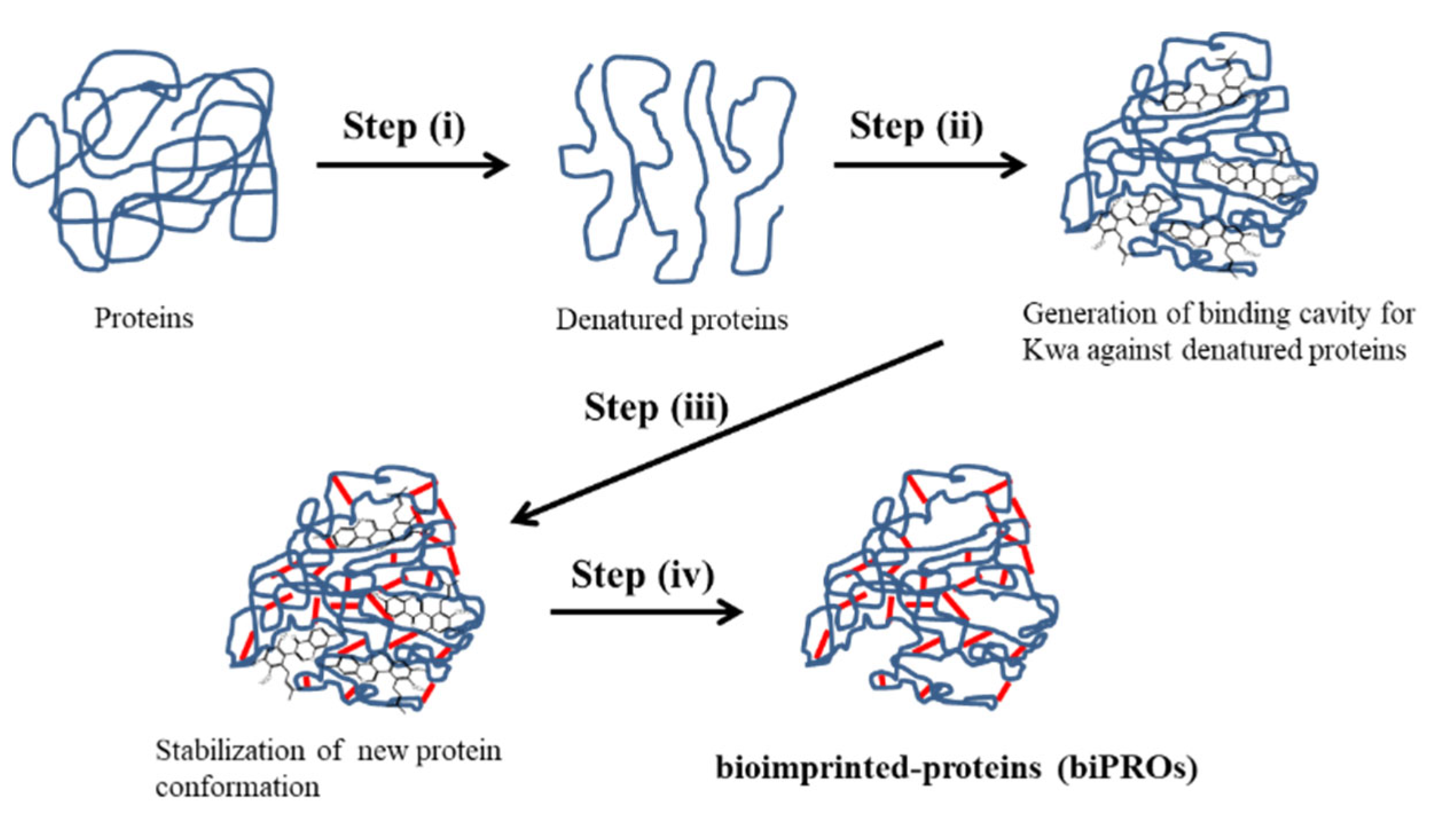
2.2. Bioimprinting Procedure
2.3. Preparation of Kwa-HSA and Kwa-HRP Conjugates
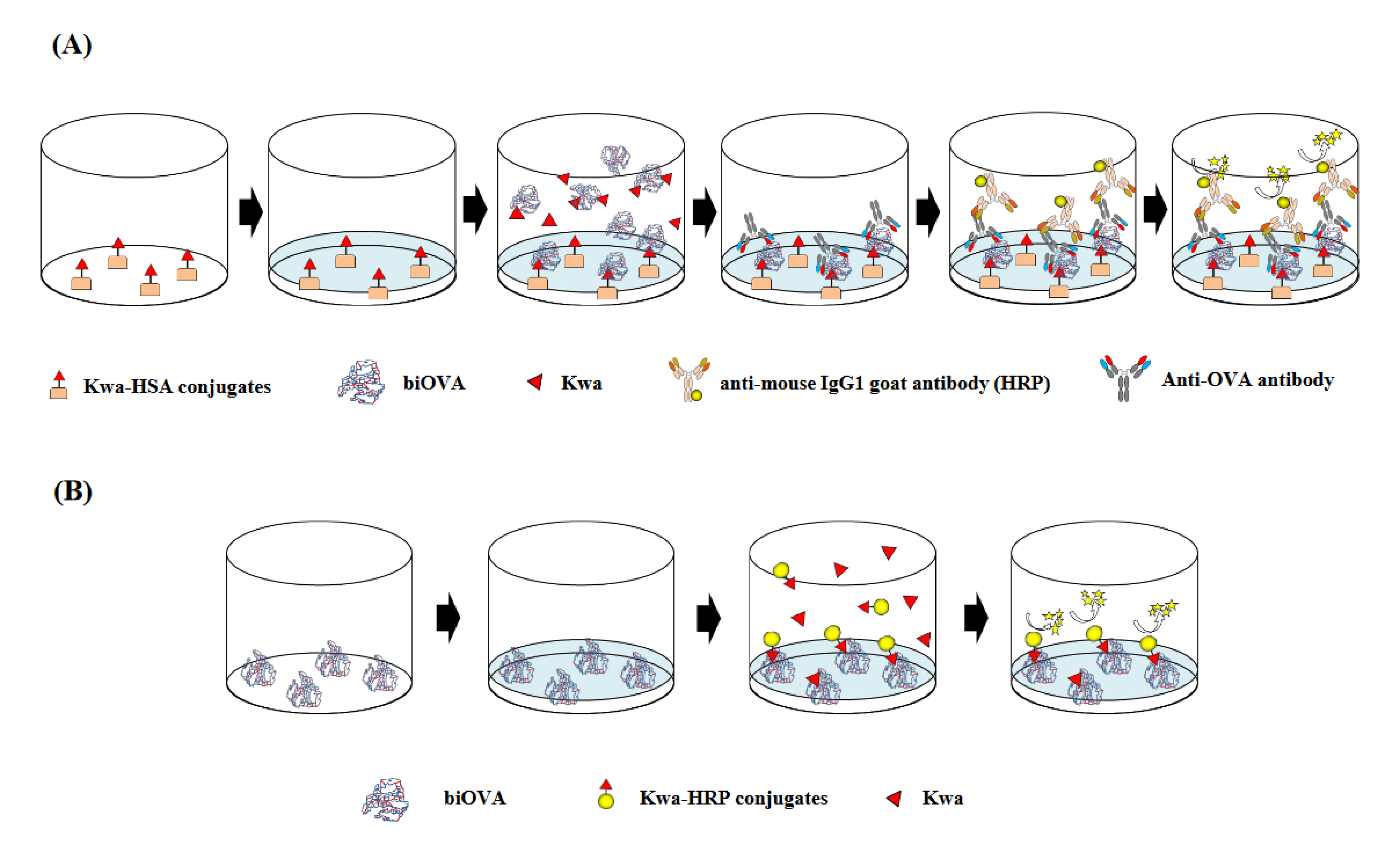
2.4. icELISA Using biOVA
2.5. Indirect ELISA (iELISA) Using biOVA
2.6. cELBIA Using biOVA
2.7. Non-Competitive ELBIA (ncELBIA) Using biOVA
3. Results
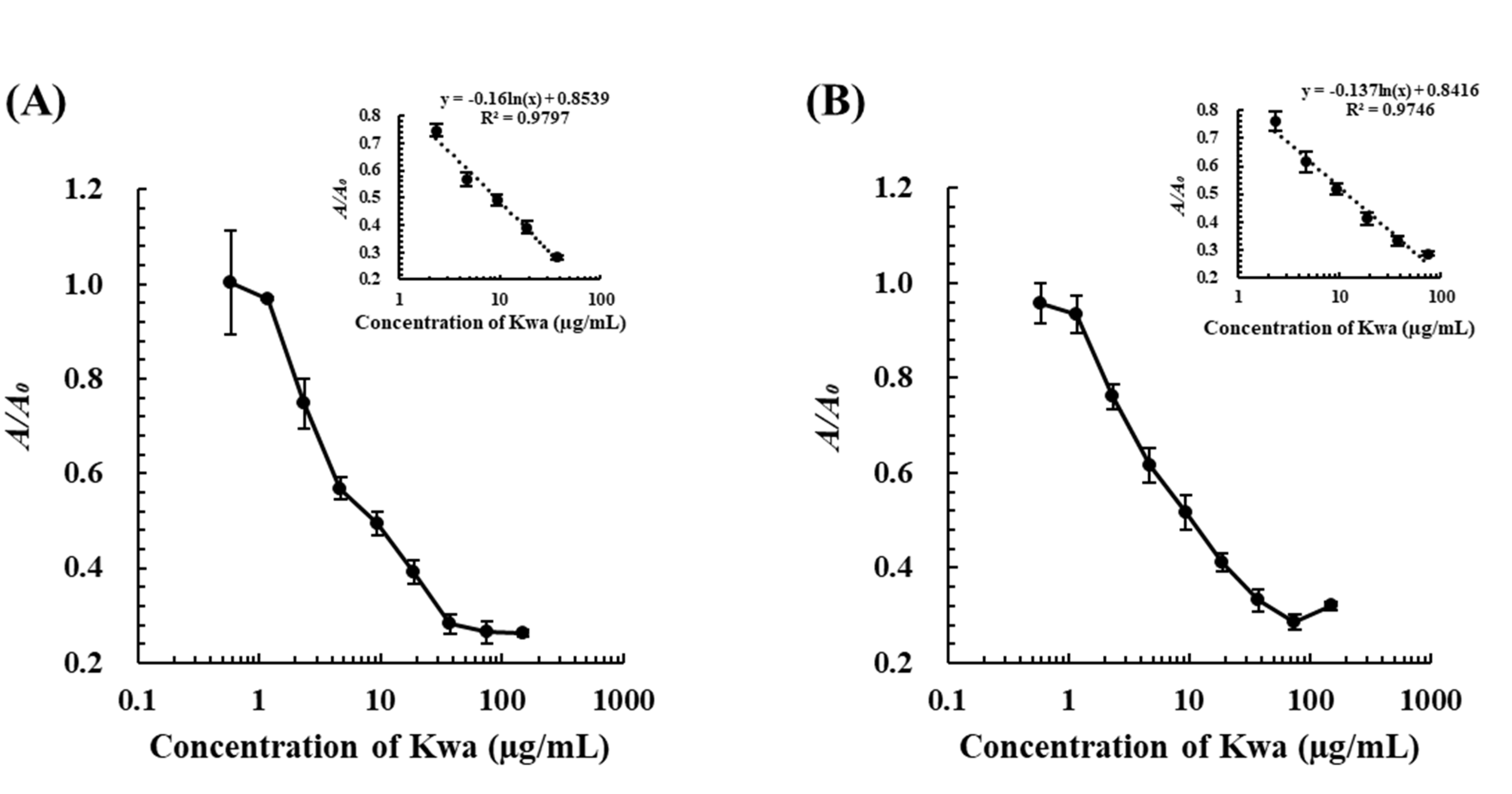
3.1. Development of the icELISA and cELBIA Using biOVA
3.1.1. Optimization of Various Parameters for icELISA
3.1.2. Optimization of Various Parameters for cELBIA
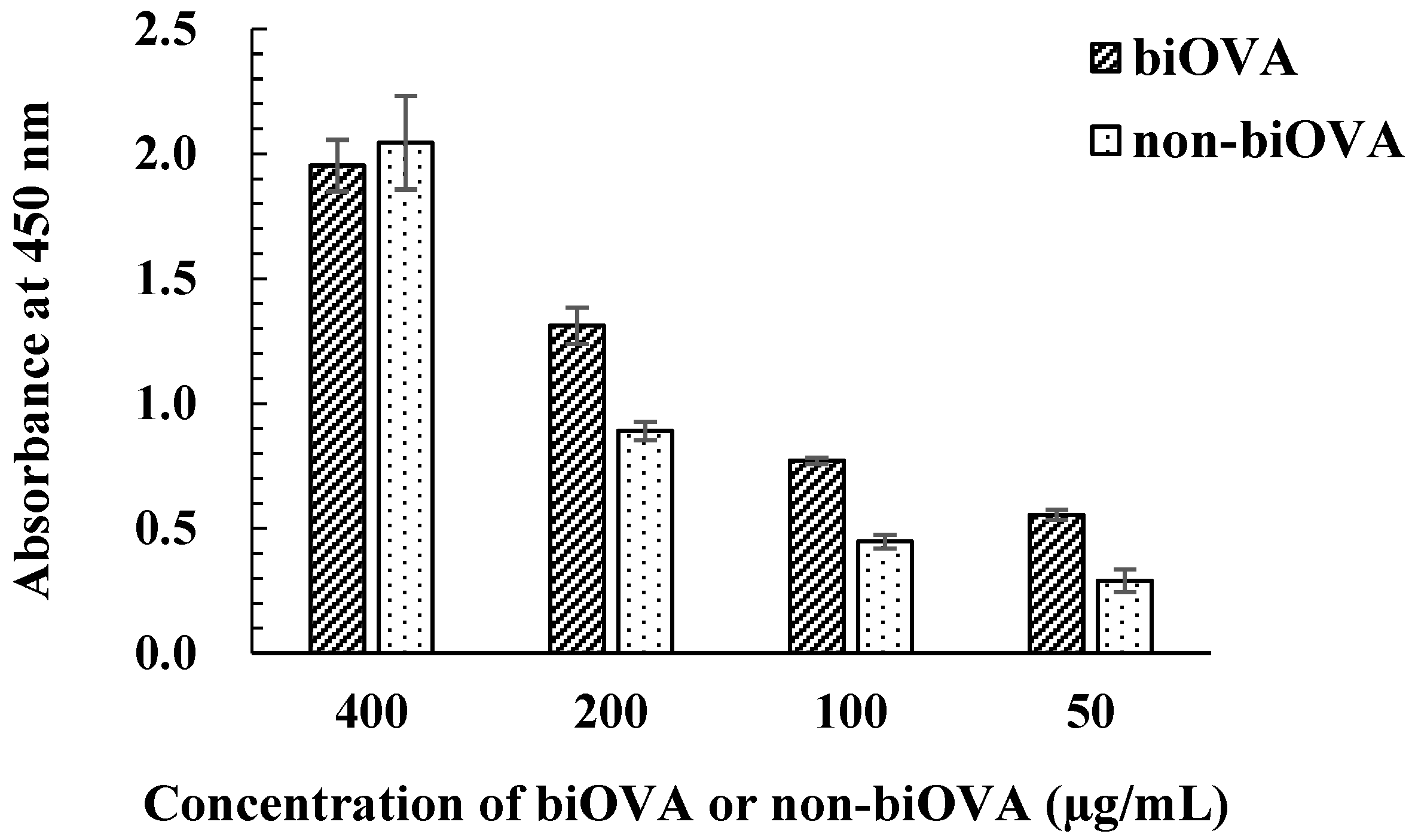
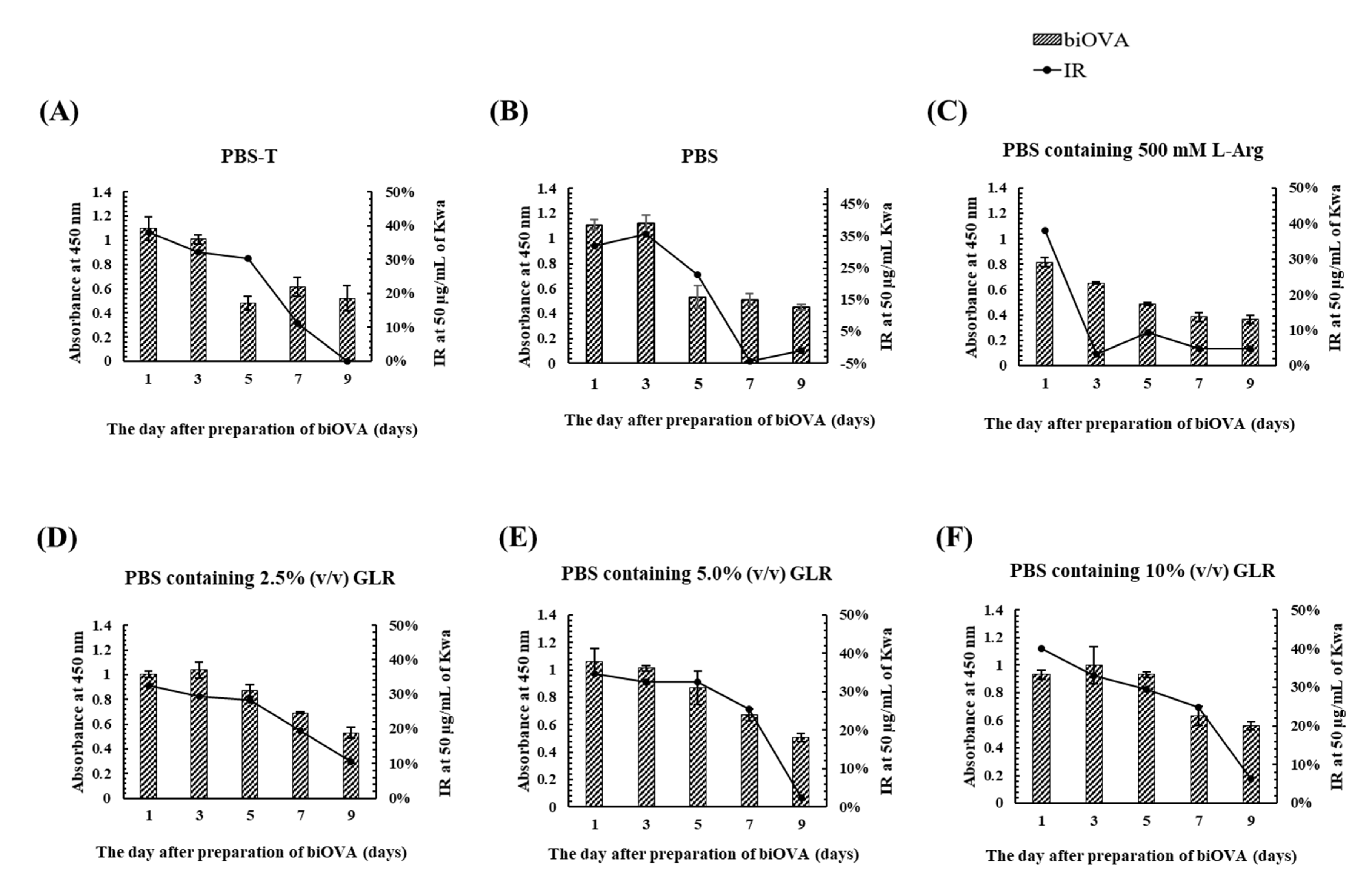
3.1.3. Characterization of biOVA
3.1.4. Determination of Kwa by the icELISA and cELBIA Using biOVA
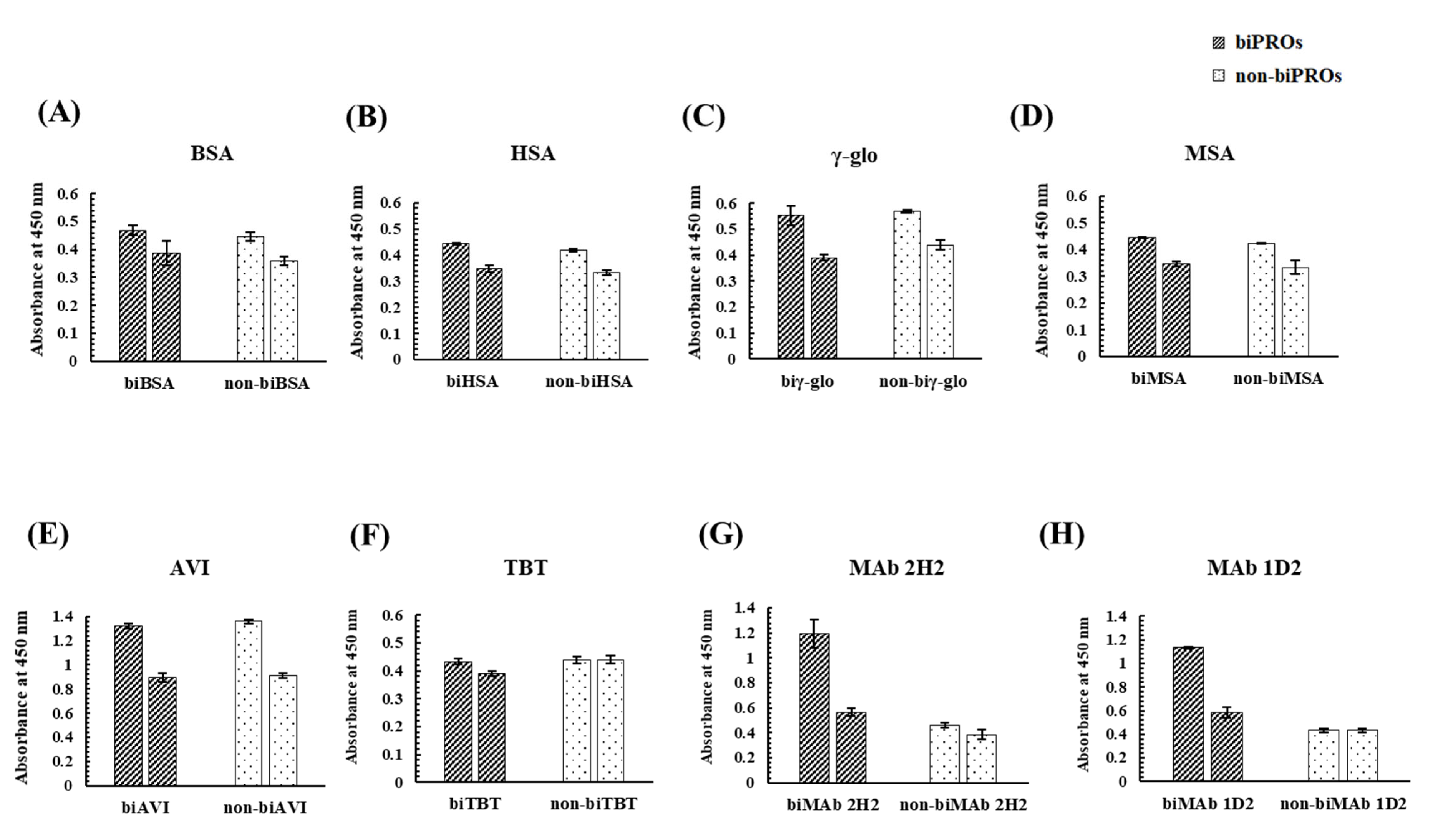
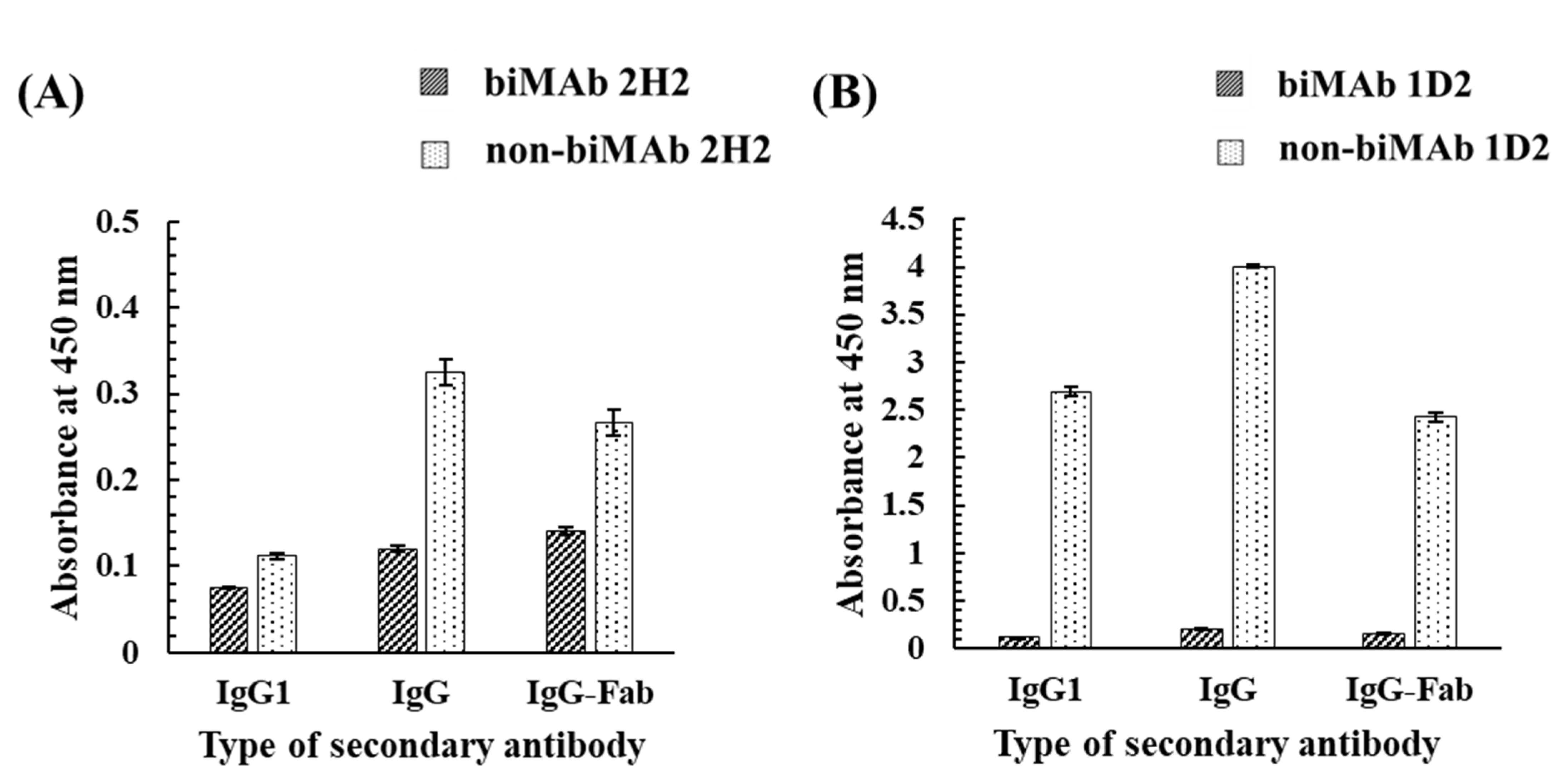
3.2. Investigation of Candidate Proteins for the Bioimprinting of Kwa
4. Discussion
4.1. Development of the icELISA and cELBIA Using biOVA for Quantitative Analysis of Kwa
4.2. Investigation of Candidate Proteins for the Bioimprinting of Kwa
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Urasopon, N.; Hamada, Y.; Cherdshewasart, W.; Malaivijitnond, S. Preventive effects of Pueraria mirifica on bone loss in ovariectomized rats. Maturitas 2008, 59, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Kittivanichkul, D.; Charoenphandhu, N.; Khemawoot, P.; Malaivijitnond, S. Pueraria mirifica alleviates cortical bone loss in naturally menopausal monkeys. J. Endocrinol. 