The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Polymer ELISA
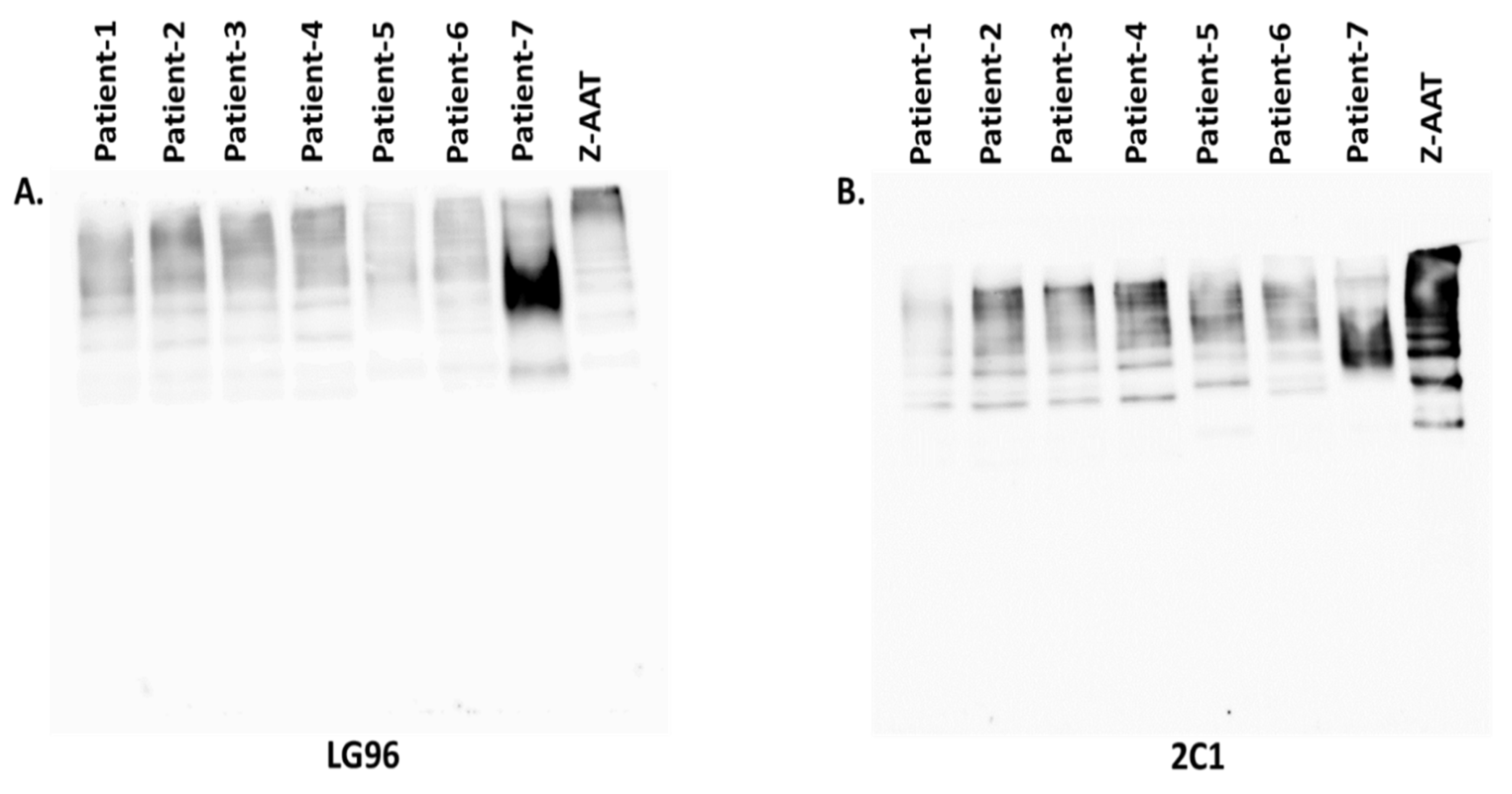
2.3. Western Blot Analyses
2.4. Statistical Analysis
3. Results
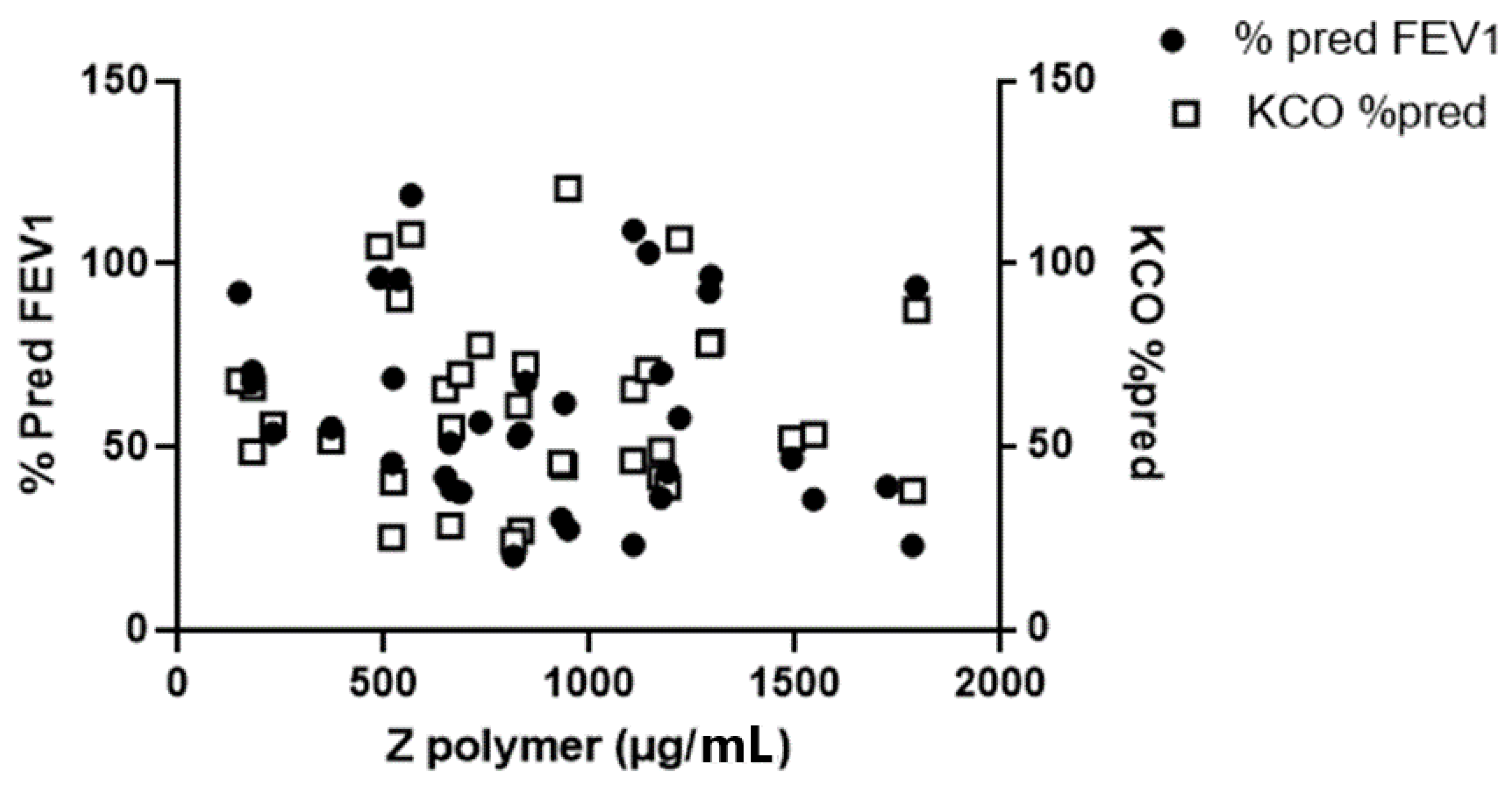
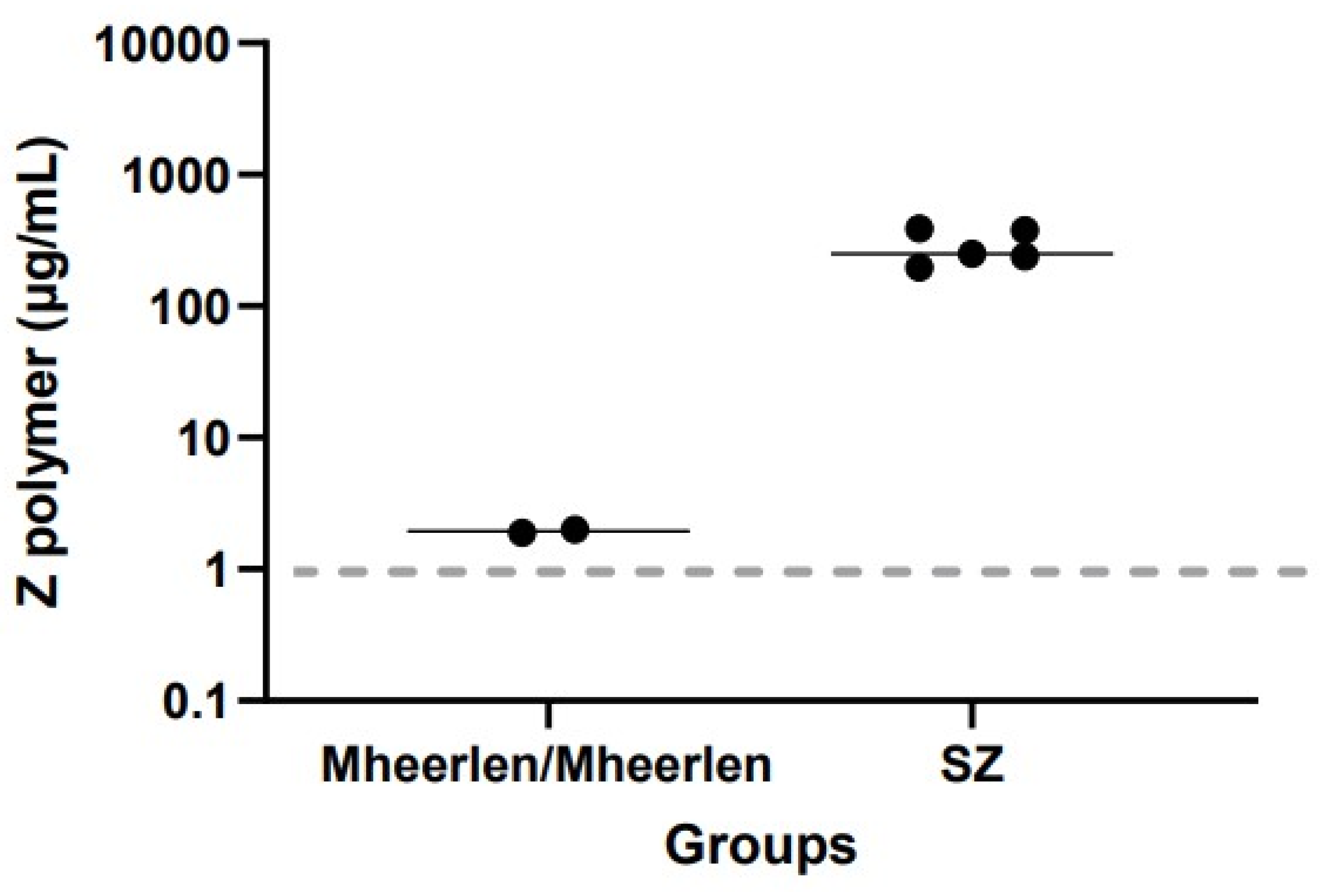
3.1. The Relationship between Plasma Z-Aat Polymers and Lung Function in Dutch Cohort
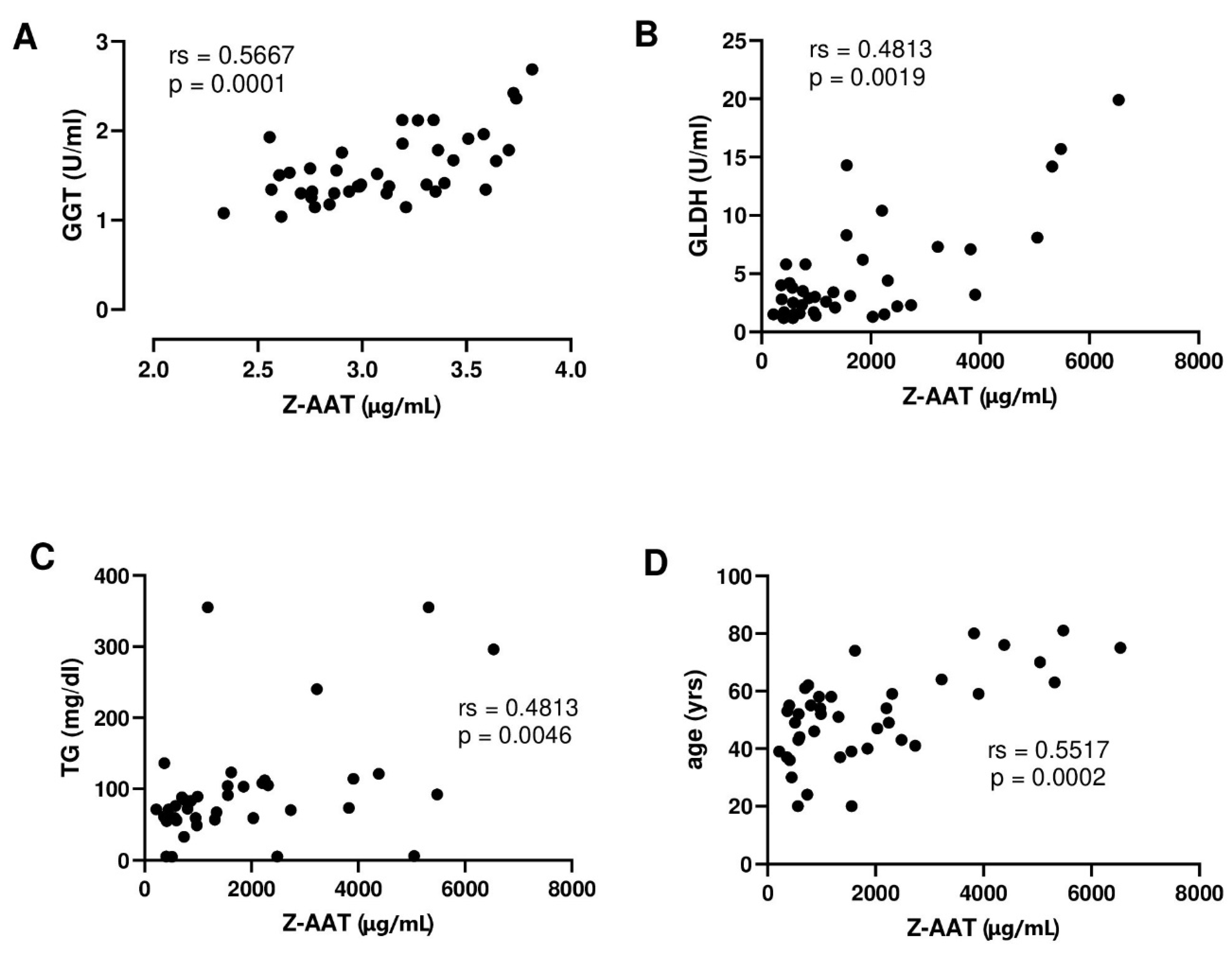
3.2. The Relationship between Plasma Z-AAT Polymers and Liver Function in the Alpha1-Liver Aachen Cohort
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bergin, D.A.; Reeves, E.P.; Hurley, K.; Wolfe, R.; Jameel, R.; Fitzgerald, S.; McElvaney, N.G. The circulating proteinase inhibitor alpha-1 antitrypsin regulates neutrophil degranulation and autoimmunity. Sci. Transl. Med. 2014, 6, 217ra1. [Google Scholar] [CrossRef]
- Jonigk, D.; Al-Omari, M.; Maegel, L.; Muller, M.; Izykowski, N.; Hong, J.; Hong, K.; Kim, S.H.; Dorsch, M.; Mahadeva, R.; et al. Anti-inflammatory and immunomodulatory properties of alpha1-antitrypsin without inhibition of elastase. Proc. Natl. Acad. Sci. USA 2013, 110, 15007–15012. [Google Scholar] [CrossRef]
- Seixas, S.; Marques, P.I. Known Mutations at the Cause of Alpha-1 Antitrypsin Deficiency an Updated Overview of SERPINA1 Variation Spectrum. Appl. Clin. Genet. 2021, 14, 173–194. [Google Scholar] [CrossRef]
- Strnad, P.; McElvaney, N.G.; Lomas, D.A. Alpha1-Antitrypsin Deficiency. N. Engl. J. Med. 2020, 382, 1443–1455. [Google Scholar] [CrossRef]
- Janciauskiene, S.; Welte, T. Well-Known and Less Well-Known Functions of Alpha-1 Antitrypsin. Its Role in Chronic Obstructive Pulmonary Disease and Other Disease Developments. Ann. Am. Thorac. Soc. 2016, 13 (Suppl S4), S280–S288. [Google Scholar] [CrossRef]
