Hyaluronic Acid: A Key Ingredient in the Therapy of Inflammation
Abstract
:1. Introduction
1.1. Biosynthesis
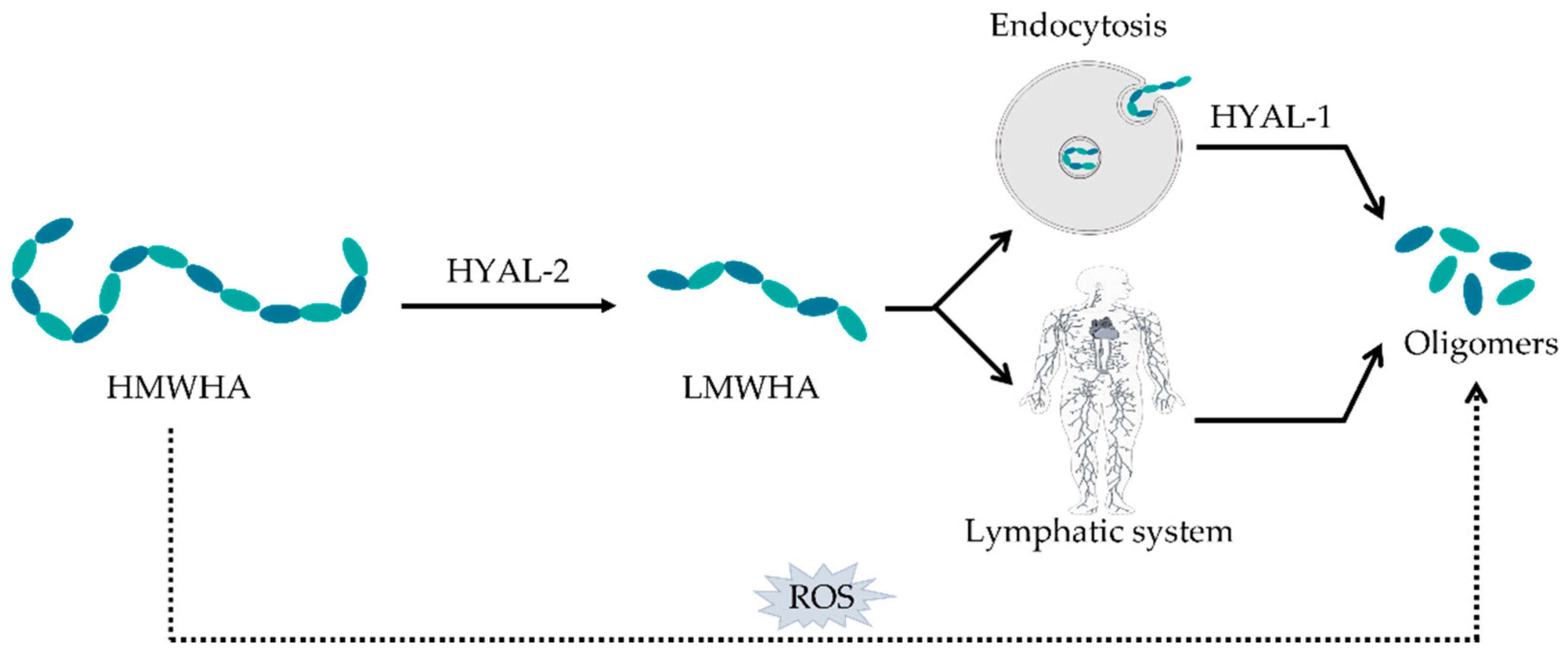
1.2. Degradation
1.3. Hydration
2. Biological Functions
3. Mechanism of Action
4. Interaction with ECM Molecules and Cell Surface Receptors
4.1. CD44
4.2. RHAMM
4.3. ICAM-1
4.4. Other Receptors
5. HA Potential as Therapeutic or Coadjutant Agent
5.1. Inflammatory Joint Diseases
5.1.1. Osteoarthritis
5.1.2. Rheumatoid Arthritis
5.1.3. Other Disorders
5.2. Tendinopathy
5.3. Intervertebal Disc injury
5.4. Inflammatory Skin Diseases
Atopic Dermatitis
5.5. Wound Healing
5.6. Inflammatory Bowel Diseases
5.7. Lung Inflammation
5.8. Inflammatory Diseases of the Oral Cavity
5.9. Bladder Inflammation
5.10. Renal Inflammation
5.11. Cardiac Inflammation
6. Conclusions and Future Outlooks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fallacara, A.; Baldini, E.; Manfredini, S.; Vertuani, S. Hyaluronic Acid in the Third Millennium. Polymers 2018, 10, 701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Litwiniuk, M.; Krejner, A.; Speyrer, M.S.; Gauto, A.R.; Grzela, T. Hyaluronic Acid in Inflammation and Tissue Regeneration. Wounds A Compend. Clin. Res. Pract. 2016, 28, 78–88. [Google Scholar]
- Bukhari, S.N.A.; Roswandi, N.L.; Waqas, M.; Habib, H.; Hussain, F.; Khan, S.; Sohail, M.; Ramli, N.A.; Thu, H.E.; Hussain, Z. Hyaluronic acid, a promising skin rejuvenating biomedicine: A review of recent updates and pre-clinical and clinical investigations on cosmetic and nutricosmetic effects. Int. J. Biol. Macromol. 2018, 120, 1682–1695. [Google Scholar] [CrossRef] [PubMed]
- Marcotti, S.; Maki, K.; Reilly, G.C.; Lacroix, D.; Adachi, T. Hyaluronic acid selective anchoring to the cytoskeleton: An atomic force microscopy study. PLoS ONE 2018, 13, e0206056. [Google Scholar] [CrossRef]
- Matarasso, S.L. Understanding and Using Hyaluronic Acid. Aesthetic Surg. J. 2004, 24, 361–364. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinology 2012, 4, 253–258. [Google Scholar] [CrossRef] [Green Version]
- Petrey, A.C.; de la Motte, C.A. Hyaluronan, a crucial regulator of inflammation. Front Immunol. 2014, 5, 101. [Google Scholar] [CrossRef] [Green Version]
- Jiang, D.; Liang, J.; Noble, P.W. Hyaluronan as an Immune Regulator in Human Diseases. Physiol. Rev. 2011, 91, 221–264. [Google Scholar] [CrossRef] [Green Version]
- Nikitovic, D.; Kouvidi, K.; Karamanos, N.K.; Tzanakakis, G.N. The Roles of Hyaluronan/RHAMM/CD44 and Their Respective Interactions along the Insidious Pathways of Fibrosarcoma Progression. BioMed Res. Int. 2013, 2013, 929531. [Google Scholar] [CrossRef]
- Stern, R.; Kogan, G.; Jedrzejas, M.J.; Šoltés, L. The many ways to cleave hyaluronan. Biotechnol. Adv. 2007, 25, 537–557. [Google Scholar] [CrossRef]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front. Vet. Sci. 2019, 6, 192. [Google Scholar] [CrossRef] [Green Version]
- Liang, J.; Jiang, D.; Noble, P.W. Hyaluronan as a therapeutic target in human diseases. Adv. Drug Deliv. Rev. 2016, 97, 186–203. [Google Scholar] [CrossRef] [Green Version]
- Gallo, N.; Nasser, H.; Salvatore, L.; Natali, M.L.; Campa, L.; Mahmoud, M.; Capobianco, L.; Sannino, A.; Madaghiele, M. Hyaluronic acid for advanced therapies: Promises and challenges. Eur. Polym. J. 2019, 117, 134–147. [Google Scholar] [CrossRef]
- Johnson, P.; Ruffell, B. CD44 and its role in inflammation and inflammatory diseases. Inflamm. Allergy Drug Targets 2009, 8, 208–220. [Google Scholar] [CrossRef]
- Salwowska, N.M.; Bebenek, K.A.; Żądło, D.A.; Wcisło-Dziadecka, D.L. Physiochemical properties and application of hyaluronic acid: A systematic review. J. Cosmet. Dermatol. 2016, 15, 520–526. [Google Scholar] [CrossRef]
- Liao, Y.H.; Jones, S.A.; Forbes, B.; Martin, G.P.; Brown, M.B. Hyaluronan: Pharmaceutical characterization and drug delivery. Drug Deliv. 2005, 12, 327–342. [Google Scholar] [CrossRef]
- Reitinger, S.; Lepperdinger, G. Hyaluronan, a Ready Choice to Fuel Regeneration: A Mini-Review. Gerontology 2013, 59, 71–76. [Google Scholar] [CrossRef]
- Cyphert, J.M.; Trempus, C.S.; Garantziotis, S. Size Matters: Molecular Weight Specificity of Hyaluronan Effects in Cell Biology. Int. J. Cell Biol. 2015, 2015, 563818. [Google Scholar] [CrossRef] [Green Version]
- Aya, K.L.; Stern, R. Hyaluronan in wound healing: Rediscovering a major player. Wound Repair Regen. 2014, 22, 579–593. [Google Scholar] [CrossRef]
- Adamia, S.; Maxwell, C.A.; Pilarski, L.M. Hyaluronan and hyaluronan synthases: Potential therapeutic targets in cancer. Curr. Drug Targets Cardiovasc. Haematol. Disord. 2005, 5, 3–14. [Google Scholar] [CrossRef] [Green Version]
- Sapudom, J.; Müller, C.D.; Nguyen, K.-T.; Martin, S.; Anderegg, U.; Pompe, T. Matrix Remodeling and Hyaluronan Production by Myofibroblasts and Cancer-Associated Fibroblasts in 3D Collagen Matrices. Gels 2020, 6, 33. [Google Scholar] [CrossRef]
- Bastow, E.R.; Byers, S.; Golub, S.B.; Clarkin, C.E.; Pitsillides, A.A.; Fosang, A.J. Hyaluronan synthesis and degradation in cartilage and bone. Cell Mol. Life Sci. 2008, 65, 395–413. [Google Scholar] [CrossRef]
- Bowman, S.; Awad, M.E.; Hamrick, M.W.; Hunter, M.; Fulzele, S. Recent advances in hyaluronic acid based therapy for osteoarthritis. Clin. Transl. Med. 2018, 7, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altman, R.; Bedi, A.; Manjoo, A.; Niazi, F.; Shaw, P.; Mease, P. Anti-Inflammatory Effects of Intra-Articular Hyaluronic Acid: A Systematic Review. Cartilage 2019, 10, 43–52. [Google Scholar] [CrossRef]
- Maytin, E.V. Hyaluronan: More than just a wrinkle filler. Glycobiology 2016, 26, 553–559. [Google Scholar] [CrossRef] [Green Version]
- Karbownik, M.S.; Nowak, J.Z. Hyaluronan: Towards novel anti-cancer therapeutics. Pharmacol. Rep. 2013, 65, 1056–1074. [Google Scholar] [CrossRef]
- Lee-Sayer, S.S.M.; Dong, Y.; Arif, A.A.; Olsson, M.; Brown, K.L.; Johnson, P. The Where, When, How, and Why of Hyaluronan Binding by Immune Cells. Front Immunol. 2015, 6, 150. [Google Scholar] [CrossRef] [Green Version]
