The Effect of Acute Intense Exercise on Activity of Antioxidant Enzymes in Smokers and Non-Smokers
Abstract
1. Introduction
2. Materials and Methods
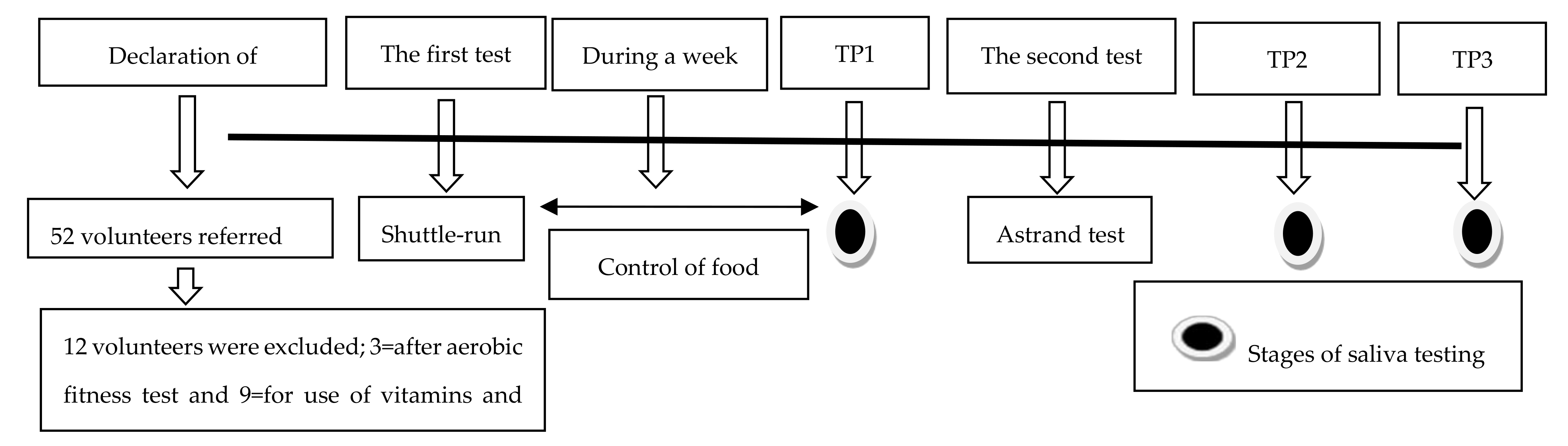
2.1. Participants
2.2. Experimental Approach to the Problem
2.3. Food Control
2.4. Procedures
2.4.1. Acute Intense Exercise
2.4.2. Monitoring Internal Load
2.4.3. Chemical Resources
2.4.4. Saliva Sampling
2.4.5. POX Activity Measurement
2.4.6. CAT Activity Measurement
2.4.7. SOD Activity Measurement
2.5. Statistical Analysis
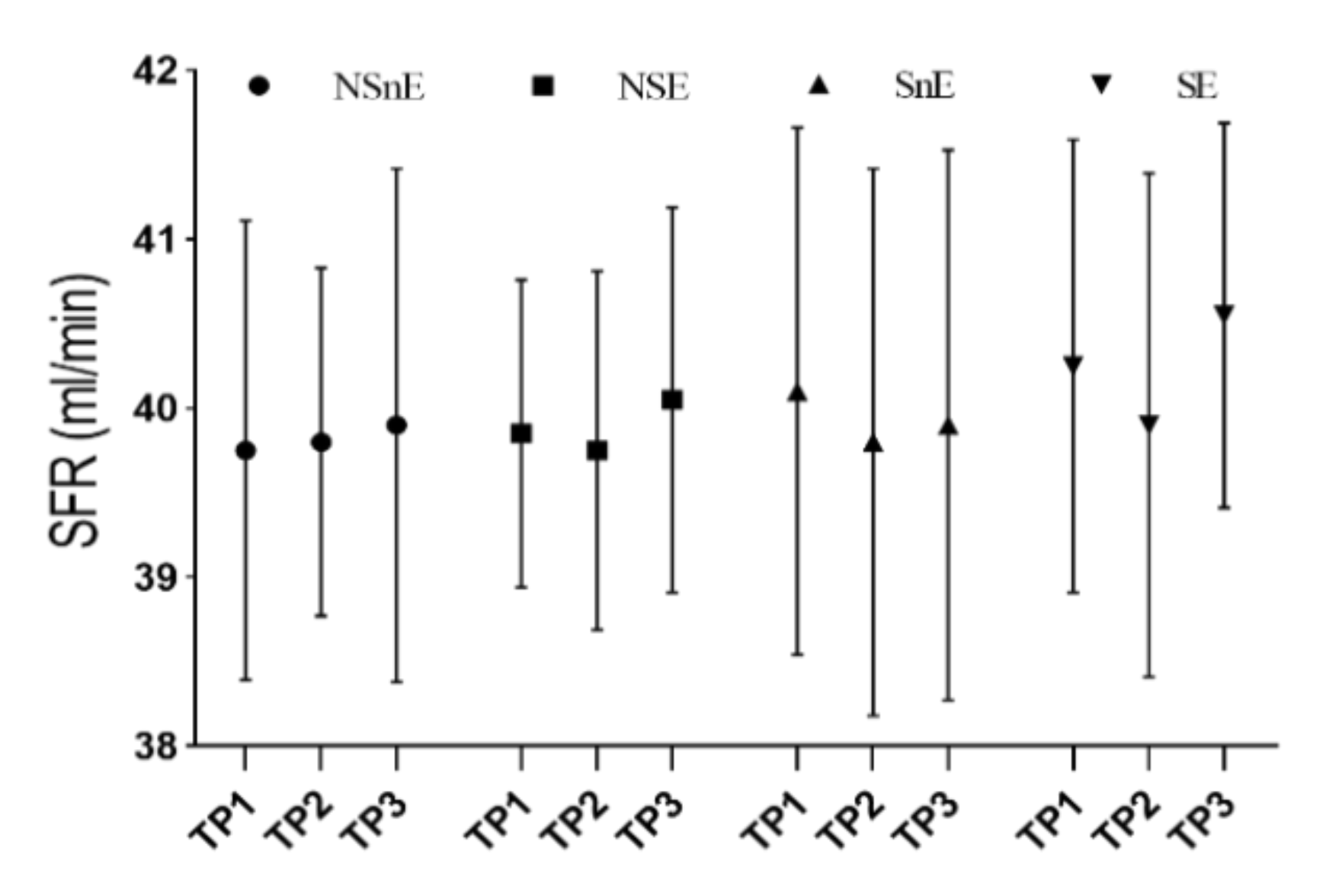
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jha, P.; Jacob, B.; Gajalakshmi, V.; Gupta, P.C.; Dhingra, N.; Kumar, R.; Sinha, D.N.; Dikshit, R.P.; Parida, D.K.; Kamadod, R. A nationally representative case—control study of smoking and death in India. N. Engl. J. Med. 2008, 358, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Ratiu, I.A.; Ligor, T.; Bocos-Bintintan, V.; Mayhew, C.A.; Buszewski, B. Volatile Organic Compounds in Exhaled Breath as Fingerprints of Lung Cancer, Asthma and COPD. J. Clin. Med. 2021, 10, 32. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.; Perfetti, T.; Garg, R.; Hansch, C. IARC carcinogens reported in cigarette mainstream smoke and their calculated log P values. Food Chem. Toxicol. 2003, 41, 807–817. [Google Scholar] [CrossRef]
- WHO Report on the Global Tobacco Epidemic 2019: Offer Help to Quit Tobacco Use; WHO: Geneva, Switzerland, 2019.
