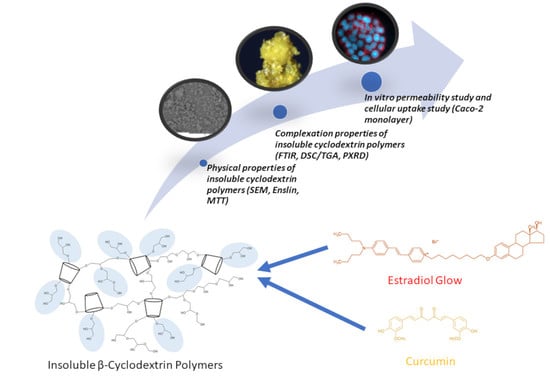
Investigation of the Drug Carrier Properties of Insoluble Cyclodextrin Polymer Microspheres
Abstract
1. Introduction
2. Materials and Methods
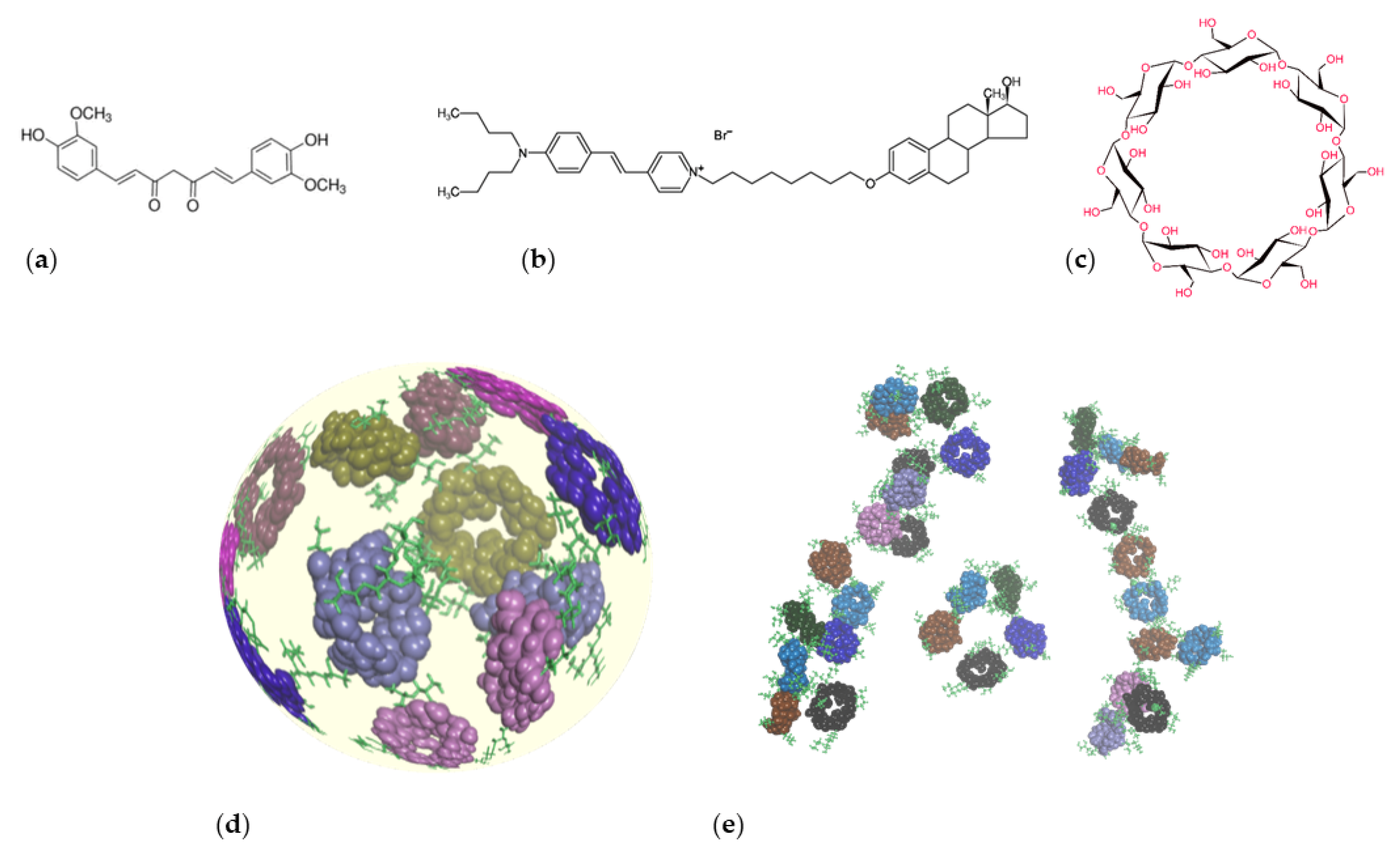
2.1. Materials
2.2. Physical Properties of Insoluble Cyclodextrin Polymers
2.2.1. Size Distribution
2.2.2. Scanning Electron Microscopy (SEM)
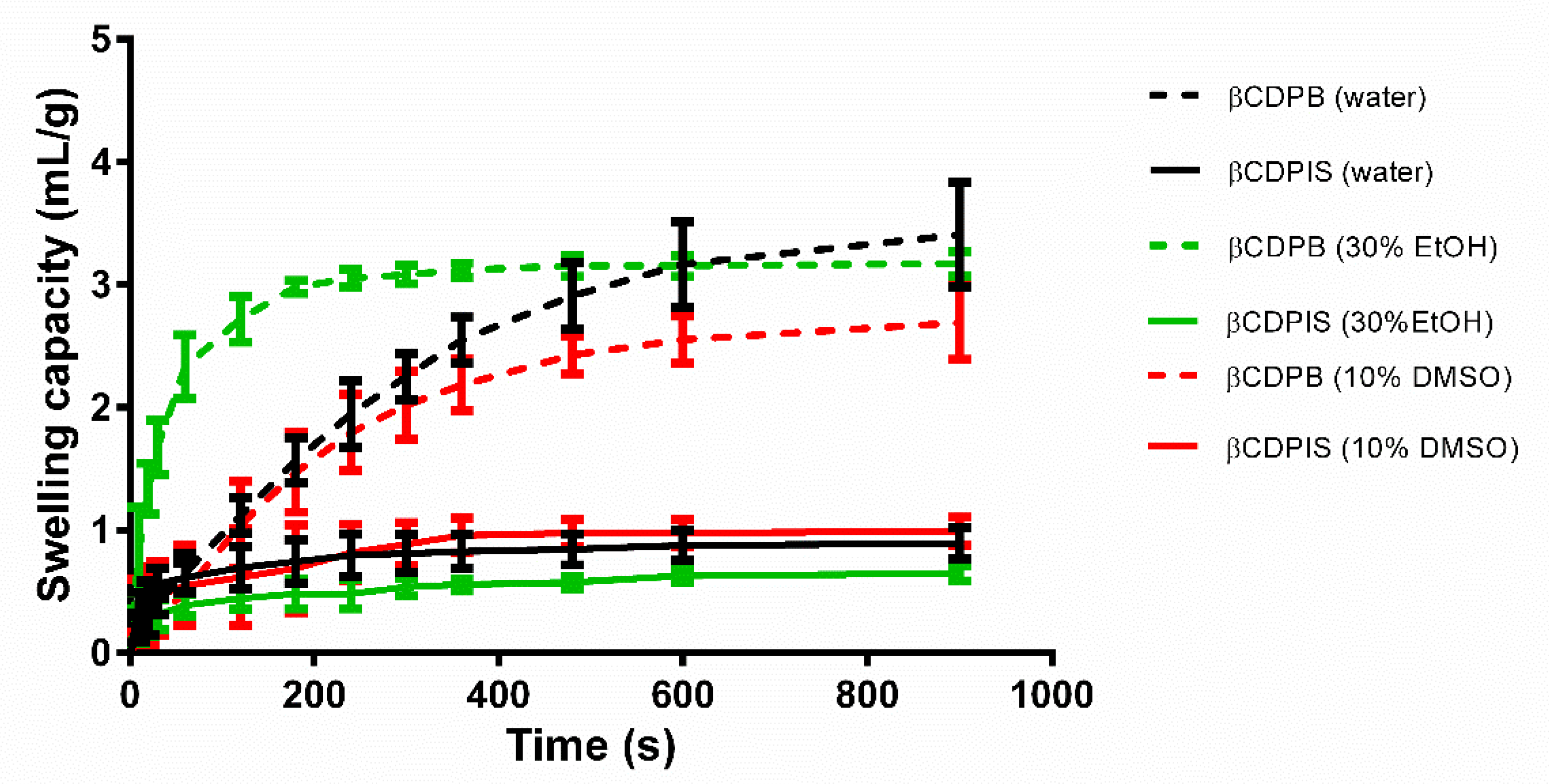
2.2.3. Swelling Capacity
2.3. Complexation Properties of Insoluble Cyclodextrin Polymers
2.3.1. Complexation Study and Preparation of Complexes
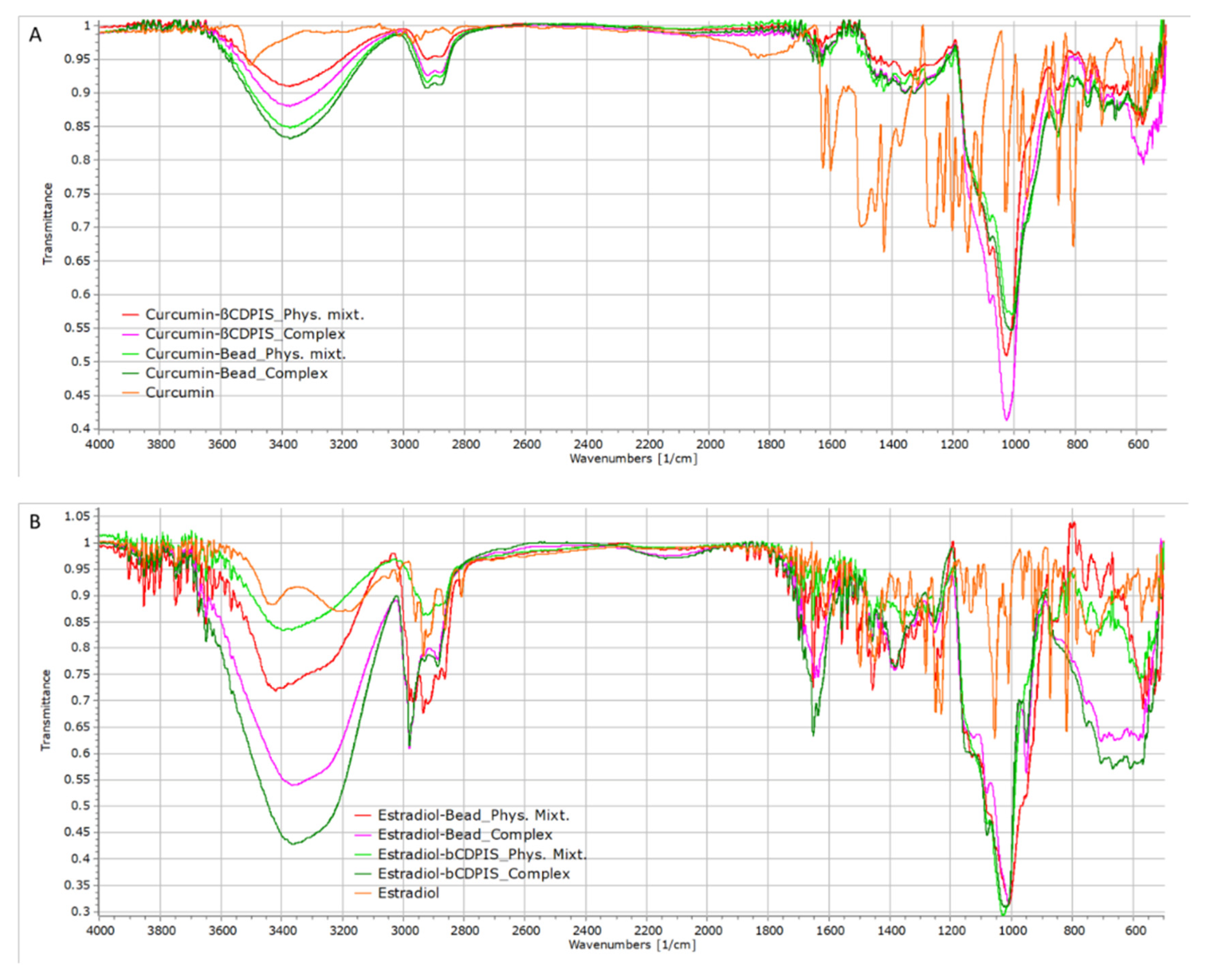
2.3.2. FTIR Analysis
2.3.3. Powder X-ray Diffraction (PXRD)
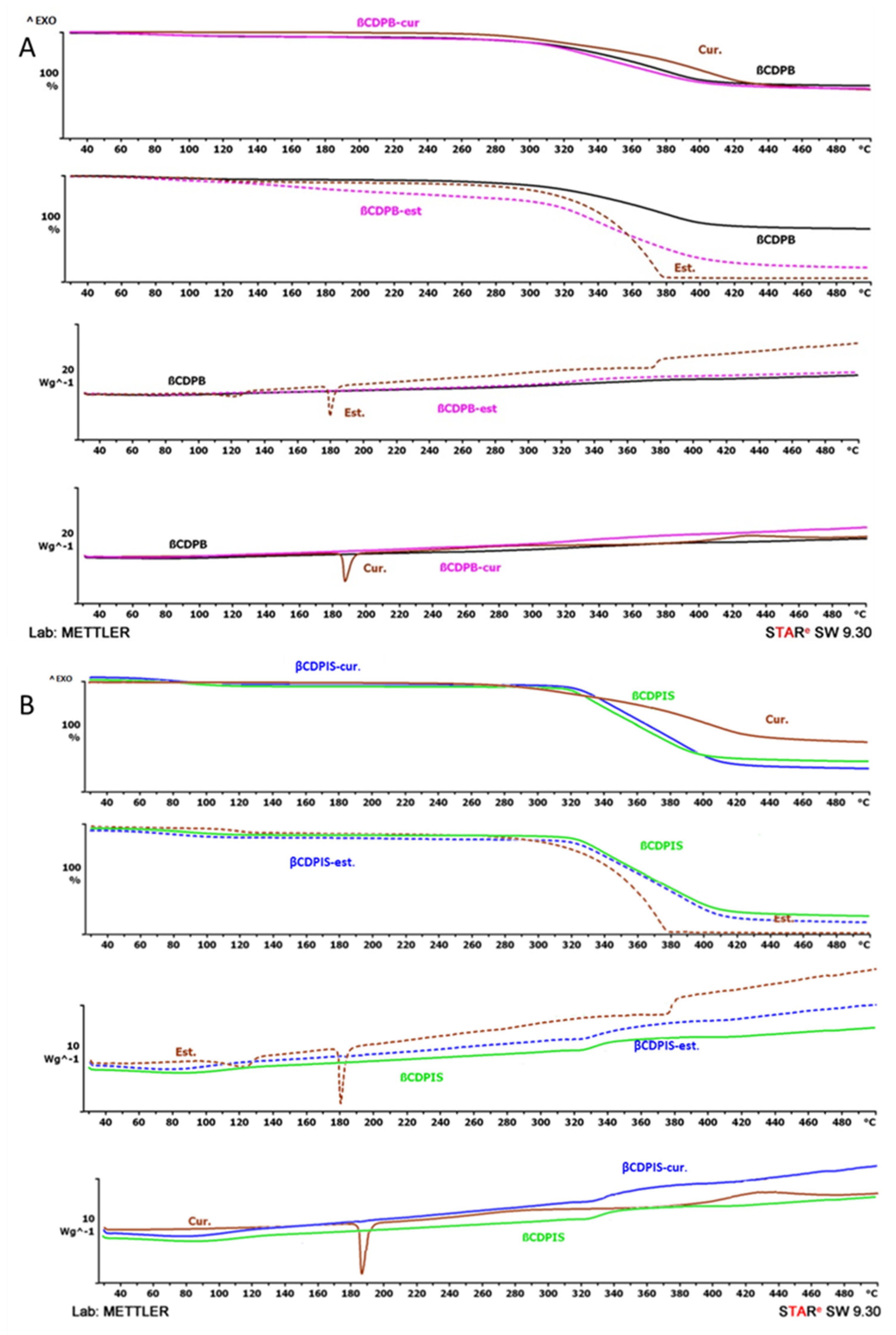
2.3.4. Thermoanalytical Methods (TGA/DSC)
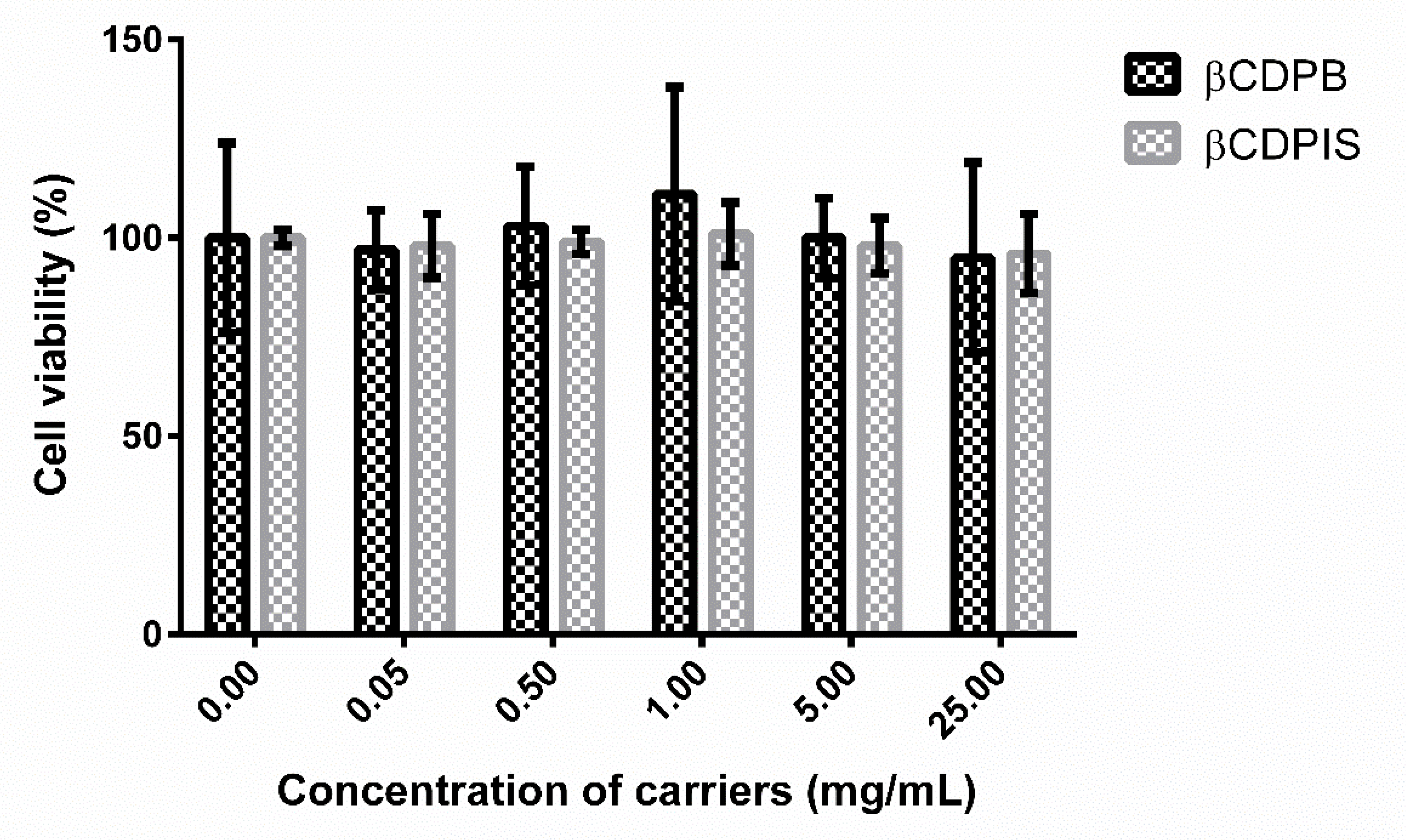
2.4. MTT (3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide) Assay
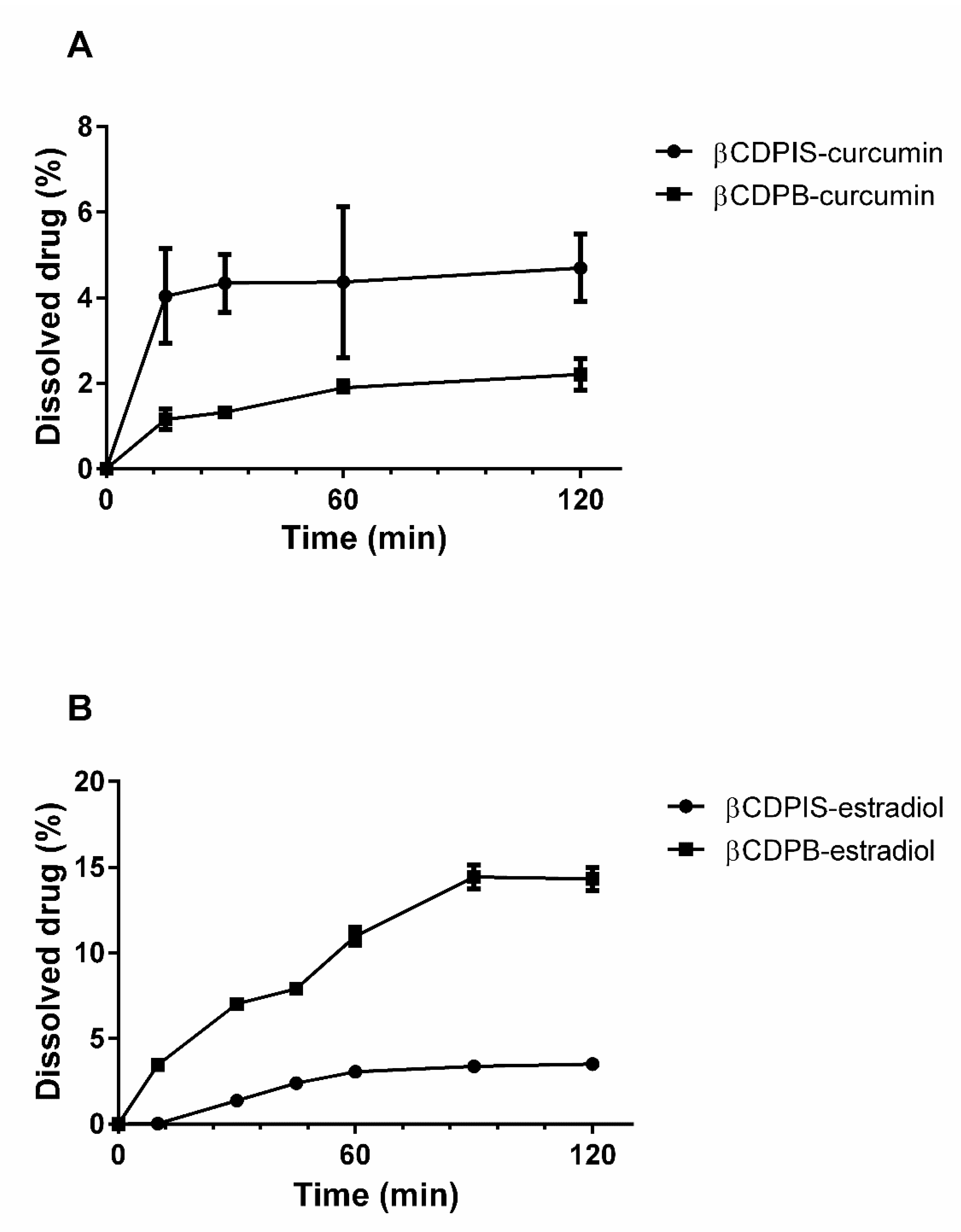
2.5. In Vitro Dissolution Test
2.6. In Vitro Permeability Study
2.7. Statistical Analysis
3. Results