2016, 231, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Suthon, S.; Jaroenporn, S.; Charoenphandhu, N.; Suntornsaratoon, P.; Malaivijitnond, S. Anti-osteoporotic effects of Pueraria candollei var.mirifica on bone mineral density and histomorphometry in estrogen-deficient rats. J. Nat. Med. 2016, 70, 225–233. [Google Scholar] [CrossRef]
- Sookvanichsilp, N.; Soonthornchareonnon, N.; Boonleang, C. Estrogenic activity of the dichloromethane extract from Pueraria mirifica. Fitoterapia 2008, 79, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Cherdshewasart, W.; Panriansaen, R.; Picha, P. Pretreatment with phytoestrogen-rich plant decreases breast tumor incidence and exhibits lower profile of mammary ERalpha and ERbeta. Maturitas 2007, 58, 174–181. [Google Scholar] [CrossRef]
- Ratanachamnong, P.; Phivthong-Ngam, L.; Namchaiw, P. Daily white kwao krua dietary supplement alleviates LDL oxidative susceptibility, plasma LDL level and improves vasculature in a hypercholesterolemia rabbit model. J. Tradit. Complement. Med. 2020, 10, 496–503. [Google Scholar] [CrossRef]
- Sucontphunt, A.; De-Eknamkul, W.; Nimmannit, U.; Dan Dimitrijevich, S.; Gracy, R.W. Protection of HT22 neuronal cells against glutamate toxicity mediated by the antioxidant activity of Pueraria candollei var. mirifica extracts. J. Nat. Med. 2011, 65, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chulikhit, Y.; Sukhano, W.; Daodee, S.; Putalun, W.; Wongpradit, R.; Khamphukdee, C.; Umehara, K.; Noguchi, H.; Matsumoto, K.; Monthakantirat, O. Effects