Lipopolysaccharide of the Yersinia pseudotuberculosis Complex
Abstract
1. Introduction
2. Y. pestis and Y. pseudotuberculosis Lipopolysaccharide Structures
2.1. Composition and Structure of the O-Specific Polysaccharides
| Serotype [Reference] | Structure of the Repeating Unit |
|---|---|
| O1a R = β-Parf [11] O2a R = α-Abep [7] O4b R = α-Tyvp [12] | 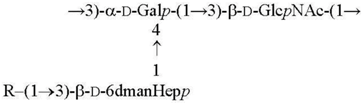 |
| O1c R = β-Parf [13] O2b R = α-Abep [14] | 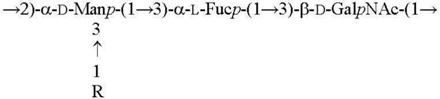 |
| O3 [15] | 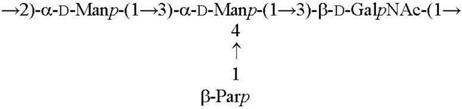 |
| O2c R = α-Abep [15] O4a R = α-Tyvp [16] | 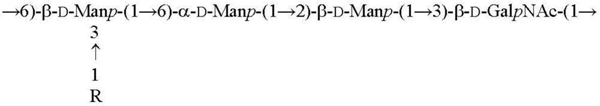 |
| O1b R = β-Parf [17] O11 R = α-l-6dAltf [18] | 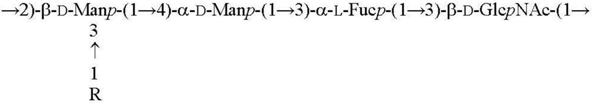 |
| O5a R = α-Ascp [19] O5b R = α-l-6dAltf [20] O15 R = β-Parf [21] |  |
| O6 [22,23] | 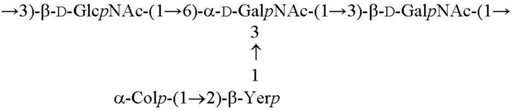 |
| O7 [24] |  |
| O10 [25] |  |
| O12 [3] |  |
| O9 [26,27] | 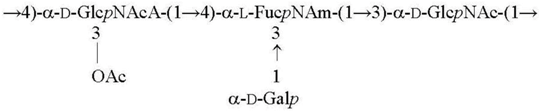 |
2.2. Structural Variants of the Core Region
2.3. Structure Variations of Lipid A
3. Genetics and Biosynthesis
4. Biological Effects of Y. pestis and Y. pseudotuberculosis Lipopolysaccharide
4.1. LPS Recognition by Innate Immunity
4.2. LPS Interaction with Antimicrobial Peptides (AMPs)—The First Line of Defense in Innate Immunity
4.3. Lipopolysaccharide and Yersiniae Virulence
5. Immunogenicity and Protective Efficacy of Yersinia Lipopolysaccharide
6. Phages Target LPS
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 6dAlt | 6-deoxyaltrose |
| 6dmanHep | 6-deoxy-d-manno-heptose |
| Abe | 3,6-dideoxy-d-xylo-hexose (abequose) |
| Asc | 3,6-dideoxy-l-arabino-hexose (ascarylose) |
| Col | 3,6-dideoxy-l-xylo-hexose (colitose) |
| FucNAm | 2-acetimidoylamino-2-deoxyfucose |
| GlcNAcA | 2-acetamido-2-deoxyglucuronic acid |
| Par | 3,6-dideoxy-d-ribo-hexose (paratose) |
| Qui | 6-deoxyglucose (quinovose) |
| Tyv | 3,6-dideoxy-d-arabino-hexose (tyvelose) |
| Yer | 4-C-[(S)-1-hydroxyethyl]-3,6-dideoxy-d-xylo-hexose (yersiniose A) |
| GlcNAc | 2-acetamido-2-deoxyglucose |
| GalNAc | 2-acetamido-2-deoxygalactose |
| EtNP | 2-aminoethyl phosphate |
| l-α-d-Hep | l-glycero-α-d-manno-heptose |
| d-α-d-Hep | d-glycero-α-d-manno-heptose |
| Gal | galactose |
| Glc | glucose |
| Kdo | 3-deoxy-d-manno-oct-2-ulosonic acid |
| Ko | d-glycero-d-talo-oct-2-ulosonic acid |
| Ara4N | 4-amino-4-deoxy-L-arabinose |
| GlcN | glucosamine |
| PEtN | phosphoethanolamine |
| UndP,UndPP | undecaprenyl phosphate/diphosphate |
| CAMP | cationic antimicrobial peptides |
| LPS | lipopolysaccharide |
| R-formLPS | rough LPS |
| S-formLPS | smooth form LPS |
| NHS | normal human serum |
| AMPs | antimicrobial peptides |
| AMCs | antimicrobial chemokines |
| AGP | aminoalkyl glucosaminide 4-phosphate |
| TLR4 | Toll-like receptor 4 |
| PAMP | pathogen-associated molecular patterns |
| DC | dendritic cell |
References
- Tsubokura, M.; Aleksic, S. A simplified antigenic scheme for serotyping of Yersinia pseudotuberculosis: Phenotypic characterization of reference strains and preparation of O and H factor sera. Contrib. Microbiol. Immunol. 1995, 13, 99–105. [Google Scholar]
