Using Vertebrate Stem and Progenitor Cells for Cellular Agriculture, State-of-the-Art, Challenges, and Future Perspectives
Abstract
1. Introduction
2. Stem Cell and Progenitor Types Relevant in Cultured Meat (Mammalian, Avian) and Cultured Seafood (Fish) Bioprocesses
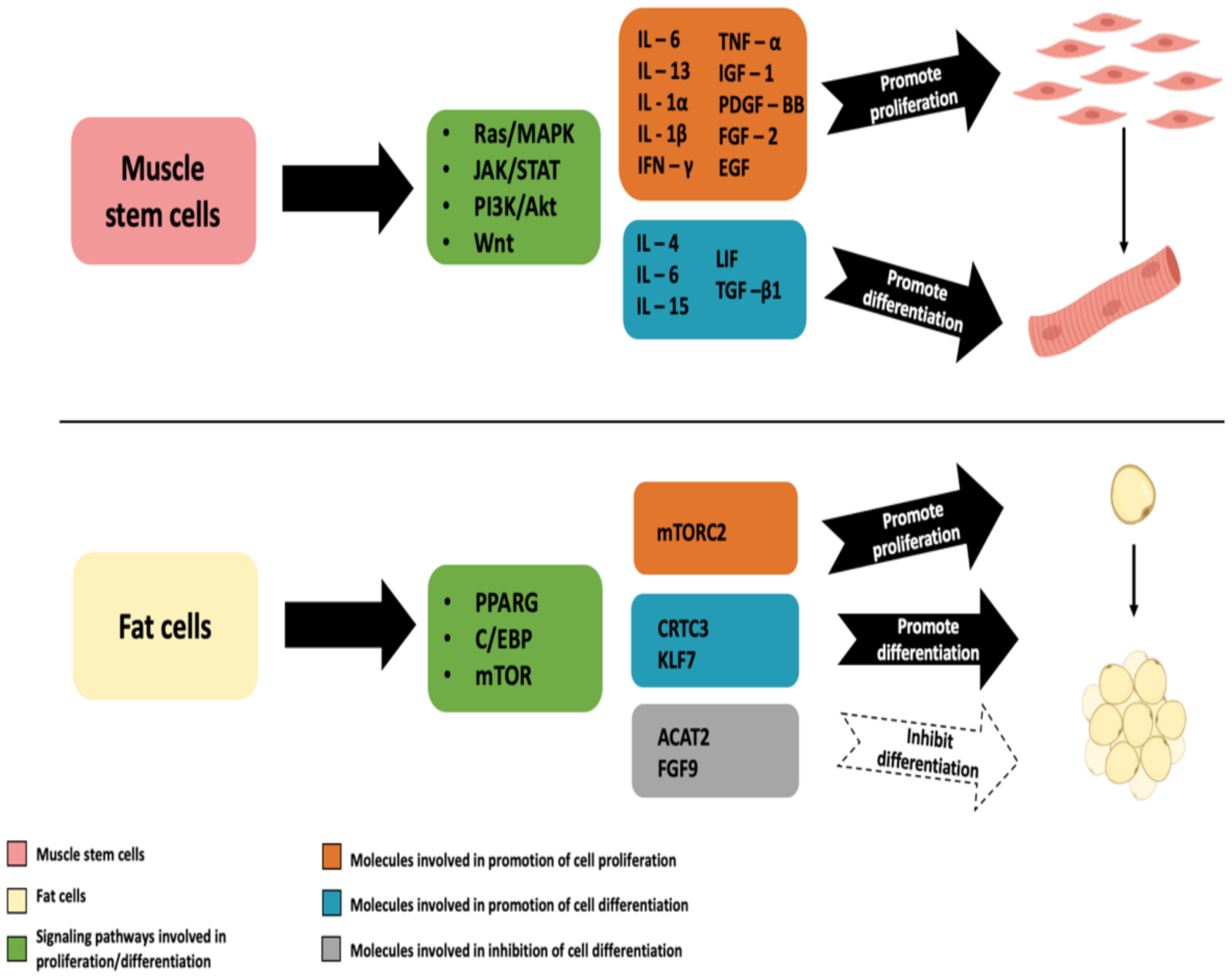
2.1. Myogenic Differentiation–Satellite Cells (SCs) and Myoblasts
2.1.1. Mammalian Myogenic Cells
2.1.2. Avian Myogenic Cells
2.1.3. Fish Myogenic Cells
2.1.4. Maintaining SCs Stemness through p38 Pathway
2.2. Embryonic Stem Cells (ESCs)
2.2.1. Mammalian ESCs
2.2.2. Avian ESCs
2.2.3. Fish ESCs
2.3. Induced Pluripotent Stem Cells (iPSCs)
2.3.1. Mammalian iPSCs
2.3.2. Avian iPSCs
2.3.3. Fish iPSCs
2.4. Mesenchymal Stem Cells (MSCs) for Myogenic and Adipogenic Differentiation
2.4.1. Mammalian MSCs for Myogenic Differentiation
2.4.2. Avian MSCs for Myogenic Differentiation
2.4.3. Mammalian MSCs for Adipogenic Differentiation
2.4.4. Fish MSCs for Adipogenic Differentiation
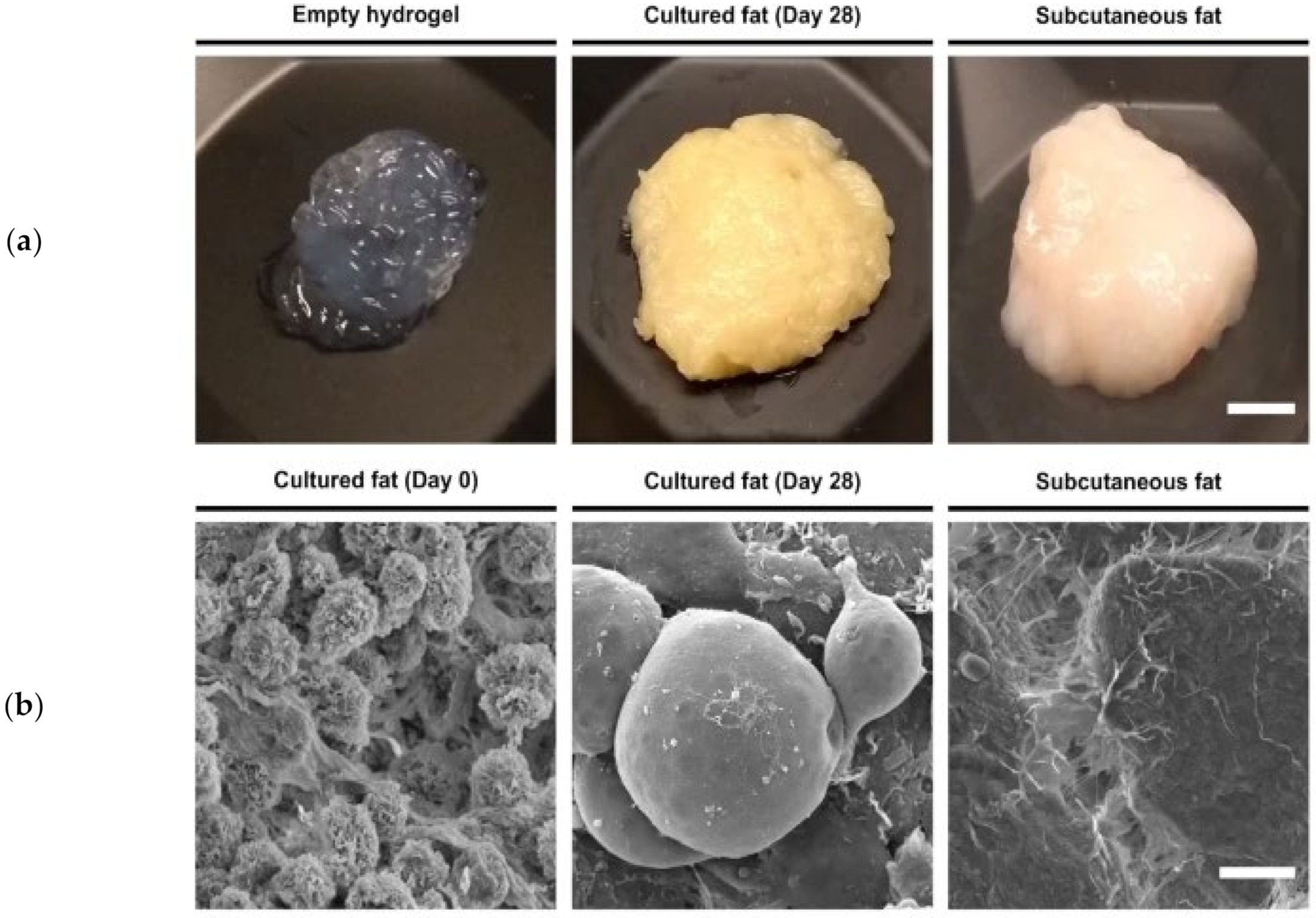
2.4.5. Fibro-Adipogenic Progenitor Cells (FAPs)
2.4.6. Dedifferentiated Fat (DFAT) Cells
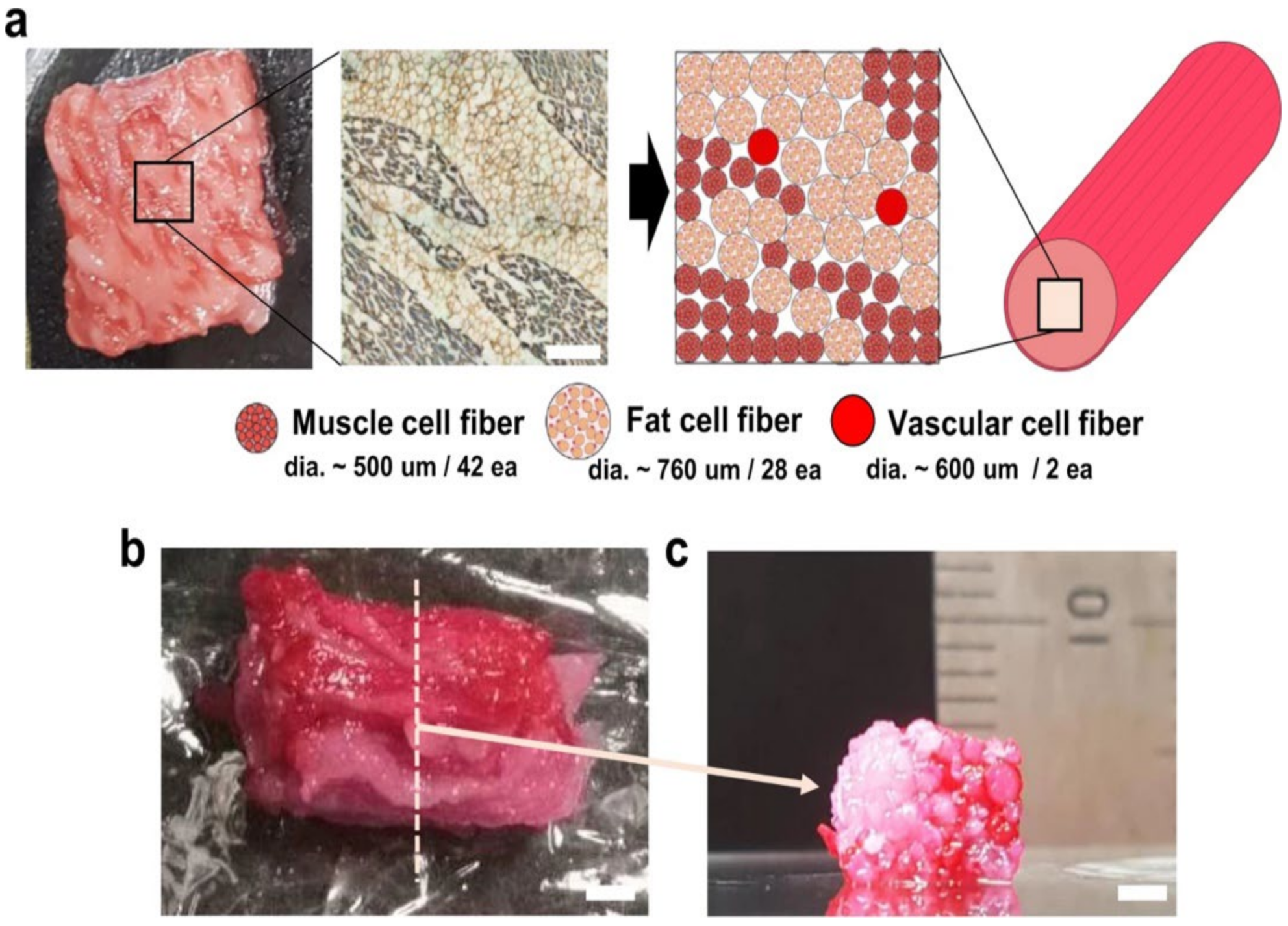
2.5. Myoblasts and Adipocytes Co-Cultivation
Need for Performing in Parallel Myogenesis, Adipogenesis, and Vasculogenesis
2.6. Stem Cell Normoxia
3. Existing Challenges concerning Stem and Progenitor Cells for CA
3.1. Need for Immortalized Cell Lines
3.2. Need for Efficient Stimulation of Differentiation
3.2.1. Physical Cues
3.2.2. Biochemical Cues
3.2.3. Using miRNAs as Stimulators of Myoblast Differentiation
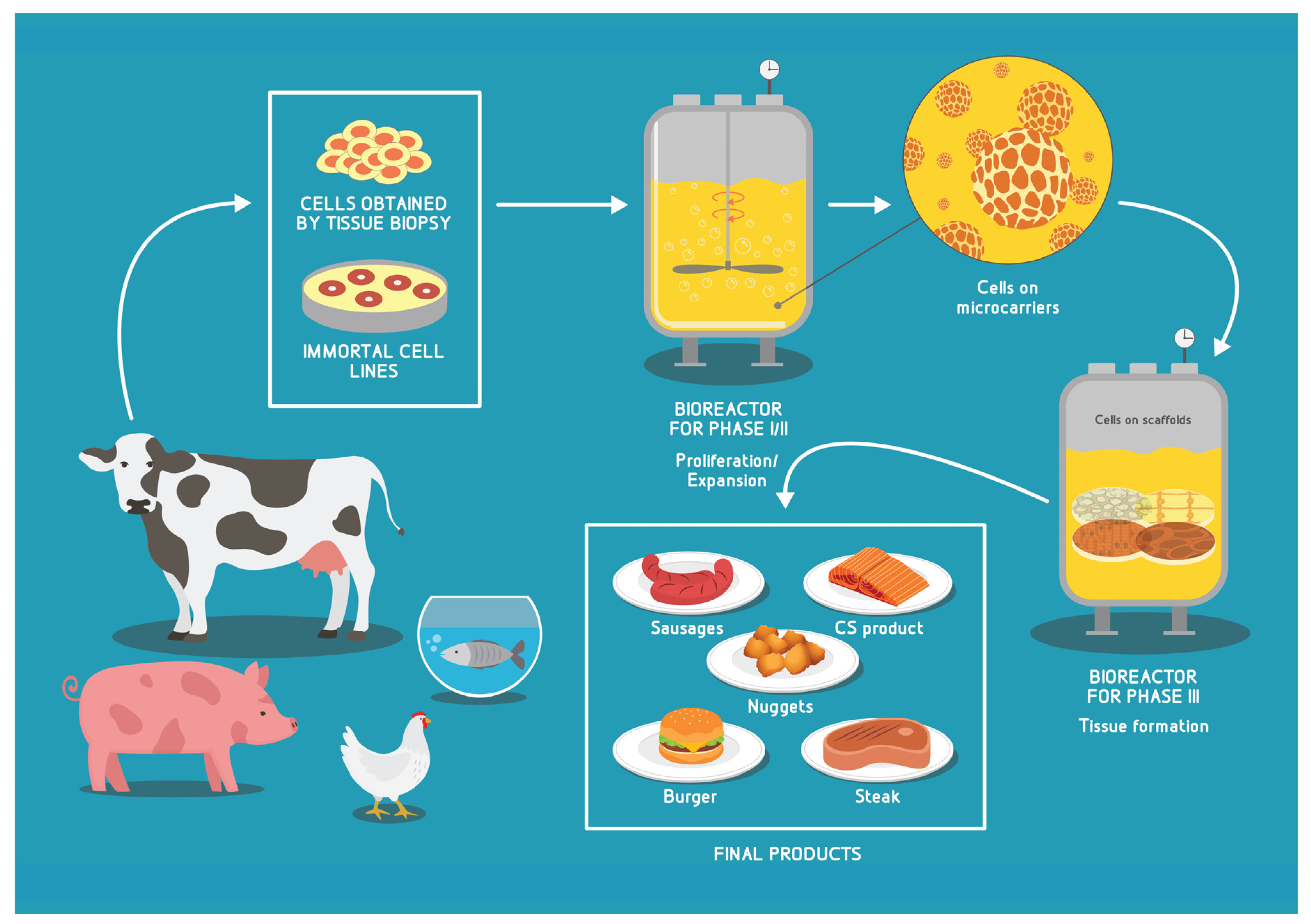
4. Achieving Industry Scale CM/CS Production
5. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACAT2 | acetyl-CoA acetyltransferase 2 |
| ACSL1 | Long-chain acyl-CoA synthetase 1 |
| ACTC1 | actin alpha cardiac muscle 1gene |
| ADSCs | adipose tissue-derived stem cells |
| AIF1L | allograft inflammatory factor 1 like |
| ALA | alpha-linolenic acid |
| APOH | apolipoprotein H |
| APs | alternative proteins |
| ASCs | adult stem cells |
| ATP | adenosine triphosphate |
| BM | bone marrow |
| BM-MSCs | bone marrow-derived mesenchymal stem cells |
| BSCs | bovine satellite cells |
| BSMC | bovine smooth muscle cells |
| CA | cellular agriculture |
| C/BEP | CCAAT/enhancer-binding protein |
| CDK4 | cyclin-dependent kinase 4 |
| Cdc25A | Cell Division Cycle 25A gene |
| C/EBPα/β | CCAAT enhancer-binding protein alpha/beta |
| CF | Cultured/cultivated fat |
| circRNA | Circular RNA |
| CM | Cultured/cultivated meat |
| c-Met | tyrosine-protein kinase Met gene |
| COL1A1 | pro-alpha1 chains of type I collagen coding gene |
| COVID-19 | Coronavirus Disease 2019 |
| CRISPR | clustered regularly interspaced short palindromic repeats |
| CRTC2 | CREB-regulated transcription coactivator 2 isoform X1 |
| CRTC3 | CREB-regulated transcription coactivator 3 |
| CS | cultivated seafood |
| CSRP3 | cysteine and glycine-rich protein 3 |
| Cx43 | connexin 43 gene |
| DEX | dexamethasone |
| DFAT | Dedifferentiated fat |
| DHA | docosahexaenoic acid |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DNA | deoxyribonucleic acid |
| DNApola | DNA polymerase alpha gene |
| DrAMPCs | Drosophila melanogaster adult muscle progenitor-like cells |
| ECM | extracellular matrix |
| ECs | endothelial cells |
| EGF | epidermal growth factor |
| ELN | Elastin gene |
| EPA | eicosapentaenoic acid |
| ESCs | embryonic stem cells |
| Ezh2 | Enhancer Of Zeste 2 Polycomb Repressive Complex 2 Subunit gene |
| E2F5 | E2F Transcription Factor 5 gene |
| FACS | fluorescence-activated cell sorting |
| FAO | Food and Agriculture Organization of the United Nations |
| FAPs | fibro-adipogenic progenitor cells |
| FBS | fetal bovine serum |
| FFCs | feather follicular cells |
| FGF-2 | fibroblast growth factor-2/basic fibroblast growth factor |
| FGF9 | fibroblast growth factor 9 |
| FGFR2 | fibroblast growth factor receptor 2 |
| FOS | fructo-oligosaccharides |
| FoxO1 | Forkhead Box O1 gene |
| Fstl1 | Follistatin-like 1 gene |
| Gal-1 | Galectin-1 |
| GelMA | Gelatin methacryloyl |
| GFI | Good Food Institute |
| GFs | growth factors |
| GLUT4 | glucose transporter type 4 |
| GM | genetically modified |
| GMO | genetically modified organism |
| GPR43 | G-Protein Coupled Receptor 43 |
| GR | glucocorticoid receptor |
| G6PD | glucose-6-phosphate dehydrogenase gene |
| HC | hydrocortisone |
| HDAC4 | Histone Deacetylase 4 gene |
| HeLa | Henrietta Lacks |
| Hox-A11 | Homeobox A11 gene |
| HP-1β | Heterochromatin Protein 1beta gene |
| IBMX | isobutylmethylxanthine |
| ID1 | Inhibitor Of DNA Binding 1 |