- Brosky, M.E. The role of saliva in oral health: Strategies for prevention and management of xerostomia. J. Supportive Oncol. 2007, 5, 215–225. [Google Scholar]
- Genestra, M. Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell. Signal. 2007, 19, 1807–1819. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; Forte, T.M.; Ames, B.N.; Cross, C.E. Gas phase oxidants of cigarette smoke induce lipid peroxidation and changes in lipoprotein properties in human blood plasma. Protective effects of ascorbic acid. Biochem. J. 1991, 277, 133–138. [Google Scholar] [CrossRef]
- Zahraei, M.; Goodarzvand, K.; Sadeghpour, H.; Kiani, A. Effects of Cigarette Smoking on Erythrocyte Antioxidative Enzyme Activities and Plasma Concentrations of Their Cofactors. Acta Med. Iran. 2005, 43, 253–258. [Google Scholar]
- Suzuki, K. Chronic inflammation as an immunological abnormality and effectiveness of exercise. Biomolecules 2019, 9, 223. [Google Scholar] [CrossRef]
- Suzuki, K.; Tominaga, T.; Ruhee, R.T.; Ma, S. Characterization and modulation of systemic inflammatory response to exhaustive exercise in relation to oxidative stress. Antioxidants 2020, 9, 401. [Google Scholar] [CrossRef]
- Goldfarb, A.H. Antioxidants: Role of supplementation to prevent exercise-induced oxidative stress. Med. Sci. Sports Exerc. 1993, 25, 232–236. [Google Scholar] [CrossRef]
- Michiels, C.; Raes, M.; Toussaint, O.; Remacle, J. Importance of Se-glutathione peroxidase, catalase, and Cu/Zn-SOD for cell survival against oxidative stress. Free Radic. Biol. Med. 1994, 17, 235–248. [Google Scholar] [CrossRef]
- Nalcakan, G.R.; Varol, S.R.; Turgay, F.; Nalcakan, M.; Ozkol, M.Z.; Karamizrak, S.O. Effects of aerobic training on serum paraoxonase activity and its relationship with PON1-192 phenotypes in women. J. Sport Health Sci. 2016, 5, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Koubaa, A.; Triki, M.; Trabelsi, H.; Baati, H.; Sahnoun, Z.; Hakim, A. The effect of a 12-week moderate intensity interval training program on the antioxidant defense capability and lipid profile in men smoking cigarettes or hookah: A cohort study. Sci. World J. 2015, 2015, 639369. [Google Scholar] [CrossRef] [PubMed]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef] [PubMed]
- Pingitore, A.; Lima, G.P.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, A.A.; Paschalis, V.; Kyparos, A.; Panayiotou, G.; Nikolaidis, M.G. Passive smoking reduces and vitamin C increases exercise-induced oxidative stress: Does this make passive smoking an anti-oxidant and vitamin C a pro-oxidant stimulus? Biochem. Biophys. Res. Commun. 2014, 454, 131–136. [Google Scholar] [CrossRef]
- Milanowski, M.; Monedeiro, F.; Złoch, M.; Ratiu, I.-A.; Pomastowski, P.; Ligor, T.; De Martinis, B.S.; Buszewski, B. Profiling of VOCs released from different salivary bacteria treated with non-lethal concentrations of silver nitrate. Anal. Biochem. 2019, 578, 36–44. [Google Scholar] [CrossRef]
- Arazi, H.; Taati, B.; Rafati Sajedi, F.; Suzuki, K. Salivary Antioxidants Status Following Progressive Aerobic Exercise: What Are the Differences between Waterpipe Smokers and Non-Smokers? Antioxidants 2019, 8, 418. [Google Scholar] [CrossRef]