- Al-Khateeb, R.; Prpic, J. Hyaluronic acid: The reason for its variety of physiological and biochemical functional properties. Appl. Clin. Res. Clin. Trials Regul. Aff. 2019, 6, 112–159. [Google Scholar] [CrossRef]
- Triggs-Raine, B.; Natowicz, M.R. Biology of hyaluronan: Insights from genetic disorders of hyaluronan metabolism. World J. Biol. Chem. 2015, 6, 110–120. [Google Scholar] [CrossRef]
- Fakhari, A.; Berkland, C. Applications and emerging trends of hyaluronic acid in tissue engineering, as a dermal filler and in osteoarthritis treatment. Acta Biomater. 2013, 9, 7081–7092. [Google Scholar] [CrossRef] [Green Version]
- Oe, M.; Tashiro, T.; Yoshida, H.; Nishiyama, H.; Masuda, Y.; Maruyama, K.; Koikeda, T.; Maruya, R.; Fukui, N. Oral hyaluronan relieves knee pain: A review. Nutr. J. 2016, 15, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasvani, S.; Kulkarni, P.; Rawtani, D. Hyaluronic acid: A review on its biology, aspects of drug delivery, route of administrations and a special emphasis on its approved marketed products and recent clinical studies. Int. J. Biol. Macromol. 2020, 151, 1012–1029. [Google Scholar] [CrossRef] [PubMed]
- Heldin, P.; Basu, K.; Olofsson, B.; Porsch, H.; Kozlova, I.; Kahata, K. Deregulation of hyaluronan synthesis, degradation and binding promotes breast cancer. J. Biochem. 2013, 154, 395–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monslow, J.; Govindaraju, P.; Puré, E. Hyaluronan—A Functional and Structural Sweet Spot in the Tissue Microenvironment. Front. Immunol. 2015, 6, 231. [Google Scholar] [CrossRef] [Green Version]
- Romagnoli, M.; Belmontesi, M. Hyaluronic acid-based fillers: Theory and practice. Clin. Dermatol. 2008, 26, 123–159. [Google Scholar] [CrossRef]
- Johnson, P.; Arif, A.A.; Lee-Sayer, S.S.M.; Dong, Y. Hyaluronan and Its Interactions with Immune Cells in the Healthy and Inflamed Lung. Front. Immunol. 2018, 9, 2787. [Google Scholar] [CrossRef]
- Dahiya, P.; Kamal, R. Hyaluronic acid: A boon in periodontal therapy. N. Am. J. Med. Sci. 2013, 5, 309–315. [Google Scholar] [CrossRef]
- Wolf, K.J.; Kumar, S. Hyaluronic Acid: Incorporating the Bio into the Material. ACS Biomater. Sci. Eng. 2019, 5, 3753–3765. [Google Scholar] [CrossRef]
- Tamer, T.M. Hyaluronan and synovial joint: Function, distribution and healing. Interdiscip. Toxicol. 2013, 6, 111–125. [Google Scholar] [CrossRef]
- Sze, J.H.; Brownlie, J.C.; Love, C.A. Biotechnological production of hyaluronic acid: A mini review. 3 Biotech 2016, 6, 67. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.H.; Xue, J.F.; Zheng, Z.Y.; Shuhaidi, M.; Thu, H.E.; Hussain, Z. Hyaluronic acid, an efficient biomacromolecule for treatment of inflammatory skin and joint diseases: A review of recent developments and critical appraisal of preclinical and clinical investigations. Int. J. Biol. Macromol. 2018, 116, 572–584. [Google Scholar] [CrossRef]
- Kreger, S.T.; Voytik-Harbin, S.L. Hyaluronan concentration within a 3D collagen matrix modulates matrix viscoelasticity, but not fibroblast response. Matrix Biol. 2009, 28, 336–346. [Google Scholar] [CrossRef] [Green Version]
- Sapudom, J.; Nguyen, K.-T.; Martin, S.; Wippold, T.; Möller, S.; Schnabelrauch, M.; Anderegg, U.; Pompe, T. Biomimetic tissue models reveal the role of hyaluronan in melanoma proliferation and invasion. Biomater. Sci. 2020, 8, 1405–1417. [Google Scholar] [CrossRef] [Green Version]
- Unnikandam Veettil, S.R.; Hwang, D.; Correia, J.; Bartlett, M.D.; Schneider, I.C. Cancer cell migration in collagen-hyaluronan composite extracellular matrices. Acta Biomater. 2021, 130, 183–198. [Google Scholar] [CrossRef]
- Shuborna, N.S.; Chaiyasamut, T.; Sakdajeyont, W.; Vorakulpipat, C.; Rojvanakarn, M.; Wongsirichat, N. Generation of novel hyaluronic acid biomaterials for study of pain in third molar intervention: A review. J. Dent. Anesth. Pain Med. 2019, 19, 11–19. [Google Scholar] [CrossRef]
- Sherman, L.S.; Matsumoto, S.; Su, W.; Srivastava, T.; Back, S.A. Hyaluronan Synthesis, Catabolism, and Signaling in Neurodegenerative Diseases. Int. J. Cell Biol. 2015, 2015, 368584. [Google Scholar] [CrossRef] [Green Version]
- Pichert, A.; Schlorke, D.; Franz, S.; Arnhold, J. Functional aspects of the interaction between interleukin-8 and sulfated glycosaminoglycans. Biomatter 2012, 2, 142–148. [Google Scholar] [CrossRef] [Green Version]
- Nordsieck, K.; Baumann, L.; Hintze, V.; Pisabarro, M.T.; Schnabelrauch, M.; Beck-Sickinger, A.; Samsonov, S. The effect of interleukin-8 truncations on its interactions with glycosaminoglycans. Biopolymers 2018, 109, e23103. [Google Scholar] [CrossRef]
- Day, A.J.; Sheehan, J.K. Hyaluronan: Polysaccharide chaos to protein organisation. Curr. Opin. Struct. Biol. 2001, 11, 617–622. [Google Scholar] [CrossRef]
- Stern, R.; Maibach, H.I. Hyaluronan in skin: Aspects of aging and its pharmacologic modulation. Clin. Dermatol. 2008, 26, 106–122. [Google Scholar] [CrossRef]
- Passi, A.; Vigetti, D. Hyaluronan as tunable drug delivery system. Adv. Drug Deliv. Rev. 2019, 146, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Garantziotis, S.; Savani, R.C. Hyaluronan biology: A complex balancing act of structure, function, location and context. Matrix Biol. 2019, 78–79, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhai, P.; Peng, X.; Li, B.; Liu, Y.; Sun, H.; Li, X. The application of hyaluronic acid in bone regeneration. Int. J. Biol. Macromol. 2020, 151, 1224–1239. [Google Scholar] [CrossRef] [PubMed]
- Stenson, W.F. Hyaluronic acid and intestinal inflammation. Curr. Opin. Gastroenterol. 2010, 26, 85–87. [Google Scholar] [CrossRef]
- Day, A.J.; Prestwich, G.D. Hyaluronan-binding Proteins: Tying Up the Giant. J. Biol. Chem. 2002, 277, 4585–4588. [Google Scholar] [CrossRef] [Green Version]
- Dicker, K.T.; Gurski, L.A.; Pradhan-Bhatt, S.; Witt, R.L.; Farach-Carson, M.C.; Jia, X. Hyaluronan: A simple polysaccharide with diverse biological functions. Acta Biomater. 2014, 10, 1558–1570. [Google Scholar] [CrossRef] [Green Version]