| IFN-γ | interferon-gamma |
| IGF-1/2 | Insulin-Like Growth Factor 1/2 gene |
| IGF1R | Insulin-Like Growth Factor 1 Receptor |
| IL-1α/β | interleukin-1 alpha/beta |
| IL-4/6/13/15 | interleukin-4/6/13/15 |
| IMF | intramuscular fat |
| IMPs | intramuscular preadipocytes |
| INSM1 | Insulinoma-associated 1 |
| iPSCs | induced pluripotent stem cells |
| IWR1 | tankyrase/Wnt inhibitor |
| KLF4 | Krüppel-like factor 4 |
| LA | linoleic acid |
| LIF | leukemia inhibitory factor |
| lncRNA | long non-coding RNA |
| LPAR1 | Lysophosphatidic Acid Receptor 1 |
| LR3-IGF-1 | long-chain human insulin growth factor-1 |
| MAPK | mitogen-activated protein kinase |
| MAs | mature lipid-containing adipocytes |
| MC | microcarriers |
| MEF | mouse embryonic fibroblast |
| Mef2a | Myocyte Enhancer Factor 2A gene |
| miRNAs | microRNAs |
| MPCs | muscle progenitor cells |
| MRF | myogenic regulatory factor |
| MRF4 | myogenic regulatory factor 4 gene |
| MSCs | mesenchymal stem cells |
| mTOR | Mechanistic Target Of Rapamycin Kinase gene |
| Myf5 | Myogenic Factor 5 gene |
| Myf6 | Myogenic Factor 6 gene |
| Myh 1/2/4 | Myosin Heavy Chain 1/2/4 gene |
| MyoD | myoblast determination protein 1 |
| NASA | National Aeronautics and Space Administration |
| NENF | neudesin neurotrophic factor |
| N-Ras | Neuroblastoma RAS Viral Oncogene Homolog gene |
| OCT4 | octamer-binding transcription factor 4 |
| OPTi-OX | optimized inducible overexpression |
| O2 | oxygen |
| PAI-1/SERPINE1 | plasminogen activator inhibitor 1 |
| Pax3/7 | Paired Box 3/7 gene |
| PDGF-BB | platelet-derived growth factor BB |
| PDGFRA/B | platelet-derived growth factor receptor A/B |
| PDLIM3/5 | PDZ And LIM Domain 3/5 |
| PDMS | polydimethylsiloxane |
| POU5F1 | POU domain, class 5, transcription factor 1 |
| PPARγ | peroxisome proliferator-activated receptor-gamma |
| PPI | pea protein isolate |
| PPP3R1 | protein phosphatase 3 regulatory subunit B, alpha |
| Pref1 | preadipocyte factor 1 |
| PTEN | Phosphatase And Tensin Homolog gene |
| Purβ | Purine Rich Element Binding Protein B |
| RGD | Arginine-Glycine-Aspartate motif |
| RNA | ribonucleic acid |
| ROCK1 | rho-associated, coiled-coil-containing protein kinase 1 gene |
| RPS6KA6 | Ribosomal Protein S6 Kinase A6 gene |
| Rybp | RING1 And YY1 Binding Protein gene |
| R&D | research and development |
| SB203580 | Adezmapimod, p38 MAPK inhibitor |
| SCs | satellite cells |
| SEM | scanning electron microscopy |
| SESN1 | Sestrin 1 gene |
| SFA | Singapore Food Agency |
| SkGM-2 | Skeletal Muscle Cell Growth Medium-2 |
| SMAD1/2/4 | SMAD Family Member 1/2/4 gene |
| SOX2 | SRY-Box Transcription Factor 2 |
| Sox6 | SRY-Box Transcription Factor 6 gene |
| SPI | soy protein isolate |
| Sp3 | Sp3 Transcription Factor gene |
| srebp2/ldlr | KLF7-Krüppel-like factor 7 |
| SRF | serum response factor |
| SRSF10 | serine and arginine-rich splicing factor 10 |
| SSEAs | stage-specific embryonic antigens |
| SVCs | stromal vascular cells |
| TAZ | transcriptional co-activator with PDZ-binding motif |
| TE | tissue engineering |
| TERT | telomerase reverse transcriptase |
| TFRC | Transferrin Receptor |
| TGF-bR1 | transforming growth factor-beta receptor type 1 gene |
| TGF-β | transforming growth factor-beta |
| TIMP3 | TIMP Metallopeptidase Inhibitor 3 gene |
| TNF-α | tumor necrosis factor-alpha |
| TORC2 | Transducer of regulated cAMP response element-binding protein (CREB) 2 |
| TSP | textured soy protein |
| UCP2 | Uncoupling Protein 2 gene |
| UFA | unsaturated fatty acid |
| US | United States |
| Utrn | Utrophin gene |
| Wnt1 | Wnt Family Member 1 gene |
| XBP1 | X-box binding protein 1 |
| YAP | Yes-associated protein |
| YY1 | Yin Yang 1 gene |
| ZNF263 | Zinc finger protein 263 |
| 5-Aza | 5-Aza-2′-deoxycytidine |
References
- FAO. The Future of Food and Agriculture—Trends and Challenges; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2017; p. 180. [Google Scholar]
- Dasgupta, S.; Robinson, E.J.Z. Impact of COVID-19 on Food Insecurity Using Multiple Waves of High Frequency Household Surveys. Sci. Rep. 2022, 12, 1865. [Google Scholar] [CrossRef] [PubMed]
- Roubík, H.; Lošťák, M.; Ketuama, C.T.; Procházka, P.; Soukupová, J.; Hakl, J.; Karlík, P.; Hejcman, M. Current Coronavirus Crisis and Past Pandemics—What Can Happen in Post-COVID-19 Agriculture? Sustain. Prod. Consum. 2022, 30, 752–760. [Google Scholar] [CrossRef] [PubMed]
- The Ukraine War: A Global Crisis? Available online: https://www.crisisgroup.org/europe-central-asia/eastern-europe/ukraine/ukraine-war-global-crisis (accessed on 30 March 2022).
