A Series of PSMA-Targeted Near-Infrared Fluorescent Imaging Agents
Abstract
1. Introduction
2. Materials and Methods
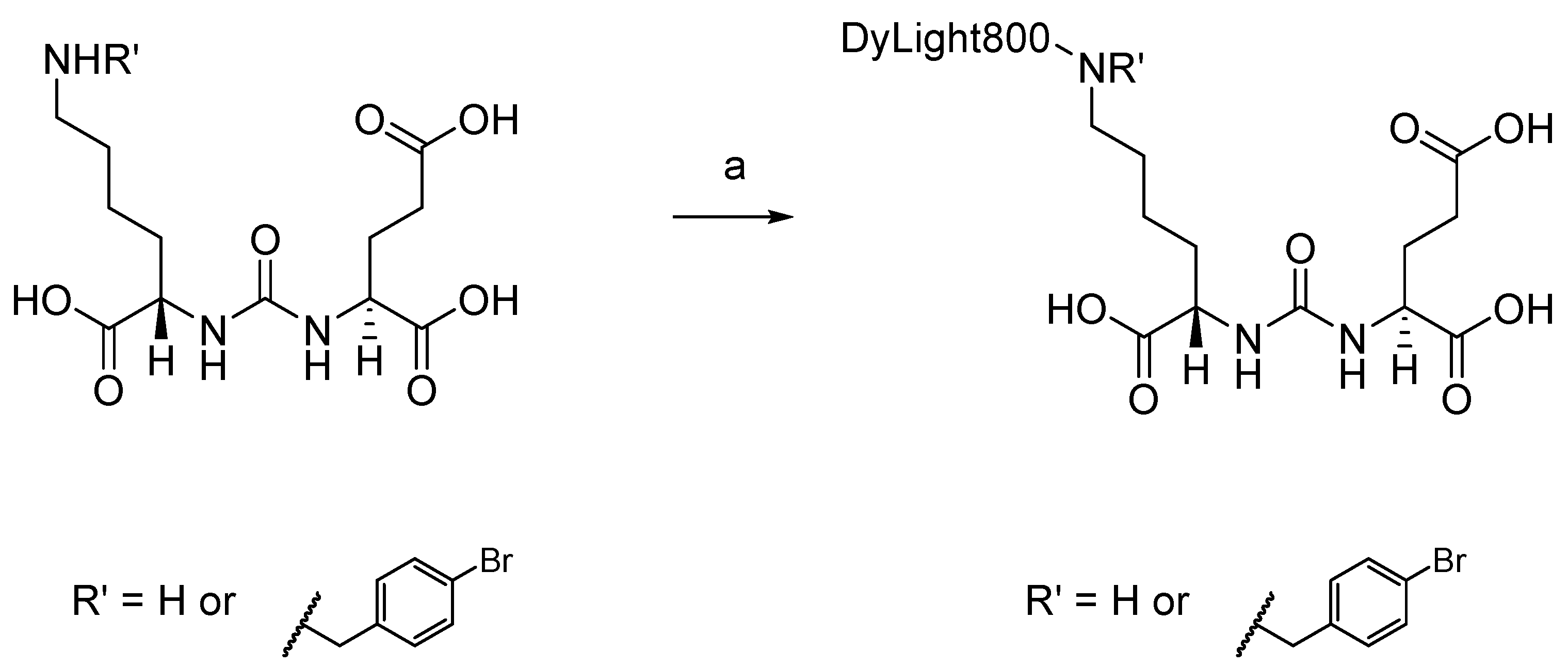
2.1. Chemistry
2.2. NAALADase Assay
2.3. Cell Lines and Tumor Models
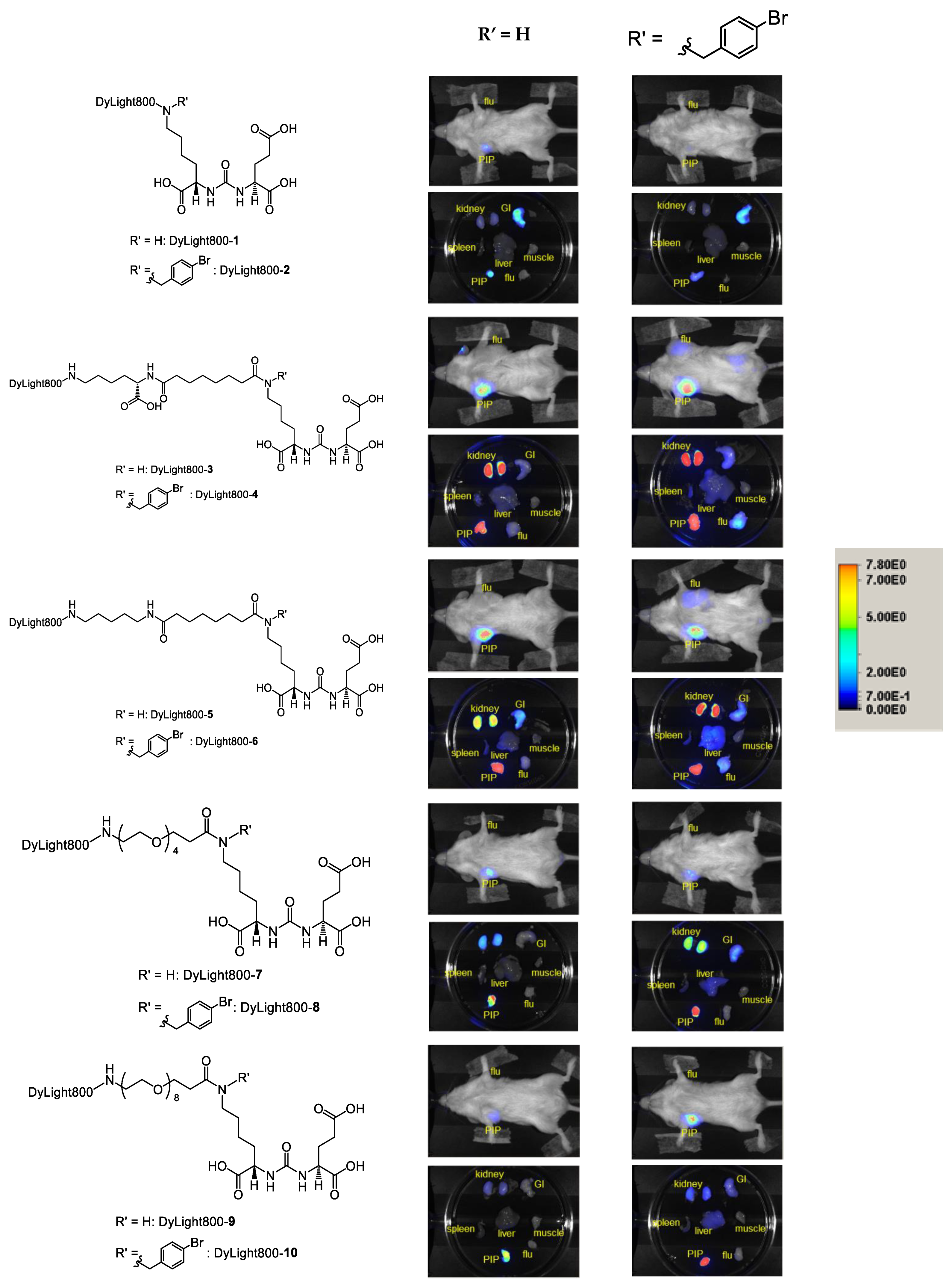
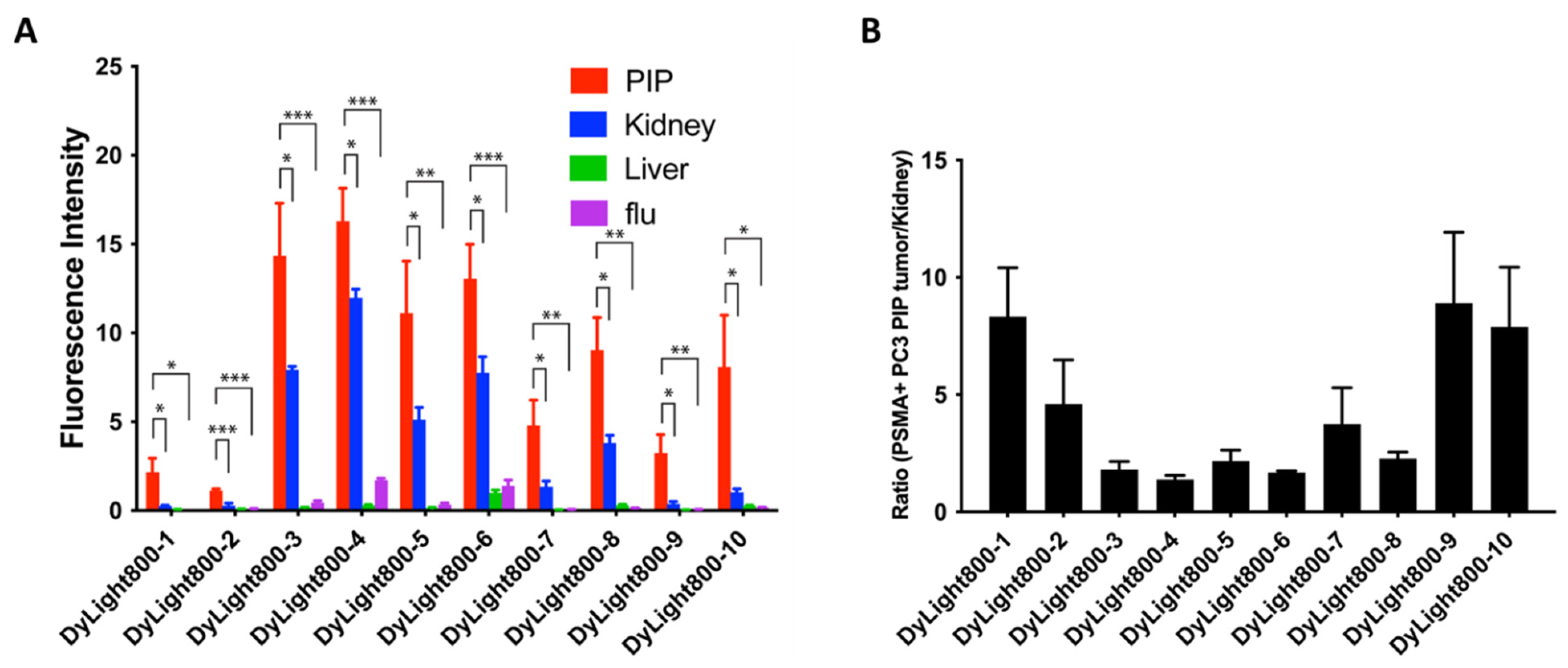
2.4. In Vivo Imaging and Ex Vivo Biodistribution
2.5. Immunohistochemistry (IHC)
2.6. Statistical Considerations
3. Results
3.1. Chemical Synthesis
3.2. In Vitro Inhibition Assay
3.3. Imaging
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kularatne, S.A.; Thomas, M.; Myers, C.H.; Gagare, P.; Kanduluru, A.K.; Crian, C.J.; Cichocki, B.N.; Crain, C.J. Evaluation of Novel Prostate-Specific Membrane Antigen-Targeted Near-Infrared Imaging Agent for Fluorescence-Guided Surgery of Prostate Cancer. Clin. Cancer Res. 2018, 25, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Lwin, T.M.; Turner, M.A.; Amirfakhri, S.; Nishino, H.; Hoffman, R.M.; Bouvet, M. Fluorescence Molecular Targeting of Colon Cancer to Visualize the Invisible. Cells 2022, 11, 249. [Google Scholar] [CrossRef]
- Nishio, N.; Berg, N.S.V.D.; Van Keulen, S.; Martin, B.A.; Fakurnejad, S.; Teraphongphom, N.; Chirita, S.U.; Oberhelman, N.J.; Lu, G.; Horton, C.; et al. Optical molecular imaging can differentiate metastatic from benign lymph nodes in head and neck cancer. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, S.L. Near infrared fluorescence for image-guided surgery. Quant. Imaging Med. Surg. 2012, 2, 177–187. [Google Scholar] [PubMed]
- Thammineedi, S.R.; Saksena, A.R.; Nusrath, S.; Iyer, R.R.; Shukla, S.; Patnaik, S.C.; Reddy, R.P.; Boleneni, N.; Sharma, R.M.; Smith, L.; et al. Fluorescence-guided cancer surgery-A new paradigm. J. Surg. Oncol. 2021, 123, 1679–1698. [Google Scholar] [CrossRef] [PubMed]
- Abrahimi, P.; McClure, T. Emerging Intraoperative Imaging Technologies in Urologic Oncology. Urol. Clin. North Am. 2021, 49, 57–63. [Google Scholar] [CrossRef]
- A Stern, L.; A Case, B.; Hackel, B.J. Alternative non-antibody protein scaffolds for molecular imaging of cancer. Curr. Opin. Chem. Eng. 2013, 2, 425–432. [Google Scholar] [CrossRef]
- Fukuda, T.; Yokomizo, S.; Casa, S.; Monaco, H.; Manganiello, S.; Wang, H.; Lv, X.; Ulumben, A.D.; Yang, C.; Kang, M.W.; et al. Fast and Durable Intraoperative Near-infrared Imaging of Ovarian Cancer Using Ultrabright Squaraine Fluorophores. Angew. Chem. 2022. [Google Scholar] [CrossRef]
- Hingorani, D.V.; A Whitney, M.; Friedman, B.; Kwon, J.-K.; Crisp, J.L.; Xiong, Q.; Gross, L.; Kane, C.J.; Tsien, R.Y.; Nguyen, Q.T. Nerve-targeted probes for fluorescence-guided intraoperative imaging. Theranostics 2018, 8, 4226–4237. [Google Scholar] [CrossRef]