- Sapudom, J.; Ullm, F.; Martin, S.; Kalbitzer, L.; Naab, J.; Möller, S.; Schnabelrauch, M.; Anderegg, U.; Schmidt, S.; Pompe, T. Molecular weight specific impact of soluble and immobilized hyaluronan on CD44 expressing melanoma cells in 3D collagen matrices. Acta Biomater. 2017, 50, 259–270. [Google Scholar] [CrossRef] [Green Version]
- Kouvidi, K.; Berdiaki, A.; Nikitovic, D.; Katonis, P.; Afratis, N.; Hascall, V.C.; Karamanos, N.K.; Tzanakakis, G.N. Role of receptor for hyaluronic acid-mediated motility (RHAMM) in low molecular weight hyaluronan (LMWHA)-mediated fibrosarcoma cell adhesion. J. Biol. Chem. 2011, 286, 38509–38520. [Google Scholar] [CrossRef] [Green Version]
- Nikitovic, D.; Kouvidi, K.; Kavasi, R.M.; Berdiaki, A.; Tzanakakis, G.N. Hyaluronan/Hyaladherins—A Promising Axis for Targeted Drug Delivery in Cancer. Curr. Drug. Deliv. 2016, 13, 500–511. [Google Scholar] [CrossRef]
- Necas, J.; Bartosikova, L.; Brauner, P.; Kolář, J. Hyaluronic acid (Hyaluronan): A review. Vet. Med. 2008, 53, 397–411. [Google Scholar] [CrossRef] [Green Version]
- Weindl, G.; Schaller, M.; Schäfer-Korting, M.; Korting, H.C. Hyaluronic acid in the treatment and prevention of skin diseases: Molecular biological, pharmaceutical and clinical aspects. Ski. Pharmacol. Physiol. 2004, 17, 207–213. [Google Scholar] [CrossRef]
- Jiang, D.; Liang, J.; Noble, P.W. Hyaluronan in tissue injury and repair. Annu. Rev. Cell Dev. Biol. 2007, 23, 435–461. [Google Scholar] [CrossRef] [Green Version]
- Nagy, N.; Kuipers, H.F.; Marshall, P.L.; Wang, E.; Kaber, G.; Bollyky, P.L. Hyaluronan in immune dysregulation and autoimmune diseases. Matrix Biol. 2019, 78–79, 292–313. [Google Scholar] [CrossRef]
- Ioannou, S.; Voulgarelis, M. Toll-Like Receptors, Tissue Injury, and Tumourigenesis. Mediat. Inflamm. 2010, 2010, 581837. [Google Scholar] [CrossRef] [Green Version]
- Suthahar, N.; Meijers, W.C.; Silljé, H.H.W.; de Boer, R.A. From Inflammation to Fibrosis—Molecular and Cellular Mechanisms of Myocardial Tissue Remodelling and Perspectives on Differential Treatment Opportunities. Curr. Heart Fail. Rep. 2017, 14, 235–250. [Google Scholar] [CrossRef] [Green Version]
- Yu, C.; Li, X.; Hou, Y.; Meng, X.; Wang, D.; Liu, J.; Sun, F.; Li, Y. Hyaluronic Acid Coated Acid-Sensitive Nanoparticles for Targeted Therapy of Adjuvant-Induced Arthritis in Rats. Molecules 2019, 24, 146. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Shin, M.; Han, S.; Kwon, W.; Hahn, S.K. Hyaluronic Acid Derivatives for Translational Medicines. Biomacromolecules 2019, 20, 2889–2903. [Google Scholar] [CrossRef]
- Mero, A.; Campisi, M. Hyaluronic Acid Bioconjuates for the Delivery of Bioactive Molecules Polymers. Wounds 2014, 6, 346–369. [Google Scholar]
- Tiwari, S.; Bahadur, P. Modified hyaluronic acid based materials for biomedical applications. Int. J. Biol. Macromol. 2019, 121, 556–571. [Google Scholar] [CrossRef]
- Huang, G.; Huang, H. Hyaluronic acid-based biopharmaceutical delivery and tumor-targeted drug delivery system. J. Control Release 2018, 278, 122–126. [Google Scholar] [CrossRef]
- Huang, G.; Huang, H. Application of hyaluronic acid as carriers in drug delivery. Drug Deliv. 2018, 25, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Choi, H.; Choi, E.S.; Park, M.-H.; Ryu, J.-H. Hyaluronic Acid-Coated Nanomedicine for Targeted Cancer Therapy. Pharmaceutics 2019, 11, 301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masuko, K.; Murata, M.; Yudoh, K.; Kato, T.; Nakamura, H. Anti-inflammatory effects of hyaluronan in arthritis therapy: Not just for viscosity. Int. J. Gen. Med. 2009, 2, 77–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kogan, G.; Soltés, L.; Stern, R.; Gemeiner, P. Hyaluronic acid: A natural biopolymer with a broad range of biomedical and industrial applications. Biotechnol. Lett. 2007, 29, 17–25. [Google Scholar] [CrossRef]
- De Souza, A.B.; Chaud, M.V.; Santana, M.H.A. Hyaluronic acid behavior in oral administration and perspectives for nanotechnology-based formulations: A review. Carbohydr. Polym. 2019, 222, 115001. [Google Scholar] [CrossRef]
- Bauer, C.; Niculescu-Morzsa, E.; Jeyakumar, V.; Kern, D.; Späth, S.S.; Nehrer, S. Chondroprotective effect of high-molecular-weight hyaluronic acid on osteoarthritic chondrocytes in a co-cultivation inflammation model with M1 macrophages. J. Inflamm. 2016, 13, 31. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Shen, J.; Zhao, W.; Wang, T.; Han, L.; Hamilton, J.L.; Im, H.-J. Osteoarthritis: Toward a comprehensive understanding of pathological mechanism. Bone Res. 2017, 5, 16044. [Google Scholar] [CrossRef]
- De Lucia, O.; Murgo, A.; Pregnolato, F.; Pontikaki, I.; De Souza, M.; Sinelli, A.; Cimaz, R.; Caporali, R. Hyaluronic Acid Injections in the Treatment of Osteoarthritis Secondary to Primary Inflammatory Rheumatic Diseases: A Systematic Review and Qualitative Synthesis. Adv. Ther. 2020, 37, 1347–1359. [Google Scholar] [CrossRef] [Green Version]
- Ren, C.D.; Gao, S.; Kurisawa, M.; Ying, J.Y. Cartilage synthesis in hyaluronic acid–tyramine constructs. J. Mater. Chem. B 2015, 3, 1942–1956. [Google Scholar] [CrossRef]
- Dong, J.; Jiang, D.; Wang, Z.; Wu, G.; Miao, L.; Huang, L. Intra-articular delivery of liposomal celecoxib–hyaluronate combination for the treatment of osteoarthritis in rabbit model. Int. J. Pharm. 2013, 441, 285–290. [Google Scholar] [CrossRef]
- Kang, M.L.; Jeong, S.Y.; Im, G.I. Hyaluronic Acid Hydrogel Functionalized with Self-Assembled Micelles of Amphiphilic PEGylated Kartogenin for the Treatment of Osteoarthritis. Tissue Eng. Part A 2017, 23, 630–639. [Google Scholar] [CrossRef]
- Kim, S.E.; Lee, J.Y.; Shim, K.-S.; Lee, S.; Min, K.; Bae, J.-H.; Kim, H.-J.; Park, K.; Song, H.-R. Attenuation of inflammation and cartilage degradation by sulfasalazine-containing hyaluronic acid on osteoarthritis rat model. Int. J. Biol. Macromol. 2018, 114, 341–348. [Google Scholar] [CrossRef]