- FAO Ukraine. Note on the Impact of the War on Food Security in Ukraine; FAO: Rome, Italy, 2022; ISBN 978-92-5-135950-1. [Google Scholar]
- Amid Inflation, Food Banks See Increased Demand for Protein-Rich Foods. Available online: https://www.publicnewsservice.org/index.php?/content/article/76377-1 (accessed on 30 March 2022).
- FAO. Food Outlook—Biannual Report on Global Food Markets; FAO: Rome, Italy, 2020; ISBN 978-92-5-132848-4. [Google Scholar]
- Costello, C.; Cao, L.; Gelcich, S.; Cisneros-Mata, M.Á.; Free, C.M.; Froehlich, H.E.; Golden, C.D.; Ishimura, G.; Maier, J.; Macadam-Somer, I.; et al. The Future of Food from the Sea. Nature 2020, 588, 95–100. [Google Scholar] [CrossRef]
- The Good Food Institute. Global Food System Transition Is Necessary to Keep Warming Below 1.5 °C—Opportunities for Alternative Proteins; The Good Food Institute: Washington, DC, USA, 2021. [Google Scholar]
- Onwezen, M.C.; Bouwman, E.P.; Reinders, M.J.; Dagevos, H. A Systematic Review on Consumer Acceptance of Alternative Proteins: Pulses, Algae, Insects, Plant-Based Meat Alternatives, and Cultured Meat. Appetite 2021, 159, 105058. [Google Scholar] [CrossRef]
- Choudhury, D.; Tseng, T.W.; Swartz, E. The Business of Cultured Meat. Trends Biotechnol. 2020, 38, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Bodiou, V.; Moutsatsou, P.; Post, M.J. Microcarriers for Upscaling Cultured Meat Production. Front. Nutr. 2020, 7, 10. [Google Scholar] [CrossRef]
- Zernov, A.; Baruch, L.; Machluf, M. Chitosan-Collagen Hydrogel Microparticles as Edible Cell Microcarriers for Cultured Meat. Food Hydrocoll. 2022, 129, 107632. [Google Scholar] [CrossRef]
- Seah, J.S.H.; Singh, S.; Tan, L.P.; Choudhury, D. Scaffolds for the Manufacture of Cultured Meat. Crit. Rev. Biotechnol. 2022, 42, 311–323. [Google Scholar] [CrossRef]
- Ianovici, I.; Zagury, Y.; Redensky, I.; Lavon, N.; Levenberg, S. 3D-Printable Plant Protein-Enriched Scaffolds for Cultivated Meat Development. Biomaterials 2022, 284, 121487. [Google Scholar] [CrossRef]
- Wollschlaeger, J.O.; Maatz, R.; Albrecht, F.B.; Klatt, A.; Heine, S.; Blaeser, A.; Kluger, P.J. Scaffolds for Cultured Meat on the Basis of Polysaccharide Hydrogels Enriched with Plant-Based Proteins. Gels 2022, 8, 94. [Google Scholar] [CrossRef]
- Bomkamp, C.; Skaalure, S.C.; Fernando, G.F.; Ben-Arye, T.; Swartz, E.W.; Specht, E.A. Scaffolding Biomaterials for 3D Cultivated Meat: Prospects and Challenges. Adv. Sci. 2022, 9, 2102908. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.; Kurisawa, M. Integrating Biomaterials and Food Biopolymers for Cultured Meat Production. Acta Biomater. 2021, 124, 108–129. [Google Scholar] [CrossRef] [PubMed]
- Pajčin, I.; Knežić, T.; Azoulay, I.S.; Vlajkov, V.; Djisalov, M.; Janjušević, L.; Grahovac, J.; Gadjanski, I. Bioengineering Outlook on Cultivated Meat Production. Micromachines 2022, 13, 402. [Google Scholar] [CrossRef]
- Djisalov, M.; Knežić, T.; Podunavac, I.; Živojević, K.; Radonic, V.; Knežević, N.Ž.; Bobrinetskiy, I.; Gadjanski, I. Cultivating Multidisciplinarity: Manufacturing and Sensing Challenges in Cultured Meat Production. Biology 2021, 10, 204. [Google Scholar] [CrossRef] [PubMed]
- Allan, S.J.; De Bank, P.A.; Ellis, M.J. Bioprocess Design Considerations for Cultured Meat Production with a Focus on the Expansion Bioreactor. Front. Sustain. Food Syst. 2019, 3, 44. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, X.; Li, X.; Du, G.; Zhou, J.; Chen, J. Challenges and Possibilities for Bio-Manufacturing Cultured Meat. Trends Food Sci. Technol. 2020, 97, 443–450. [Google Scholar] [CrossRef]
- Biechele, P.; Busse, C.; Solle, D.; Scheper, T.; Reardon, K. Sensor Systems for Bioprocess Monitoring. Eng. Life Sci. 2015, 15, 469–488. [Google Scholar] [CrossRef]
- Handral, H.K.; Hua Tay, S.; Wan Chan, W.; Choudhury, D. 3D Printing of Cultured Meat Products. Crit. Rev. Food Sci. Nutr. 2022, 62, 272–281. [Google Scholar] [CrossRef]
- Holmes, J.T.; Jaberansari, Z.; Collins, W.; Latour, M.L.; Modulevsky, D.J.; Pelling, A.E. Homemade Bread: Repurposing an Ancient Technology for in Vitro Tissue Engineering. Biomaterials 2022, 280, 121267. [Google Scholar] [CrossRef]
- Shaikh, S.; Lee, E.; Ahmad, K.; Ahmad, S.-S.; Chun, H.; Lim, J.; Lee, Y.; Choi, I. Cell Types Used for Cultured Meat Production and the Importance of Myokines. Foods 2021, 10, 2318. [Google Scholar] [CrossRef]
- Reiss, J.; Robertson, S.; Suzuki, M. Cell Sources for Cultivated Meat: Applications and Considerations throughout the Production Workflow. Int. J. Mol. Sci. 2021, 22, 7513. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Zhou, J.; Du, G.; Chen, J. Bioprocessing Technology of Muscle Stem Cells: Implications for Cultured Meat. Trends Biotechnol. 2021, in press. [CrossRef] [PubMed]
- Humbird, D. Scale-Up Economics for Cultured Meat: Techno-Economic Analysis and Due Diligence. Biotechnol. Bioeng. 2021, 118, 3239–3250. [Google Scholar] [CrossRef]
- Mauro, A. Satellite Cell of Skeletal Muscle Fibers. J. Biophys. Biochem. Cytol. 1961, 9, 493–495. [Google Scholar] [CrossRef]
- Yin, H.; Price, F.; Rudnicki, M.A. Satellite Cells and the Muscle Stem Cell Niche. Physiol. Rev. 2013, 93, 23–67. [Google Scholar] [CrossRef] [PubMed]
- Cornelison, D.D.; Olwin, B.B.; Rudnicki, M.A.; Wold, B.J. MyoD(−/−) Satellite Cells in Single-Fiber Culture Are Differentiation Defective and MRF4 Deficient. Dev. Biol. 2000, 224, 122–137. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Swennen, G.N.M.; Messmer, T.; Gagliardi, M.; Molin, D.G.M.; Li, C.; Zhou, G.; Post, M.J. Maintaining Bovine Satellite Cells Stemness through P38 Pathway. Sci. Rep. 2018, 8, 10808. [Google Scholar] [CrossRef]
- Furuhashi, M.; Morimoto, Y.; Shima, A.; Nakamura, F.; Ishikawa, H.; Takeuchi, S. Formation of Contractile 3D Bovine Muscle Tissue for Construction of Millimetre-Thick Cultured Steak. Npj Sci. Food 2021, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Verbruggen, S.; Luining, D.; van Essen, A.; Post, M.J. Bovine Myoblast Cell Production in a Microcarriers-Based System. Cytotechnology 2018, 70, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Haraguchi, Y.; Yoshida, A.; Takahashi, H.; Yamanaka, K.; Sawamura, N.; Asahi, T.; Shimizu, T. Proliferation and Differentiation of Primary Bovine Myoblasts Using Chlorella Vulgaris Extract for Sustainable Production of Cultured Meat. Biotechnol. Prog. 2022, e3239. [Google Scholar] [CrossRef]
- Haraguchi, Y.; Shimizu, T. Three-Dimensional Tissue Fabrication System by Co-Culture of Microalgae and Animal Cells for Production of Thicker and Healthy Cultured Food. Biotechnol. Lett. 2021, 43, 1117–1129. [Google Scholar] [CrossRef] [PubMed]
- MacQueen, L.A.; Alver, C.G.; Chantre, C.O.; Ahn, S.; Cera, L.; Gonzalez, G.M.; O’Connor, B.B.; Drennan, D.J.; Peters, M.M.; Motta, S.E.; et al. Muscle Tissue Engineering in Fibrous Gelatin: Implications for Meat Analogs. Npj Sci. Food 2019, 3, 20. [Google Scholar] [CrossRef] [PubMed]
- Wilschut, K.J.; Jaksani, S.; Van Den Dolder, J.; Haagsman, H.P.; Roelen, B.A.J. Isolation and Characterization of Porcine Adult Muscle-Derived Progenitor Cells. J. Cell. Biochem. 2008, 105, 1228–1239. [Google Scholar] [CrossRef] [PubMed]
- Baquero-Perez, B.; Kuchipudi, S.V.; Nelli, R.K.; Chang, K.-C. A Simplified but Robust Method for the Isolation of Avian and Mammalian Muscle Satellite Cells. BMC Cell Biol. 2012, 13, 16. [Google Scholar] [CrossRef] [PubMed]
- Greene, E.A.; Raub, R.H. Procedures for Harvesting Satellite Cells from Equine Skeletal Muscle. J. Equine Vet. Sci. 1992, 12, 33–35. [Google Scholar] [CrossRef]
- Wang, S.; Li, K.; Gao, H.; Liu, Z.; Shi, S.; Tan, Q.; Wang, Z. Ubiquitin-Specific Peptidase 8 Regulates Proliferation and Early Differentiation of Sheep Skeletal Muscle Satellite Cells. Czech J. Anim. Sci. 2021, 66, 87–96. [Google Scholar] [CrossRef]
- Wang, Y.; Xiao, X.; Wang, L. In Vitro Characterization of Goat Skeletal Muscle Satellite Cells. Anim. Biotechnol. 2020, 31, 115–121. [Google Scholar] [CrossRef]
- Yamanouchi, K.; Hosoyama, T.; Murakami, Y.; Nakano, S.-I.; Nishihara, M. Satellite Cell Differentiation in Goat Skeletal Muscle Single Fiber Culture. J. Reprod. Dev. 2009, 55, 252–255. [Google Scholar] [CrossRef]
- Yamanouchi, K.; Hosoyama, T.; Murakami, Y.; Nishihara, M. Myogenic and Adipogenic Properties of Goat Skeletal Muscle Stem Cells. J. Reprod. Dev. 2006, 53, 51–58. [Google Scholar] [CrossRef]
- Jankowski, M.; Mozdziak, P.; Petitte, J.; Kulus, M.; Kempisty, B. Avian Satellite Cell Plasticity. Animals 2020, 10, 1322. [Google Scholar] [CrossRef]
- McFarland, D.C.; Doumit, M.E.; Minshall, R.D. The Turkey Myogenic Satellite Cell: Optimization of in Vitro Proliferation and Differentiation. Tissue Cell 1988, 20, 899–908. [Google Scholar] [CrossRef]
- Nihashi, Y.; Umezawa, K.; Shinji, S.; Hamaguchi, Y.; Kobayashi, H.; Kono, T.; Ono, T.; Kagami, H.; Takaya, T. Distinct Cell Proliferation, Myogenic Differentiation, and Gene Expression in Skeletal Muscle Myoblasts of Layer and Broiler Chickens. Sci. Rep. 2019, 9, 16527. [Google Scholar] [CrossRef] [PubMed]
- Gursel, I.V.; Sturme, M.; Hugenholtz, J.; Bruins, M. Review and Analysis of Studies on Sustainability of Cultured Meat; Wageningen Food & Biobased Research: Wageningen, The Netherlands, 2022. [Google Scholar]
- Powell, R.L.; Dodson, M.V.; Cloud, J.G. Cultivation and Differentiation of Satellite Cells From Skeletal Muscle of the Rainbow Trout Salmo Gairdneri. J. Exp. Zool. 1989, 250, 333–338. [Google Scholar] [CrossRef]