- Wang, L.G.; Barth, C.W.; Kitts, C.H.; Mebrat, M.D.; Montaño, A.R.; House, B.J.; McCoy, M.E.; Antaris, A.L.; Galvis, S.N.; McDowall, I.; et al. Near-infrared nerve-binding fluorophores for buried nerve tissue imaging. Sci. Transl. Med. 2020, 12. [Google Scholar] [CrossRef]
- Kaplan-Marans, E.; Fulla, J.; Tomer, N.; Bilal, K.; Palese, M. Indocyanine Green (ICG) in Urologic Surgery. Urology 2019, 132, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, G.M.; Themelis, G.; Crane, L.M.; Harlaar, N.J.; Pleijhuis, R.G.; Kelder, W.; Sarantopoulos, A.; De Jong, J.S.; Arts, H.J.; Van Der Zee, A.G.; et al. Intraoperative tumor-specific fluorescence imaging in ovarian cancer by folate receptor-α targeting: First in-human results. Nat. Med. 2011, 17, 1315–1319. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Dhara, S.; Banerjee, S.R.; Byun, Y.; Pullambhatla, M.; Mease, R.C.; Pomper, M.G. A low molecular weight PSMA-based fluorescent imaging agent for cancer. Biochem. Biophys. Res. Commun. 2009, 390, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.R.; Pullambhatla, M.; Byun, Y.; Nimmagadda, S.; Foss, C.A.; Green, G.; Fox, J.J.; Lupold, S.E.; Mease, R.C.; Pomper, M.G. Sequential SPECT and Optical Imaging of Experimental Models of Prostate Cancer with a Dual Modality Inhibitor of the Prostate-Specific Membrane Antigen. Angew. Chem. Int. Ed. 2011, 50, 9167–9170. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pullambhatla, M.; Banerjee, S.R.; Byun, Y.; Stathis, M.; Rojas, C.; Slusher, B.S.; Mease, R.C.; Pomper, M.G. Synthesis and Biological Evaluation of Low Molecular Weight Fluorescent Imaging Agents for the Prostate-Specific Membrane Antigen. Bioconjugate Chem. 2012, 23, 2377–2385. [Google Scholar] [CrossRef][Green Version]
- Kelderhouse, L.E.; Chelvam, V.; Wayua, C.; Mahalingam, S.; Poh, S.; Kularatne, S.A.; Low, P.S. Development of Tumor-Targeted Near Infrared Probes for Fluorescence Guided Surgery. Bioconjugate Chem. 2013, 24, 1075–1080. [Google Scholar] [CrossRef]
- Wang, X.; Huang, S.S.; Heston, W.D.; Guo, H.; Wang, B.-C.; Basilion, J.P. Development of Targeted Near-Infrared Imaging Agents for Prostate Cancer. Mol. Cancer Ther. 2014, 13, 2595–2606. [Google Scholar] [CrossRef]
- Baranski, A.-C.; Schäfer, M.; Bauder-Wüst, U.; Roscher, M.; Schmidt, J.; Stenau, E.; Simpfendörfer, T.; Teber, D.; Maier-Hein, L.; Hadaschik, B.; et al. PSMA-11–Derived Dual-Labeled PSMA Inhibitors for Preoperative PET Imaging and Precise Fluorescence-Guided Surgery of Prostate Cancer. J. Nucl. Med. 2017, 59, 639–645. [Google Scholar] [CrossRef]