- Mohammadi, F.; Tanideh, N.; Mohammadi Samani, S.; Ahmadi, F. Efficacy of a hybrid system of hyaluronic acid and collagen loaded with prednisolone and TGF-β3 for cartilage regeneration in rats. J. Drug Deliv. Sci. Technol. 2019, 51, 55–62. [Google Scholar] [CrossRef]
- Homma, A.; Sato, H.; Okamachi, A.; Emura, T.; Ishizawa, T.; Kato, T.; Matsuura, T.; Sato, S.; Tamura, T.; Higuchi, Y.; et al. Novel hyaluronic acid-methotrexate conjugates for osteoarthritis treatment. Bioorg. Med. Chem. 2009, 17, 4647–4656. [Google Scholar] [CrossRef]
- Tamura, T.; Higuchi, Y.; Kitamura, H.; Murao, N.; Saitoh, R.; Morikawa, T.; Sato, H. Novel hyaluronic acid–methotrexate conjugate suppresses joint inflammation in the rat knee: Efficacy and safety evaluation in two rat arthritis models. Arthritis Res. Ther. 2016, 18, 79. [Google Scholar] [CrossRef] [Green Version]
- Siracusa, R.; Impellizzeri, D.; Cordaro, M.; Peritore, A.; Gugliandolo, E.; D’Amico, R.; Fusco, R.; Crupi, R.; Rizzarelli, E.; Cuzzocrea, S.; et al. Protective Effect of New Formulation of Carnosine+Hyaluronic Acid on the Inflammation and Cartilage Degradation in the Experimental Model of Osteoarthritis. Appl. Sci. 2020, 10, 1324. [Google Scholar] [CrossRef] [Green Version]
- Zhou, P.; Qiu, B.; Deng, R.; Li, H.; Xu, X.; Shang, X. Chondroprotective Effects of Hyaluronic Acid-Chitosan Nanoparticles Containing Plasmid DNA Encoding Cytokine Response Modifier A in a Rat Knee Osteoarthritis Model. Cell. Physiol. Biochem. 2018, 47, 1207–1216. [Google Scholar] [CrossRef]
- Zerrillo, L.; Que, I.; Vepris, O.; Morgado, L.N.; Chan, A.; Bierau, K.; Li, Y.; Galli, F.; Bos, E.; Censi, R.; et al. pH-responsive poly(lactide-co-glycolide) nanoparticles containing near-infrared dye for visualization and hyaluronic acid for treatment of osteoarthritis. J. Control Release 2019, 309, 265–276. [Google Scholar] [CrossRef]
- Mota, A.H.; Direito, R.; Carrasco, M.P.; Rijo, P.; Ascensão, L.; Viana, A.S.; Rocha, J.; Eduardo-Figueira, M.; Rodrigues, M.J.; Custódio, L.; et al. Combination of hyaluronic acid and PLGA particles as hybrid systems for viscosupplementation in osteoarthritis. Int. J. Pharm. 2019, 559, 13–22. [Google Scholar] [CrossRef]
- Tolba, Y.M.; Omar, S.S.; Nagui, D.A.; Nawwar, M.A. Effect of high molecular weight hyaluronic acid in treatment of osteoarthritic temporomandibular joints of rats. Arch. Oral Biol. 2020, 110, 104618. [Google Scholar] [CrossRef]
- Faust, H.J.; Sommerfeld, S.D.; Rathod, S.; Rittenbach, A.; Ray Banerjee, S.; Tsui, B.M.W.; Pomper, M.; Amzel, M.L.; Singh, A.; Elisseeff, J.H. A hyaluronic acid binding peptide-polymer system for treating osteoarthritis. Biomaterials 2018, 183, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Koh, R.H.; Kim, S.-H.; Kim, K.M.; Park, G.K.; Hwang, N.S. Injectable anti-inflammatory hyaluronic acid hydrogel for osteoarthritic cartilage repair. Mater. Sci. Eng. C 2020, 115, 111096. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Yang, J.; Liang, J.; Xu, X.; Cui, W.; Deng, L.; Zhang, H. Bioinspired Hyaluronic Acid/Phosphorylcholine Polymer with Enhanced Lubrication and Anti-Inflammation. Biomacromolecules 2019, 20, 4135–4142. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, Y.; Shou, X.; Ni, D.; Kong, T.; Zhao, Y. Bio-inspired lubricant drug delivery particles for the treatment of osteoarthritis. Nanoscale 2020, 12, 17093–17102. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; He, Z.; Shu, L.; Li, X.; Ma, M.; Ye, C. Intra-Articular Platelet-Rich Plasma Combined with Hyaluronic Acid Injection for Knee Osteoarthritis Is Superior to Platelet-Rich Plasma or Hyaluronic Acid Alone in Inhibiting Inflammation and Improving Pain and Function. Arthrosc. J. Arthrosc. Relat. Surg. 2021, 37, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.C.; Chiang, P.F.; Kuo, Y.J.; Peng, C.L.; Chen, K.Y.; Chiang, Y.C. Hyaluronan-Loaded Liposomal Dexamethasone-Diclofenac Nanoparticles for Local Osteoarthritis Treatment. Int. J. Mol. Sci. 2021, 22, 665. [Google Scholar] [CrossRef]
- El-Gogary, R.I.; Khattab, M.A.; Abd-Allah, H. Intra-articular multifunctional celecoxib loaded hyaluronan nanocapsules for the suppression of inflammation in an osteoarthritic rat model. Int. J. Pharm. 2020, 583, 119378. [Google Scholar] [CrossRef]
- Cai, Z.; Hong, M.; Xu, L.; Yang, K.; Li, C.; Sun, T.; Feng, Y.; Zeng, H.; Lu, W.W.; Chiu, K.-Y. Prevent action of magnoflorine with hyaluronic acid gel from cartilage degeneration in anterior cruciate ligament transection induced osteoarthritis. Biomed. Pharmacother. 2020, 126, 109733. [Google Scholar] [CrossRef]
- Mou, D.; Yu, Q.; Zhang, J.; Zhou, J.; Li, X.; Zhuang, W.; Yang, X. Intra-articular Injection of Chitosan-Based Supramolecular Hydrogel for Osteoarthritis Treatment. Tissue Eng. Regen. Med. 2021, 18, 113–125. [Google Scholar] [CrossRef]
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038. [Google Scholar] [CrossRef]
- Zhou, M.; Hou, J.; Zhong, Z.; Hao, N.; Lin, Y.; Li, C. Targeted delivery of hyaluronic acid-coated solid lipid nanoparticles for rheumatoid arthritis therapy. Drug Deliv. 2018, 25, 716–722. [Google Scholar] [CrossRef]
- Gouveia, V.M.; Lopes-de-Araújo, J.; Lima, S.A.C.; Nunes, C.; Reis, S. Hyaluronic acid-conjugated pH-sensitive liposomes for targeted delivery of prednisolone on rheumatoid arthritis therapy. Nanomedicine 2018, 13, 1037–1049. [Google Scholar] [CrossRef]
- Heo, R.; Park, J.S.; Jang, H.J.; Kim, S.H.; Shin, J.M.; Suh, Y.D.; Jeong, J.H.; Jo, D.G.; Park, J.H. Hyaluronan nanoparticles bearing γ-secretase inhibitor: In vivo therapeutic effects on rheumatoid arthritis. J. Control Release 2014, 192, 295–300. [Google Scholar] [CrossRef]
- Alam, M.M.; Han, H.S.; Sung, S.; Kang, J.H.; Sa, K.H.; Al Faruque, H.; Hong, J.; Nam, E.J.; Kim, I.S.; Park, J.H.; et al. Endogenous inspired biomineral-installed hyaluronan nanoparticles as pH-responsive carrier of methotrexate for rheumatoid arthritis. J. Control. Release 2017, 252, 62–72. [Google Scholar] [CrossRef]