- Koumans, J.T.M.; Akster, H.A.; Dulos, G.J.; Osse, J.W.M. Myosatellite Cells of Cyprinus Carpio (Teleostei) in Vitro: Isolation, Recognition and Differentiation. Cell Tissue Res. 1990, 261, 173–181. [Google Scholar] [CrossRef]
- Froehlich, J.M.; Seiliez, I.; Gabillard, J.-C.; Biga, P.R. Preparation of Primary Myogenic Precursor Cell/Myoblast Cultures from Basal Vertebrate Lineages. J. Vis. Exp. 2014, 86, e51354. [Google Scholar] [CrossRef]
- Benjaminson, M.A.; Gilchriest, J.A.; Lorenz, M. In Vitro Edible Muscle Protein Production System (MPPS): Stage 1, Fish. Acta Astronaut. 2002, 51, 879–889. [Google Scholar] [CrossRef]
- Segalés, J.; Perdiguero, E.; Muñoz-Cánoves, P. Regulation of Muscle Stem Cell Functions: A Focus on the P38 MAPK Signaling Pathway. Front. Cell Dev. Biol. 2016, 4, 91. [Google Scholar] [CrossRef]
- Choi, K.-H.; Yoon, J.W.; Kim, M.; Jeong, J.; Ryu, M.; Park, S.; Jo, C.; Lee, C.-K. Optimization of Culture Conditions for Maintaining Pig Muscle Stem Cells in Vitro. Food Sci. Anim. Resour. 2020, 40, 659–667. [Google Scholar] [CrossRef]
- Yin, H.; Zhao, J.; He, H.; Chen, Y.; Wang, Y.; Li, D.; Zhu, Q. Gga-MiR-3525 Targets PDLIM3 through the MAPK Signaling Pathway to Regulate the Proliferation and Differentiation of Skeletal Muscle Satellite Cells. Int. J. Mol. Sci. 2020, 21, 5573. [Google Scholar] [CrossRef]
- He, H.; Yin, H.; Yu, X.; Zhang, Y.; Ma, M.; Li, D.; Zhu, Q. PDLIM5 Affects Chicken Skeletal Muscle Satellite Cell Proliferation and Differentiation via the P38-MAPK Pathway. Animals 2021, 11, 1016. [Google Scholar] [CrossRef]
- Aubel, P.; Pain, B. Chicken Embryonic Stem Cells: Establishment and Characterization. In Epiblast Stem Cells: Methods and Protocols; Alberio, R., Ed.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2013; pp. 137–150. ISBN 978-1-62703-628-3. [Google Scholar]
- Hong, Y.; Winkler, C.; Schartl, M. Pluripotency and Differentiation of Embryonic Stem Cell Lines from the Medakafish (Oryzias Latipes). Mech. Dev. 1996, 60, 33–44. [Google Scholar] [CrossRef]
- Bogliotti, Y.S.; Wu, J.; Vilarino, M.; Okamura, D.; Soto, D.A.; Zhong, C.; Sakurai, M.; Sampaio, R.V.; Suzuki, K.; Izpisua Belmonte, J.C.; et al. Efficient Derivation of Stable Primed Pluripotent Embryonic Stem Cells from Bovine Blastocysts. Proc. Natl. Acad. Sci. USA 2018, 115, 2090–2095. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.-H.; Lee, D.-K.; Oh, J.-N.; Kim, S.-H.; Lee, M.; Woo, S.-H.; Kim, D.-Y.; Lee, C.-K. Pluripotent Pig Embryonic Stem Cell Lines Originating from in Vitro-Fertilized and Parthenogenetic Embryos. Stem Cell Res. 2020, 49, 102093. [Google Scholar] [CrossRef] [PubMed]
- Bhat, Z.F.; Bhat, H.; Kumar, S. Cultured Meat—A Humane Meat Production System. In Principles of Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1369–1388. [Google Scholar]
- Gandolfi, F.; Pennarossa, G.; Maffei, S.; Brevini, T.A.L. Why Is It so Difficult to Derive Pluripotent Stem Cells in Domestic Ungulates? Reprod. Domest. Anim. 2012, 47, 11–17. [Google Scholar] [CrossRef]
- Navarro, M.; Soto, D.A.; Pinzon, C.A.; Wu, J.; Ross, P.J.; Navarro, M.; Soto, D.A.; Pinzon, C.A.; Wu, J.; Ross, P.J. Livestock Pluripotency Is Finally Captured in Vitro. Reprod. Fertil. Dev. 2020, 32, 11–39. [Google Scholar] [CrossRef]
- Soto, D.A.; Navarro, M.; Zheng, C.; Halstead, M.M.; Zhou, C.; Guiltinan, C.; Wu, J.; Ross, P.J. Simplification of Culture Conditions and Feeder-Free Expansion of Bovine Embryonic Stem Cells. Sci. Rep. 2021, 11, 11045. [Google Scholar] [CrossRef]
- Vilarino, M.; Alba Soto, D.; Soledad Bogliotti, Y.; Yu, L.; Zhang, Y.; Wang, C.; Paulson, E.; Zhong, C.; Jin, M.; Carlos Izpisua Belmonte, J.; et al. Derivation of Sheep Embryonic Stem Cells under Optimized Conditions. Reproduction 2020, 160, 761–772. [Google Scholar] [CrossRef]
- Eyal-Giladi, H.; Kochav, S. From Cleavage to Primitive Streak Formation: A Complementary Normal Table and a New Look at the First Stages of the Development of the Chick: I. General Morphology. Dev. Biol. 1976, 49, 321–337. [Google Scholar] [CrossRef]
- Xiong, C.; Wang, M.; Ling, W.; Xie, D.; Chu, X.; Li, Y.; Huang, Y.; Li, T.; Otieno, E.; Qiu, X.; et al. Advances in Isolation and Culture of Chicken Embryonic Stem Cells In Vitro. Cell. Reprogram. 2020, 22, 43–54. [Google Scholar] [CrossRef]
- Yi, M.; Hong, N.; Hong, Y. Generation of Medaka Fish Haploid Embryonic Stem Cells. Science 2009, 326, 430–433. [Google Scholar] [CrossRef]
- Hong, Y.; Chen, S.; Schartl, M. Embryonic Stem Cells in Fish: Current Status and Perspectives. Fish Physiol. Biochem. 2000, 22, 165–170. [Google Scholar] [CrossRef]
- Hong, N.; Li, Z.; Hong, Y. Fish Stem Cell Cultures. Int. J. Biol. Sci. 2011, 7, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Yu, J.; Vodyanik, M.A.; Smuga-Otto, K.; Antosiewicz-Bourget, J.; Frane, J.L.; Tian, S.; Nie, J.; Jonsdottir, G.A.; Ruotti, V.; Stewart, R.; et al. Induced Pluripotent Stem Cell Lines Derived from Human Somatic Cells. Science 2007, 318, 1917–1920. [Google Scholar] [CrossRef]
- Talluri, T.R.; Kumar, D.; Glage, S.; Garrels, W.; Ivics, Z.; Debowski, K.; Behr, R.; Niemann, H.; Kues, W.A. Derivation and Characterization of Bovine Induced Pluripotent Stem Cells by Transposon-Mediated Reprogramming. Cell. Reprogram. 2015, 17, 131–140. [Google Scholar] [CrossRef]
- Su, Y.; Wang, L.; Fan, Z.; Liu, Y.; Zhu, J.; Kaback, D.; Oudiz, J.; Patrick, T.; Yee, S.P.; Tian, X.C.; et al. Establishment of Bovine-Induced Pluripotent Stem Cells. Int. J. Mol. Sci. 2021, 22, 10489. [Google Scholar] [CrossRef]
- Gao, X.; Nowak-Imialek, M.; Chen, X.; Chen, D.; Herrmann, D.; Ruan, D.; Chen, A.C.H.; Eckersley-Maslin, M.A.; Ahmad, S.; Lee, Y.L.; et al. Establishment of Porcine and Human Expanded Potential Stem Cells. Nat. Cell Biol. 2019, 21, 687–699. [Google Scholar] [CrossRef]
- Sandmaier, S.E.S.; Nandal, A.; Powell, A.; Garrett, W.; Blomberg, L.; Donovan, D.M.; Talbot, N.; Telugu, B.P. Generation of Induced Pluripotent Stem Cells from Domestic Goats. Mol. Reprod. Dev. 2015, 82, 709–721. [Google Scholar] [CrossRef]
- Breton, A.; Sharma, R.; Diaz, A.C.; Parham, A.G.; Graham, A.; Neil, C.; Whitelaw, C.B.; Milne, E.; Donadeu, F.X. Derivation and Characterization of Induced Pluripotent Stem Cells from Equine Fibroblasts. Stem Cells Dev. 2013, 22, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Balehosur, D.; Murray, B.; Kelly, J.M.; Sumer, H.; Verma, P.J. Generation and Characterization of Reprogrammed Sheep Induced Pluripotent Stem Cells. Theriogenology 2012, 77, 338–346.e1. [Google Scholar] [CrossRef]
- Amilon, K.R.; Cortes-Araya, Y.; Moore, B.; Lee, S.; Lillico, S.; Breton, A.; Esteves, C.L.; Donadeu, F.X. Generation of Functional Myocytes from Equine Induced Pluripotent Stem Cells. Cell. Reprogram. 2018, 20, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Scarfone, R.A.; Pena, S.M.; Russell, K.A.; Betts, D.H.; Koch, T.G. The Use of Induced Pluripotent Stem Cells in Domestic Animals: A Narrative Review. BMC Vet. Res. 2020, 16, 477. [Google Scholar] [CrossRef] [PubMed]
- Genovese, N.J.; Domeier, T.L.; Telugu, B.P.V.L.; Roberts, R.M. Enhanced Development of Skeletal Myotubes from Porcine Induced Pluripotent Stem Cells. Sci. Rep. 2017, 7, 41833. [Google Scholar] [CrossRef]
- Rosselló, R.A.; Chen, C.-C.; Dai, R.; Howard, J.T.; Hochgeschwender, U.; Jarvis, E.D. Mammalian Genes Induce Partially Reprogrammed Pluripotent Stem Cells in Non-Mammalian Vertebrate and Invertebrate Species. eLife 2013, 2, e00036. [Google Scholar] [CrossRef]
- Pain, B.; Kress, C.; Rival-Gervier, S. Pluripotency in Avian Species. Int. J. Dev. Biol. 2018, 62, 245–255. [Google Scholar] [CrossRef]
- Yu, P.; Lu, Y.; Jordan, B.J.; Liu, Y.; Yang, J.-Y.; Hutcheson, J.M.; Ethridge, C.L.; Mumaw, J.L.; Kinder, H.A.; Beckstead, R.B.; et al. Nonviral Minicircle Generation of Induced Pluripotent Stem Cells Compatible with Production of Chimeric Chickens. Cell. Reprogram. 2014, 16, 366–378. [Google Scholar] [CrossRef]
- Kim, Y.M.; Park, Y.H.; Lim, J.M.; Jung, H.; Han, J.Y. Technical Note: Induction of Pluripotent Stem Cell-like Cells from Chicken Feather Follicle Cells. J. Anim. Sci. 2017, 95, 3479. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Zhou, Y.; Xu, W.; Jiang, M.; Li, H.; Long, M.; Liu, W.; Liu, J.; Zhao, X.; Xiao, Y. Generation of Stable Induced Pluripotent Stem-like Cells from Adult Zebra Fish Fibroblasts. Int. J. Biol. Sci. 2019, 15, 2340–2349. [Google Scholar] [CrossRef]
- Xu, W.; Li, H.; Peng, L.; Pu, L.; Xiang, S.; Li, Y.; Tao, L.; Liu, W.; Liu, J.; Xiao, Y.; et al. Fish Pluripotent Stem-Like Cell Line Induced by Small-Molecule Compounds From Caudal Fin and Its Developmental Potentiality. Front. Cell Dev. Biol. 2021, 9, 817779. [Google Scholar] [CrossRef]
- Hernandez, J. GMO Is out, “Bioengineered” Is in, as New U.S. Food Labeling Rules Take Effect; NPR: Washington, DC, USA, 2022. [Google Scholar]
- Kim, Y.; Kim, S.; Arora, N. GMO Labeling Policy and Consumer Choice. J. Mark. 2022, 86, 21–39. [Google Scholar] [CrossRef]
- Pawlowski, M.; Ortmann, D.; Bertero, A.; Tavares, J.M.; Pedersen, R.A.; Vallier, L.; Kotter, M.R.N. Inducible and Deterministic Forward Programming of Human Pluripotent Stem Cells into Neurons, Skeletal Myocytes, and Oligodendrocytes. Stem Cell Rep. 2017, 8, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Du, M.; Yin, J.; Zhu, M.J. Cellular Signaling Pathways Regulating the Initial Stage of Adipogenesis and Marbling of Skeletal Muscle. Meat Sci. 2010, 86, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Das, A.K.; Yang, Q.-Y.; Zhu, M.-J.; Du, M. Zfp423 Promotes Adipogenic Differentiation of Bovine Stromal Vascular Cells. PLoS ONE 2012, 7, e47496. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Espinosa, J.J.; González-Dávalos, L.; Shimada, A.; Piña, E.; Varela-Echavarria, A.; Mora, O. Bovine (Bos Taurus) Bone Marrow Mesenchymal Cell Differentiation to Adipogenic and Myogenic Lineages. Cells Tissues Organs 2016, 201, 51–64. [Google Scholar] [CrossRef]
