Gut Microbiota as a Potential Player in Mn-Induced Neurotoxicity
Abstract
:1. Introduction
2. A Brief Introduction to Mn Neurotoxicity: Gut-Independent Mechanisms
3. The Role of Gut Microbiota and Its Metabolites in Brain Functioning and Neurodegeneration
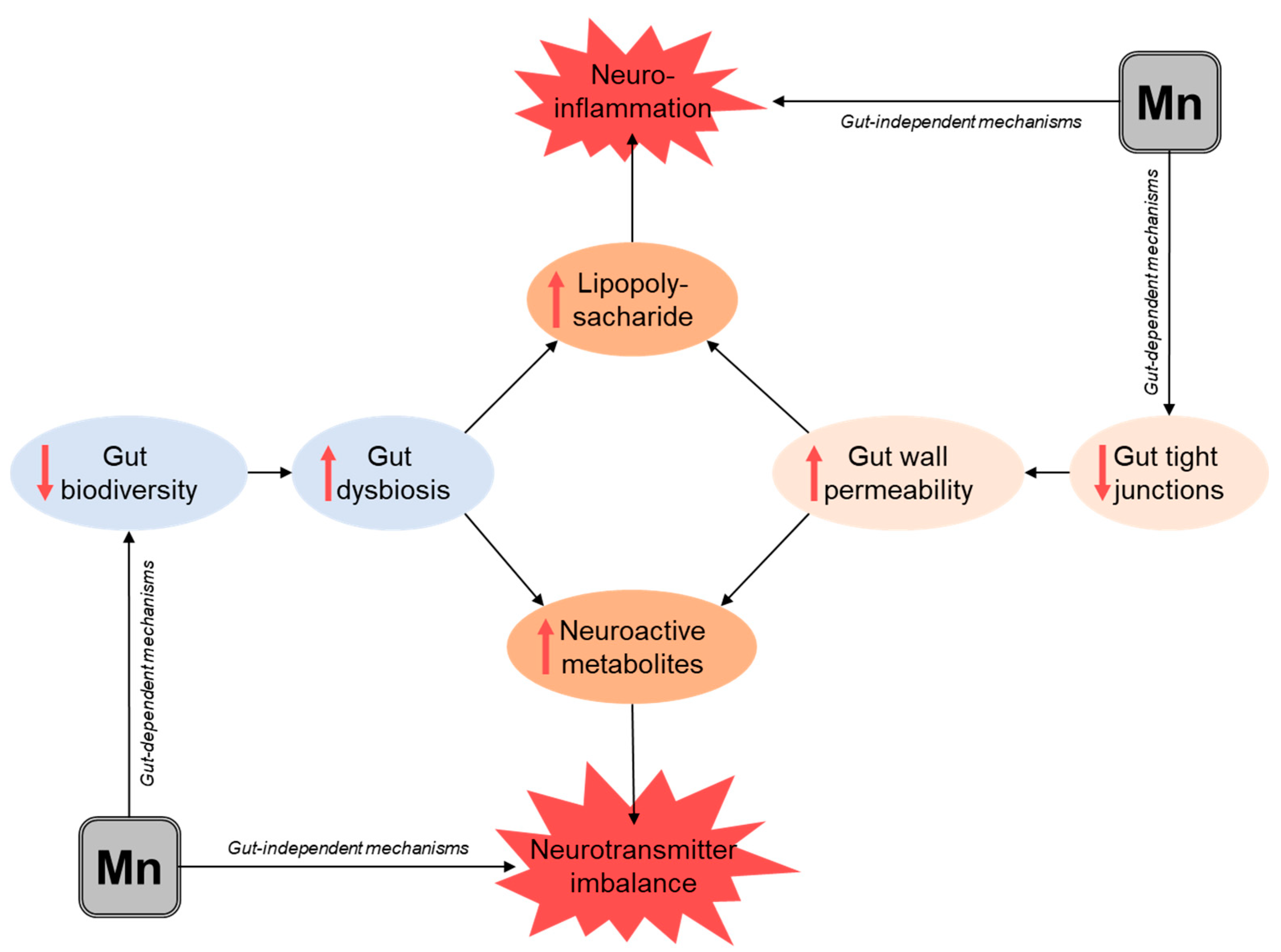
4. Mn and Gut Microbiota
5. Mn and Gut Wall Permeability
6. Mn as a Player in Nutritional Immunity
7. Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martins, A.C., Jr.; Gubert, P.; Villas Boas, G.R.; Meirelles Paes, M.; Santamaria, A.; Lee, E.; Tinkov, A.A.; Bowman, A.B.; Aschner, M. Manganese-induced neurodegenerative diseases and possible therapeutic approaches. Expert Rev. Neurother. 2020, 20, 1109–1121. [Google Scholar] [CrossRef]
- Mergler, D.; Huel, G.; Bowler, R.; Iregren, A.; Belanger, S.; Baldwin, M.; Tardif, R.; Smargiassi, A.; Martin, L. Nervous system dysfunction among workers with long-term exposure to manganese. Environ. Res. 1994, 64, 151–180. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, A.B.; Cushing, C.A.; Antonini, J.M.; Finley, B.L.; Mowat, F.S. State-of-the-science review: Does manganese exposure during welding pose a neurological risk? J. Toxicol. Environ. Health B Crit. Rev. 2007, 10, 417–465. [Google Scholar] [CrossRef] [PubMed]
- Roth, J.A. Are there common biochemical and molecular mechanisms controlling manganism and parkisonism. Neuromol. Med. 2009, 11, 281–296. [Google Scholar] [CrossRef] [PubMed]
- Aschner, M.; Dorman, D.C. Manganese: Pharmacokinetics and molecular mechanisms of brain uptake. Toxicol. Rev. 2006, 25, 147–154. [Google Scholar] [CrossRef]
- Aschner, M.; Erikson, K.M.; Herrero Hernandez, E.; Tjalkens, R. Manganese and its role in Parkinson’s disease: From transport to neuropathology. Neuromol. Med. 2009, 11, 252–266. [Google Scholar] [CrossRef]
- Martins, A.C.; Krum, B.N.; Queiros, L.; Tinkov, A.A.; Skalny, A.V.; Bowman, A.B.; Aschner, M. Manganese in the Diet: Bioaccessibility, Adequate Intake, and Neurotoxicological Effects. J. Agric. Food Chem. 2020, 68, 12893–12903. [Google Scholar] [CrossRef]
- Food and Drug Administration, H.H.S. Food Labeling: Revision of the Nutrition and Supplement Facts Labels. Final rule. Fed. Regist. 2016, 81, 33741–33999. [Google Scholar]
- Sachse, B.; Kolbaum, A.E.; Ziegenhagen, R.; Andres, S.; Berg, K.; Dusemund, B.; Hirsch-Ernst, K.I.; Kappenstein, O.; Muller, F.; Rohl, C.; et al. Dietary Manganese Exposure in the Adult Population in Germany-What Does it Mean in Relation to Health Risks? Mol. Nutr. Food Res. 2019, 63, e1900065. [Google Scholar] [CrossRef]
- Benedetto, A.; Au, C.; Aschner, M. Manganese-induced dopaminergic neurodegeneration: Insights into mechanisms and genetics shared with Parkinson’s disease. Chem. Rev. 2009, 109, 4862–4884. [Google Scholar] [CrossRef]