- Liu, H.; Moy, P.; Kim, S.; Xia, Y.; Rajasekaran, A.; Navarro, V.; Knudsen, B.; Bander, N.H. Monoclonal antibodies to the extracellular domain of prostate-specific membrane antigen also react with tumor vascular endothelium. Cancer Res. 1997, 57, 3629–3634. [Google Scholar]
- Chang, S.S.; E Reuter, V.; Heston, W.D.; Bander, N.H.; Grauer, L.S.; Gaudin, P.B. Five different anti-prostate-specific membrane antigen (PSMA) antibodies confirm PSMA expression in tumor-associated neovasculature. Cancer Res. 1999, 59, 3192–3198. [Google Scholar]
- Neuman, B.P.; Eifler, J.B.; Castanares, M.; Chowdhury, W.; Chen, Y.; Mease, R.C.; Ma, R.; Mukherjee, A.; Lupold, S.E.; Pomper, M.G.; et al. Real-time, Near-Infrared Fluorescence Imaging with an Optimized Dye/Light Source/Camera Combination for Surgical Guidance of Prostate Cancer. Clin. Cancer Res. 2015, 21, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.K.; Chen, Y.; Kang, J.; Lisok, A.; Minn, I.; Pomper, M.G.; Boctor, E.M. Prostate-specific membrane antigen-targeted photoacoustic imaging of prostate cancer in vivo. J. Biophotonics 2018, 11, e201800021. [Google Scholar] [CrossRef] [PubMed]
- ThermoFisher Scientific Product Guide. Available online: https://www.thermofisher.com/us/en/home/brands/thermo-scientific/pierce-protein-biology.html?cid=fl-ts-pierce (accessed on 22 February 2022).