- Fan, Z.; Li, J.; Liu, J.; Jiao, H.; Liu, B. Anti-Inflammation and Joint Lubrication Dual Effects of a Novel Hyaluronic Acid/Curcumin Nanomicelle Improve the Efficacy of Rheumatoid Arthritis Therapy. ACS Appl. Mater. Interfaces 2018, 10, 23595–23604. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Siracusa, R.; Cordaro, M.; Peritore, A.F.; Gugliandolo, E.; D’amico, R.; Fusco, R.; Crupi, R.; Rizzarelli, E.; Cuzzocrea, S.; et al. Protective effect of a new hyaluronic acid -carnosine conjugate on the modulation of the inflammatory response in mice subjected to collagen-induced arthritis. Biomed. Pharmacother. 2020, 125, 110023. [Google Scholar] [CrossRef]
- Schmitt, F.; Lagopoulos, L.; Käuper, P.; Rossi, N.; Busso, N.; Barge, J.; Wagnières, G.; Laue, C.; Wandrey, C.; Juillerat-Jeanneret, L. Chitosan-based nanogels for selective delivery of photosensitizers to macrophages and improved retention in and therapy of articular joints. J. Control Release 2010, 144, 242–250. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.M.; Kim, S.-H.; Thambi, T.; You, D.G.; Jeon, J.; Lee, J.O.; Chung, B.Y.; Jo, D.-G.; Park, J.H. A hyaluronic acid–methotrexate conjugate for targeted therapy of rheumatoid arthritis. Chem. Commun. 2014, 50, 7632–7635. [Google Scholar] [CrossRef]
- Joshi, N.; Yan, J.; Levy, S.; Bhagchandani, S.; Slaughter, K.V.; Sherman, N.E.; Amirault, J.; Wang, Y.; Riegel, L.; He, X.; et al. Towards an arthritis flare-responsive drug delivery system. Nat. Commun. 2018, 9, 1275. [Google Scholar] [CrossRef]
- Pandey, S.; Rai, N.; Mahtab, A.; Mittal, D.; Ahmad, F.J.; Sandal, N.; Neupane, Y.R.; Verma, A.K.; Talegaonkar, S. Hyaluronate-functionalized hydroxyapatite nanoparticles laden with methotrexate and teriflunomide for the treatment of rheumatoid arthritis. Int. J. Biol. Macromol. 2021, 171, 502–513. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, J.; Yang, Y.; Pu, X.; Zhao, J.; Zhang, N. Targeted and Combined TPCA-1-Gold Nanocage Therapy for In Vivo Treatment of Inflammatory Arthritis. AAPS PharmSciTech 2020, 21, 298. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Liu, Z.; Song, Y.; Hu, C. Hyaluronic acid-functionalized bilosomes for targeted delivery of tripterine to inflamed area with enhancive therapy on arthritis. Drug Deliv. 2019, 26, 820–830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Storozhylova, N.; Crecente-Campo, J.; Cabaleiro, D.; Lugo, L.; Dussouy, C.; Simões, S.; Monteiro, M.; Grandjean, C.; Alonso, M.J. An In Situ Hyaluronic Acid-Fibrin Hydrogel Containing Drug-Loaded Nanocapsules for Intra-Articular Treatment of Inflammatory Joint Diseases. Regen. Eng. Transl. Med. 2020, 6, 201–216. [Google Scholar] [CrossRef]
- Ragab, G.; Elshahaly, M.; Bardin, T. Gout: An old disease in new perspective—A review. J. Adv. Res. 2017, 8, 495–511. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, D.; Wang, X.; Bai, R.; Wang, C.; Gao, Y.; Anastassiades, T. N-Butyrylated hyaluronic acid ameliorates gout and hyperuricemia in animal models. Pharm. Biol. 2019, 57, 717–728. [Google Scholar] [CrossRef] [Green Version]
- Bernetti, A.; Francesco, A.; Alviti, F.; Mangone, M.; Santilli, V.; Marco, P. Hyaluronic Acid Injections in the Management of Tendinopathies. A Descriptive Review. EC Orthop. 2018, 9, 225–232. [Google Scholar]
- Loiacono, C.; Palermi, S.; Massa, B.; Belviso, I.; Romano, V.; Gregorio, A.D.; Sirico, F.; Sacco, A.M. Tendinopathy: Pathophysiology, Therapeutic Options, and Role of Nutraceutics. A Narrative Literature Review. Medicina 2019, 55, 447. [Google Scholar] [CrossRef] [Green Version]
- Lipman, K.; Wang, C.; Ting, K.; Soo, C.; Zheng, Z. Tendinopathy: Injury, repair, and current exploration. Drug Des. Dev. Ther. 2018, 12, 591–603. [Google Scholar] [CrossRef] [Green Version]
- Abate, M.; Schiavone, C.; Salini, V. The Use of Hyaluronic Acid after Tendon Surgery and in Tendinopathies. BioMed Res. Int. 2014, 2014, 783632. [Google Scholar] [CrossRef] [Green Version]
- Fogli, M.; Giordan, N.; Mazzoni, G. Efficacy and safety of hyaluronic acid (500–730 kDa) Ultrasound-guided injections on painful tendinopathies: A prospective, open label, clinical study. Muscles Ligaments Tendons J. 2017, 7, 388–395. [Google Scholar] [CrossRef]
- Chen, C.-T.; Chen, C.-H.; Sheu, C.; Chen, J.-P. Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation. Int. J. Mol. Sci. 2019, 20, 5038. [Google Scholar] [CrossRef] [Green Version]
- Rea, W.; Kapur, S.; Mutagi, H. Intervertebral disc as a source of pain. Contin. Educ. Anaesth. Crit. Care Pain 2012, 12, 279–282. [Google Scholar] [CrossRef]
- Nedresky, D.; Reddy, V.; Singh, G. Anatomy, Back, Nucleus Pulposus; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Mohd Isa, I.L.; Abbah, S.A.; Kilcoyne, M.; Sakai, D.; Dockery, P.; Finn, D.P.; Pandit, A. Implantation of hyaluronic acid hydrogel prevents the pain phenotype in a rat model of intervertebral disc injury. Sci. Adv. 2018, 4, eaaq0597. [Google Scholar] [CrossRef] [Green Version]
- Isa, I.L.M.; Srivastava, A.; Tiernan, D.; Owens, P.; Rooney, P.; Dockery, P.; Pandit, A. Hyaluronic Acid Based Hydrogels Attenuate Inflammatory Receptors and Neurotrophins in Interleukin-1β Induced Inflammation Model of Nucleus Pulposus Cells. Biomacromolecules 2015, 16, 1714–1725. [Google Scholar] [CrossRef]
- Kazezian, Z.; Sakai, D.; Pandit, A. Hyaluronic Acid Microgels Modulate Inflammation and Key Matrix Molecules toward a Regenerative Signature in the Injured Annulus Fibrosus. Adv. Biosyst. 2017, 1, 1700077. [Google Scholar] [CrossRef]
- Neuman, M.G.; Nanau, R.M.; Oruña, L.; Coto, G. In vitro anti-inflammatory effects of hyaluronic acid in ethanol-induced damage in skin cells. J. Pharm. Pharm. Sci. 2011, 14, 425–437. [Google Scholar] [CrossRef] [Green Version]
- Pleguezuelos-Villa, M.; Nácher, A.; Hernández, M.J.; Ofelia Vila Buso, M.A.; Ruiz Sauri, A.; Díez-Sales, O. Mangiferin nanoemulsions in treatment of inflammatory disorders and skin regeneration. Int. J. Pharm. 2019, 564, 299–307. [Google Scholar] [CrossRef]