of Pueraria candollei var mirifica (Airy Shaw and Suvat.) Niyomdham on ovariectomy-induced cognitive impairment and oxidative stress in the mouse brain. Molecules 2021, 26, 3442. [Google Scholar] [CrossRef]
- Anukulthanakorn, K.; Parhar, I.S.; Jaroenporn, S.; Kitahashi, T.; Watanbe, G.; Malaivijitnond, S. Neurotherapeutic effects of Pueraria mirifica extract in early- and late-stage cognitive impaired rats. Phytother. Res. 2016, 30, 929–939. [Google Scholar] [CrossRef]
- Cain, J.C. Miroestrol: An oestrogen from the plant Pueraria mirifica. Nature 1960, 188, 774–777. [Google Scholar] [CrossRef] [PubMed]
- Chansakaow, S.; Ishikawa, T.; Seki, H.; Sekine, K.; Okada, M.; Chaichantipyuth, C. Identification of deoxymiroestrol as the actual rejuvenating principle of “Kwao Keur”, Pueraria mirifica. The known miroestrol may be an artifact. J. Nat. Prod. 2000, 63, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Yusakul, G.; Udomsin, O.; Juengwatanatrakul, T.; Tanaka, H.; Chaichantipyuth, C.; Putalun, W. Highly selective and sensitive determination of deoxymiroestrol using a polyclonal antibody-based enzyme-linked immunosorbent assay. Talanta 2013, 114, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Chansakaow, S.; Ishikawa, T.; Sekine, K.; Okada, M.; Higuchi, Y.; Kudo, M.; Chaichantipyuth, C. Isoflavonoids from Pueraria mirifica and their estrogenic activity. Planta Med. 2000, 66, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Pongkitwitoon, B.; Sakamoto, S.; Tanaka, H.; Tsuchihashi, R.; Kinjo, J.; Morimoto, S.; Putalun, W. Development of an enzyme-linked immunosorbent assay to determine puerarin and its aglycone daidzein. J. Nat. Med. 2011, 65, 31–36. [Google Scholar] [CrossRef]
- Yusakul, G.; Togita, R.; Minami, K.; Chanpokapaiboon, K.; Juengwatanatrakul, T.; Putalun, W.; Tanaka, H.; Sakamoto, S.; Morimoto, S. An indirect competitive enzyme-linked immunosorbent assay toward the standardization of Pueraria candollei based on its unique isoflavonoid, kwakhurin. Fitoterapia 2019, 133, 23–28. [Google Scholar] [CrossRef]
- Chanpokapaiboon, K.; Khoonrit, P.; Yusakul, G.; Juengwatanatrakul, T.; Putalun, W.; Tanaka, H.; Sakamoto, S.; Morimoto, S. A recombinant Fab antibody against kwakhurin as a tool for sensitive indirect competitive ELISA. Curr. Pharm. Biotechnol. 2018, 19, 1170–1176. [Google Scholar] [CrossRef]
- Sakamoto, S.; Eto, R.; Nuntawong, P.; Yusakul, G.; Juengwatanatrakul, T.; Putalun, W.; Fujii, S.; Tanaka, H.; Morimoto, S. Kwakhurin-magnetic particles conjugates enable fast enzyme immunoassay for the detection of kwakhurin in Pueraria candollei. Phytochem. Anal. 2020, 31, 930–936. [Google Scholar] [CrossRef]
- Phaisan, S.; Yusakul, G.; Nuntawong, P.; Sakamoto, S.; Putalun, W.; Morimoto, S.; Tanaka, H. Immunochromatographic assay for the detection of kwakhurin and its application for the identification of Pueraria candollei varmirifica (Airy Shaw & Suvat.). Niyomdham. Phytochem. Anal. 2021, 32, 503–511. [Google Scholar] [CrossRef]