- Nobari, H.; Aquino, R.; Clemente, F.M.; Khalafi, M.; Adsuar, J.C.; Pérez-Gómez, J. Description of acute and chronic load, training monotony and strain over a season and its relationships with well-being status: A study in elite under-16 soccer players. Physiol. Behav. 2020, 113117. [Google Scholar] [CrossRef]
- Jackson, A.S.; Pollock, M.L. Generalized equations for predicting body density of men. Br. J. Nutr. 1978, 40, 497–504. [Google Scholar] [CrossRef]
- Arazi, H.; Mirzaei, B.; Nobari, H. Anthropometric profile, body composition and somatotyping of national Iranian cross-country runners. Turk. J. Sport Exerc. 2015, 17, 35–41. [Google Scholar] [CrossRef]
- Rahmat, A.J.; Arsalan, D.; Bahman, M.; Hadi, N. Anthropometrical profile and bio-motor abilities of young elite wrestlers. Phys. Educ. Stud. 2016, 20, 63–69. [Google Scholar] [CrossRef]
- Leger, L.A.; Mercier, D.; Gadoury, C.; Lambert, J. The multistage 20 metre shuttle run test for aerobic fitness. J. Sports Sci. 1988, 6, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Nobari, H.; Ahmadi, M.; Pérez-Gómez, J.; Clemente, M.; Adsuar, J.; Minasian, V.; Afonso, J. The effect of two types of combined training on bio-motor ability adaptations in sedentary females. J. Sports Med. Phys. Fit. 2020. [Google Scholar] [CrossRef]
- Stang, J.; Story, M.; Feldman, S. Nutrition in adolescent pregnancy. Int. J. Childbirth Educ. 2005, 20, 4. [Google Scholar]
- Astrand, P.-O. Experimental Studies of Physical Work Capacity in Relation to Sex and Age. Ph.D. Thesis, Kungliga Gymnastiska Centralinstitutet, Stockholm, Sweden, 1952. [Google Scholar]
- Kang, J.; Chaloupka, E.C.; Mastrangelo, M.A.; Biren, G.B.; Robertson, R.J. Physiological comparisons among three maximal treadmill exercise protocols in trained and untrained individuals. Eur. J. Appl. Physiol. 2001, 84, 291–295. [Google Scholar] [CrossRef]
- Foster, C.; Florhaug, J.A.; Franklin, J.; Gottschall, L.; Hrovatin, L.A.; Parker, S.; Doleshal, P.; Dodge, C. A new approach to monitoring exercise training. J. Strength Cond. Res. 2001, 15, 109–115. [Google Scholar]
- Nobari, H.; Tubagi Polito, L.F.; Clemente, F.M.; Pérez-Gómez, J.; Ahmadi, M.; Garcia-Gordillo, M.Á.; Silva, A.F.; Adsuar, J.C. Relationships Between Training Workload Parameters with Variations in Anaerobic Power and Change of Direction Status in Elite Youth Soccer Players. Int. J. Environ. Res. Public Health 2020, 17, 7934. [Google Scholar] [CrossRef]
- Damirchi, A.; Kiani, M.; Jafarian, V.; Sariri, R. Response of salivary peroxidase to exercise intensity. Eur. J. Appl. Physiol. 2010, 108, 1233–1237. [Google Scholar] [CrossRef]
- Urbanska, A. Location and variability of catalase activity within aphids. Electron. J. Pol. Agric. Univ. Ser. Biol. 2007, 10, 4. [Google Scholar]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Ighodaro, O.; Akinloye, O. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Farhadi, H.; Siakuhian, M.; Dolatkhah, H.; Rahimifardin, S.; Salemi, S.N.P. Effect of short-term garlic supplementation on DNA damage after exhaustive exercise in non-athlete men. Eur. J. Exp. Biol. 2013, 3, 455–459. [Google Scholar]