- Laukkanen-Ninios, R.; Didelot, X.; Jolley, K.A.; Morelli, G.; Sangal, V.; Kristo, P.; Brehony, C.; Imori, P.F.; Fukushima, H.; Siitonen, A.; et al. Population structure of the Yersinia pseudotuberculosis complex according to multilocus sequence typing. Environ. Microbiol. 2011, 13, 3114–3127. [Google Scholar] [CrossRef] [PubMed]
- De Castro, C.; Kenyon, J.J.; Cunneen, M.M.; Molinaro, A.; Holst, O.; Skurnik, P.R. The O-specific polysaccharide structure and gene cluster of serotype O:12 of the Yersinia pseudotuberculosis complex, and the identification of a novel L-quinovose biosynthesis gene. Glycobiology 2012, 2, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Kenyon, J.J.; Cunneen, M.M.; Reeves, P.R. Genetics and evolution of Yersinia pseudotuberculosis O-specific polysaccharides: A novel pattern of O-antigen diversity. FEMS Microbiol. Rev. 2017, 41, 200–217. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Kondakova, A.N.; Bystrova, O.V.; Lindner, B.; Shaikhutdinova, R.Z.; Dentovskaya, S.V.; Anisimov, A.P. New features of Yersinia lipopolysaccharide structures as revealed by high-resolution electrospray ionization mass spectrometry. Adv. Sci. Lett. 2008, 1, 192–198. [Google Scholar] [CrossRef]
- Skurnik, M.; Bengoechea, J.A. The biosynthesis and biological role of lipopolysaccharide O-antigens of pathogenic Yersiniae. Carbohydr. Res. 2003, 338, 2521–2529. [Google Scholar] [CrossRef]
- Kondakova, A.N.; Ho, N.; Bystrova, O.V.; Shashkov, A.S.; Lindner, B.; Creuzenet, C.; Knirel, Y.A. Structural studies of the O-antigens of Yersinia pseudotuberculosis O:2a and mutants thereof with impaired 6-deoxy-d-manno-heptose biosynthesis pathway. Carbohydr. Res. 2008, 343, 1383–1389. [Google Scholar] [CrossRef]
- Wang, L.; Curd, H.; Qu, W.J.; Reeves, P.R. Sequencing of Escherichia coli O111 O-antigen gene cluster and identification of O111-specific genes. J. Clin. Microbiol. 1998, 36, 3182–3187. [Google Scholar] [CrossRef]
- Kenne, L.; Lindberg, B. Bacterial polysaccharides. In The Polysaccharides; Aspinall, G.O., Ed.; Academic Press: New York, NY, USA, 1983; pp. 287–363. [Google Scholar]
- Vinogradov, E.; Nossova, L.; Radziejewska-Lebrecht, J. The structure of the O-specific polysaccharide from Salmonella cerro (serogroup K, O:6,14,18). Carbohydr. Res. 2004, 339, 2441–2443. [Google Scholar] [CrossRef]
- Kondakova, A.N.; Sevillano, A.M.; Shaikhutdinova, R.Z.; Lindner, B.; Komandrova, N.A.; Dentovskaya, S.V.; Shashkov, A.S.; Anisimov, A.P.; Skurnik, M.; Knirel, Y.A. Revision of the O-polysaccharide structure of Yersinia pseudotuberculosis O:1a; confirmation of the function of WbyM as paratosyltransferase. Carbohydr. Res. 2012, 350, 98–102. [Google Scholar] [CrossRef]
- Kondakova, A.N.; Bystrova, O.V.; Shaikhutdinova, R.Z.; Ivanov, S.A.; Dentovskaya, S.V.; Shashkov, A.S.; Knirel, Y.A.; Anisimov, A.P. Structure of the O-antigen of Yersinia pseudotuberculosis O:4b. Carbohydr. Res. 2009, 344, 152–154. [Google Scholar] [CrossRef] [PubMed]
- De Castro, C.; Kenyon, J.; Cunneen, M.M.; Reeves, P.R.; Molinaro, A.; Holst, O.; Skurnik, M. Genetic characterization and structural analysis of the O-specific polysaccharide of Yersinia pseudotuberculosis serotype O:1c. Innate Immun. 2011, 17, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Kondakova, A.N.; Bystrova, O.V.; Shaikhutdinova, R.Z.; Ivanov, S.A.; Dentovskaya, S.V.; Shashkov, A.S.; Knirel, Y.A.; Anisimov, A.P. Structure of the O-polysaccharide of Yersinia pseudotuberculosis O:2b. Carbohydr. Res. 2009, 344, 405–407. [Google Scholar] [CrossRef] [PubMed]