- Glowacki, R.W.P.; Martens, E.C. In sickness and health: Effects of gut microbial metabolites on human physiology. PLoS Pathog. 2020, 16, e1008370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toor, D.; Wsson, M.K.; Kumar, P.; Karthikeyan, G.; Kaushik, N.K.; Goel, C.; Singh, S.; Kumar, A.; Prakash, H. Dysbiosis Disrupts Gut Immune Homeostasis and Promotes Gastric Diseases. Int. J. Mol. Sci. 2019, 20, 2432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerhardt, S.; Mohajeri, M.H. Changes of Colonic Bacterial Composition in Parkinson’s Disease and Other Neurodegenerative Diseases. Nutrients 2018, 10, 708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ojeda, J.; Avila, A.; Vidal, P.M. Gut Microbiota Interaction with the Central Nervous System throughout Life. J. Clin. Med. 2021, 10, 1299. [Google Scholar] [CrossRef]
- Sharma, V.K.; Singh, T.G.; Garg, N.; Dhiman, S.; Gupta, S.; Rahman, M.H.; Najda, A.; Walasek-Janusz, M.; Kamel, M.; Albadrani, G.M.; et al. Dysbiosis and Alzheimer’s Disease: A Role for Chronic Stress? Biomolecules 2021, 11, 678. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.C.; Bhargava, R.; Vaziriyan-Sani, A.S.; Pourciau, C.; Donahue, E.T.; Dove, A.S.; Gebhardt, M.J.; Ellward, G.L.; Romeo, T.; Czyz, D.M. Colonization of the Caenorhabditis elegans gut with human enteric bacterial pathogens leads to proteostasis disruption that is rescued by butyrate. PLoS Pathog. 2021, 17, e1009510. [Google Scholar] [CrossRef]
- Huang, Y.; Liao, J.; Liu, X.; Zhong, Y.; Cai, X.; Long, L. Review: The Role of Intestinal Dysbiosis in Parkinson’s Disease. Front. Cell. Infect. Microbiol. 2021, 11, 615075. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; de Vos, R.A.; Bohl, J.; Del Tredici, K. Gastric alpha-synuclein immunoreactive inclusions in Meissner’s and Auerbach’s plexuses in cases staged for Parkinson’s disease-related brain pathology. Neurosci. Lett. 2006, 396, 67–72. [Google Scholar] [CrossRef]
- Tinkov, A.A.; Paoliello, M.M.B.; Mazilina, A.N.; Skalny, A.V.; Martins, A.C.; Voskresenskaya, O.N.; Aaseth, J.; Santamaria, A.; Notova, S.V.; Tsatsakis, A.; et al. Molecular Targets of Manganese-Induced Neurotoxicity: A Five-Year Update. Int. J. Mol. Sci. 2021, 22, 4646. [Google Scholar] [CrossRef]
- Graumann, R.; Paris, I.; Martinez-Alvarado, P.; Rumanque, P.; Perez-Pastene, C.; Cardenas, S.P.; Marin, P.; Diaz-Grez, F.; Caviedes, R.; Caviedes, P.; et al. Oxidation of dopamine to aminochrome as a mechanism for neurodegeneration of dopaminergic systems in Parkinson’s disease. Possible neuroprotective role of DT-diaphorase. Pol. J. Pharmacol. 2002, 54, 573–580. [Google Scholar]
- Gavin, C.E.; Gunter, K.K.; Gunter, T.E. Manganese and calcium efflux kinetics in brain mitochondria. Relevance to manganese toxicity. Biochem. J. 1990, 266, 329–334. [Google Scholar] [CrossRef]
- Morello, M.; Canini, A.; Mattioli, P.; Sorge, R.P.; Alimonti, A.; Bocca, B.; Forte, G.; Martorana, A.; Bernardi, G.; Sancesario, G. Sub-cellular localization of manganese in the basal ganglia of normal and manganese-treated rats An electron spectroscopy imaging and electron energy-loss spectroscopy study. Neurotoxicology 2008, 29, 60–72. [Google Scholar] [CrossRef]
- Zhang, S.; Fu, J.; Zhou, Z. In vitro effect of manganese chloride exposure on reactive oxygen species generation and respiratory chain complexes activities of mitochondria isolated from rat brain. Toxicol. In Vitro 2004, 18, 71–77. [Google Scholar] [CrossRef]
- Gunter, T.E.; Pfeiffer, D.R. Mechanisms by which mitochondria transport calcium. Am. J. Physiol. 1990, 258, C755–C786. [Google Scholar] [CrossRef]