- Mulgrew, A.T.; Taggart, C.C.; Lawless, M.W.; Greene, C.M.; Brantly, M.L.; O’Neill, S.J.; McElvaney, N.G. Z alpha1-antitrypsin polymerizes in the lung and acts as a neutrophil chemoattractant. Chest 2004, 125, 1952–1957. [Google Scholar] [CrossRef]
- Tumpara, S.; Ballmaier, M.; Wrenger, S.; Konig, M.; Lehmann, M.; Lichtinghagen, R.; Martinez-Delgado, B.; Korenbaum, E.; DeLuca, D.; Jedicke, N.; et al. Polymerization of misfolded Z alpha-1 antitrypsin protein lowers CX3CR1 expression in human PBMCs. eLife 2021, 10, e64881. [Google Scholar] [CrossRef]
- Patel, D.; McAllister, S.L.; Teckman, J.H. Alpha-1 antitrypsin deficiency liver disease. Transl. Gastroenterol. Hepatol. 2021, 6, 23. [Google Scholar] [CrossRef]
- Wewers, M.D.; Casolaro, M.A.; Sellers, S.E.; Swayze, S.C.; McPhaul, K.M.; Wittes, J.T.; Crystal, R.G. Replacement therapy for alpha 1-antitrypsin deficiency associated with emphysema. N. Engl. J. Med. 1987, 316, 1055–1062. [Google Scholar] [CrossRef]
- Janciauskiene, S.; Dominaitiene, R.; Sternby, N.H.; Piitulainen, E.; Eriksson, S. Detection of circulating and endothelial cell polymers of Z and wild type alpha 1-antitrypsin by a monoclonal antibody. J. Biol. Chem. 2002, 277, 26540–26546. [Google Scholar] [CrossRef]
- Tan, L.; Dickens, J.A.; Demeo, D.L.; Miranda, E.; Perez, J.; Rashid, S.T.; Day, J.; Ordonez, A.; Marciniak, S.J.; Haq, I.; et al. Circulating polymers in alpha1-antitrypsin deficiency. Eur. Respir. J. 2014, 43, 1501–1504. [Google Scholar] [CrossRef]