- Maresca, K.P.; Hillier, S.M.; Femia, F.J.; Keith, D.; Barone, C.; Joyal, J.L.; Zimmerman, C.N.; Kozikowski, A.P.; Barrett, J.A.; Eckelman, W.C.; et al. A Series of Halogenated Heterodimeric Inhibitors of Prostate Specific Membrane Antigen (PSMA) as Radiolabeled Probes for Targeting Prostate Cancer. J. Med. Chem. 2009, 52, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Tykvart, J.; Schimer, J.; Bařinková, J.; Pachl, P.; Poštová-Slavětínská, L.; Majer, P.; Konvalinka, J.; Šácha, P. Rational design of urea-based glutamate carboxypeptidase II (GCPII) inhibitors as versatile tools for specific drug targeting and delivery. Bioorg. Med. Chem. 2014, 22, 4099–4108. [Google Scholar] [CrossRef]
- Chen, Y.; Pullambhatla, M.; Foss, C.A.; Byun, Y.; Nimmagadda, S.; Senthamizhchelvan, S.; Sgouros, G.; Mease, R.C.; Pomper, M.G. 2-(3-{1-Carboxy-5-[(6-[18F]fluoro-pyridine-3-carbonyl)-amino]-pentyl}-ureido)-pen tanedioic acid, [18F]DCFPyL, a PSMA-based PET imaging agent for prostate cancer. Clin. Cancer Res. An Off. J. Am. Assoc. Cancer Res. 2011, 17, 7645–7653. [Google Scholar] [CrossRef]
- Cheng, Y.; Prusoff, W.H. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem. Pharmacol. 1973, 22, 3099–3108. [Google Scholar]
- Boinapally, S.; Ahn, H.-H.; Cheng, B.; Brummet, M.; Nam, H.; Gabrielson, K.L.; Banerjee, S.R.; Minn, I.; Pomper, M.G. A prostate-specific membrane antigen (PSMA)-targeted prodrug with a favorable in vivo toxicity profile. Sci. Rep. 2021, 11, 1–10. [Google Scholar] [CrossRef]
- Rowe, S.P.; Drzezga, A.; Neumaier, B.; Dietlein, M.; Gorin, M.A.; Zalutsky, M.R.; Pomper, M.G. Prostate-Specific Membrane Antigen–Targeted Radiohalogenated PET and Therapeutic Agents for Prostate Cancer. J. Nucl. Med. 2016, 57, 90S–96S. [Google Scholar] [CrossRef]
- Barth, C.W.; Gibbs, S.L. Fluorescence Image-Guided Surgery—A Perspective on Contrast Agent Development. Proc. SPIE Int. Soc. Opt. Eng. 2020, 11222, 112220J. [Google Scholar]
- Banerjee, S.R.; Kumar, V.; Lisok, A.; Chen, J.; Minn, I.; Brummet, M.; Boinapally, S.; Cole, M.; Ngen, E.; Wharram, B.; et al. 177Lu-labeled low-molecular-weight agents for PSMA-targeted radiopharmaceutical therapy. Eur. J. Pediatr. 2019, 46, 2545–2557. [Google Scholar] [CrossRef]
- Rowe, S.P.; Gorin, M.A.; Hammers, H.J.; Som Javadi, M.; Hawasli, H.; Szabo, Z.; Cho, S.Y.; Pomper, M.G.; Allaf, M.E. Imaging of metastatic clear cell renal cell carcinoma with PSMA-targeted (1)(8)F-DCFPyL PET/CT. Ann. Nucl. Med. 2015, 29, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Rowe, S.P.; Gorin, M.A.; Hammers, H.J.; Pomper, M.G.; Allaf, M.E.; Javadi, M.S. Detection of 18F-FDG PET/CT Occult Lesions With 18F-DCFPyL PET/CT in a Patient With Metastatic Renal Cell Carcinoma. Clin. Nucl. Med. 2016, 41, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.R.; Carducci, M.A.; Denmeade, S.R.; Markowski, M.C.; Pomper, M.G.; Pierorazio, P.M.; Allaf, M.E.; Rowe, S.P.; Gorin, M.A. Improved identification of patients with oligometastatic clear cell renal cell carcinoma with PSMA-targeted (18)F-DCFPyL PET/CT. Ann. Nucl. Med. 2019, 33, 617–623. [Google Scholar] [CrossRef]