- Milatovic, D.; Yin, Z.; Gupta, R.C.; Sidoryk, M.; Albrecht, J.; Aschner, J.L.; Aschner, M. Manganese induces oxidative impairment in cultured rat astrocytes. Toxicol. Sci. 2007, 98, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Ijomone, O.M.; Aluko, O.M.; Okoh, C.O.A.; Martins, A.C., Jr.; Aschner, M. Role for calcium signaling in manganese neurotoxicity. J. Trace Elem. Med. Biol. 2019, 56, 146–155. [Google Scholar] [CrossRef]
- Malecki, E.A. Manganese toxicity is associated with mitochondrial dysfunction and DNA fragmentation in rat primary striatal neurons. Brain Res. Bull. 2001, 55, 225–228. [Google Scholar] [CrossRef]
- Korsmeyer, S.J.; Wei, M.C.; Saito, M.; Weiler, S.; Oh, K.J.; Schlesinger, P.H. Pro-apoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that result in the release of cytochrome c. Cell Death Differ. 2000, 7, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Aschner, J.L.; dos Santos, A.P.; Aschner, M. Mitochondrial-dependent manganese neurotoxicity in rat primary astrocyte cultures. Brain Res. 2008, 1203, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Cao, R.; Cai, T.; Aschner, M.; Zhao, F.; Yao, T.; Chen, Y.; Cao, Z.; Luo, W.; Chen, J. The role of autophagy dysregulation in manganese-induced dopaminergic neurodegeneration. Neurotox. Res. 2013, 24, 478–490. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Su, P.; Luo, W.; Chen, J. Role of LRRK2 in manganese-induced neuroinflammation and microglial autophagy. Biochem. Biophys. Res. Commun. 2018, 498, 171–177. [Google Scholar] [CrossRef]
- Ghavami, S.; Shojaei, S.; Yeganeh, B.; Ande, S.R.; Jangamreddy, J.R.; Mehrpour, M.; Christoffersson, J.; Chaabane, W.; Moghadam, A.R.; Kashani, H.H.; et al. Autophagy and apoptosis dysfunction in neurodegenerative disorders. Prog. Neurobiol. 2014, 112, 24–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Yan, J.; Bowman, A.B.; Bryan, M.R.; Singh, R.; Aschner, M. Dysregulation of TFEB contributes to manganese-induced autophagic failure and mitochondrial dysfunction in astrocytes. Autophagy 2020, 16, 1506–1523. [Google Scholar] [CrossRef]
- Bryan, M.R.; O’Brien, M.T.; Nordham, K.D.; Rose, D.I.R.; Foshage, A.M.; Joshi, P.; Nitin, R.; Uhouse, M.A.; Di Pardo, A.; Zhang, Z.; et al. Acute manganese treatment restores defective autophagic cargo loading in Huntington’s disease cell lines. Hum. Mol. Genet. 2019, 28, 3825–3841. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, J.; Jiang, W.; Cao, Z.; Zhao, F.; Cai, T.; Aschner, M.; Luo, W. The role of NLRP3-CASP1 in inflammasome-mediated neuroinflammation and autophagy dysfunction in manganese-induced, hippocampal-dependent impairment of learning and memory ability. Autophagy 2017, 13, 914–927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Z.; Wang, C.; Liu, C.; Yan, D.Y.; Deng, Y.; Liu, W.; Yang, T.Y.; Xu, Z.F.; Xu, B. The role S-nitrosylation in manganese-induced autophagy dysregulation in SH-SY5Y cells. Environ. Toxicol. 2017, 32, 2428–2439. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.Y.; Xu, B. The Role of Autophagy in Manganese-Induced Neurotoxicity. Front. Neurosci. 2020, 14, 574750. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yan, D.Y.; Wang, C.; Ma, Z.; Deng, Y.; Liu, W.; Xu, B. IRE1 signaling pathway mediates protective autophagic response against manganese-induced neuronal apoptosis in vivo and in vitro. Sci. Total Environ. 2020, 712, 136480. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Lei, M.Y.; Liu, Z.Q.; Liu, Z.F.; Ma, Z.; Liu, K.; Li, J.; Deng, Y.; Liu, W.; Xu, B. Resveratrol attenuates manganese-induced oxidative stress and neuroinflammation through SIRT1 signaling in mice. Food Chem. Toxicol. 2021, 153, 112283. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Zhang, Z.; Wang, Q.; Zhang, J.; Wang, L.; Zhang, Q.; Li, H.; Wu, S. Manganese chloride induces histone acetylation changes in neuronal cells: Its role in manganese-induced damage. Neurotoxicology 2018, 65, 255–263. [Google Scholar] [CrossRef]
- Searles Nielsen, S.; Checkoway, H.; Criswell, S.R.; Farin, F.M.; Stapleton, P.L.; Sheppard, L.; Racette, B.A. Inducible nitric oxide synthase gene methylation and parkinsonism in manganese-exposed welders. Parkinsonism Relat. Disord. 2015, 21, 355–360. [Google Scholar] [CrossRef] [Green Version]
- Tarale, P.; Sivanesan, S.; Daiwile, A.P.; Stoger, R.; Bafana, A.; Naoghare, P.K.; Parmar, D.; Chakrabarti, T.; Kannan, K. Global DNA methylation profiling of manganese-exposed human neuroblastoma SH-SY5Y cells reveals epigenetic alterations in Parkinson’s disease-associated genes. Arch. Toxicol. 2017, 91, 2629–2641. [Google Scholar] [CrossRef]
- Lee, E.; Karki, P.; Johnson, J., Jr.; Hong, P.; Aschner, M. Manganese Control of Glutamate Transporters’ Gene Expression. Adv. Neurobiol. 2017, 16, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Sidoryk-Wegrzynowicz, M.; Lee, E.; Aschner, M. Mechanism of Mn(II)-mediated dysregulation of glutamine-glutamate cycle: Focus on glutamate turnover. J. Neurochem. 2012, 122, 856–867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, B.; Wang, X.; Li, L. Human gut microbiome: The second genome of human body. Protein Cell 2010, 1, 718–725. [Google Scholar] [CrossRef] [Green Version]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Xu, J.; Mahowald, M.A.; Ley, R.E.; Lozupone, C.A.; Hamady, M.; Martens, E.C.; Henrissat, B.; Coutinho, P.M.; Minx, P.; Latreille, P.; et al. Evolution of symbiotic bacteria in the distal human intestine. PLoS Biol. 2007, 5, e156. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.M.; Surette, M.; Bercik, P. The interplay between the intestinal microbiota and the brain. Nat. Rev. Microbiol. 2012, 10, 735–742. [Google Scholar] [CrossRef]
- Gallo, P.M.; Rapsinski, G.J.; Wilson, R.P.; Oppong, G.O.; Sriram, U.; Goulian, M.; Buttaro, B.; Caricchio, R.; Gallucci, S.; Tukel, C. Amyloid-DNA Composites of Bacterial Biofilms Stimulate Autoimmunity. Immunity 2015, 42, 1171–1184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhan, X.; Stamova, B.; Jin, L.W.; DeCarli, C.; Phinney, B.; Sharp, F.R. Gram-negative bacterial molecules associate with Alzheimer disease pathology. Neurology 2016, 87, 2324–2332. [Google Scholar] [CrossRef] [Green Version]
- Paley, E.L.; Merkulova-Rainon, T.; Faynboym, A.; Shestopalov, V.I.; Aksenoff, I. Geographical Distribution and Diversity of Gut Microbial NADH:Ubiquinone Oxidoreductase Sequence Associated with Alzheimer’s Disease. J. Alzheimers Dis. 2018, 61, 1531–1540. [Google Scholar] [CrossRef]