- Sakamoto, S.; Putalun, W.; Vimolmangkang, S.; Phoolcharoen, W.; Shoyama, Y.; Tanaka, H.; Morimoto, S. Enzyme-linked immunosorbent assay for the quantitative/qualitative analysis of plant secondary metabolites. J. Nat. Med. 2018, 72, 32–42. [Google Scholar] [CrossRef] [Green Version]
- Nuntawong, P.; Putalun, W.; Tanaka, H.; Morimoto, S.; Sakamoto, S. Lateral flow immunoassay for small-molecules detection in phytoproducts: A review. J. Nat. Med. 2022, 76, 521–545. [Google Scholar] [CrossRef]
- Vlatakis, G.; Andersson, L.I.; Müller, R.; Mosbach, K. Drug assay using antibody mimics made by molecular imprinting. Nature 1993, 361, 645–647. [Google Scholar] [CrossRef] [PubMed]
- Mingarro, I.; Abad, C.; Braco, L. Interfacial activation-based molecular bioimprinting of lipolytic enzymes. Proc. Natl. Acad. Sci. USA 1995, 92, 3308–3312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gutierrez, A.; Hedström, M.; Mattiasson, B. Bioimprinting as a tool for the detection of aflatoxin B1 using a capacitive biosensor. Biotechnol. Rep. 2016, 11, 12–17. [Google Scholar] [CrossRef] [Green Version]
- Beloglazova, N.; Lenain, P.; Tessier, M.; Goryacheva, I.; Hens, Z.; Saeger, S.D. Bioimprinting for multiplex luminescent detection of deoxynivalenol and zearalenone. Talanta 2019, 192, 169–174. [Google Scholar] [CrossRef]