- Williamson, S.; Merritt, J.; De Benedetto, A. Atopic dermatitis in the elderly. Br. J. Dermatol. 2020, 182, e21. [Google Scholar] [CrossRef] [Green Version]
- Sayaseng, K.Y.; Vernon, P. Pathophysiology and Management of Mild to Moderate Pediatric Atopic Dermatitis. J. Pediatr. Health Care 2018, 32, S2–S12. [Google Scholar] [CrossRef] [Green Version]
- Zhuo, F.; Abourehab, M.A.S.; Hussain, Z. Hyaluronic acid decorated tacrolimus-loaded nanoparticles: Efficient approach to maximize dermal targeting and anti-dermatitis efficacy. Carbohydr. Polym. 2018, 197, 478–489. [Google Scholar] [CrossRef]
- Ghatak, S.; Maytin, E.V.; Mack, J.A.; Hascall, V.C.; Atanelishvili, I.; Moreno Rodriguez, R.; Markwald, R.R.; Misra, S. Roles of Proteoglycans and Glycosaminoglycans in Wound Healing and Fibrosis. Int. J. Cell Biol. 2015, 2015, 834893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iacopetti, I.; Perazzi, A.; Martinello, T.; Gemignani, F.; Patruno, M. Hyaluronic acid, Manuka honey and Acemannan gel: Wound-specific applications for skin lesions. Res. Vet. Sci. 2020, 129, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.C.d.O.; Costa, T.F.; Andrade, Z.d.A.; Medrado, A.R.A.P. Wound healing—A literature review. Anais Brasileiros de Dermatologia 2016, 91, 614–620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, R.; Myers, S.; Leigh, I.; Navsaria, H. The Role of Hyaluronic Acid in Wound Healing. Am. J. Clin. Dermatol. 2005, 6, 393–402. [Google Scholar] [CrossRef]
- Gao, Y.; Sun, Y.; Yang, H.; Qiu, P.; Cong, Z.; Zou, Y.; Song, L.; Guo, J.; Anastassiades, T.P. A Low Molecular Weight Hyaluronic Acid Derivative Accelerates Excisional Wound Healing by Modulating Pro-Inflammation, Promoting Epithelialization and Neovascularization, and Remodeling Collagen. Int. J. Mol. Sci. 2019, 20, 3722. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Li, A.; Feng, F.; Jiang, Q.; Sun, H.; Chai, Y.; Yang, R.; Wang, Z.; Hou, J.; Li, R. Effect of the hyaluronic acid-poloxamer hydrogel on skin-wound healing: In vitro and in vivo studies. Anim. Models Exp. Med. 2019, 2, 107–113. [Google Scholar] [CrossRef]
- Zhao, W.; Li, Y.; Zhang, X.; Zhang, R.; Hu, Y.; Boyer, C.; Xu, F.-J. Photo-responsive supramolecular hyaluronic acid hydrogels for accelerated wound healing. J. Control Release 2020, 323, 24–35. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, Q.; Yu, J.; Shao, N.; Lu, H.; Guo, J.; Qiu, X.; Zhou, D.; Huang, Y. Absorbable Thioether Grafted Hyaluronic Acid Nanofibrous Hydrogel for Synergistic Modulation of Inflammation Microenvironment to Accelerate Chronic Diabetic Wound Healing. Adv. Healthc. Mater. 2020, 9, 2000198. [Google Scholar] [CrossRef]
- Chiu, C.-T.; Kuo, S.-N.; Hung, S.-W.; Yang, C.-Y. Combined Treatment with Hyaluronic Acid and Mesalamine Protects Rats from Inflammatory Bowel Disease Induced by Intracolonic Administration of Trinitrobenzenesulfonic Acid. Molecules 2017, 22, 904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vafaei, S.Y.; Esmaeili, M.; Amini, M.; Atyabi, F.; Ostad, S.N.; Dinarvand, R. Self assembled hyaluronic acid nanoparticles as a potential carrier for targeting the inflamed intestinal mucosa. Carbohydr. Polym. 2016, 144, 371–381. [Google Scholar] [CrossRef]
- Fakhoury, M.; Negrulj, R.; Mooranian, A.; Al-Salami, H. Inflammatory bowel disease: Clinical aspects and treatments. J. Inflamm. Res. 2014, 7, 113–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seyedian, S.S.; Nokhostin, F.; Malamir, M.D. A review of the diagnosis, prevention, and treatment methods of inflammatory bowel disease. J. Med. Life 2019, 12, 113–122. [Google Scholar] [PubMed]
- Hendrickson, B.A.; Gokhale, R.; Cho, J.H. Clinical aspects and pathophysiology of inflammatory bowel disease. Clin. Microbiol. Rev. 2002, 15, 79–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, B.; Xu, Z.; Viennois, E.; Zhang, Y.; Zhang, Z.; Zhang, M.; Han, M.K.; Kang, Y.; Merlin, D. Orally Targeted Delivery of Tripeptide KPV via Hyaluronic Acid-Functionalized Nanoparticles Efficiently Alleviates Ulcerative Colitis. Mol. Ther. 2017, 25, 1628–1640. [Google Scholar] [CrossRef] [Green Version]
- Sammarco, G.; Shalaby, M.; Elangovan, S.; Petti, L.; Roda, G.; Restelli, S.; Arena, V.; Ungaro, F.; Fiorino, G.; Day, A.J.; et al. Hyaluronan Accelerates Intestinal Mucosal Healing through Interaction with TSG-6. Cells 2019, 8, 1074. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Mahaseth, M.; Zhang, Y. Hyaluronic acid as a rescue therapy for trinitrobenzene sulfonic acid-induced colitis through Cox-2 and PGE2 in a Toll-like receptor 4-dependent way. J. Zhejiang Univ. Sci. B 2011, 12, 712–719. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.; Sugihara, K.; Gillilland, M.G.; Jon, S.; Kamada, N.; Moon, J.J. Hyaluronic acid–bilirubin nanomedicine for targeted modulation of dysregulated intestinal barrier, microbiome and immune responses in colitis. Nat. Mater. 2020, 19, 118–126. [Google Scholar] [CrossRef]
- Fallacara, A.; Busato, L.; Pozzoli, M.; Ghadiri, M.; Ong, H.X.; Young, P.M.; Manfredini, S.; Traini, D. Combination of urea-crosslinked hyaluronic acid and sodium ascorbyl phosphate for the treatment of inflammatory lung diseases: An in vitro study. Eur. J. Pharm. Sci. 2018, 120, 96–106. [Google Scholar] [CrossRef]
- Moldoveanu, B.; Otmishi, P.; Jani, P.; Walker, J.; Sarmiento, X.; Guardiola, J.; Saad, M.; Yu, J. Inflammatory mechanisms in the lung. J. Inflamm. Res. 2009, 2, 1–11. [Google Scholar]
- Boehme, J.D.; Stegemann-Koniszewski, S.; Autengruber, A.; Peters, N.; Wissing, J.; Jänsch, L.; Jeron, A.; Bruder, D. Chronic lung inflammation primes humoral immunity and augments antipneumococcal resistance. Sci. Rep. 2017, 7, 4972. [Google Scholar] [CrossRef]
- Pignataro, L.; Marchisio, P.; Ibba, T.; Torretta, S. Topically administered hyaluronic acid in the upper airway: A narrative review. Int. J. Immunopathol. Pharmacol. 2018, 32, 2058738418766739. [Google Scholar] [CrossRef]