3.1. Physical Properties of Insoluble Cyclodextrin Polymers
3.1.1. Size Distribution
3.1.2. SEM
3.1.3. Swelling Capacity
3.2. Complexation Properties of Insoluble Cyclodextrin Polymers
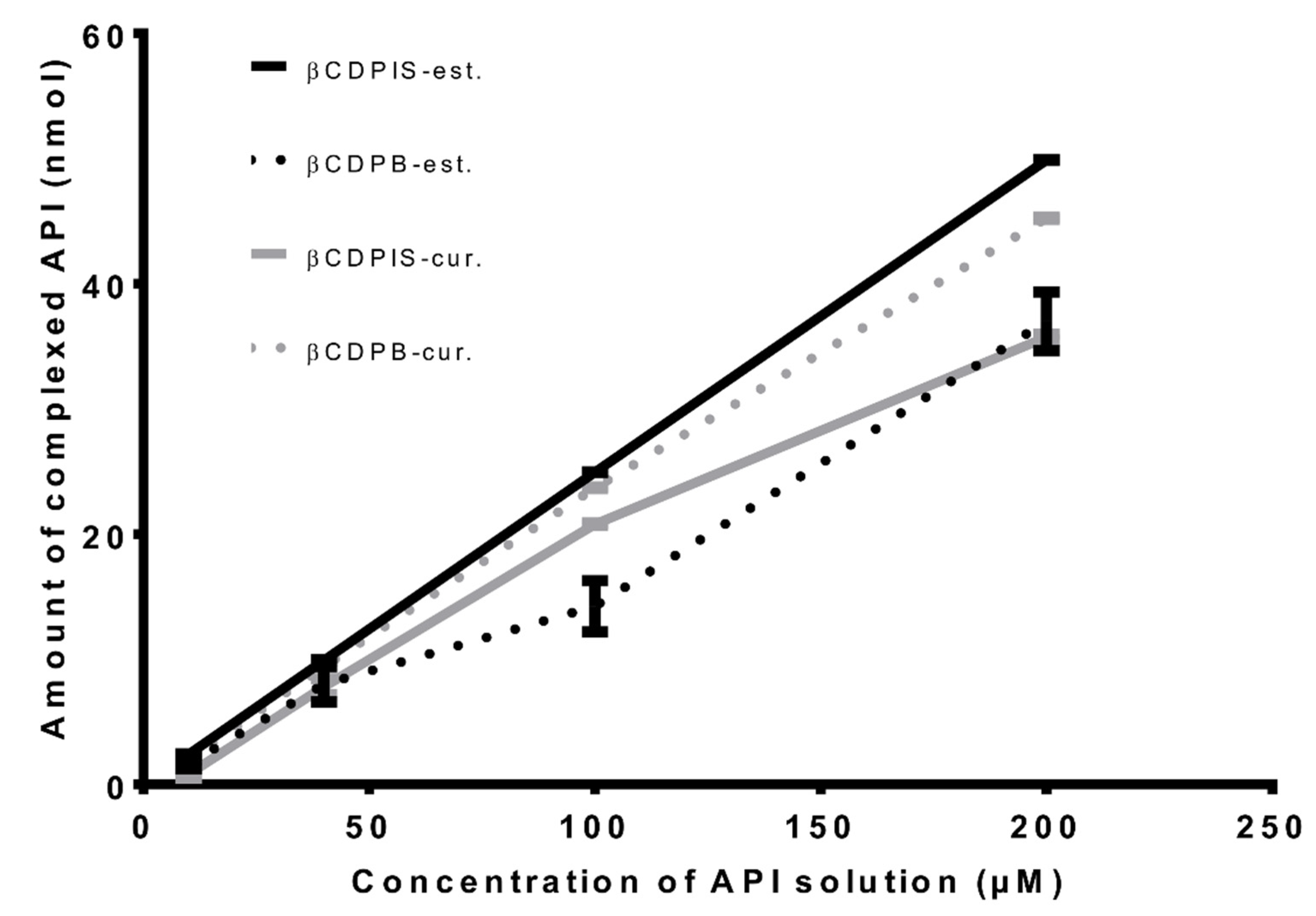
3.2.1. Complexation Study
3.2.2. FTIR Analysis
3.2.3. PXRD
3.2.4. Thermoanalytical Investigations (TGA/DSC)
3.2.5. MTT Assay
3.2.6. In Vitro Dissolution Study
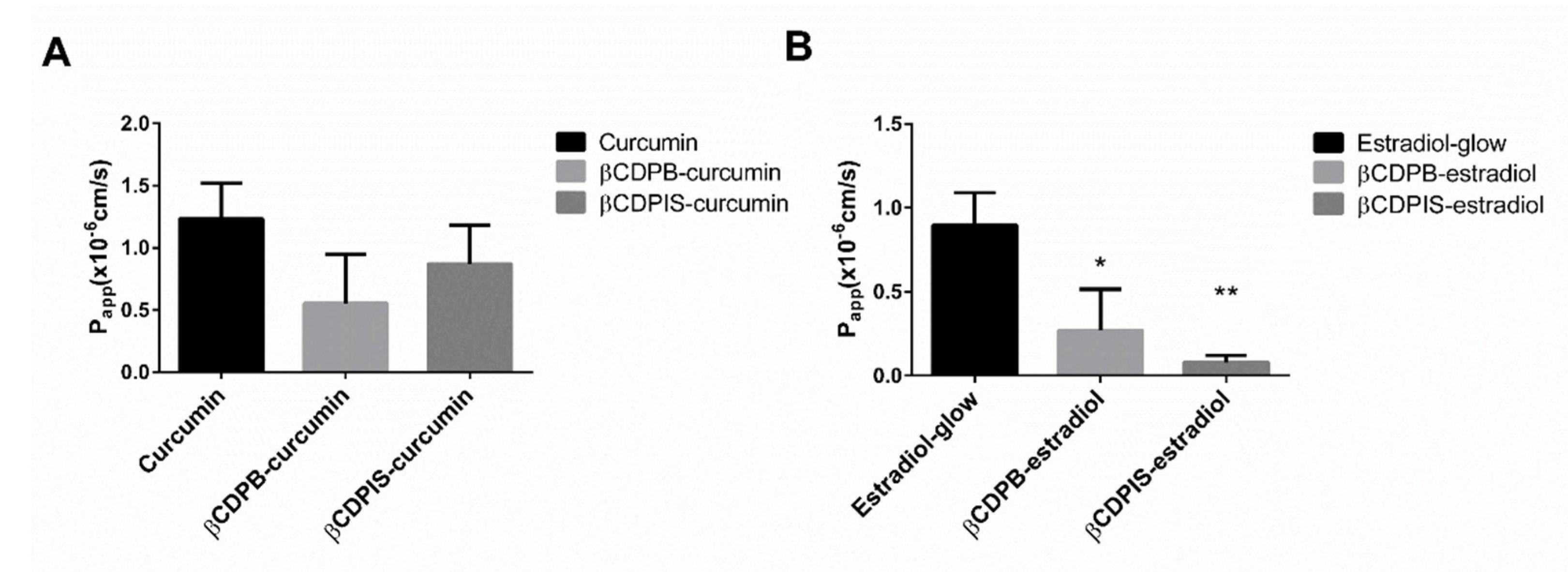
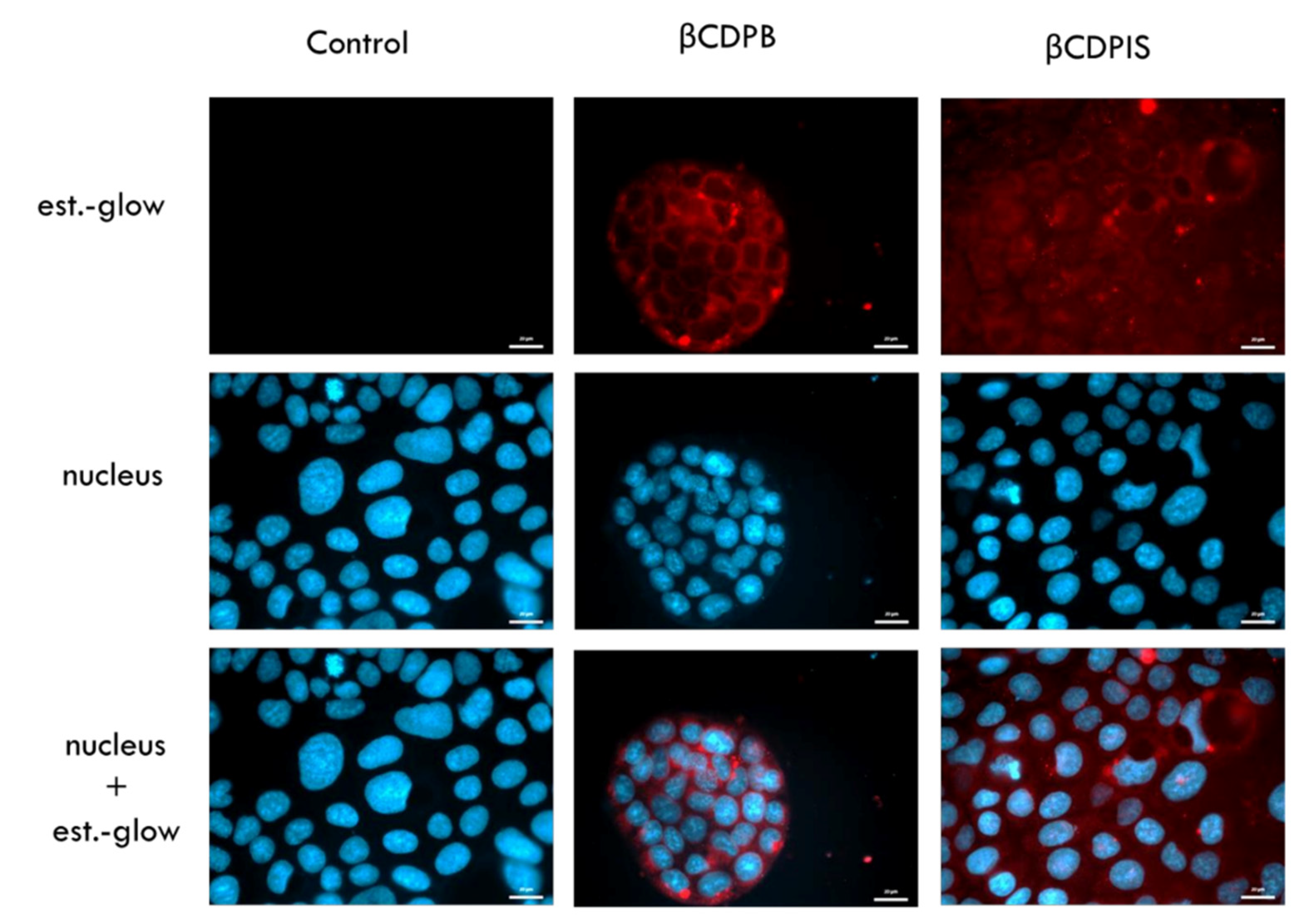
3.2.7. In Vitro Permeability Study and Cellular Uptake of Estradiol-Glow
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Szejtli, J. Introduction and General Overview of Cyclodextrin Chemistry. Chem. Rev. 1998, 98, 1743–1754. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, V.B.; Patel, J.K. Cyclodextrin Inclusion Complex to Enhance Solubility of Poorly Water Soluble Drugs: A Review. Int. J. Pharm. Sci. Res. 2013, 4, 68–76. [Google Scholar]