- Gross, B.; Grebe, M.; Wencker, M.; Stoller, J.K.; Bjursten, L.M.; Janciauskiene, S. New Findings in PiZZ alpha1-antitrypsin deficiency-related panniculitis. Demonstration of skin polymers and high dosing requirements of intravenous augmentation therapy. Dermatology 2009, 218, 370–375. [Google Scholar] [CrossRef]
- Mahadeva, R.; Atkinson, C.; Li, Z.; Stewart, S.; Janciauskiene, S.; Kelley, D.G.; Parmar, J.; Pitman, R.; Shapiro, S.D.; Lomas, D.A. Polymers of Z alpha1-antitrypsin co-localize with neutrophils in emphysematous alveoli and are chemotactic in vivo. Am. J. Pathol. 2005, 166, 377–386. [Google Scholar] [CrossRef]
- Parmar, J.S.; Mahadeva, R.; Reed, B.J.; Farahi, N.; Cadwallader, K.A.; Keogan, M.T.; Bilton, D.; Chilvers, E.R.; Lomas, D.A. Polymers of alpha(1)-antitrypsin are chemotactic for human neutrophils: A new paradigm for the pathogenesis of emphysema. Am. J. Respir. Cell Mol. Biol. 2002, 26, 723–730. [Google Scholar] [CrossRef]
- Clemmensen, S.N.; Jacobsen, L.C.; Rorvig, S.; Askaa, B.; Christenson, K.; Iversen, M.; Jorgensen, M.H.; Larsen, M.T.; van Deurs, B.; Ostergaard, O.; et al. Alpha-1-antitrypsin is produced by human neutrophil granulocytes and their precursors and liberated during granule exocytosis. Eur. J. Haematol. 2011, 86, 517–530. [Google Scholar] [CrossRef]
- Ekeowa, U.I.; Freeke, J.; Miranda, E.; Gooptu, B.; Bush, M.F.; Perez, J.; Teckman, J.; Robinson, C.V.; Lomas, D.A. Defining the mechanism of polymerization in the serpinopathies. Proc. Natl. Acad. Sci. USA 2010, 107, 17146–17151. [Google Scholar] [CrossRef]