- Courey, M.S.; Pletcher, S.D. 49—Upper Airway Disorders. In Murray and Nadel’s Textbook of Respiratory Medicine, 6th ed.; Broaddus, V.C., Mason, R.J., Ernst, J.D., King, T.E., Lazarus, S.C., Murray, J.F., Nadel, J.A., Slutsky, A.S., Gotway, M.B., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2016; pp. 877–896.e875. [Google Scholar]
- Hasturk, H.; Kantarci, A.; Van Dyke, T.E. Oral inflammatory diseases and systemic inflammation: Role of the macrophage. Front Immunol. 2012, 3, 118. [Google Scholar] [CrossRef] [Green Version]
- Casale, M.; Moffa, A.; Vella, P.; Sabatino, L.; Capuano, F.; Salvinelli, B.; Lopez, M.A.; Carinci, F.; Salvinelli, F. Hyaluronic acid: Perspectives in dentistry. A systematic review. Int. J. Immunopathol. Pharmacol. 2016, 29, 572–582. [Google Scholar] [CrossRef]
- Shaw, L.; Harjunmaa, U.; Doyle, R.; Mulewa, S.; Charlie, D.; Maleta, K.; Callard, R.; Walker, A.S.; Balloux, F.; Ashorn, P.; et al. Distinguishing the Signals of Gingivitis and Periodontitis in Supragingival Plaque: A Cross-Sectional Cohort Study in Malawi. Appl. Environ. Microbiol. 2016, 82, 6057–6067. [Google Scholar] [CrossRef] [Green Version]
- Jain, Y. Clinical evaluation of 0.2% hyaluronic acid containing gel in the treatment of gingivitis. Med. J. Dr. D.Y. Patil. Univ. 2013, 6, 416–420. [Google Scholar] [CrossRef]
- Chen, M.; Li, L.; Wang, Z.; Li, P.; Feng, F.; Zheng, X. High molecular weight hyaluronic acid regulates P. gingivalis–induced inflammation and migration in human gingival fibroblasts via MAPK and NF-κB signaling pathway. Arch. Oral Biol. 2019, 98, 75–80. [Google Scholar] [CrossRef]
- Pilloni, A.; Annibali, S.; Dominici, F.; Di Paolo, C.; Papa, M.; Cassini, M.A.; Polimeni, A. Evaluation of the efficacy of an hyaluronic acid-based biogel on periodontal clinical parameters. A randomized-controlled clinical pilot study. Ann. Stomatol. 2011, 2, 3–9. [Google Scholar]
- Al-Shammari, N.M.; Shafshak, S.M.; Ali, M.S. Effect of 0.8% Hyaluronic Acid in Conventional Treatment of Moderate to Severe Chronic Periodontitis. J. Contemp. Dent. Pract. 2018, 19, 527–534. [Google Scholar] [CrossRef]
- Suchánek, J.; Ivančaková, R.K.; Mottl, R.; Browne, K.Z.; Pilneyová, K.C.; Pilbauerová, N.; Schmidt, J.; Suchánková Kleplová, T. Hyaluronic Acid-Based Medical Device for Treatment of Alveolar Osteitis—Clinical Study. Int. J. Environ. Res. Public Health 2019, 16, 3698. [Google Scholar] [CrossRef] [Green Version]
- Veale, B. Alveolar osteitis: A critical review of the aetiology and management. Oral Surg. 2015, 8, 68–77. [Google Scholar] [CrossRef]
- Chow, O.; Wang, R.; Ku, D.; Huang, W. Alveolar Osteitis: A Review of Current Concepts. J. Oral Maxillofac. Surg. 2020, 78, 1288–1296. [Google Scholar] [CrossRef]
- Kapitán, M.; Schmidt, J.; Mottl, R.; Pilbauerová, N. Initial Observation of Factors Interfering with the Treatment of Alveolar Osteitis Using Hyaluronic Acid with Octenidine—A Series of Case Reports. Biomolecules 2021, 11, 1157. [Google Scholar] [CrossRef]
- Stellavato, A.; Pirozzi, A.V.A.; Diana, P.; Reale, S.; Vassallo, V.; Fusco, A.; Donnarumma, G.; De Rosa, M.; Schiraldi, C. Hyaluronic acid and chondroitin sulfate, alone or in combination, efficiently counteract induced bladder cell damage and inflammation. PLoS ONE 2019, 14, e0218475. [Google Scholar] [CrossRef] [Green Version]
- Cervigni, M.; Natale, F.; Nasta, L.; Mako, A. Intravesical hyaluronic acid and chondroitin sulphate for bladder pain syndrome/interstitial cystitis: Long-term treatment results. Int. Urogynecol. J. 2012, 23, 1187–1192. [Google Scholar] [CrossRef]
- Sahiner, I.F.; Soylu, H.; Ates, E.; Acar, N.; Ustunel, I.; Danisman, A. Impact of intravesical hyaluronic acid treatment on bladder inflammation in interstitial cystitis rat model. Int. Braz. J. Urol. 2018, 44, 1014–1022. [Google Scholar] [CrossRef]
- Yıldız, N.; Alpay, H.; Tuğtepe, H.; Özdemir Kumral, Z.N.; Akakın, D.; İlki, A.; Şener, G.; Ç Yeğen, B. Intravesical hyaluronic acid treatment improves bacterial cystitis and reduces cystitis-induced hypercontractility in rats. Int. J. Urol. 2015, 22, 598–603. [Google Scholar] [CrossRef]
- Rooney, P.R.; Kannala, V.K.; Kotla, N.G.; Benito, A.; Dupin, D.; Loinaz, I.; Quinlan, L.R.; Rochev, Y.; Pandit, A. A high molecular weight hyaluronic acid biphasic dispersion as potential therapeutics for interstitial cystitis. J. Biomed. Mater. Res. B Appl. Biomater. 2021, 109, 864–876. [Google Scholar] [CrossRef]
- Hu, J.-B.; Li, S.-J.; Kang, X.-Q.; Qi, J.; Wu, J.-H.; Wang, X.-J.; Xu, X.-L.; Ying, X.-Y.; Jiang, S.-P.; You, J.; et al. CD44-targeted hyaluronic acid-curcumin prodrug protects renal tubular epithelial cell survival from oxidative stress damage. Carbohydr. Polym. 2018, 193, 268–280. [Google Scholar] [CrossRef]
- Bonafè, F.; Govoni, M.; Giordano, E.; Caldarera, C.M.; Guarnieri, C.; Muscari, C. Hyaluronan and cardiac regeneration. J. Biomed. Sci. 2014, 21, 100. [Google Scholar] [CrossRef] [Green Version]
- Wang, N.; Liu, C.; Wang, X.; He, T.; Li, L.; Liang, X.; Wang, L.; Song, L.; Wei, Y.; Wu, Q.; et al. Hyaluronic Acid Oligosaccharides Improve Myocardial Function Reconstruction and Angiogenesis against Myocardial Infarction by Regulation of Macrophages. Theranostics 2019, 9, 1980–1992. [Google Scholar] [CrossRef]
- Yoon, S.; Fang, Y.-H.; Lim, C.; Kim, B.; Son, H.; Park, Y.; Sun, K. Regeneration of Ischemic Heart Using Hyaluronic Acid-Based Injectable Hydrogel. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 91, 163–171. [Google Scholar] [CrossRef] [PubMed]


| Formulation | Studies | Administration Route | Main Results | Ref. | |
|---|---|---|---|---|---|
| HA-Clx | Liposomes | In vitro and in vivo | IA | Pain control, and cartilage protection | [80] |
| HA/PEG/KGN | Micelles | In vitro and in vivo | IA | Suppression of OA progression | [81] |
| SASP/HA | Conjugate | In vitro and in vivo | IA | Inhibition of cartilage destruction, and OA progression | [82] |