- Haimhoffer, A.; Rusznyák, A.; Réti-Nagy, K.; Vasvári, G.; Váradi, J.; Vecsernyés, M.; Bácskay, I.; Fehér, P.; Ujhelyi, Z.; Fenyvesi, F. Cyclodextrins in Drug Delivery Systems and their Effects on Biological Barriers. Sci. Pharm. 2019, 87, 33. [Google Scholar] [CrossRef]
- Khan, A.R.; Forgo, P.; Stine, K.J.; D’Souza, V.T. Methods for Selective Modifications of Cyclodextrins. Chem. Rev. 1998, 98, 1977–1996. [Google Scholar] [CrossRef] [PubMed]
- Hattori, K.; Ikeda, H. Modification Reactions of Cyclodextrins and the Chemistry of Modified Cyclodextrins. In Cyclodextrins and Their Complexes; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2006; pp. 31–64. ISBN 3527312803. [Google Scholar]
- Adeoye, O.; Cabral-Marques, H. Cyclodextrin Nanosystems in Oral Drug Delivery: A mini review. Int. J. Pharm. 2017, 531, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Eastburn, S.D.; Tao, B.Y. Applications of Modified Cyclodextrins. Biotechnol. Adv. 1994, 12, 325–339. [Google Scholar] [CrossRef]