- Kondakova, A.N.; Bystrova, O.V.; Shaikhutdinova, R.Z.; Ivanov, S.A.; Dentovskaya, S.V.; Shashkov, A.S.; Knirel, Y.A.; Anisimov, A.P. Reinvestigation of the O-antigens of Yersinia pseudotuberculosis: Revision of the O2c and confirmation of the O3 antigen structures. Carbohydr. Res. 2008, 343, 2486–2488. [Google Scholar] [CrossRef] [PubMed]
- Kondakova, A.N.; Bystrova, O.V.; Shaikhutdinova, R.Z.; Ivanov, S.A.; Dentovskaya, S.V.; Shashkov, A.S.; Knirel, Y.A.; Anisimov, A.P. Structure of the O-antigen of Yersinia pseudotuberculosis O:4a revised. Carbohydr. Res. 2009, 344, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Kondakova, A.N.; Shaikhutdinova, R.Z.; Ivanov, S.A.; Dentovskaya, S.V.; Shashkov, A.S.; Anisimov, A.P.; Knirel, Y.A. Revision of the O-polysaccharide structure of Yersinia pseudotuberculosis O:1b. Carbohydr. Res. 2009, 344, 2421–2423. [Google Scholar] [CrossRef]
- Cunneen, M.M.; De Castro, C.; Kenyon, J.; Parrilli, M.; Reeves, P.R.; Molinaro, A.; Holst, O.; Skurnik, M. The O-specific polysaccharide structure and biosynthetic gene cluster of Yersinia pseudotuberculosis serotype O:11. Carbohydr. Res. 2009, 344, 1533–1540. [Google Scholar] [CrossRef]
- Gorshkova, R.P.; Korchagina, N.I.; Ovodov, Y.S. Structural studies on the O-specific side-chain polysaccharide of lipopolysaccharide from Yersinia pseudotuberculosis VA serovar. Eur. J. Biochem. 1983, 131, 345–347. [Google Scholar] [CrossRef]
- Korchagina, N.I.; Gorshkova, R.P.; Ovodov, Y.S. Studies on O-specific polysaccharide from Yersinia pseudotuberculosis VB serovar. Bioorg. Khim. 1982, 8, 1666–1669. [Google Scholar]
- De Castro, C.; Skurnik, M.; Molinaro, A.; Holst, O. Characterization of the O-polysaccharide structure and biosynthetic gene cluster of Yersinia pseudotuberculosis serotype O:15. Innate Immun. 2009, 15, 351–359. [Google Scholar] [CrossRef]
- Gorshkova, R.P.; Zubkov, V.A.; Isakov, V.V.; Ovodov, Y.S. Structural features of O-specific polysaccharide from lipopolysaccharide of Yersinia pseudotuberculosis VI serovar. Bioorg. Khim. 1983, 9, 1068–1073. [Google Scholar] [PubMed]
- Zubkov, V.A.; Gorshkova, R.P.; Ovodov, Y.S.; Sviridov, A.F.; Shashkov, A.S. Synthesis of 3,6-dideoxy-4-C-(41-hydroxyethyl)hexopyranoses (yersinioses) from 1,6-anhydro-β-d-glycopyranose. Carbohydr. Res. 1992, 225, 189–207. [Google Scholar] [CrossRef]
- Komandrova, N.A.; Gorshkova, R.P.; Zubkov, V.A.; Ovodov, Y.S. Structure of the O-specific polysaccharide chain of lipopolysaccharide of Yersinia pseudotuberculosis serovar VII. Bioorg. Khim. 1989, 15, 104–110. [Google Scholar]
- Kenyon, J.J.; De Castro, C.; Cunneen, M.M.; Reeves, P.R.; Molinaro, A.; Holst, O.; Skurnik, M. The genetics and structure of the O-specific polysaccharide of Yersinia pseudotuberculosis serotype O:10 and its relationship to Escherichia coli O111 and Salmonella enterica O35. Glycobiology 2011, 21, 1131–11139. [Google Scholar] [CrossRef]