- Miranda, E.; Perez, J.; Ekeowa, U.I.; Hadzic, N.; Kalsheker, N.; Gooptu, B.; Portmann, B.; Belorgey, D.; Hill, M.; Chambers, S.; et al. A novel monoclonal antibody to characterize pathogenic polymers in liver disease associated with alpha1-antitrypsin deficiency. Hepatology 2010, 52, 1078–1088. [Google Scholar] [CrossRef]
- Lomas, D.A. An ECLIPSE View of Alpha-1 Antitrypsin Deficiency. Ann. Am. Thorac. Soc. 2016, 13 (Suppl S4), S326–S331. [Google Scholar] [CrossRef]
- Luisetti, M.; Miravitlles, M.; Stockley, R.A. Alpha1-antitrypsin deficiency: A report from the 2nd meeting of the Alpha One International Registry, Rapallo (Genoa, Italy), 2001. Eur. Respir. J. 2002, 20, 1050–1056. [Google Scholar] [CrossRef]
- Cotes, J.E.; Chinn, D.J.; Quanjer, P.H.; Roca, J.; Yernault, J.C. Standardization of the measurement of transfer factor (diffusing capacity). Report Working Party Standardization of Lung Function Tests, European Community for Steel and Coal. Official Statement of the European Respiratory Society. Eur. Respir. J. Suppl. 1993, 16, 41–52. [Google Scholar] [CrossRef]
- Quanjer, P.H.; Tammeling, G.J.; Cotes, J.E.; Pedersen, O.F.; Peslin, R.; Yernault, J.C. Lung volumes and forced ventilatory flows. Report Working Party Standardization of Lung Function Tests, European Community for Steel and Coal. Official Statement of the European Respiratory Society. Eur. Respir. J. Suppl. 1993, 16, 5–40. [Google Scholar] [CrossRef]