- Petitjean, M.; García-Zubiri, I.X.; Isasi, J.R. History of Cyclodextrin—Based Polymers in Food and Pharmacy: A Review. Environ. Chem. Lett. 2021, 19, 3465–3476. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Winterton, P.; Fourmentin, S.; Wilson, L.D.; Fenyvesi, É.; Crini, G. Water-insoluble β-cyclodextrin–epichlorohydrin Polymers for Removal of Pollutants from Aqueous Solutions by Sorption Processes Using Batch Studies: A review of inclusion mechanisms. Prog. Polym. Sci. 2018, 78, 1–23. [Google Scholar] [CrossRef]
- Tian, B.; Liu, J. The Classification and Application of Cyclodextrin Polymers: A review. N. J. Chem. 2020, 44, 9137–9148. [Google Scholar] [CrossRef]
- Martel, B.; Ruffin, D.; Weltrowski, M.; Lekchiri, Y.; Morcellet, M. Water-soluble Polymers and Gels from the Polycondensation Between Cyclodextrins and Poly(carboxylic acid)s: A study of the Preparation Parameters. J. Appl. Polym. Sci. 2005, 97, 433–442. [Google Scholar] [CrossRef]
- Sainz-Rozas, P.R.; Isasi, J.R.; González-Gaitano, G. Binding of dibenzofuran and its derivatives to water-soluble β-cyclodextrin polymers. J. Photochem. Photobiol. A Chem. 2005, 173, 248–257. [Google Scholar] [CrossRef]
- Chen, J.; Qin, X.; Zhong, S.; Chen, S.; Su, W.; Liu, Y. Characterization of Curcumin/Cyclodextrin Polymer Inclusion Complex and Investigation on its Antioxidant and Antiproliferative Activities. Molecules 2018, 23, 1179. [Google Scholar] [CrossRef] [PubMed]
- Szeman, J.; Ueda, H.; Szejtli, J.; Fenyvesi, E.; Machida, Y.; Nagai, T. Complexation of Several Cyclodextrin Drugs with Polymer. Chem. Pharm. Bull. 1987, 35, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Gong, X.; Cai, Y.; Zhang, C.; Yu, X.; Fan, J.; Diao, G. Investigation of Water-soluble Inclusion Complex of Hypericin with β-cyclodextrin Polymer. Carbohydr. Polym. 2013, 95, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Nojavan, S.; Yazdanpanah, M. Micro-solid phase extraction of benzene, toluene, ethylbenzene and xylenes from aqueous solutions using water-insoluble β-cyclodextrin