- Pidenko, P.; Zhang, H.; Lenain, P.; Goryacheva, I.; Saeger, S.D.; Beloglazova, N. Imprinted proteins as a receptor for detection of zearalenone. Anal. Chim. Acta. 2018, 1040, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.; Morinaga, O.; Uto, T.; Nomura, S.; Shoyama, Y. Simultaneous determination of glycyrrhizin and liquiritin in licorice roots and Kampo medicines by combination enzyme-linked immunosorbent assay using anti-glycyrrhizin and anti-liquiritin monoclonal antibodies. J. Immunoassay. Immunochem. 2017, 38, 285–298. [Google Scholar] [CrossRef]
- Sakamoto, S.; Yusakul, G.; Tsuneura, Y.; Putalun, W.; Usui, K.; Miyamoto, T.; Tanaka, H.; Morimoto, S. Sodium periodate-mediated conjugation of harringtonine enabling the production of a highly specific monoclonal antibody, and the development of a sensitive quantitative analysis method. Analyst 2017, 142, 1140–1148. [Google Scholar] [CrossRef]
- Weiler, E.W.; Zenk, M.H. Radioimmunoassay for the detection of digoxin and related compounds in Digitalis lanata. Phytochemistry 1976, 15, 1537–1545. [Google Scholar] [CrossRef]
- Nuntawong, P.; Tanaka, H.; Sakamoto, S.; Morimoto, S. ELISA for the detection of the prohibited doping agent higenamine. Planta Med. 2020, 86, 760–766. [Google Scholar] [CrossRef]
- Sakamoto, S.; Wada, S.; Morita, Y.; Yamaguchi, T.; Tanaka, H.; Morimoto, S. Magnetic particles-based enzyme immunoassay for rapid determination of secoiridoid glycoside, amarogentin. Talanta 2019, 194, 731–736. [Google Scholar] [CrossRef]