- Souza, A.V.; Giolo, J.S.; Teixeira, R.R.; Vilela, D.D.; Peixoto, L.G.; Justino, A.B.; Caixeta, D.C.; Puga, G.M.; Espindola, F.S. Salivary and Plasmatic Antioxidant Profile following Continuous, Resistance, and High-Intensity Interval Exercise: Preliminary Study. Oxidative Med. Cell. Longev. 2019, 2019, 5425021. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Nakaji, S.; Yamada, M.; Totsuka, M.; Sato, K.; Sugawara, K. Systemic inflammatory response to exhaustive exercise. Cytokine kinetics. Exerc. Immunol. Rev. 2002, 8, 6. [Google Scholar] [PubMed]
- Anbarasi, K.; Vani, G.; Balakrishna, K.; Devi, C.S. Effect of bacoside A on brain antioxidant status in cigarette smoke exposed rats. Life Sci. 2006, 78, 1378–1384. [Google Scholar] [CrossRef]
- Klein, I.; Nagler, R.M.; Toffler, R.; van Der Vliet, A.; Reznick, A.Z. Effect of cigarette smoke on oral peroxidase activity in human saliva: Role of hydrogen cyanide. Free Radic. Biol. Med. 2003, 35, 1448–1452. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Cavas, L.; Arpinar, P.; Yurdakoc, K. Possible interactions between antioxidant enzymes and free sialic acids in saliva: A preliminary study on elite judoists. Int. J. Sports Med. 2005, 26, 832–835. [Google Scholar] [CrossRef]
- Suzuki, K.; Nakaji, S.; Yamada, M.; Liu, Q.; Kurakake, S.; Okamura, N.; Kumae, T.; Umeda, T.; Sugawara, K. Impact of a competitive marathon race on systemic cytokine and neutrophil responses. Med. Sci. Sports Exerc. 2003, 35, 348. [Google Scholar] [CrossRef]
- Bogdanis, G.; Stavrinou, P.; Fatouros, I.; Philippou, A.; Chatzinikolaou, A.; Draganidis, D.; Ermidis, G.; Maridaki, M. Short-term high-intensity interval exercise training attenuates oxidative stress responses and improves antioxidant status in healthy humans. Food Chem. Toxicol. 2013, 61, 171–177. [Google Scholar] [CrossRef] [PubMed]
- González, D.; Marquina, R.; Rondón, N.; Rodríguez-Malaver, A.J.; Reyes, R. Effects of aerobic exercise on uric acid, total antioxidant activity, oxidative stress, and nitric oxide in human saliva. Res. Sports Med. 2008, 16, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.; Maughan, R.; Duthie, G.; Morrice, P. Increased blood antioxidant systems of runners in response to training load. Clin. Sci. 1991, 80, 611–618. [Google Scholar] [CrossRef]
- Bogdanska, J.; Korneti, P.; Todorova, B. Erythrocyte superoxide dismutase, glutathione peroxidase and catalase activities in healthy male subjects in Republic of Macedonia. Bratisl. Lek. Listy 2003, 104, 108–114. [Google Scholar] [PubMed]
- Duarte, T.L.; Lunec, J. When is an antioxidant not an antioxidant? A review of novel actions and reactions of vitamin C. Free Radic. Res. 2005, 39, 671–686. [Google Scholar] [CrossRef] [PubMed]
- Goldfarb, A.H.; Bloomer, R.J.; Mckenzie, M.J. Combined antioxidant treatment effects on blood oxidative stress after eccentric exercise. Med. Sci. Sports Exerc. 2005, 37, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Suriyaprom, K.; Sirikulchayanonta, C.; Tungtrongchitr, R.; Namjuntra, P.; Phraephan, S.; Sornwatana, T. The Relationship between Characteristics of Cigarette Smoking and Cytokine/Antioxidant Status in Thai Males. J. Med. Assoc. Thail. 2020, 103, 38–45. [Google Scholar]