- Beczala, A.; Ovchinnikova, O.G.; Duda, K.A.; Skurnik, M.; Radziejewska-Lebrecht, J.; Holst, O. Structure of Yersinia pseudotuberculosis O:9 O-specific polysaccharide repeating unit resolved. In Proceedings of the Abstracts of the 15th European Carbohydrate Symposium, Vienna, Austria, 19–24 July 2009; pp. 19–24. [Google Scholar]
- Beczala, A.; Ovchinnikova, O.G.; Datta, N.; Mattinen, L.; Knapska, K.; Radziejewska-Lebrecht, J.; Holst, O.; Skurnik, M. Structure and genetic basis of Yersinia similis serotype O:9 O-specific polysaccharide. Innate Immun. 2015, 21, 3–16. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Lindner, B.; Vinogradov, E.V.; Kocharova, N.A.; Senchenkova, S.N.; Shaikhutdinova, R.Z.; Dentovskaya, S.V.; Fursova, N.K.; Bakhteeva, I.V.; Titareva, G.M.; et al. Temperature-dependent variations and intraspecies diversity of the structure of the lipopolysaccharide of Yersinia pestis. Biochemistry 2005, 44, 1731–1743. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Lindner, B.; Vinogradov, E.V.; Shaikhutdinova, R.Z.; Senchenkova, S.N.; Kocharova, N.A.; Holst, O.; Pier, G.B.; Anisimov, A.P. Cold temperature-induced modifications to the composition and structure of the lipopolysaccharide of Yersinia pestis. Carbohydr. Res. 2008, 343, 1625–1630. [Google Scholar] [CrossRef] [PubMed]
- Hitchen, P.G.; Prior, J.L.; Oyston, P.C.; Panico, M.; Wren, B.W.; Titball, R.W.; Morris, H.R.; Dell, A. Structural characterization of lipo-oligosaccharide (LOS) from Yersinia pestis: Regulation of LOS structure by the PhoPQ system. Mol. Microbiol. 2002, 44, 1637–1650. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Dentovskaya, S.V.; Senchenkova, S.N.; Shaikhutdinova, R.Z.; Kocharova, N.A.; Anisimov, A.P. Structural features and structural variability of the lipopolysaccharide of Yersinia pestis, the cause of plague. J. Endotoxin Res. 2006, 12, 3–9. [Google Scholar] [PubMed]
- Rebeil, R.; Ernst, R.K.; Gowen, B.B.; Miller, S.I.; Hinnebusch, B.J. Variation in lipid A structure in the pathogenic yersiniae. Mol. Microbiol. 2004, 52, 1363–1373. [Google Scholar] [CrossRef]
- Therisod, H.; Karibian, D.; Perry, M.B.; Caroff, M. Structural analysis of Yersinia pseudotuberculosis ATCC 29833 lipid A. Int. J. Mass Spectrom. 2002, 219, 549–557. [Google Scholar] [CrossRef]
- Raetz, C.R.; Whitfield, C. Lipopolysaccharide endotoxins. Annu Rev. Biochem. 2002, 71, 635–700. [Google Scholar] [CrossRef] [PubMed]
- Dentovskaya, S.V.; Anisimov, A.P.; Kondakova, A.N.; Bystrova, O.V.; Lindner, B.; Svetoch, T.E.; Shaikhutdinova, R.Z.; Ivanov, S.A.; Bakhteeva, I.V.; Titareva, G.M.; et al. Functional characterization and biological significance of Yersinia pestis lipopolysaccharide biosynthesis genes. Biochemistry 2011, 76, 808–822. [Google Scholar] [CrossRef] [PubMed]
- Bishop, R.E. The lipid A palmitoyltransferase PagP: Molecular mechanisms and role in bacterial pathogenesis. Mol. Microbiol. 2005, 57, 900–912. [Google Scholar] [CrossRef] [PubMed]
- Rebeil, R.; Ernst, R.K.; Jarrett, C.O.; Adams, K.N.; Miller, S.I.; Hinnebusch, B.J. Characterization of late acyltransferase genes of Yersinia pestis and their role in temperature-dependent lipid A variation. J. Bacteriol. 2006, 188, 1381–1388. [Google Scholar] [CrossRef] [PubMed]