- Okamura, L.H.; Cordero, P.; Palomino, J.; Parraguez, V.H.; Torres, C.G.; Peralta, O.A. Myogenic Differentiation Potential of Mesenchymal Stem Cells Derived from Fetal Bovine Bone Marrow. Anim. Biotechnol. 2018, 29, 1–11. [Google Scholar] [CrossRef]
- Bosnakovski, D.; Mizuno, M.; Kim, G.; Takagi, S.; Okumura, M.; Fujinaga, T. Isolation and Multilineage Differentiation of Bovine Bone Marrow Mesenchymal Stem Cells. Cell Tissue Res. 2005, 319, 243–253. [Google Scholar] [CrossRef]
- Ben-Arye, T.; Levenberg, S. Tissue Engineering for Clean Meat Production. Front. Sustain. Food Syst. 2019, 3, 46. [Google Scholar] [CrossRef]
- Hill, A.B.T.; Bressan, F.F.; Murphy, B.D.; Garcia, J.M. Applications of Mesenchymal Stem Cell Technology in Bovine Species. Stem Cell Res. Ther. 2019, 10, 44. [Google Scholar] [CrossRef]
- Gao, Y.; Guo, Y.; Duan, A.; Cheng, D.; Zhang, S.; Wang, H. Optimization of Culture Conditions for Maintaining Porcine Induced Pluripotent Stem Cells. DNA Cell Biol. 2014, 33, 1–11. [Google Scholar] [CrossRef]
- Sampaio, R.V.; Chiaratti, M.R.; Santos, D.C.N.; Bressan, F.F.; Sangalli, J.R.; Sá, A.L.A.; Silva, T.V.G.; Costa, N.N.; Cordeiro, M.S.; Santos, S.S.D.; et al. Generation of Bovine (Bos Indicus) and Buffalo (Bubalus Bubalis) Adipose Tissue Derived Stem Cells: Isolation, Characterization, and Multipotentiality. Genet. Mol. Res. 2015, 14, 53–62. [Google Scholar] [CrossRef]
- De la Fuente-Hernandez, M.A.; Sarabia-Sanchez, M.A.; Melendez-Zajgla, J.; Maldonado-Lagunas, V. LncRNAs in Mesenchymal Differentiation. Am. J. Physiol.-Cell Physiol. 2022, 322, C421–C460. [Google Scholar] [CrossRef] [PubMed]
- Lavial, F.; Acloque, H.; Bertocchini, F.; Macleod, D.J.; Boast, S.; Bachelard, E.; Montillet, G.; Thenot, S.; Sang, H.M.; Stern, C.D.; et al. The Oct4 Homologue PouV and Nanog Regulate Pluripotency in Chicken Embryonic Stem Cells. Development 2007, 134, 3549–3563. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, R.; Chen, C.; Waters, E.; West, F.D.; Kim, W.K. Isolation and Differentiation of Mesenchymal Stem Cells from Broiler Chicken Compact Bones. Front. Physiol. 2018, 9, 1892. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, J.J.; Ji, H.; Guan, W.; Zhao, Y. Isolation, Culture, and Characterization of Chicken Lung-Derived Mesenchymal Stem Cells. Can. J. Vet. Res. 2018, 82, 225–235. [Google Scholar]
- Zhou, Z.; Zhao, C.; Cai, B.; Ma, M.; Kong, S.; Zhang, J.; Zhang, X.; Nie, Q. Myogenic Differentiation Potential of Chicken Mesenchymal Stem Cells from Bone Marrow. Res. Sq. 2021, 1–21. [Google Scholar] [CrossRef]
- Pu, Y.; Veiga-Lopez, A. PPAR\gamma Agonist through the Terminal Differentiation Phase Is Essential for Adipogenic Differentiation of Fetal Ovine Preadipocytes. Cell. Mol. Biol. Lett. 2017, 22, 6. [Google Scholar] [CrossRef] [PubMed]
- Heidari, B.; Shirazi, A.; Akhondi, M.M.; Hassanpour, H.; Behzadi, B.; Naderi, M.M.; Sarvari, A.; Borjian, S. Comparison of Proliferative and Multilineage Differentiation Potential of Sheep Mesenchymal Stem Cells Derived from Bone Marrow, Liver, and Adipose Tissue. Avicenna J. Med. Biotechnol. 2013, 5, 104–117. [Google Scholar]
- Riera-Heredia, N.; Lutfi, E.; Gutiérrez, J.; Navarro, I.; Capilla, E. Fatty Acids from Fish or Vegetable Oils Promote the Adipogenic Fate of Mesenchymal Stem Cells Derived from Gilthead Sea Bream Bone Potentially through Different Pathways. PLoS ONE 2019, 14, e0215926. [Google Scholar] [CrossRef]
- Li, X.; Fu, X.; Yang, G.; Du, M. Enhancing Intramuscular Fat Development via Targeting Fibro-Adipogenic Progenitor Cells in Meat Animals. Animal 2020, 14, 312–321. [Google Scholar] [CrossRef]
- Guan, L.; Hu, X.; Liu, L.; Xing, Y.; Zhou, Z.; Liang, X.; Yang, Q.; Jin, S.; Bao, J.; Gao, H. Bta-MiR-23a Involves in Adipogenesis of Progenitor Cells Derived from Fetal Bovine Skeletal Muscle. Sci. Rep. 2017, 7, 43716. [Google Scholar] [CrossRef]
- Joe, A.W.B.; Yi, L.; Natarajan, A.; Le Grand, F.; So, L.; Wang, J.; Rudnicki, M.A.; Rossi, F.M.V. Muscle Injury Activates Resident Fibro/Adipogenic Progenitors That Facilitate Myogenesis. Nat. Cell Biol. 2010, 12, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Tichy, E.D.; Zhong, L.; Mohanty, S.; Wang, L.; Ai, E.; Yang, S.; Mourkioti, F.; Qin, L. Gli1 Defines a Subset of Fibro-adipogenic Progenitors That Promote Skeletal Muscle Regeneration with Less Fat Accumulation. J. Bone Miner. Res. 2021, 36, 1159–1173. [Google Scholar] [CrossRef] [PubMed]
- Dohmen, R.G.J.; Hubalek, S.; Melke, J.; Messmer, T.; Cantoni, F.; Mei, A.; Hueber, R.; Mitic, R.; Remmers, D.; Moutsatsou, P.; et al. Muscle-Derived Fibro-Adipogenic Progenitor Cells for Production of Cultured Bovine Adipose Tissue. Npj Sci. Food 2022, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Contreras, O.; Rossi, F.M.; Brandan, E. Adherent Muscle Connective Tissue Fibroblasts Are Phenotypically and Biochemically Equivalent to Stromal Fibro/Adipogenic Progenitors. Matrix Biol. Plus 2019, 2, 100006. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Song, T.; Hu, X.; Zhou, Y.; Wei, H.; Peng, J.; Jiang, S. Phenotypic and Functional Properties of Porcine Dedifferentiated Fat Cells during the Long-Term Culture In Vitro. BioMed Res. Int. 2015, 2015, 673651. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Duarte, M.S.; Du, M.; Paulino, P.V.R.; Jiang, Z.; Albrecht, E.; Fernyhough-Culver, M.; Zan, L.; Hausman, G.J.; Dodson, M.V. Bovine Mature Adipocytes Readily Return to a Proliferative State. Tissue Cell 2012, 44, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Oki, Y.; Hagiwara, R.; Matsumaru, T.; Kano, K. Effect of Volatile Fatty Acids on Adipocyte Differentiation in Bovine Dedifferentiated Fat (DFAT) Cells in Vitro. Genes Cells 2022, 27, 5–13. [Google Scholar] [CrossRef]
- Kumar, D.; Talluri, T.R.; Selokar, N.L.; Hyder, I.; Kues, W.A. Perspectives of Pluripotent Stem Cells in Livestock. World J. Stem Cells 2021, 13, 1–29. [Google Scholar] [CrossRef]
- Han, X.; Han, J.; Ding, F.; Cao, S.; Lim, S.S.; Dai, Y.; Zhang, R.; Zhang, Y.; Lim, B.; Li, N. Generation of Induced Pluripotent Stem Cells from Bovine Embryonic Fibroblast Cells. Cell Res. 2011, 21, 1509–1512. [Google Scholar] [CrossRef]
- Bai, C.; Li, X.; Gao, Y.; Yuan, Z.; Hu, P.; Wang, H.; Liu, C.; Guan, W.; Ma, Y. Melatonin Improves Reprogramming Efficiency and Proliferation of Bovine-Induced Pluripotent Stem Cells. J. Pineal Res. 2016, 61, 154–167. [Google Scholar] [CrossRef]
- Xiong, H.; Bai, C.; Wu, S.; Gao, Y.; Lu, T.; Hu, Q.; Guan, W.; Ma, Y. Biological Characterization of Mesenchymal Stem Cells from Bovine Umbilical Cord. Anim. Cells Syst. 2014, 18, 59–67. [Google Scholar] [CrossRef][Green Version]
- Bai, C.; Hou, L.; Ma, Y.; Chen, L.; Zhang, M.; Guan, W. Isolation and Characterization of Mesenchymal Stem Cells from Chicken Bone Marrow. Cell Tissue Bank. 2013, 14, 437–451. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.; Li, X.; Hou, L.; Zhang, M.; Guan, W.; Ma, Y. Biological Characterization of Chicken Mesenchymal Stem/Progenitor Cells from Umbilical Cord Wharton’s Jelly. Mol. Cell. Biochem. 2013, 376, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Huang, J.; Wang, X.; Ma, Y. Transcription Factors Regulate Adipocyte Differentiation in Beef Cattle. Anim. Genet. 2020, 51, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Pei, W.; Wang, K.; Zhang, S.; Chen, F.; Wu, Y.; Guan, W. In Vitro Culture and Biological Properties of Broiler Adipose-Derived Stem Cells. Exp. Ther. Med. 2018, 16, 2399–2407. [Google Scholar] [CrossRef] [PubMed]
- Blanton, J.; Grant, A.; McFarland, D.; Robinson, J.; Bidwell, C. Isolation of Two Populations of Myoblasts from Porcine Skeletal Muscle. Muscle Nerve 1999, 22, 43–50. [Google Scholar] [CrossRef]
- Li, B.-J.; Li, P.-H.; Huang, R.-H.; Sun, W.-X.; Wang, H.; Li, Q.-F.; Chen, J.; Wu, W.-J.; Liu, H.-L. Isolation, Culture and Identification of Porcine Skeletal Muscle Satellite Cells. Asian-Australas. J. Anim. Sci. 2015, 28, 1171–1177. [Google Scholar] [CrossRef]
- Miersch, C.; Stange, K.; Röntgen, M. Effects of Trypsinization and of a Combined Trypsin, Collagenase, and DNase Digestion on Liberation and in Vitro Function of Satellite Cells Isolated from Juvenile Porcine Muscles. In Vitro Cell. Dev. Biol. Anim. 2018, 54, 406–412. [Google Scholar] [CrossRef]
- Kubis; Haller; Wetzel; Gros Adult Fast Myosin Pattern and Ca2+-Induced Slow Myosin Pattern in Primary Skeletal Muscle Culture. Proc. Natl. Acad. Sci. USA 1997, 94, 4205–4210. [CrossRef]
- McFarland, D.C.; Gilkerson, K.K.; Pesall, J.E.; Ferrin, N.H.; Wellenreiter, R.H. In Vitro Characteristics of Myogenic Satellite Cells Derived from the Pectoralis Major and Biceps Femoris Muscles of the Chicken. Cytobios 1997, 91, 45–52. [Google Scholar]
- Doumit, M.E.; McFarland, D.C.; Minshall, R.D. Satellite Cells of Growing Turkeys: Influence of Donor Age and Sex on Proliferation and Differentiation in Vitro. Exp. Cell Res. 1990, 189, 81–86. [Google Scholar] [CrossRef]
- McFarland; Pesall; Gilkerson The Influence of Growth Factors on Turkey Embryonic Myoblasts and Satellite Cells in Vitro. Gen. Comp. Endocrinol. 1993, 89, 415–424. [CrossRef]