- Stolk, J.; Veldhuisen, B.; Annovazzi, L.; Zanone, C.; Versteeg, E.M.; van Kuppevelt, T.H.; Berden, J.H.; Nieuwenhuizen, W.; Iadarola, P.; Luisetti, M. Short-term variability of biomarkers of proteinase activity in patients with emphysema associated with type Z alpha-1-antitrypsin deficiency. Respir. Res. 2005, 6, 47. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ferrarotti, I.; Thun, G.A.; Zorzetto, M.; Ottaviani, S.; Imboden, M.; Schindler, C.; von Eckardstein, A.; Rohrer, L.; Rochat, T.; Russi, E.W.; et al. Serum levels and genotype distribution of alpha1-antitrypsin in the general population. Thorax 2012, 67, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Fromme, M.; Schneider, C.V.; Trautwein, C.; Brunetti-Pierri, N.; Strnad, P. Alpha-1 antitrypsin deficiency: A re-surfacing adult liver disorder. J. Hepatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Aldonyte, R.; Eriksson, S.; Piitulainen, E.; Wallmark, A.; Janciauskiene, S. Analysis of systemic biomarkers in COPD patients. COPD J. Chronic Obstr. Pulm. Dis. 2004, 1, 155–164. [Google Scholar] [CrossRef]
- Nunez, A.; Belmonte, I.; Miranda, E.; Barrecheguren, M.; Farago, G.; Loeb, E.; Pons, M.; Rodriguez-Frias, F.; Gabriel-Medina, P.; Rodriguez, E.; et al. Association between circulating alpha-1 antitrypsin polymers and lung and liver disease. Respir. Res. 2021, 22, 244. [Google Scholar] [CrossRef]
- Whitfield, J.B. Gamma glutamyl transferase. Crit. Rev. Clin. Lab. Sci. 2001, 38, 263–355. [Google Scholar] [CrossRef]
- Clark, V.C.; Marek, G.; Liu, C.; Collinsworth, A.; Shuster, J.; Kurtz, T.; Nolte, J.; Brantly, M. Clinical and histologic features of adults with alpha-1 antitrypsin deficiency in a non-cirrhotic cohort. J. Hepatol. 2018, 69, 1357–1364. [Google Scholar] [CrossRef]