- Dentovskaya, S.V.; Shaikhutdinova, R.Z.; Knirel, Y.A.; Ivanov, S.L.; Anisimov, A.P. Generation of Vaccine Strains of Gram-Negative Bacteria with Reduced Adverse Reactions. Mol. Gen. Mikrobiol. Virusol. 2006, 2, 3–8. [Google Scholar]
- Gorzelak, P.; Klein, G.; Raina, S. Molecular Basis of Essentiality of Early Critical Steps in the Lipopolysaccharide Biogenesis in Escherichia coli K-12: Requirement of MsbA, Cardiolipin, LpxL, LpxM and GcvB. Int. J. Mol. Sci. 2021, 22, 5099. [Google Scholar] [CrossRef]
- Reynolds, C.M.; Raetz, C.R. Replacement of lipopolysaccharide with free lipid A molecules in Escherichia coli mutants lacking all core sugars. Biochemistry 2009, 48, 9627–9640. [Google Scholar] [CrossRef]
- Klein, G.; Lindner, B.; Brabetz, W.; Brade, H.; Raina, S. Escherichia coli K-12 Suppressor-free Mutants Lacking Early Glycosyltransferases and Late Acyltransferases: Minimal lipopolysaccharide structure and induction of envelope stress response. J. Biol. Chem. 2009, 284, 15369–15389. [Google Scholar] [CrossRef]
- Guo, L.; Lim, K.B.; Poduje, C.M.; Daniel, M.; Gunn, J.S.; Hackett, M.; Miller, S.I. Lipid A acylation and bacterial resistance against vertebrate antimicrobial peptides. Cell 1998, 95, 189–198. [Google Scholar] [CrossRef]
- Aussel, L.; Therisod, H.; Karibian, D.; Perry, M.B.; Bruneteau, M.; Caroff, M. Novel variation of lipid A structures in strains of different Yersinia species. FEBS Lett. 2000, 465, 87–92. [Google Scholar] [CrossRef]
- Trent, M.S.; Ribeiro, A.A.; Lin, S.; Cotter, R.J.; Raetz, C.R. An inner membrane enzyme in Salmonella and Escherichia coli that transfers 4-amino-4-deoxy-L-arabinose to lipid A: Induction on polymyxin-resistant mutants and role of a novel lipid-linked donor. J. Biol. Chem. 2001, 276, 43122–43131. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, A.P.; Dentovskaya, S.V.; Kondakova, A.N.; Lindner, B.; Shaikhutdinova, R.Z.; Kocharova, N.A.; Senchenkova, S.N.; Knirel, Y.A. Yersinia pestis lipopolysaccharide in host-pathogen interactions. In The Challenge of Highly Pathogenic Microorganisms. Mechanisms of Virulence and Novel Medical Countermeasures; Shafferman, A., Ordentlich, A., Velan, B., Eds.; Springer: Dordrecht, The Netherlands; London, UK; New York, NY, USA, 2010; pp. 77–87. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Dentovskaya, S.V.; Bystrova, O.V.; Kocharova, N.A.; Senchenkova, S.N.; Shaikhutdinova, R.Z.; Titareva, G.M.; Bakhteeva, I.V.; Lindner, B.; Pier, G.B.; et al. Relationship of the lipopolysaccharide structure of Yersinia pestis to resistance to antimicrobial factors. Adv. Exp. Med. Biol. 2007, 603, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Winfield, M.D.; Latifi, T.; Groisman, E.A. Transcriptional regulation of the 4-amino-4-deoxy-L-arabinose biosynthetic genes in Yersinia pestis. J. Biol. Chem. 2005, 280, 14765–14772. [Google Scholar] [CrossRef]
- Chung, H.S.; Raetz, C.R. Dioxygenases in Burkholderia ambifaria and Yersinia pestis that hydroxylate the outer Kdo unit of lipopolysaccharide. Proc. Natl. Acad. Sci. USA 2011, 108, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.S.; Yang, E.G.; Hwang, D.; Lee, J.E.; Guan, Z.; Raetz, C.R. Kdo hydroxylase is an inner core assembly enzyme in the Ko-containing lipopolysaccharide biosynthesis. Biochem. Biophys. Res. Commun. 2014, 452, 789–794. [Google Scholar] [CrossRef][Green Version]