- Tokmakidis, S.P.; Volaklis, K.A. Training and detraining effects of a combined-strength and aerobic exercise program on blood lipids in patients with coronary artery disease. J. Cardiopulm. Rehabil. Prev. 2003, 23, 193–200. [Google Scholar] [CrossRef]
- Di Mascio, P.; Murphy, M.E.; Sies, H. Antioxidant defense systems: The role of carotenoids, tocopherols, and thiols. Am. J. Clin. Nutr. 1991, 53, 194S–200S. [Google Scholar] [CrossRef]
- Salminen, A.; VIHKO, V. Endurance training reduces the susceptibility of mouse skeletal muscle to lipid peroxidation in vitro. Acta Physiol. Scand. 1983, 117, 109–113. [Google Scholar] [CrossRef]


| Groups | Age (Years) | Height (m) | Weight (kg) | BMI (kg/m2) | Body Fat % | VO2max |
|---|---|---|---|---|---|---|
| NSnE | 21.7 ± 1.9 | 1.78 ± 0.06 | 75.4 ± 4.45 | 23.84 ± 1.35 | 15.73 ± 1.28 | 42.1 ± 3.7 |
| NSE | 22.5 ± 1.6 | 1.77 ± 0.04 | 70.8 ± 3.90 | 22.41 ± 0.49 | 12.08 ± 0.88 | 55.9 ± 4.0 |
| SnE | 23.9 ± 1.5 | 1.76 ± 0.03 | 72.3 ± 6.58 | 23.25 ± 2.26 | 14.94 ± 2.38 | 40.8 ± 4.2 |
| SE | 23.6 ± 1.2 | 1.78 ± 0.02 | 69.9 ± 2.47 | 22.07 ± 0.88 | 12.71 ± 0.85 | 54.9 ± 4.2 |
| Groups | NSnE | NSE | SnE | SE | MFT |
|---|---|---|---|---|---|
| Variable | M ± SD | M ± SD | M ± SD | M ± SD | p |
| Calorie Intake (kcals/day) | 2667.80 ± 156.09 | 2733.27 ± 122.55 | 2657.23 ± 144.64 | 2616.07 ± 70.4 | 0.15 |
| Carbohydrate (g) | 377.88 ± 23.06 | 387 ± 17.47 | 377.97 ± 19.19 | 371.12 ± 10.71 | 0.16 |
| Protein (g) | 94.55 ± 5.61 | 96.25 ± 4.81 | 93.73 ± 6 | 92.48 ± 3.05 | 0.37 |
| Fat (g) | 86.45 ± 4.99 | 88.48 ± 4.10 | 86.29 ± 5.06 | 84.63 ± 2.31 | 0.26 |
| Vitamin C (mg) | 73.04 ± 2.7 | 71.86 ± 3.6 | 72.08 ± 4.10 | 72.23 ± 3.11 | 0.87 |
| Vitamin E (mg) | 16.07 ± 1.38 | 16.05 ± 1.25 | 15.57 ± 1.49 | 16.04 ± 1.13 | 0.35 |
| Enzymes | Groups | Mean Difference | 95% CI for Difference | Bonferroni | ||
|---|---|---|---|---|---|---|
| Comparison | Lower | Upper | p | |||
| TP1 | OX (U/mL) | NSnE vs NSE | −0.007 | −0.018 | 0.0031 | 0.341 |
| NSnE > SnE | 0.016 # | 0.005 | 0.026 | 0.001 * | ||
| NSnE > SE | 0.012 # | 0.002 | 0.022 | 0.015 * | ||
| NSE > SnE | 0.023 # | 0.012 | 0.033 | <0.001 * | ||
| NSE > SE | 0.019 # | 0.009 | 0.030 | <0.001 * | ||
| SnE vs SE | −0.003 | −0.014 | 0.007 | 1 | ||
| TP2 | POX (U/mL) | NSnE < NSE | −0.031 # | −0.043 | −0.018 | <0.001 * |
| NSnE > SnE | 0.019 # | 0.007 | 0.032 | 0.001 * | ||
| NSnE > SE | 0.011 | −0.001 | 0.024 | 0.106 | ||
| NSE > SnE | 0.050 # | 0.037 | 0.063 | <0.001 * | ||
| NSE > SE | 0.042 # | 0.029 | 0.055 | <0.001 * | ||
| SnE vs SE | −0.008 | −0.021 | 0.005 | 0.536 | ||
| TP3 | POX (U/mL) | NSnE < NSE | −0.018 # | −0.030 | −0.006 | 0.001 * |
| NSnE > SnE | 0.013 # | 0.001 | 0.025 | 0.026 * | ||
| NSnE > SE | 0.009 | −0.003 | 0.021 | 0.253 | ||
| NSE > SnE | 0.031 # | 0.019 | 0.043 | <0.001 * | ||
| NSE > SE | 0.027 # | 0.015 | 0.039 | <0.001 * | ||
| SnE vs SE | −0.004 | −0.016 | 0.008 | 1 | ||