- Giovannini, M.G.; Lana, D.; Traini, C.; Vannucchi, M.G. The Microbiota-Gut-Brain Axis and Alzheimer Disease. From Dysbiosis to Neurodegeneration: Focus on the Central Nervous System Glial Cells. J. Clin. Med. 2021, 10, 2358. [Google Scholar] [CrossRef]
- Erickson, M.A.; Hartvigson, P.E.; Morofuji, Y.; Owen, J.B.; Butterfield, D.A.; Banks, W.A. Lipopolysaccharide impairs amyloid beta efflux from brain: Altered vascular sequestration, cerebrospinal fluid reabsorption, peripheral clearance and transporter function at the blood-brain barrier. J. Neuroinflamm. 2012, 9, 150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galloway, S.; Takechi, R.; Nesbit, M.; Pallebage-Gamarallage, M.M.; Lam, V.; Mamo, J.C.L. The differential effects of fatty acids on enterocytic abundance of amyloid-beta. Lipids Health Dis. 2019, 18, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galloway, S.; Takechi, R.; Pallebage-Gamarallage, M.M.; Dhaliwal, S.S.; Mamo, J.C. Amyloid-beta colocalizes with apolipoprotein B in absorptive cells of the small intestine. Lipids Health Dis. 2009, 8, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scheperjans, F.; Aho, V.; Pereira, P.A.; Koskinen, K.; Paulin, L.; Pekkonen, E.; Haapaniemi, E.; Kaakkola, S.; Eerola-Rautio, J.; Pohja, M.; et al. Gut microbiota are related to Parkinson’s disease and clinical phenotype. Mov. Disord. 2015, 30, 350–358. [Google Scholar] [CrossRef]
- Heinzel, S.; Aho, V.T.E.; Suenkel, U.; von Thaler, A.K.; Schulte, C.; Deuschle, C.; Paulin, L.; Hantunen, S.; Brockmann, K.; Eschweiler, G.W.; et al. Gut microbiome signatures of risk and prodromal markers of Parkinson’s disease. Ann. Neurol. 2021, 88, 320–331. [Google Scholar] [CrossRef]
- Hasegawa, S.; Goto, S.; Tsuji, H.; Okuno, T.; Asahara, T.; Nomoto, K.; Shibata, A.; Fujisawa, Y.; Minato, T.; Okamoto, A.; et al. Intestinal Dysbiosis and Lowered Serum Lipopolysaccharide-Binding Protein in Parkinson’s Disease. PLoS ONE 2015, 10, e0142164. [Google Scholar] [CrossRef] [Green Version]
- Hopfner, F.; Kunstner, A.; Muller, S.H.; Kunzel, S.; Zeuner, K.E.; Margraf, N.G.; Deuschl, G.; Baines, J.F.; Kuhlenbaumer, G. Gut microbiota in Parkinson disease in a northern German cohort. Brain Res. 2017, 1667, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, X.; Hu, X.; Wang, T.; Liang, S.; Duan, Y.; Jin, F.; Qin, B. Structural changes of gut microbiota in Parkinson’s disease and its correlation with clinical features. Sci. China Life Sci. 2017, 60, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.F.; Zhu, Y.L.; Zhou, Z.L.; Jia, X.B.; Xu, Y.D.; Yang, Q.; Cui, C.; Shen, Y.Q. Neuroprotective effects of fecal microbiota transplantation on MPTP-induced Parkinson’s disease mice: Gut microbiota, glial reaction and TLR4/TNF-alpha signaling pathway. Brain Behav. Immun. 2018, 70, 48–60. [Google Scholar] [CrossRef]
- Ghebretatios, M.; Schaly, S.; Prakash, S. Nanoparticles in the Food Industry and Their Impact on Human Gut Microbiome and Diseases. Int. J. Mol. Sci. 2021, 22, 1942. [Google Scholar] [CrossRef] [PubMed]