| Disks and injectable thermosensitive gels with PD and TGF-β3 | Scaffolds | In vitro and in vivo | IA | Decrease in osteoarthritis score, and global score | [83] |
| HA-MTX | Conjugate | In vivo | IA | Reduction of swelling, and inflammation | [84] |
| DK226 | Conjugate | In vivo | IA | Reduction of swelling, and inflammation | [85] |
| FidHycarn | Conjugate | In vivo | Oral | Reduced levels of cytokines, chemokines, and oxidative damage | [86] |
| HA/CS-CrmA | Polymeric nanoparticles | In vitro | Reduction of cartilage damage and inflammation | [87] | |
| pH-responsive nanoparticles with NH4CO3 and HA | Polymeric nanoparticles | In vitro and in vivo | IA | Reduction disease progression | [88] |
| HA-loaded PLGA particles | Hydrogel | In vitro and in vivo | IA | Inhibition of inflammation | [89] |
| HMWHA | In vivo | IA | Restoration of normal joint structure | [90] | |
| HABP2-8-arm PEG-COLBP | Peptide-polymer platform | In vivo | IA | Reduced levels of pro-inflammatory cytokines | [91] |
| HTG-E | Hydrogel | In vitro and in vivo | IA | Induction of chondrogenic regeneration and loss of minimized cartilage | [92] |
| HAMPC | Lubricated polymer | In vitro | Lubrication capacity and reduction of inflammation | [93] | |
| pNIPAM | Hydrogel | In vitro and in vivo | IA | Lubrication capacity and ability of intelligently releasing | [94] |
| PRP+HA | In vivo | IA | Inhibition of inflammation | [95] | |
| HA-Lipo-DIC/DEX | Liposomes | In vitro and in vivo | IA | Inhibition of inflammation and increased cell numbers | [96] |
| Celecoxib-HA | Nanocapsules | In vitro and in vivo | IA | Inhibition of inflammation and | [97] |
| HA-gel+magnoflorine | In vitro and in vivo | IA | Protects effect on the cartilage matrix | [98] | |
| In situ crosslinking N-chitosan, ADH and HA-ALD | Hydrogel | In vitro and in vivo | IA | Prevention of cartilage destruction and pain relief | [99] |
| Formulation | Studies | Administration Route | Main Results | Ref. | |
|---|---|---|---|---|---|
| HA-SLN/PD | Lipid nanoparticles | In vitro and in vivo | Intravenous | Bone preservation and reduced levels of pro-inflammatory cytokines | [101] |
| pH-sensitive liposomes with PDP | Liposomes | In vitro | Increased bioavailability and effectiveness of PDP | [102] | |
| HAPNPs/Dex | Polymeric nanoparticles | In vitro and in vivo | IA | Reduction in inflammatory cell infiltration, bone damage and cartilage | [66] |
| DNPs | Polymeric nanoparticles | In vivo | Systemic | Reduced levels of pro-inflammatory cytokines | [103] |
| MP-HANPs | Polymeric nanoparticles | In vitro and in vivo | Systemic | Accumulation at the inflammation site | [104] |
| HA/Cur | Nanomicelles | In vitro and in vivo | IA | Reduction of inflammation and protection of cartilage | [105] |
| FidHycarn | Conjugate | In vivo | oral | Reduced levels of pro-inflammatory cytokines and chemokines | [106] |
| Chitosan nanogels for the delivery of PS | Nanogels | In vitro and in vivo | IA | PS retention in the joint and inflammation reduction | [107] |
| HA-MTX | Conjugate | In vitro and in vivo | Intravenous | Reduction of inflammatory cytokine levels and cartilage damage | [108] |
| Flare-responsive TG-18 and TA delivery system | Hydrogel | In vitro and in vivo | IA | Reduced the arthritis activity | [109] |
| HYA-HAMT-NPs | Polymeric nanoparticles | In vitro and in vivo | IA | Preventing disease progression and promoting joint regeneration | [110] |
| HA-AuNCs/T/P | Gold nanocages | In vivo | Intravenous | Bone and cartilage preservation | [111] |
| Formulation | Studies | Administration Route | Main Results | Ref. | |
|---|---|---|---|---|---|
| HA, MH and AG | In vivo | Topic | Reduced healing time with the use of HA | [133] | |
| BHA | N-butyrylated LMW-HA | In vitro and in vivo | Topic | Reduction in pro-inflammatory cytokines levels; increased expression of growth factors | [136] |
| HA-POL | Hydrogel | In vitro and in vivo | Topic | Increase in protein accumulation; improvement in tissue repair | [137] |
| photo-responsive supramolecular hydrogels for EGF delivery | Hydrogel | In vitro and in vivo | Topic | Granulation tissue formation, and growth of neoepidermis | [138] |
| FHHA-S/Fe | Hydrogel | In vitro and in vivo | Topic | Decreased average wound area | [139] |
| Formulation | Studies | Administration Route | Main Results | Ref. | |
|---|---|---|---|---|---|
| IBD98-M | Conjugate | Ex vivo and in vivo | Injected into the ligated area of the distal colon | Promotion of healing of the intestinal mucosa; reduction in intestinal inflammation | [140] |
| HANPs with BDS | Polymeric Nanoparticles | In vitro | Inflammation reduction | [141] | |
| HA-KPV-NPs | Polymeric nanoparticles | In vitro and in vivo | Oral | Inflammation relief; mucosal healing; combination with hydrogel exhibits increased ability to prevent mucosal damage | [145] |
| HMWHA | In vitro and in vivo | Local application | Improvement in tissue regeneration | [146] | |
| HMWHA | In vivo | Systemic | Relief of UC symptoms; increased expression of COX-2 and PGE2 | [147] | |
| HABN | Conjugate | In vivo | Oral | Colon epithelium barrier restored | [148] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marinho, A.; Nunes, C.; Reis, S. Hyaluronic Acid: A Key Ingredient in the Therapy of Inflammation. Biomolecules 2021, 11, 1518. https://doi.org/10.3390/biom11101518
Marinho A, Nunes C, Reis S. Hyaluronic Acid: A Key Ingredient in the Therapy of Inflammation. Biomolecules. 2021; 11(10):1518. https://doi.org/10.3390/biom11101518
Chicago/Turabian StyleMarinho, Andreia, Cláudia Nunes, and Salette Reis. 2021. "Hyaluronic Acid: A Key Ingredient in the Therapy of Inflammation" Biomolecules 11, no. 10: 1518. https://doi.org/10.3390/biom11101518
APA StyleMarinho, A., Nunes, C., & Reis, S. (2021). Hyaluronic Acid: A Key Ingredient in the Therapy of Inflammation. Biomolecules, 11(10), 1518. https://doi.org/10.3390/biom11101518