polymer as sorbent. J. Chromatogr. A 2017, 1525, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Morin-Crini, N.; Crini, G. Environmental applications of water-insoluble β-cyclodextrin- epichlorohydrin polymers. Prog. Polym. Sci. 2013, 38, 344–368. [Google Scholar] [CrossRef]
- Moulahcene, L.; Skiba, M.; Senhadji, O.; Milon, N.; Benamor, M.; Lahiani-Skiba, M. Inclusion and removal of pharmaceutical residues from aqueous solution using water-insoluble cyclodextrin polymers. Chem. Eng. Res. Des. 2015, 97, 145–158. [Google Scholar] [CrossRef]
- Fenyvesi, É. Cyclodextrin polymers in the pharmaceutical industry. J. Incl. Phenom. 1988, 6, 537–545. [Google Scholar] [CrossRef]
- Szejtli, J.; Fenyvesi, É.; Zoltán, S.; Zsadon, B.; Túdos, F. Cyclodextrin-Polyvinyl Alcohol Polymers and a Process for the Preparation Thereof in Pearl, Foil, Fiber or Block Form. US Patent US4274985A, 23 June 1981. [Google Scholar]
- Fenyvesi, É.; Ujházy, A.; Szejtli, J.; Pütter, S.; Gan, T.G. Controlled release of drugs from CD polymers substituted with ionic groups. J. Incl. Phenom. Mol. Recognit. Chem. 1996, 25, 185–189. [Google Scholar] [CrossRef]
- Felméray, I.; Fenyvesi, É.; Neumark, T.; Takács, J.; Gerlóczy, A.; Szejtli, J. Effect of Cyclodextrin Bead Polymer on Wound Healing. In Proceedings of the Eighth International Symposium on Cyclodextrins, Budapest, Hungary, 31 March–2 April 1996; pp. 491–494. [Google Scholar] [CrossRef]
- Jicsinszky, L.; Calsolaro, F.; Martina, K.; Bucciol, F.; Manzoli, M.; Cravotto, G. Reaction of oxiranes with cyclodextrins under high-energy ball-milling conditions. Beilstein J. Org. Chem. 2019, 15, 1448–1459. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 969516, Curcumin. 2021. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Curcumin (accessed on 18 August 2021).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 5757, Estradiol. 2021. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Estradiol (accessed on 18 August 2021).