- Matschak, T.W.; Stickland, N.C. The Growth of Atlantic Salmon (Salmo Salar L.) Myosatellite Cells in Culture at Two Different Temperatures. Experientia 1995, 51, 260–266. [Google Scholar] [CrossRef]
- Mulvaney, D.R.; Cyrino, J.E. Establishment of Channel Catfish Satellite Cell Cultures. Basic Appl. Myol. BAM 1995, 5, 65–70. [Google Scholar]
- Millan-Cubillo, A.F.; Martin-Perez, M.; Ibarz, A.; Fernandez-Borras, J.; Gutiérrez, J.; Blasco, J. Proteomic Characterization of Primary Cultured Myocytes in a Fish Model at Different Myogenesis Stages. Sci. Rep. 2019, 9, 14126. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Du, M.; Jiang, Z.; Duarte, M.S.; Fernyhough-Culver, M.; Albrecht, E.; Will, K.; Zan, L.; Hausman, G.J.; Elabd, E.M.Y.; et al. Bovine Dedifferentiated Adipose Tissue (DFAT) Cells: DFAT Cell Isolation. Adipocyte 2013, 2, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Chung, K.Y.; Johnson, B.J.; Go, G.W.; Kim, K.H.; Choi, C.W.; Smith, S.B. Co-culture of bovine muscle satellite cells with preadipocytes increases PPARγ and C/EBPβ gene expression in differentiated myoblasts and increases GPR43 gene expression in adipocytes. J. Nutr. Biochem. 2013, 24, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.; Suzuki, T.; Kobayashi, K.; Nishimura, T. Adipocytes Suppress Differentiation of Muscle Cells in a Co-Culture System. Anim. Sci. J. 2019, 90, 423–434. [Google Scholar] [CrossRef]
- Chu, W.; Wei, W.; Yu, S.; Han, H.; Shi, X.; Sun, W.; Gao, Y.; Zhang, L.; Chen, J. C2C12 Myotubes Inhibit the Proliferation and Differentiation of 3T3-L1 Preadipocytes by Reducing the Expression of Glucocorticoid Receptor Gene. Biochem. Biophys. Res. Commun. 2016, 472, 68–74. [Google Scholar] [CrossRef]
- Xu, X.; Zhao, R.; Ma, W.; Zhao, Q.; Zhang, G. Comparison of Lipid Deposition of Intramuscular Preadipocytes in Tan Sheep Co-Cultured with Satellite Cells or Alone. J. Anim. Physiol. Anim. Nutr. 2021, in press. [CrossRef]
- Yan, X.; Zhu, M.-J.; Dodson, M.V.; Du, M. Developmental Programming of Fetal Skeletal Muscle and Adipose Tissue Development. J. Genom. 2013, 1, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Su, X.; Tian, Y.; Song, G.; Zan, L.; Wang, H. Effect of Actin Alpha Cardiac Muscle 1 on the Proliferation and Differentiation of Bovine Myoblasts and Preadipocytes. Animals 2021, 11, 3468. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Wang, Y.; Li, A.; Zan, L.; Wang, H. Neudesin Neurotrophic Factor Promotes Bovine Preadipocyte Differentiation and Inhibits Myoblast Myogenesis. Animals 2019, 9, 1109. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Su, X.; Wang, Y.; Cheng, G.; Zan, L.; Wang, H. Effect of Neudesin Neurotrophic Factor on Differentiation of Bovine Preadipocytes and Myoblasts in a Co-Culture System. Animals 2020, 11, 34. [Google Scholar] [CrossRef]
- Cui, H.X.; Guo, L.P.; Zhao, G.P.; Liu, R.R.; Li, Q.H.; Zheng, M.Q.; Wen, J. Method Using a Co-Culture System with High-Purity Intramuscular Preadipocytes and Satellite Cells from Chicken Pectoralis Major Muscle. Poult. Sci. 2018, 97, 3691–3697. [Google Scholar] [CrossRef] [PubMed]
- Shima, A.; Itou, A.; Takeuchi, S. Cell Fibers Promote Proliferation of Co-Cultured Cells on a Dish. Sci. Rep. 2020, 10, 288. [Google Scholar] [CrossRef] [PubMed]
- Jo, B.; Nie, M.; Shima, A.; Morimoto, Y.; Takeuchi, S. Micro Tissue Assembly for Co-Culturing 3D Skeletal Muscle and Adipose Tissues. In Proceedings of the 2020 IEEE 33rd International Conference on Micro Electro Mechanical Systems (MEMS), Vancouver, BC, Canada, 18–22 January 2020; pp. 459–460. [Google Scholar]
- Du, M.; Wang, B.; Fu, X.; Yang, Q.; Zhu, M.-J. Fetal Programming in Meat Production. Meat Sci. 2015, 109, 40–47. [Google Scholar] [CrossRef]
- Freiman, A.; Shandalov, Y.; Rosenfeld, D.; Shor, E.; Ben-David, D.; Meretzki, S.; Levenberg, S.; Egozi, D. Engineering Vascularized Flaps Using Adipose-Derived Microvascular Endothelial Cells and Mesenchymal Stem Cells. J. Tissue Eng. Regen. Med. 2018, 12, e130–e141. [Google Scholar] [CrossRef]
- Ma, Y.N.; Wang, B.; Wang, Z.X.; Gomez, N.A.; Zhu, M.J.; Du, M. Three-Dimensional Spheroid Culture of Adipose Stromal Vascular Cells for Studying Adipogenesis in Beef Cattle. Animal 2018, 12, 2123–2129. [Google Scholar] [CrossRef]
- Kayabolen, A.; Keskin, D.; Aykan, A.; Karslıoglu, Y.; Zor, F.; Tezcaner, A. Native Extracellular Matrix/Fibroin Hydrogels for Adipose Tissue Engineering with Enhanced Vascularization. Biomed. Mater. 2017, 12, 035007. [Google Scholar] [CrossRef]
- Kang, D.-H.; Louis, F.; Liu, H.; Shimoda, H.; Nishiyama, Y.; Nozawa, H.; Kakitani, M.; Takagi, D.; Kasa, D.; Nagamori, E.; et al. Engineered Whole Cut Meat-like Tissue by the Assembly of Cell Fibers Using Tendon-Gel Integrated Bioprinting. Nat. Commun. 2021, 12, 5059. [Google Scholar] [CrossRef] [PubMed]
- Ben-Arye, T.; Shandalov, Y.; Ben-Shaul, S.; Landau, S.; Zagury, Y.; Ianovici, I.; Lavon, N.; Levenberg, S. Textured Soy Protein Scaffolds Enable the Generation of Three-Dimensional Bovine Skeletal Muscle Tissue for Cell-Based Meat. Nat. Food 2020, 1, 210–220. [Google Scholar] [CrossRef]
- Ivanovic, Z. Hypoxia or in Situ Normoxia: The Stem Cell Paradigm. J. Cell. Physiol. 2009, 219, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Urbani, L.; Piccoli, M.; Franzin, C.; Pozzobon, M.; De Coppi, P. Hypoxia Increases Mouse Satellite Cell Clone Proliferation Maintaining Both in Vitro and in Vivo Heterogeneity and Myogenic Potential. PLoS ONE 2012, 7, e49860. [Google Scholar] [CrossRef]
- Elashry, M.I.; Kinde, M.; Klymiuk, M.C.; Eldaey, A.; Wenisch, S.; Arnhold, S. The Effect of Hypoxia on Myogenic Differentiation and Multipotency of the Skeletal Muscle-Derived Stem Cells in Mice. Stem Cell Res. Ther. 2022, 13, 56. [Google Scholar] [CrossRef]
- Redshaw, Z.; Loughna, P.T. Oxygen Concentration Modulates the Differentiation of Muscle Stem Cells toward Myogenic and Adipogenic Fates. Differ. Res. Biol. Divers. 2012, 84, 193–202. [Google Scholar] [CrossRef]
- Nilsson, G.E.; Ostlund-Nilsson, S. Hypoxia in Paradise: Widespread Hypoxia Tolerance in Coral Reef Fishes. Proc. Biol. Sci. R. Soc. 2004, 271 (Suppl. 3), S30–S33. [Google Scholar] [CrossRef]
- Fish, K.D.; Rubio, N.R.; Stout, A.J.; Yuen, J.S.K.; Kaplan, D.L. Prospects and Challenges for Cell-Cultured Fat as a Novel Food Ingredient. Trends Food Sci. Technol. 2020, 98, 53–67. [Google Scholar] [CrossRef]
- Soice, E.; Johnston, J. Immortalizing Cells for Human Consumption. Int. J. Mol. Sci. 2021, 22, 11660. [Google Scholar] [CrossRef]
- Cell Line Repositories | Solution | GFI—The Good Food Institute. Available online: https://gfi.org/solutions/establishment-of-cell-line-repositories-and-standardized-isolation-protocols/ (accessed on 24 March 2022).
- Species-Specific Genomic Studies Enabling Assay Development for Regulatory Standards and Cell Line Optimization. The Good Food Institute. Available online: https://gfi.org/solutions/species-specific-genomic-studies-enabling-assay-development-for-regulatory-standards-and-cell-line-optimization/ (accessed on 24 March 2022).
- Melzener, L.; Verzijden, K.E.; Buijs, A.J.; Post, M.J.; Flack, J.E. Cultured Beef: From Small Biopsy to Substantial Quantity. J. Sci. Food Agric. 2021, 101, 7–14. [Google Scholar] [CrossRef]
- Compassion in World Farming (CIWF). RE: Advance Notice of Proposed Rulemaking, Labeling of Meat or Poultry Products Comprised of or Containing Cultured Animal Cells; Posted 3 September 2021; Docket No. FSIS-2020-0036; CIWF: New York, NY, USA, 2021; Available online: https://www.ciwf.com/media/7448126/ciwf-comments-on-docket-fsis-2020-0036-final.pdf (accessed on 30 March 2022).
- Choi, K.-H.; Yoon, J.W.; Kim, M.; Lee, H.J.; Jeong, J.; Ryu, M.; Jo, C.; Lee, C.-K. Muscle Stem Cell Isolation and in Vitro Culture for Meat Production: A Methodological Review. Compr. Rev. Food Sci. Food Saf. 2021, 20, 429–457. [Google Scholar] [CrossRef] [PubMed]
- C2C12|ATCC. Available online: https://www.atcc.org/products/crl-1772 (accessed on 28 March 2022).
- L6|ATCC. Available online: https://www.atcc.org/products/crl-1458 (accessed on 28 March 2022).
- Albini, S.; Coutinho, P.; Malecova, B.; Giordani, L.; Savchenko, A.; Forcales, S.V.; Puri, P.L. Epigenetic Reprogramming of Human Embryonic Stem Cells into Skeletal Muscle Cells and Generation of Contractile Myospheres. Cell Rep. 2013, 3, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Specht, E.A.; Welch, D.R.; Rees Clayton, E.M.; Lagally, C.D. Opportunities for Applying Biomedical Production and Manufacturing Methods to the Development of the Clean Meat Industry. Biochem. Eng. J. 2018, 132, 161–168. [Google Scholar] [CrossRef]
- Genovese, N.; Nicole, D.; Schulze, E. Methods for Extending the Replicative Capacity of Somatic Cells during an Ex Vivo Cultivation Process; United States Department of Commerce/US Patent and Trademark Office: Alexandria, VA, USA, 2017; pp. 22313–22450.
- Stadler, G.; Chen, J.C.; Wagner, K.; Robin, J.D.; Shay, J.W.; Emerson, C.P., Jr.; Wright, W.E. Establishment of Clonal Myogenic Cell Lines from Severely Affected Dystrophic Muscles-CDK4 Maintains the Myogenic Population. Skelet. Muscle 2011, 1, 12. [Google Scholar] [CrossRef] [PubMed]
- Genovese, N.J.; Roberts, R.M.; Telugu, B.P.V.L. Method for Scalable Skeletal Muscle Lineage Specification and Cultivation; United States Department of Commerce/US Patent and Trademark Office: Alexandria, VA, USA, 2014; pp. 22313–22450.