- Erickson, D.L.; Lew, C.S.; Kartchner, B.; Porter, N.T.; McDaniel, S.W.; Jones, N.M.; Mason, S.; Wu, E.; Wilson, E. Lipopolysaccharide biosynthesis genes of Yersinia pseudotuberculosis promote resistance to antimicrobial chemokines. PLoS ONE 2016, 11, e0157092. [Google Scholar] [CrossRef]
- Reynolds, C.M.; Kalb, S.R.; Cotter, R.J.; Raetz, C.R. A phosphoethanolamine transferase specific for the outer 3-deoxy-D-manno-octulosonic acid residue of Escherichia coli lipopolysaccharide. Identification of the eptB gene and Ca2+ hypersensitivity of an eptB deletion mutant. J. Biol. Chem. 2005, 280, 21202–21211. [Google Scholar] [CrossRef]
- Kislichkina, A.A.; Platonov, M.E.; Vagaiskaya, A.S.; Bogun, A.G.; Dentovskaya, S.V.; Anisimov, A.P. Rational Taxonomy of Yersinia pestis. Mol. Genet. Microbiol. Virol. 2019, 34, 110–117. [Google Scholar] [CrossRef]
- Savin, C.; Martin, L.; Bouchier, C.; Filali, S.; Chenau, J.; Zhou, Z.; Becher, F.; Fukushima, H.; Thomson, N.R.; Scholz, H.C.; et al. The Yersinia pseudotuberculosis complex: Characterization and delineation of a new species, Yersinia wautersii. Int. J. Med. Microbiol. 2014, 304, 452–463. [Google Scholar] [CrossRef]
- Thal, E.; Knapp, W. A revised antigenic scheme of Yersinia pseudotuberculosis. Symp. Ser. Immunobiol. Standard. 1971, 13, 219–222. [Google Scholar]
- Bogdanovich, T.; Carniel, E.; Fukushima, H.; Skurnik, M. Use of O-antigen gene cluster-specific PCRs for the identification and O-genotyping of Yersinia pseudotuberculosis and Yersinia pestis. J. Clin. Microbiol. 2003, 41, 5103–5112. [Google Scholar] [CrossRef] [PubMed]
- Duan, R.; Liang, J.; Shi, G.; Cui, Z.; Hai, R.; Wang, P.; Xiao, Y.; Li, K.; Qiu, H.; Gu, W.; et al. Homology analysis of pathogenic Yersinia: Yersinia enterocolitica, Yersinia pseudotuberculosis, and Yersinia pestis based on multilocus sequence typing. J. Clin. Microbiol. 2014, 52, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Skurnik, M.; Peippo, A.; Ervela, E. Characterization of the O-antigen gene clusters of Yersinia pseudotuberculosis and the cryptic O-antigen gene cluster of Yersinia pestis shows that the plague bacillus is most closely related to and has evolved from Y. pseudotuberculosis serotype O:1b. Mol. Microbiol. 2000, 37, 316–330. [Google Scholar] [CrossRef] [PubMed]
- Matsura, M. Structural modifications of bacterial lipopolysaccharide that facilitate Gram-negative bacteria evasion of host innate immunity. Front. Immunol. 2013, 4, 109. [Google Scholar] [CrossRef] [PubMed]
- Porat, R.; McCabe, W.R.; Brubaker, R.R. Lipopolysaccharide associated resistance to killing of yersiniae by complement. J. Endotoxin Res. 1995, 2, 91–97. [Google Scholar] [CrossRef]
- Karlyshev, A.V.; Oyston, P.C.; Williams, K.; Clark, G.C.; Titball, R.W.; Winzeler, E.A.; Wren, B.W. Application of high-density array-based signature-tagged mutagenesis to discover novel Yersinia virulence-associated genes. Infect. Immun. 2001, 69, 7810–7819. [Google Scholar] [CrossRef]
- Mecsas, J.; Bilis, I.; Falkow, S. Identification of attenuated Yersinia pseudotuberculosis strains and characterization of an orogastric infection in BALB/c mice on day 5 postinfection by signature-tagged mutagenesis. Infect. Immun. 2001, 69, 2779–2787. [Google Scholar] [CrossRef]