- Jicsinszky, L.; Bucciol, F.; Manzoli, M.; Cravotto, G. Comparative Studies of Mechanochemically Synthesized Insoluble Beta-Cyclodextrin Polymers. Curr. Org. Chem. 2021, 25, 1923–1936. [Google Scholar] [CrossRef]
- Wu, J.-S.; Ho, H.-O.; Sheu, M.-T. A statistical design to evaluate the influence of manufacturing factors on the material properties and functionalities of microcrystalline cellulose. Eur. J. Pharm. Sci. 2001, 12, 417–425. [Google Scholar] [CrossRef]
- Kaufhold, S.; Dohrmann, R. Comparison of the Traditional Enslin-Neff method and the Modified Dieng Method for Measuring Water-uptake Capacity. Clays Clay Miner. 2008, 56, 686–692. [Google Scholar] [CrossRef]
- Gholibegloo, E.; Mortezazadeh, T.; Salehian, F.; Ramazani, A.; Amanlou, M.; Khoobi, M. Improved Curcumin Loading, Release, Solubility and Toxicity by Tuning the Molar Ratio of Cross-linker to β-cyclodextrin. Carbohydr. Polym. 2019, 213, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Fenyvesi, F.; Le, T.; Nguyen, P.; Ruszny, Á.; Judit, V. Cyclodextrin Complexation Improves the Solubility and Caco-2 Permeability of Chrysin. Materials 2020, 13, 3618. [Google Scholar] [CrossRef]
- Siregar, C.; Martono, S.; Rohman, A. Application of Fourier transform infrared (FTIR) spectroscopy coupled with multivariate calibration for quantitative analysis of curcuminoid in tablet dosage form. J. Appl. Pharm. Sci. 2018, 8, 151–156. [Google Scholar] [CrossRef]
- Ratna, W.; Martono, S.; Rohman, A.; Sudjadi. Liquid Chromatography and Fourier Transform Infrared Spectroscopy for quantitative analysis of individual and total curcuminoid in Curcuma longa extract. J. Appl. Pharm. Sci. 2018, 8, 107–113. [Google Scholar] [CrossRef]
- Mishra, D.P.; Srivastava, A.; Shukla, R.K. Spectroscopic and chemical reactivity analysis of D-Myo-Inositol using quantum chemical approach and its experimental verification. Pramana 2017, 89, 12. [Google Scholar] [CrossRef]
- Khalique Ahmed, M.; McLeod, M.P.; Nézivar, J.; Giuliani, A.W. Fourier transform infrared and near-infrared spectroscopic methods for the detection of toxic diethylene glycol (DEG) contaminant in glycerin based cough syrup. Spectroscopy 2010, 24, 608749. [Google Scholar]
- Minaev, B.F.; Minaeva, V.A. Study of IR spectrum of the 17β-estradiol using quantum-chemical density functional theory. Biopolym. Cell 2006, 22, 363–374. [Google Scholar] [CrossRef][Green Version]
- Cakmak, G.; Togan, I.; Severcan, F. 17β-Estradiol induced compositional, structural and functional changes in rainbow trout liver, revealed by FT-IR spectroscopy: A comparative study with nonylphenol. Aquat. Toxicol. 2006, 77, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Minaeva, V.A.; Minaev, B.F.; Hovorun, D.M. Vibrational spectra of the steroid hormones, estradiol and estriol, calculated by density functional theory. The role of low-frequency vibrations. Ukr. BiokhimichnyiZhurnal 2008, 80, 82–95. [Google Scholar]
- Tappa, K.; Jammalamadaka, U.; Ballard, D.H.; Bruno, T.; Israel, M.R.; Vemula, H.; Meacham, J.M.; Mills, D.K.; Woodard, P.K.; Weisman, J.A. Medication Eluting Devices for the Field of OBGYN (MEDOBGYN): 3D Printed Biodegradable Hormone Eluting Constructs, a Proof of Concept Study. PLoS ONE 2017, 12, e0182929. [Google Scholar] [CrossRef] [PubMed]
- Cas, M.D.; Ghidoni, R. Dietary Curcumin: Correlation Between Bioavailability and Health Potential. Nutrients 2019, 11, 2147. [Google Scholar] [CrossRef] [PubMed]



| Type of Cyclodextrin Polymer | Size ± SD |
|---|---|
| βCDPB in water after 15 min | 218.4 ± 38.04 µm |
| βCDPB in paraffin oil | 154.1 ± 21.38 µm |
| βCDPIS in water | 4.1 ± 2.5 µm |
| βCDPB | βCDPIS | |
|---|---|---|
| Curcumin loading efficiency (%) | 90.5 ± 0.2% | 71.5 ± 0.5% |
| Estradiol-glow loading efficiency (%) | 74.0 ± 4.7% | 99.8 ± 0.1% |
| Curcumin loading capacity (‰) | 0.66 ± 0.001‰ | 0.52 ± 0.003‰ |
| Estradiol-glow loading capacity (‰) | 1.14 ± 0.07‰ | 1.54 ± 0.0008‰ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haimhoffer, Á.; Vas, A.; Árvai, G.; Fenyvesi, É.; Jicsinszky, L.; Budai, I.; Bényei, A.; Regdon, G., Jr.; Rusznyák, Á.; Vasvári, G.; et al. Investigation of the Drug Carrier Properties of Insoluble Cyclodextrin Polymer Microspheres. Biomolecules 2022, 12, 931. https://doi.org/10.3390/biom12070931
Haimhoffer Á, Vas A, Árvai G, Fenyvesi É, Jicsinszky L, Budai I, Bényei A, Regdon G Jr., Rusznyák Á, Vasvári G, et al. Investigation of the Drug Carrier Properties of Insoluble Cyclodextrin Polymer Microspheres. Biomolecules. 2022; 12(7):931. https://doi.org/10.3390/biom12070931
Chicago/Turabian StyleHaimhoffer, Ádám, Alexandra Vas, Gabriella Árvai, Éva Fenyvesi, László Jicsinszky, István Budai, Attila Bényei, Géza Regdon, Jr., Ágnes Rusznyák, Gábor Vasvári, and et al. 2022. "Investigation of the Drug Carrier Properties of Insoluble Cyclodextrin Polymer Microspheres" Biomolecules 12, no. 7: 931. https://doi.org/10.3390/biom12070931
APA StyleHaimhoffer, Á., Vas, A., Árvai, G., Fenyvesi, É., Jicsinszky, L., Budai, I., Bényei, A., Regdon, G., Jr., Rusznyák, Á., Vasvári, G., Váradi, J., Bácskay, I., Vecsernyés, M., & Fenyvesi, F. (2022). Investigation of the Drug Carrier Properties of Insoluble Cyclodextrin Polymer Microspheres. Biomolecules, 12(7), 931. https://doi.org/10.3390/biom12070931