- Fournier, E.; Etienne-Mesmin, L.; Grootaert, C.; Jelsbak, L.; Syberg, K.; Blanquet-Diot, S.; Mercier-Bonin, M. Microplastics in the human digestive environment: A focus on the potential and challenges facing in vitro gut model development. J. Hazard. Mater. 2021, 415, 125632. [Google Scholar] [CrossRef] [PubMed]
- Giambo, F.; Teodoro, M.; Costa, C.; Fenga, C. Toxicology and Microbiota: How Do Pesticides Influence Gut Microbiota? A Review. Int. J. Environ. Res. Public Health 2021, 18, 5510. [Google Scholar] [CrossRef]
- Tian, Y.; Gui, W.; Rimal, B.; Koo, I.; Smith, P.B.; Nichols, R.G.; Cai, J.; Liu, Q.; Patterson, A.D. Metabolic impact of persistent organic pollutants on gut microbiota. Gut Microbes 2020, 12, 1–16. [Google Scholar] [CrossRef]
- Martins, A.C.; Ke, T.; Bowman, A.B.; Aschner, M. New insights on mechanisms underlying methylmercury-induced and manganese-induced neurotoxicity. Curr. Opin. Toxicol. 2021, 25, 30–35. [Google Scholar] [CrossRef]
- Chi, L.; Gao, B.; Bian, X.; Tu, P.; Ru, H.; Lu, K. Manganese-induced sex-specific gut microbiome perturbations in C57BL/6 mice. Toxicol. Appl. Pharmacol. 2017, 331, 142–153. [Google Scholar] [CrossRef] [Green Version]
- Weng, Y.J.; Gan, H.Y.; Li, X.; Huang, Y.; Li, Z.C.; Deng, H.M.; Chen, S.Z.; Zhou, Y.; Wang, L.S.; Han, Y.P.; et al. Correlation of diet, microbiota and metabolite networks in inflammatory bowel disease. J. Dig. Dis. 2019, 20, 447–459. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, H.; Wang, B.; Zhang, B.; Li, W. Effects of manganese and Bacillus subtilis on the reproductive performance, egg quality, antioxidant capacity, and gut microbiota of breeding geese during laying period. Poult. Sci. 2020, 99, 6196–6204. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Guo, J.; Shi, J.; Shi, Z.; Zhang, G.; Zhang, J. Stress response of Lymantria dispar asiatica (Lepidoptera: Erebidae) larvae and its gut microbiota to manganese ion. J. For. Res. 2020, 32, 1241–1251. [Google Scholar] [CrossRef]
- Ghaisas, S.; Maher, J.; Kanthasamy, A. Gut microbiome in health and disease: Linking the microbiome-gut-brain axis and environmental factors in the pathogenesis of systemic and neurodegenerative diseases. Pharmacol. Ther. 2016, 158, 52–62. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Zhang, S.; Yang, F.; Xin, R.; Wang, S.; Cui, D.; Sun, Y. The gut microbiota confers protection in the CNS against neurodegeneration induced by manganism. Biomed. Pharmacother. 2020, 127, 110150. [Google Scholar] [CrossRef]
- Wang, H.; Yang, F.; Xin, R.; Cui, D.; He, J.; Zhang, S.; Sun, Y. The gut microbiota attenuate neuroinflammation in manganese exposure by inhibiting cerebral NLRP3 inflammasome. Biomed. Pharmacother. 2020, 129, 110449. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Lukiw, W.J. Bacteroidetes Neurotoxins and Inflammatory Neurodegeneration. Mol. Neurobiol. 2018, 55, 9100–9107. [Google Scholar] [CrossRef] [PubMed]
- Batista, C.R.A.; Gomes, G.F.; Candelario-Jalil, E.; Fiebich, B.L.; de Oliveira, A.C.P. Lipopolysaccharide-Induced Neuroinflammation as a Bridge to Understand Neurodegeneration. Int. J. Mol. Sci. 2019, 20, 2293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, P.; Lokuta, K.M.; Turner, D.E.; Liu, B. Synergistic dopaminergic neurotoxicity of manganese and lipopolysaccharide: Differential involvement of microglia and astroglia. J. Neurochem. 2010, 112, 434–443. [Google Scholar] [CrossRef] [Green Version]