- Schomaker, S.; Warner, R.; Bock, J.; Johnson, K.; Potter, D.; Van Winkle, J.; Aubrecht, J. Assessment of emerging biomarkers of liver injury in human subjects. Toxicol. Sci. 2013, 132, 276–283. [Google Scholar] [CrossRef]
- Voss, C.M.; Arildsen, L.; Nissen, J.D.; Waagepetersen, H.S.; Schousboe, A.; Maechler, P.; Ott, P.; Vilstrup, H.; Walls, A.B. Glutamate Dehydrogenase Is Important for Ammonia Fixation and Amino Acid Homeostasis in Brain During Hyperammonemia. Front. Neurosci. 2021, 15, 646291. [Google Scholar] [CrossRef]
- Kuscuoglu, D.; Janciauskiene, S.; Hamesch, K.; Haybaeck, J.; Trautwein, C.; Strnad, P. Liver—Master and servant of serum proteome. J. Hepatol. 2018, 69, 512–524. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Byrne, C.D. Circulating Markers of Liver Function and Cardiovascular Disease Risk. Arter. Thromb. Vasc. Biol. 2015, 35, 2290–2296. [Google Scholar] [CrossRef] [PubMed]
- Wooddell, C.I.; Blomenkamp, K.; Peterson, R.M.; Subbotin, V.M.; Schwabe, C.; Hamilton, J.; Chu, Q.; Christianson, D.R.; Hegge, J.O.; Kolbe, J.; et al. Development of an RNAi therapeutic for alpha-1-antitrypsin liver disease. JCI Insight 2020, 5, e135348. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | ZZ-AATD | SZ-AATD | Rare AATD |
|---|---|---|---|
| N male/female | 26/30 | 2/3 | 2/4 |
| Age (mean ± SD) | 56 ± 7 | 51 ± 5 | 49 ± 9 |
| FEV1 % predicted (mean ± SD) | 58 ± 23 | 63 ± 17 | 53 ± 19 |
| Kco % predicted (mean ± SD) | 61 ± 27 | 60 ± 11 | 56 ± 18 |
| Serum ALT > ULN (%) | 0 | 0 | 0 |
| Serum AST > ULN (%) | 0 | 0 | 0 |
| Serum GGT > ULN (%) | 0 | 0 | 0 |
| Characteristics | Pi*ZZ (n = 40), Mean ± SD |
|---|---|
| Age (years) | 51.3 ± 15.3 |