- Zhang, J.; Rakhimbekova, A.; Duan, X.; Yin, Q.; Foss, C.A.; Fan, Y.; Xu, Y.; Li, X.; Cai, X.; Kutil, Z.; et al. A prostate-specific membrane antigen activated molecular rotor for real-time fluorescence imaging. Nat. Commun. 2021, 12, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.J.; Olson, J.M. In Vivo Bio-imaging Using Chlorotoxin-based Conjugates. Curr. Pharm. Des. 2011, 17, 4362–4371. [Google Scholar]
- Barth, C.W.; Gibbs, S.L. Direct Administration of Nerve-Specific Contrast to Improve Nerve Sparing Radical Prostatectomy. Theranostics 2017, 7, 573–593. [Google Scholar] [CrossRef]
- Tung, C.-H.; Han, M.S.; Shen, Z.; Gray, B.D.; Pak, K.Y.; Wang, J. Near-Infrared Fluorogenic Spray for Rapid Tumor Sensing. ACS Sens. 2021, 6, 3657–3666. [Google Scholar] [CrossRef]
- Tynan, C.J.; Clarke, D.; Coles, B.C.; Rolfe, D.; Martin-Fernandez, M.; Webb, S.E.D. Multicolour Single Molecule Imaging in Cells with Near Infra-Red Dyes. PLoS ONE 2012, 7, e36265. [Google Scholar] [CrossRef]
- Schreiber, C.L.; Li, D.-H.; Smith, B.D. High-Performance Near-Infrared Fluorescent Secondary Antibodies for Immunofluorescence. Anal. Chem. 2021, 93, 3643–3651. [Google Scholar] [CrossRef]




| Compound | Ki (nM) | 95% Confidence Interval of Ki |
|---|---|---|
| DyLight800-1 | 0.37 | 0.26–0.52 |
| DyLight800-2 | 0.16 | 0.11–0.22 |
| DyLight800-3 | 0.10 | 0.06–0.15 |
| DyLight800-4 | 0.29 | 0.17–0.51 |
| DyLight800-5 | 0.45 | 0.31–0.65 |
| DyLight800-6 | 0.68 | 0.40–1.17 |
| DyLight800-7 | 0.85 | 0.52–1.36 |
| DyLight800-8 | 0.50 | 0.34–0.73 |
| DyLight800-9 | 2.19 | 1.22–2.71 |
| DyLight800-10 | 0.83 | 0.73–0.94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Minn, I.; Rowe, S.P.; Lisok, A.; Chatterjee, S.; Brummet, M.; Banerjee, S.R.; Mease, R.C.; Pomper, M.G. A Series of PSMA-Targeted Near-Infrared Fluorescent Imaging Agents. Biomolecules 2022, 12, 405. https://doi.org/10.3390/biom12030405
Chen Y, Minn I, Rowe SP, Lisok A, Chatterjee S, Brummet M, Banerjee SR, Mease RC, Pomper MG. A Series of PSMA-Targeted Near-Infrared Fluorescent Imaging Agents. Biomolecules. 2022; 12(3):405. https://doi.org/10.3390/biom12030405
Chicago/Turabian StyleChen, Ying, Il Minn, Steven P. Rowe, Alla Lisok, Samit Chatterjee, Mary Brummet, Sangeeta Ray Banerjee, Ronnie C. Mease, and Martin G. Pomper. 2022. "A Series of PSMA-Targeted Near-Infrared Fluorescent Imaging Agents" Biomolecules 12, no. 3: 405. https://doi.org/10.3390/biom12030405
APA StyleChen, Y., Minn, I., Rowe, S. P., Lisok, A., Chatterjee, S., Brummet, M., Banerjee, S. R., Mease, R. C., & Pomper, M. G. (2022). A Series of PSMA-Targeted Near-Infrared Fluorescent Imaging Agents. Biomolecules, 12(3), 405. https://doi.org/10.3390/biom12030405