- Ahirwar, R.; Bariar, S.; Balakrishnana, A.; Nahar, P. BSA blocking in enzyme-linked immunosorbent assays is a non-mandatory step: A perspective study on mechanism of BSA blocking in common ELISA protocols. RSC Adv. 2015, 5, 100077–100083. [Google Scholar] [CrossRef]
- Ikai, A. Thermostability and aliphatic index of globular proteins. J. Biochem. 1980, 88, 1895–1898. [Google Scholar] [PubMed]
- Kyte, J.; Doolittle, R.F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 1982, 157, 105–132. [Google Scholar] [CrossRef] [Green Version]

| CRs (%) | |||
|---|---|---|---|
| Class | Compound | biOVA | MAb 11F a |
| Isoflavonoids | Kwa | 100 | 100 |
| Daidzein | 23.4 | <0.005 | |
| Genistein | 25.5 | <0.005 | |
| Isoflavonoid glycosides | Daidzin | 9.4 | <0.005 |
| Genistin | 5.2 | <0.005 | |
| Puerarin | <0.1 | <0.005 | |
| Sample Name | icELISA Using biOVA | cELBIA Using biOVA | icELISA Using MAb 11F | |||
|---|---|---|---|---|---|---|
| Kwa Amount (% wt./dry wt.) | CV (%) | Kwa Amount (% wt./dry wt.) | CV (%) | Kwa Amount (% wt./dry wt.) | CV (%) | |
| P. candollei root without bark | 4.23 × 10−4 | 11.8 | 4.49 × 10−4 | 9.0 | 1.94 × 10−4 | 7.0 |
| P. candollei root bark 1 | 1.56 × 10−2 | 12.1 | 1.43 × 10−2 | 7.2 | 1.09 × 10−2 | 8.2 |
| P. candollei root bark 2 | 1.22 × 10−3 | 6.2 | 1.11 × 10−3 | 9.3 | 1.29 × 10−3 | 0.4 |
| Supplement 1 | 9.19 × 10−5 | 4.3 | ND | ND | 9.94 × 10−6 | 10.3 |
| Supplement 2 | 6.89 × 10−3 | 11.7 | 9.65 × 10−3 | 4.9 | 6.23 × 10−3 | 4.5 |
| Supplement 3 | 2.04 × 10−3 | 8.1 | 2.14 × 10−3 | 9.3 | 1.27 × 10−3 | 5.0 |