- Alt-Seafood: Cell-Based Seafood in the European Union. Available online: http://collinealfieri.com/alternative-seafood-cell-based-seafood-in-the-european-union-bluu-biosciences-sebastian-rakers.html (accessed on 25 March 2022).
- Liu, J.; Hocquette, É.; Ellies-Oury, M.-P.; Chriki, S.; Hocquette, J.-F. Chinese Consumers’ Attitudes and Potential Acceptance toward Artificial Meat. Foods 2021, 10, 353. [Google Scholar] [CrossRef]
- Kerafast Blog. Good Food Institute Partnership Links Researchers; Kerafast Blog; Kerafast Inc.: Boston, MA, USA, 2021; p. 02210. [Google Scholar]
- Buonocore, F.; Libertini, A.; Prugnoli, D.; Mazzini, M.; Scapigliati, G. Production and Characterization of a Continuous Embryonic Cell Line from Sea Bass (Dicentrarchus Labrax L.). Mar. Biotechnol. 2006, 8, 80–85. [Google Scholar] [CrossRef]
- Furuichi, Y.; Manabe, Y.; Takagi, M.; Aoki, M.; Fujii, N.L. Evidence for Acute Contraction-Induced Myokine Secretion by C2C12 Myotubes. PLoS ONE 2018, 13, e0206146. [Google Scholar] [CrossRef]
- Cooper, R.N.; Tajbakhsh, S.; Mouly, V.; Cossu, G.; Buckingham, M.; Butler-Browne, G.S. In Vivo Satellite Cell Activation via Myf5 and MyoD in Regenerating Mouse Skeletal Muscle. J. Cell Sci. 1999, 112 Pt 17, 2895–2901. [Google Scholar] [CrossRef]
- Dey, B.K.; Gagan, J.; Dutta, A. MiR-206 and -486 Induce Myoblast Differentiation by Downregulating Pax7. Mol. Cell. Biol. 2011, 31, 203–214. [Google Scholar] [CrossRef]
- Messmer, T.; Klevernic, I.; Furquim, C.; Ovchinnikova, E.; Dogan, A.; Cruz, H.; Post, M.J.; Flack, J.E. A Serum-Free Media Formulation for Cultured Meat Production Supports Bovine Satellite Cell Differentiation in the Absence of Serum Starvation. Nat. Food 2022, 3, 74–85. [Google Scholar] [CrossRef]
- Langelaan, M.L.; Boonen, K.J.; Rosaria-Chak, K.Y.; van der Schaft, D.W.; Post, M.J.; Baaijens, F.P. Advanced Maturation by Electrical Stimulation: Differences in Response between C2C12 and Primary Muscle Progenitor Cells. J. Tissue Eng. Regen. Med. 2011, 5, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Collinsworth, A.M.; Zhang, S.; Kraus, W.E.; Truskey, G.A. Apparent Elastic Modulus and Hysteresis of Skeletal Muscle Cells throughout Differentiation. Am. J. Physiol. Cell Physiol. 2002, 283, C1219–C1227. [Google Scholar] [CrossRef] [PubMed]
- Zidarič, T.; Milojević, M.; Vajda, J.; Vihar, B.; Maver, U. Cultured Meat: Meat Industry Hand in Hand with Biomedical Production Methods. Food Eng. Rev. 2020, 12, 498–519. [Google Scholar] [CrossRef]
- David, V.; Martin, A.; Lafage-Proust, M.-H.; Malaval, L.; Peyroche, S.; Jones, D.B.; Vico, L.; Guignandon, A. Mechanical Loading Down-Regulates Peroxisome Proliferator-Activated Receptor γ in Bone Marrow Stromal Cells and Favors Osteoblastogenesis at the Expense of Adipogenesis. Endocrinology 2007, 148, 2553–2562. [Google Scholar] [CrossRef]
- Shoham, N.; Mor-Yossef Moldovan, L.; Benayahu, D.; Gefen, A. Multiscale Modeling of Tissue-Engineered Fat: Is There a Deformation-Driven Positive Feedback Loop in Adipogenesis? Tissue Eng. Part A 2015, 21, 1354–1363. [Google Scholar] [CrossRef]
- Li, R.; Liang, L.; Dou, Y.; Huang, Z.; Mo, H.; Wang, Y.; Yu, B. Mechanical Stretch Inhibits Mesenchymal Stem Cell Adipogenic Differentiation through TGFβ1/Smad2 Signaling. J. Biomech. 2015, 48, 3656–3662. [Google Scholar] [CrossRef]
- Tanabe, Y.; Koga, M.; Saito, M.; Matsunaga, Y.; Nakayama, K. Inhibition of Adipocyte Differentiation by Mechanical Stretching through ERK-Mediated Downregulation of PPARγ2. J. Cell Sci. 2004, 117, 3605–3614. [Google Scholar] [CrossRef]
- Sen, B.; Xie, Z.; Case, N.; Ma, M.; Rubin, C.; Rubin, J. Mechanical Strain Inhibits Adipogenesis in Mesenchymal Stem Cells by Stimulating a Durable β-Catenin Signal. Endocrinology 2008, 149, 6065–6075. [Google Scholar] [CrossRef]
- Shoham, N.; Gottlieb, R.; Sharabani-Yosef, O.; Zaretsky, U.; Benayahu, D.; Gefen, A. Static Mechanical Stretching Accelerates Lipid Production in 3T3-L1 Adipocytes by Activating the MEK Signaling Pathway. Am. J. Physiol.-Cell Physiol. 2012, 302, C429–C441. [Google Scholar] [CrossRef]
- O’Donnell, B.T.; Al-Ghadban, S.; Ives, C.J.; L’Ecuyer, M.P.; Monjure, T.A.; Romero-Lopez, M.; Li, Z.; Goodman, S.B.; Lin, H.; Tuan, R.S. Adipose Tissue-Derived Stem Cells Retain Their Adipocyte Differentiation Potential in Three-Dimensional Hydrogels and Bioreactors. Biomolecules 2020, 10, 1070. [Google Scholar] [CrossRef]
- Liu, L.; Liu, X.; Liu, M.; Jihu, Y.; Xie, D.; Yan, H. Mechanical Signals Induces Reprogramming of Mature Adipocytes through the YAP/TAZ-Binding Motif. Exp. Cell Res. 2022, 415, 113109. [Google Scholar] [CrossRef] [PubMed]
- Totaro, A.; Zhuang, Q.; Panciera, T.; Battilana, G.; Azzolin, L.; Brumana, G.; Gandin, A.; Brusatin, G.; Cordenonsi, M.; Piccolo, S. Cell Phenotypic Plasticity Requires Autophagic Flux Driven by YAP/TAZ Mechanotransduction. Proc. Natl. Acad. Sci. USA 2019, 116, 17848–17857. [Google Scholar] [CrossRef] [PubMed]
- Paredes, J.; Cortizo-Lacalle, D.; Imaz, A.M.; Aldazabal, J.; Vila, M. Application of Texture Analysis Methods for the Characterization of Cultured Meat. Sci. Rep. 2022, 12, 3898. [Google Scholar] [CrossRef] [PubMed]
- Lei, Q.; Li, M.; Du, G.; Zhou, J.; Guan, X. An Effective Cytokine Combination for Ex Vivo Expansion of Porcine Muscle Stem Cells. Food Biosci. 2022, 46, 101571. [Google Scholar] [CrossRef]
- Steyn, P.J.; Dzobo, K.; Smith, R.I.; Myburgh, K.H. Interleukin-6 Induces Myogenic Differentiation via JAK2-STAT3 Signaling in Mouse C2C12 Myoblast Cell Line and Primary Human Myoblasts. Int. J. Mol. Sci. 2019, 20, 5273. [Google Scholar] [CrossRef]
- Quinn, L.S.; Haugk, K.L.; Damon, S.E. Interleukin-15 Stimulates C2 Skeletal Myoblast Differentiation. Biochem. Biophys. Res. Commun. 1997, 239, 6–10. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, Y.; Li, W.; Wang, G.; Song, Y.; Yang, G.; Han, X.; Du, Z.; Sun, L.; Ma, K. STAT3 Induces Muscle Stem Cell Differentiation by Interaction with MyoD. Cytokine 2009, 46, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Horsley, V.; Jansen, K.M.; Mills, S.T.; Pavlath, G.K. IL-4 Acts as a Myoblast Recruitment Factor during Mammalian Muscle Growth. Cell 2003, 113, 483–494. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Tsai, J.-N.; Chen, T.-L.; Ho, K.-T.; Cheng, H.-Y.; Hsiao, C.-W.; Shiau, M.-Y. Interleukin-4 Promotes Myogenesis and Boosts Myocyte Insulin Efficacy. Mediat. Inflamm. 2019, 2019, 4182015. [Google Scholar] [CrossRef] [PubMed]
- Otis, J.S.; Niccoli, S.; Hawdon, N.; Sarvas, J.L.; Frye, M.A.; Chicco, A.J.; Lees, S.J. Pro-Inflammatory Mediation of Myoblast Proliferation. PLoS ONE 2014, 9, e92363. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Xiao, J.; Wei, Y.; Li, S.; Liu, Y.; Yin, J.; Sun, K.; Sun, H.; Wang, H.; Zhang, Z.; et al. Combination of Inflammation-Related Cytokines Promotes Long-Term Muscle Stem Cell Expansion. Cell Res. 2015, 25, 655–673. [Google Scholar] [CrossRef] [PubMed]
- Krieger, J.; Park, B.-W.; Lambert, C.R.; Malcuit, C. 3D Skeletal Muscle Fascicle Engineering Is Improved with TGF-\beta1 Treatment of Myogenic Cells and Their Co-Culture with Myofibroblasts. PeerJ 2018, 6, e4939. [Google Scholar] [CrossRef] [PubMed]
- Stout, A.J.; Mirliani, A.B.; White, E.C.; Yuen, J.S.K.; Kaplan, D.L. Simple and Effective Serum-Free Medium for Sustained Expansion of Bovine Satellite Cells for Cell Cultured Meat. BioRxiv 2021. [CrossRef]
- Kolkmann, A.M.; Post, M.J.; Rutjens, M.A.M.; van Essen, A.L.M.; Moutsatsou, P. Serum-Free Media for the Growth of Primary Bovine Myoblasts. Cytotechnology 2020, 72, 111–120. [Google Scholar] [CrossRef]
- Rubert Pérez, C.M.; Álvarez, Z.; Chen, F.; Aytun, T.; Stupp, S.I. Mimicking the Bioactivity of Fibroblast Growth Factor-2 Using Supramolecular Nanoribbons. ACS Biomater. Sci. Eng. 2017, 3, 2166–2175. [Google Scholar] [CrossRef]
- Venkatesan, M.; Semper, C.; DiLeo, R.; Mesa, N.; Stogios, P.J.; Savchenko, A. Recombinant Production of Growth Factors for Application in Cell Culture. BioRxiv 2022. [CrossRef]
- Venter, C.; Niesler, C. A Triple Co-Culture Method to Investigate the Effect of Macrophages and Fibroblasts on Myoblast Proliferation and Migration. Biotechniques 2018, 64, 52–58. [Google Scholar] [CrossRef]
- Fei, W.; Pang, E.; Hou, L.; Dai, J.; Liu, M.; Wang, X.; Xie, B.; Wang, J. Hydrogen Enhanced the Myogenic Differentiation of Adipose Mesenchymal Stem Cells through P38 MAPK Signaling Pathway. Res. Sq. 2021, 1–23. [Google Scholar] [CrossRef]
- Du, Y.; Wang, Y.; Xu, Q.; Zhu, J.; Lin, Y. TMT-Based Quantitative Proteomics Analysis Reveals the Key Proteins Related with the Differentiation Process of Goat Intramuscular Adipocytes. BMC Genom. 2021, 22, 417. [Google Scholar] [CrossRef]
- Khan, R.; Raza, S.H.A.; Junjvlieke, Z.; Xiaoyu, W.; Garcia, M.; Elnour, I.E.; Hongbao, W.; Linsen, Z. Function and Transcriptional Regulation of Bovine TORC2 Gene in Adipocytes: Roles of C/EBPγ, XBP1, INSM1 and ZNF263. Int. J. Mol. Sci. 2019, 20, 4338. [Google Scholar] [CrossRef]
- Yue, B.; Yang, H.; Wu, J.; Wang, J.; Ru, W.; Cheng, J.; Huang, Y.; Lei, C.; Lan, X.; Chen, H. Characterization and Transcriptome Analysis of Exosomal and Nonexosomal RNAs in Bovine Adipocytes. Int. J. Mol. Sci. 2020, 21, 9313. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Bai, Y.; Tian, H.; Shi, B.; Li, X.; Luo, Y.; Wang, J.; Hu, J.; Abbas Raza, S.H. Interference with ACSL1 Gene in Bovine Adipocytes: Transcriptome Profiling of CircRNA Related to Unsaturated Fatty Acid Production. Genomics 2021, 113, 3967–3977. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Raza, S.H.A.; Tian, H.; Shi, B.; Luo, Y.; Wang, J.; Liu, X.; Li, S.; Bai, Y.; Hu, J. Effects of Overexpression of ACSL1 Gene on the Synthesis of Unsaturated Fatty Acids in Adipocytes of Bovine. Arch. Biochem. Biophys. 2020, 695, 108648. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, L.; Chen, W.; Li, J.; Shan, T. CRTC3 Regulates the Lipid Metabolism and Adipogenic Differentiation of Porcine Intramuscular and Subcutaneous Adipocytes by Activating the Calcium Pathway. J. Agric. Food Chem. 2021, 69, 7243–7255. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Zhao, Y.; Yu, W.; Melak, S.; Niu, Y.; Wei, W.; Zhang, L.; Chen, J. ACAT2 Is a Novel Negative Regulator of Pig Intramuscular Preadipocytes Differentiation. Biomolecules 2022, 12, 237. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Wang, Y.; Zhu, J.; Xiong, Y.; Lin, Y. Regulation of Fibroblast Growth Factor 9 on the Differentiation of Goat Intramuscular Adipocytes. Anim. Sci. J. 2021, 92, e13627. [Google Scholar] [CrossRef]