- Breton, J.; Massart, S.; Vandamme, P.; De Brandt, E.; Pot, B.; Foligne, B. Ecotoxicology inside the gut: Impact of heavy metals on the mouse microbiome. BMC Pharmacol. Toxicol. 2013, 14, 62. [Google Scholar] [CrossRef] [Green Version]
- Lin, X.; Zhao, J.; Zhang, W.; He, L.; Wang, L.; Chang, D.; Cui, L.; Gao, Y.; Li, B.; Chen, C.; et al. Acute oral methylmercury exposure perturbs the gut microbiome and alters gut-brain axis related metabolites in rats. Ecotoxicol. Environ. Saf. 2020, 190, 110130. [Google Scholar] [CrossRef]
- Xia, Y.; Zhu, J.; Xu, Y.; Zhang, H.; Zou, F.; Meng, X. Effects of ecologically relevant concentrations of cadmium on locomotor activity and microbiota in zebrafish. Chemosphere 2020, 257, 127220. [Google Scholar] [CrossRef] [PubMed]
- Paschall, M.; Seo, Y.A.; Choi, E.K. Low Dietary Manganese Levels Exacerbate Experimental Colitis in Mice. Curr. Dev. Nutr. 2020, 4, 1831. [Google Scholar] [CrossRef]
- Choi, E.K.; Aring, L.; Das, N.K.; Solanki, S.; Inohara, N.; Iwase, S.; Samuelson, L.C.; Shah, Y.M.; Seo, Y.A. Impact of dietary manganese on experimental colitis in mice. FASEB J. 2020, 34, 2929–2943. [Google Scholar] [CrossRef] [Green Version]
- Jiang, W.D.; Tang, R.J.; Liu, Y.; Kuang, S.Y.; Jiang, J.; Wu, P.; Zhao, J.; Zhang, Y.A.; Tang, L.; Tang, W.N.; et al. Manganese deficiency or excess caused the depression of intestinal immunity, induction of inflammation and dysfunction of the intestinal physical barrier, as regulated by NF-kappaB, TOR and Nrf2 signalling, in grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol. 2015, 46, 406–416. [Google Scholar] [CrossRef]
- Ding, Z.; Chen, X.; Kong, Y.; Shao, X.; Zhang, Y.; Ye, J. Dietary manganese requirement and its effects on antioxidant enzyme activities, intestinal morphology and microbiota in oriental river prawn Macrobrachium nipponense (De Haan). Aquaculture 2020, 516, 734622. [Google Scholar] [CrossRef]
- Nakata, T.; Creasey, E.A.; Kadoki, M.; Lin, H.; Selig, M.K.; Yao, J.; Lefkovith, A.; Daly, M.J.; Graham, D.B.; Xavier, R.J. A missense variant in SLC39A8 confers risk for Crohn’s disease by disrupting manganese homeostasis and intestinal barrier integrity. Proc. Natl. Acad. Sci. USA 2020, 117, 28930–28938. [Google Scholar] [CrossRef] [PubMed]
- Collij, V.; Imhann, F.; Vich Vila, A.; Fu, J.; Dijkstra, G.; Festen, E.A.M.; Voskuil, M.D.; Daly, M.J.; Xavier, R.J.; Wijmenga, C.; et al. SLC39A8 missense variant is associated with Crohn’s disease but does not have a major impact on gut microbiome composition in healthy subjects. PLoS ONE 2019, 14, e0211328. [Google Scholar] [CrossRef]
- Fredericks, J.; Senapati, S.; Wannemuehler, M.J. Cytotoxic effects of manganese oxide nanoparticles in combination with microbial components on intestinal epithelial cells. F1000Research 2020, 9, 975. [Google Scholar] [CrossRef]