| Supplement 4 | 3.36 × 10−4 | 8.7 | 4.12 × 10−4 | 6.5 | 2.38 × 10−4 | 9.0 |
| Supplement 5 | ND | ND | ND | ND | 5.73 × 10−6 | 9.4 |
| CRs (%) | |||
|---|---|---|---|
| Class | Compound | biMAb 2H2 | biMAb 1D2 |
| Isoflavonoids | Kwa | 100 | 100 |
| Daidzein | <0.1 | 0.2 | |
| Genistein | <0.1 | 7.1 | |
| Isoflavonoid glycosides | Daidzin | <0.1 | 5.4 |
| Genistin | 0.1 | 19.6 | |
| Puerarin | 7.0 | 57.3 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakamoto, S.; Minami, K.; Nuntawong, P.; Yusakul, G.; Putalun, W.; Tanaka, H.; Fujii, S.; Morimoto, S. Bioimprinting as a Receptor for Detection of Kwakhurin. Biomolecules 2022, 12, 1064. https://doi.org/10.3390/biom12081064
Sakamoto S, Minami K, Nuntawong P, Yusakul G, Putalun W, Tanaka H, Fujii S, Morimoto S. Bioimprinting as a Receptor for Detection of Kwakhurin. Biomolecules. 2022; 12(8):1064. https://doi.org/10.3390/biom12081064
Chicago/Turabian StyleSakamoto, Seiichi, Kei Minami, Poomraphie Nuntawong, Gorawit Yusakul, Waraporn Putalun, Hiroyuki Tanaka, Shunsuke Fujii, and Satoshi Morimoto. 2022. "Bioimprinting as a Receptor for Detection of Kwakhurin" Biomolecules 12, no. 8: 1064. https://doi.org/10.3390/biom12081064
APA StyleSakamoto, S., Minami, K., Nuntawong, P., Yusakul, G., Putalun, W., Tanaka, H., Fujii, S., & Morimoto, S. (2022). Bioimprinting as a Receptor for Detection of Kwakhurin. Biomolecules, 12(8), 1064. https://doi.org/10.3390/biom12081064