| Women (%) | 15 (37.5) |
| BMI (kg/m2) | 26.0 ± 4.2 |
| BMI ≥ 30 (%) | 7 (17.5) |
| Diabetes mellitus (%) | 1 (2.5) |
| Liver status | . |
| Median stiffness (kPa) | 7.8 ± 11.0 |
| LSM ≥ 7.1 kPa (%) | 7 (17.5) |
| LSM ≥ 10 kPa (%) | 4 (10) |
| CAP (dB/m) | 274.8 ± 50.8 |
| ALT ≥ ULN (%) | 9 (22.5) |
| AST ≥ ULN (%) | 6 (15) |
| GGT ≥ ULN (%) | 13 (32.5) |
| ALP ≥ ULN (%) | 6 (15) |
| GLDH ≥ ULN (%) | 9 (22.5) |
| Bilirubin (mg/dL) | 0.65 ± 0.40 |
| INR | 1.07 ± 0.21 |
| Triglycerides (mg/dL) | 100.7 ± 63.8 |
| HbA1c (%) | 5.1 ± 0.3 |
| Ferritin | 185.1 ± 165.1 |
| AAT serum level (mg/dL) | 40.6 ± 30.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sark, A.D.; Fromme, M.; Olejnicka, B.; Welte, T.; Strnad, P.; Janciauskiene, S.; Stolk, J. The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients. Biomolecules 2022, 12, 380. https://doi.org/10.3390/biom12030380
Sark AD, Fromme M, Olejnicka B, Welte T, Strnad P, Janciauskiene S, Stolk J. The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients. Biomolecules. 2022; 12(3):380. https://doi.org/10.3390/biom12030380
Chicago/Turabian StyleSark, Annelot D., Malin Fromme, Beata Olejnicka, Tobias Welte, Pavel Strnad, Sabina Janciauskiene, and Jan Stolk. 2022. "The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients" Biomolecules 12, no. 3: 380. https://doi.org/10.3390/biom12030380
APA StyleSark, A. D., Fromme, M., Olejnicka, B., Welte, T., Strnad, P., Janciauskiene, S., & Stolk, J. (2022). The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients. Biomolecules, 12(3), 380. https://doi.org/10.3390/biom12030380