| TP1 | CAT (U/mL) | NSnE vs NSE | −0.001 | −0.004 | 0.002 | 1 |
| NSnE > SnE | 0.003 # | 0.00 | 0.007 | 0.019 * | ||
| NSnE > SE | 0.004 # | 0.001 | 0.007 | 0.007 * | ||
| NSE > SnE | 0.004 # | 0.001 | 0.007 | 0.003 * | ||
| NSE > SE | 0.005 # | 0.002 | 0.008 | 0.001 * | ||
| SnE vs SE | < 0.001 * | −0.003 | 0.003 | 1 | ||
| TP2 | CAT (U/mL) | NSnE < NSE | −0.009 # | −0.015 | −0.003 | 0.001 * |
| NSnE vs SnE | −0.001 | −0.008 | 0.005 | 1 | ||
| NSnE < SE | < 0.001 * | −0.006 | 0.006 | 1 | ||
| NSE > SnE | 0.008 # | 0.002 | 0.014 | 0.005 * | ||
| NSE > SE | 0.009 # | 0.003 | 0.015 | 0.001 * | ||
| SnE < SE | 0.001 | −0.005 | 0.008 | 1 | ||
| TP3 | CAT (U/mL) | NSnE vs NSE | −0.004 | −0.009 | 0.00 | 0.056 |
| NSnE vs SnE | 0.002 | −0.002 | 0.007 | 0.907 | ||
| NSnE vs SE | 0.00 | 0.004 | 0.005 | 1 | ||
| NSE > SnE | 0.007 # | 0.002 | 0.011 | 0.001 * | ||
| NSE > SE | 0.005 # | 0.00 | 0.009 | 0.041 * | ||
| SnE vs SE | −0.002 | −0.007 | 0.002 | 1 | ||
| TP1 | SOD (U/mL) | NSnE vs NSE | −0.030 | −0.096 | 0.037 | 1 |
| NSnE > SnE | 0.129 # | 0.062 | 0.196 | <0.001 * | ||
| NSnE > SE | 0.098 # | 0.032 | 0.165 | 0.001 * | ||
| NSE > SnE | 0.158 # | 0.091 | 0.225 | <0.001 * | ||
| NSE > SE | 0.128 # | 0.061 | 0.195 | <0.001 * | ||
| SnE vs SE | −0.030 | −0.097 | 0.037 | 1 | ||
| TP2 | SOD (U/mL) | NSnE < NSE | −0.109 # | −0.190 | −0.029 | 0.003 * |
| NSnE > SnE | 0.139 # | 0.059 | 0.019 | <0.001 * | ||
| NSnE vs SE | 0.036 | −0.044 | 0.016 | 1 | ||
| NSE > SnE | 0.248 # | 0.168 | 0.329 | <0.001 * | ||
| NSE > SE | 0.145 # | 0.065 | 0.226 | <0.001 * | ||
| SnE < SE | −0.103 # | −0.183 | −0.023 | 0.006 * | ||
| TP3 | SOD (U/mL) | NSnE < NSE | −0.084 # | −0.154 | −0.014 | 0.011 * |
| NSnE > SnE | 0.132 # | 0.062 | 0.202 | <0.001 * | ||
| NSnE > SE | 0.093 # | 0.023 | 0.163 | 0.004 * | ||
| NSE > SnE | 0.216 # | 0.146 | 0.286 | <0.001 * | ||
| NSE > SE | 0.177 # | 0.107 | 0.247 | <0.001 * | ||
| SnE vs SE | −0.039 | −0.109 | 0.031 | 0.776 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nobari, H.; Nejad, H.A.; Kargarfard, M.; Mohseni, S.; Suzuki, K.; Carmelo Adsuar, J.; Pérez-Gómez, J. The Effect of Acute Intense Exercise on Activity of Antioxidant Enzymes in Smokers and Non-Smokers. Biomolecules 2021, 11, 171. https://doi.org/10.3390/biom11020171
Nobari H, Nejad HA, Kargarfard M, Mohseni S, Suzuki K, Carmelo Adsuar J, Pérez-Gómez J. The Effect of Acute Intense Exercise on Activity of Antioxidant Enzymes in Smokers and Non-Smokers. Biomolecules. 2021; 11(2):171. https://doi.org/10.3390/biom11020171
Chicago/Turabian StyleNobari, Hadi, Hamzeh Abdi Nejad, Mehdi Kargarfard, Soghra Mohseni, Katsuhiko Suzuki, José Carmelo Adsuar, and Jorge Pérez-Gómez. 2021. "The Effect of Acute Intense Exercise on Activity of Antioxidant Enzymes in Smokers and Non-Smokers" Biomolecules 11, no. 2: 171. https://doi.org/10.3390/biom11020171
APA StyleNobari, H., Nejad, H. A., Kargarfard, M., Mohseni, S., Suzuki, K., Carmelo Adsuar, J., & Pérez-Gómez, J. (2021). The Effect of Acute Intense Exercise on Activity of Antioxidant Enzymes in Smokers and Non-Smokers. Biomolecules, 11(2), 171. https://doi.org/10.3390/biom11020171