- Singh, G.B.; Cowan, D.B.; Wang, D.-Z. Tiny Regulators of Massive Tissue: Micrornas in Skeletal Muscle Development, Myopathies, and Cancer Cachexia. Front. Oncol. 2020, 10, 598964. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, S.W.; Han, J.S.; Shin, S.P.; Lee, S.I.; Park, T.S. Functional Analyses of MiRNA-146b-5p during Myogenic Proliferation and Differentiation in Chicken Myoblasts. BMC Mol. Cell Biol. 2020, 21, 40. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, N. Regulatory Role of MicroRNAs in Muscle Atrophy during Exercise Intervention. Int. J. Mol. Sci. 2018, 19, 405. [Google Scholar] [CrossRef]
- Sweetman, D.; Goljanek, K.; Rathjen, T.; Oustanina, S.; Braun, T.; Dalmay, T.; Münsterberg, A. Specific Requirements of MRFs for the Expression of Muscle Specific MicroRNAs, MiR-1, MiR-206 and MiR-133. Dev. Biol. 2008, 321, 491–499. [Google Scholar] [CrossRef]
- Jiang, A.; Dong, C.; Li, B.; Zhang, Z.; Chen, Y.; Ning, C.; Wu, W.; Liu, H. MicroRNA-206 Regulates Cell Proliferation by Targeting G6PD in Skeletal Muscle. FASEB J. 2019, 33, 14083–14094. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-F.; Tao, Y.; Li, J.; Deng, Z.; Yan, Z.; Xiao, X.; Wang, D.-Z. MicroRNA-1 and MicroRNA-206 Regulate Skeletal Muscle Satellite Cell Proliferation and Differentiation by Repressing Pax7. J. Cell Biol. 2010, 190, 867–879. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, J.; Liu, W.; Wang, H.; Zhao, L.; Liu, S.; Li, P.; Zhang, S.; Sun, C.; Wu, Y.; et al. MicroRNA-378 Promotes Autophagy and Inhibits Apoptosis in Skeletal Muscle. Proc. Natl. Acad. Sci. USA 2018, 115, E10849–E10858. [Google Scholar] [CrossRef]
- Bem, J.; Grabowska, I.; Daniszewski, M.; Zawada, D.; Czerwinska, A.M.; Bugajski, L.; Piwocka, K.; Fogtman, A.; Ciemerych, M.A. Transient MicroRNA Expression Enhances Myogenic Potential of Mouse Embryonic Stem Cells. Stem Cells 2018, 36, 655–670. [Google Scholar] [CrossRef] [PubMed]
- MicroRNA, MiR-374b, Directly Targets Myf6 and Negatively Regulates C2C12 Myoblasts Differentiation. Biochem. Biophys. Res. Commun. 2015, 467, 670–675. [CrossRef] [PubMed]
- Contreras, O.; Córdova-Casanova, A.; Brandan, E. PDGF-PDGFR Network Differentially Regulates the Fate, Migration, Proliferation, and Cell Cycle Progression of Myogenic Cells. Cell. Signal. 2021, 84, 110036. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, G.; Zhao, X.; Zhou, J.; Du, G.; Chen, J. A Conceptual Air-Lift Reactor Design for Large Scale Animal Cell Cultivation in the Context of in Vitro Meat Production. Chem. Eng. Sci. 2020, 211, 115269. [Google Scholar] [CrossRef]
- Chen, L.; Guttieres, D.; Koenigsberg, A.; Barone, P.W.; Sinskey, A.J.; Springs, S.L. Large-Scale Cultured Meat Production: Trends, Challenges and Promising Biomanufacturing Technologies. Biomaterials 2022, 280, 121274. [Google Scholar] [CrossRef]
- Bellani, C.F.; Ajeian, J.; Duffy, L.; Miotto, M.; Groenewegen, L.; Connon, C.J. Scale-Up Technologies for the Manufacture of Adherent Cells. Front. Nutr. 2020, 7, 575146. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Li, Y.; Chen, C.; Stoelzel, K.; Kaufmann, A.M.; Albers, A.E. Human Skeletal Muscle-Derived Stem Cells Retain Stem Cell Properties after Expansion in Myosphere Culture. Exp. Cell Res. 2011, 317, 1016–1027. [Google Scholar] [CrossRef]
- Muller, S.; Ader, I.; Creff, J.; Leménager, H.; Achard, P.; Casteilla, L.; Sensebé, L.; Carrière, A.; Deschaseaux, F. Human Adipose Stromal-Vascular Fraction Self-Organizes to Form Vascularized Adipose Tissue in 3D Cultures. Sci. Rep. 2019, 9, 7250. [Google Scholar] [CrossRef] [PubMed]
- Hanga, M.P.; de la Raga, F.A.; Moutsatsou, P.; Hewitt, C.J.; Nienow, A.W.; Wall, I. Scale-up of an Intensified Bioprocess for the Expansion of Bovine Adipose-Derived Stem Cells (BASCs) in Stirred Tank Bioreactors. Biotechnol. Bioeng. 2021, 118, 3175–3186. [Google Scholar] [CrossRef] [PubMed]
- Hanga, M.P.; Ali, J.; Moutsatsou, P.; de la Raga, F.A.; Hewitt, C.J.; Nienow, A.; Wall, I. Bioprocess Development for Scalable Production of Cultivated Meat. Biotechnol. Bioeng. 2020, 117, 3029–3039. [Google Scholar] [CrossRef] [PubMed]
- Specht, L. An Analysis of Culture Medium Costs and Production Volumes for Cultivated Meat; The Good Food Institute: Washington, DC, USA, 2020. [Google Scholar]
- Chriki, S.; Hocquette, J.-F. The Myth of Cultured Meat: A Review. Front. Nutr. 2020, 7, 7. [Google Scholar] [CrossRef]
| Cell Type | CM/CF/CS Relevant Cells | Isolated from | |
|---|---|---|---|
| Pluripotent stem cells | Embryonic stem cells (ESCs) | Mammalian ESCs | Cow [60,65]; Sheep [66] |
| Avian ESCs | Chicken eggs [68] | ||
| Fish ESCs | Medaka fish [70] | ||
| Induced pluripotent stem cells (iPSCs) | Mammalian iPSCs | Horse [80,81]; Pig [82,118]; Cow [60,75,119,120]; Sheep [79]; Goat [77] | |
| Avian iPSCs | Chicken [85,86] | ||
| Fish iPSCs | Koi fish [88] | ||
| Adult stem cells (ASCs) | Mesenchymal stem cells (MSCs) | Mammalian MSCs for myogenic differentiation | Cow [100,121] Horse [98] |
| Avian MSCs for myogenic differentiation | Chicken [103,104,122,123] | ||
| Mammalian MSCs for adipogenic differentiation | Sheep [107]; Cow [124] | ||
| Fish MSCs for adipogenic differentiation | Gilt-head sea bream [108] | ||
| Adipose tissue-derived stem cells (ADSCs) | Avian ADSCs | Chicken [125] | |
| Fibro-adipogenic progenitors (FAP) | Mammalian FAP | Cow [93] | |
| Resident muscle stem cells/muscle satellite cells (SCs) | Mammalian myogenic cells | Cow [34,35,36,37,38] Pig [40,126,127,128] Horse [40,41] Rabbit [129] | |
| Avian myogenic cells | Chicken [40,46,130]; Duck [40]; Turkey [131,132] | ||
| Fish myogenic cells | Rainbow trout [50] Common carp [51] Atlantic salmon [133] Channel catfish [134] Gilthead sea bream [52,135] Danioninae [52] Goldfish [53] | ||
| Dedifferentiated fat (DFAT) cells | Mammalian DFAT | Cow [117,136]; Pig [115] |
| microRNA | Target Gene(s) | Function |
|---|---|---|
| miR-1a | HDAC4, Cx43, Pax7, c-Met, G6PD | Increased expression upon myoblast differentiation |
| miR-16-5p | SESN1 | Represses myoblast differentiation |
| miR-22 | TGF-bR1 | Promotes myocyte differentiation |
| miR-23a | Myh 1, 2 and 4 | Inhibits myoblast differentiation |
| miR-24 | SMAD2 | Regulates myogenic differentiation |
| miR-26 | SMAD1, SMAD4, and Ezh2 | Promotes differentiation of myoblasts |
| miR-26a | Ezh2 | Increased expression upon myoblast differentiation |
| miR-27b | Pax3 | Increased expression upon myoblast differentiation |
| miR-29 | YY1, Rybp | Promotes myoblast differentiation |
| miR-29b/c | YY1, COL1A1, ELN, | Increased expression upon myoblast differentiation |
| miR-98 | E2F5 | Represses myoblast differentiation |
| miR-125b | IGF-II | Decreased expression upon myoblast differentiation |
| miR-133 | SRF, nPTB, UCP2 | Increased expression upon myoblast differentiation |
| miR-139 | Wnt1 | Represses differentiation |
| miR-148a | ROCK1 | Promotes myoblast differentiation |
| miR-155 | Mef2a | Inhibits myoblast differentiation |
| miR-181 | Hox-A11 | Enhances muscle differentiation |
| miR-186 | Myog 4 | Inhibits myoblast differentiation |
| miR-199-3p | IGF-1, mTOR, RPS6KA6 | Represses myoblast differentiation |
| miR-206a | DNApola, Fstl1, Utrn, Cx43, TIMP3, Pax7, c-Met, HDAC4 | Increased expression upon myoblast differentiation |
| miR-208b/499 | Sox6, Purβ, Sp3, HP-1β | Increased expression upon myoblast differentiation |
| miR-214 | Ezh2, N-Ras | Increased expression upon myoblast differentiation |
| miR-221/222 | p27 | Modulate differentiation and maturation of MSC |
| miR-322/424 | Cdc25A | Promotes cell cycle quiescence and differentiation |
| miR-374 | Myf6 | Represses myoblast differentiation |
| miR-378a-3p | HDAC4 | Promotes myoblasts differentiation |
| miR-431 | SMAD4 | Promotes myoblasts differentiation |
| miR-486 | FoxO1, PTEN, Pax7 | Increased expression upon myoblast differentiation |
| miR-503 | Cdc25A | Increased expression upon myoblast differentiation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knežić, T.; Janjušević, L.; Djisalov, M.; Yodmuang, S.; Gadjanski, I. Using Vertebrate Stem and Progenitor Cells for Cellular Agriculture, State-of-the-Art, Challenges, and Future Perspectives. Biomolecules 2022, 12, 699. https://doi.org/10.3390/biom12050699
Knežić T, Janjušević L, Djisalov M, Yodmuang S, Gadjanski I. Using Vertebrate Stem and Progenitor Cells for Cellular Agriculture, State-of-the-Art, Challenges, and Future Perspectives. Biomolecules. 2022; 12(5):699. https://doi.org/10.3390/biom12050699
Chicago/Turabian StyleKnežić, Teodora, Ljiljana Janjušević, Mila Djisalov, Supansa Yodmuang, and Ivana Gadjanski. 2022. "Using Vertebrate Stem and Progenitor Cells for Cellular Agriculture, State-of-the-Art, Challenges, and Future Perspectives" Biomolecules 12, no. 5: 699. https://doi.org/10.3390/biom12050699
APA StyleKnežić, T., Janjušević, L., Djisalov, M., Yodmuang, S., & Gadjanski, I. (2022). Using Vertebrate Stem and Progenitor Cells for Cellular Agriculture, State-of-the-Art, Challenges, and Future Perspectives. Biomolecules, 12(5), 699. https://doi.org/10.3390/biom12050699