- Ogunniyi, A.D.; Mahdi, L.K.; Jennings, M.P.; McEwan, A.G.; McDevitt, C.A.; Van der Hoek, M.B.; Bagley, C.J.; Hoffmann, P.; Gould, K.A.; Paton, J.C. Central role of manganese in regulation of stress responses, physiology, and metabolism in Streptococcus pneumoniae. J. Bacteriol. 2010, 192, 4489–4497. [Google Scholar] [CrossRef] [Green Version]
- Colomer-Winter, C.; Flores-Mireles, A.L.; Baker, S.P.; Frank, K.L.; Lynch, A.J.L.; Hultgren, S.J.; Kitten, T.; Lemos, J.A. Manganese acquisition is essential for virulence of Enterococcus faecalis. PLoS Pathog. 2018, 14, e1007102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diaz-Ochoa, V.E.; Lam, D.; Lee, C.S.; Klaus, S.; Behnsen, J.; Liu, J.Z.; Chim, N.; Nuccio, S.P.; Rathi, S.G.; Mastroianni, J.R.; et al. Salmonella Mitigates Oxidative Stress and Thrives in the Inflamed Gut by Evading Calprotectin-Mediated Manganese Sequestration. Cell Host Microbe 2016, 19, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Brophy, M.B.; Nolan, E.M. Manganese and microbial pathogenesis: Sequestration by the Mammalian immune system and utilization by microorganisms. ACS Chem. Biol. 2015, 10, 641–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lisher, J.P.; Giedroc, D.P. Manganese acquisition and homeostasis at the host-pathogen interface. Front. Cell. Infect. Microbiol. 2013, 3, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cassat, J.E.; Skaar, E.P. Metal ion acquisition in Staphylococcus aureus: Overcoming nutritional immunity. Semin. Immunopathol. 2012, 34, 215–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, N.J.; Chiu, I.M. Bacterial Signaling to the Nervous System through Toxins and Metabolites. J. Mol. Biol. 2017, 429, 587–605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Chiara, G.; Marcocci, M.E.; Sgarbanti, R.; Civitelli, L.; Ripoli, C.; Piacentini, R.; Garaci, E.; Grassi, C.; Palamara, A.T. Infectious agents and neurodegeneration. Mol. Neurobiol. 2012, 46, 614–638. [Google Scholar] [CrossRef] [Green Version]
- Duan, H.; Yu, L.; Tian, F.; Zhai, Q.; Fan, L.; Chen, W. Gut microbiota: A target for heavy metal toxicity and a probiotic protective strategy. Sci. Total Environ. 2020, 742, 140429. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tinkov, A.A.; Martins, A.C.; Avila, D.S.; Gritsenko, V.A.; Skalny, A.V.; Santamaria, A.; Lee, E.; Bowman, A.B.; Aschner, M. Gut Microbiota as a Potential Player in Mn-Induced Neurotoxicity. Biomolecules 2021, 11, 1292. https://doi.org/10.3390/biom11091292
Tinkov AA, Martins AC, Avila DS, Gritsenko VA, Skalny AV, Santamaria A, Lee E, Bowman AB, Aschner M. Gut Microbiota as a Potential Player in Mn-Induced Neurotoxicity. Biomolecules. 2021; 11(9):1292. https://doi.org/10.3390/biom11091292
Chicago/Turabian StyleTinkov, Alexey A., Airton C. Martins, Daiana Silva Avila, Victor A. Gritsenko, Anatoly V. Skalny, Abel Santamaria, Eunsook Lee, Aaron B. Bowman, and Michael Aschner. 2021. "Gut Microbiota as a Potential Player in Mn-Induced Neurotoxicity" Biomolecules 11, no. 9: 1292. https://doi.org/10.3390/biom11091292
APA StyleTinkov, A. A., Martins, A. C., Avila, D. S., Gritsenko, V. A., Skalny, A. V., Santamaria, A., Lee, E., Bowman, A. B., & Aschner, M. (2021). Gut Microbiota as a Potential Player in Mn-Induced Neurotoxicity. Biomolecules, 11(9), 1292. https://doi.org/10.3390/biom11091292