- Flashner, Y.; Mamroud, E.; Tidhar, A.; Ber, R.; Aftalion, M.; Gur, D.; Lazar, S.; Zvi, A.; Bino, T.; Ariel, N.; et al. Generation of Yersinia pestis attenuated strains by signature-tagged mutagenesis in search of novel vaccine candidates. Infect. Immun. 2004, 72, 908–915. [Google Scholar] [CrossRef]
- Wang, X.; Quinn, P.J. Lipopolysaccharide: Biosynthetic pathway and structure modification. Prog. Lipid Res. 2010, 49, 97–107. [Google Scholar] [CrossRef]
- Akira, S.; Hemmi, H. Recognition of pathogen-associated molecular patterns by TLR family. Immunol Lett. 2003, 85, 85–95. [Google Scholar] [CrossRef]
- Weiss, J.; Barker, J. Diverse pro-inflammatory endotoxin recognition systems of mammalian innate immunity. F1000 Res. 2018, 7, 516. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, K.; Tsukano, H.; Watanabe, H.; Lindner, B.; Matsuura, M. Modification of the structure and activity of lipid A in Yersinia pestis lipopolysaccharide by growth temperature. Infect. Immun. 2002, 70, 4092–4098. [Google Scholar] [CrossRef]
- Prior, J.L.; Hitchen, P.G.; Williamson, D.E.; Reason, A.J.; Morris, H.R.; Dell, A.; Wren, B.W.; Titball, R.W. Characterization of the lipopolysaccharide of Yersinia pestis. Microb. Pathog. 2001, 30, 49–57. [Google Scholar] [CrossRef]
- Korneev, K.V.; Kondakova, A.N.; Arbatsky, N.P.; Novototskaya-Vlasova, K.A.; Rivkina, E.M.; Anisimov, A.P.; Kruglov, A.A.; Kuprash, D.V.; Nedospasov, S.A.; Knirel, Y.A.; et al. Distinct biological activity of lipopolysaccharides with different lipid A acylation status from mutant strains of Yersinia pestis and some members of genus Psychrobacter. Biochemistry 2014, 79, 1333–1338. [Google Scholar] [CrossRef]
- Steinstraesser, L.; Kraneburg, U.; Jacobsen, F.; Al-Benna, S. Host defense peptides and their antimicrobial-immunomodulatory duality. Immunobiology 2011, 216, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Gunn, J.S.; Lim, K.B.; Krueger, J.; Kim, K.; Guo, L.; Hackett, M.; Miller, S.I. PmrA-PmrB-regulated genes necessary for 4-aminoarabinose lipid A modification and polymyxin resistance. Mol. Microbiol. 1998, 27, 1171–1182. [Google Scholar] [CrossRef] [PubMed]
- Holst, O. The structures of core regions from enterobacterial lipopolysaccharides–an update. FEMS Microbiol. Lett. 2007, 271, 3–11. [Google Scholar] [CrossRef]
- Klein, K.A.; Fukuto, H.S.; Pelletier, M.; Romanov, G.; Grabenstein, J.P.; Palmer, L.E.; Ernst, R.; Bliska, J.B. A transposon site. hybridization screen identifies galU and wecBC as important for. survival of Yersinia pestis in murine macrophages. J. Bacteriol. 2012, 194, 653–662. [Google Scholar] [CrossRef]
- Mathew, B.; Aoyagi, K.L.; Fisher, M.A. Yersinia pestis lipopolysaccharide remodeling confers resistance to a Xenopsylla cheopis cecropin. bioRxiv 2021, 7, 2536–2545. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Anisimov, A.P. Lipopolysaccharide of Yersinia pestis, the Cause of Plague: Structure, Genetics, Biological Properties. Acta Nat. 2012, 4, 46–58. [Google Scholar] [CrossRef]
- Band, V.I.; Weiss, D.S. Mechanisms of Antimicrobial Peptide Resistance in Gram-Negative Bacteria. Antibiotics 2015, 4, 18–41. [Google Scholar] [CrossRef] [PubMed]
- Shaikhutdinova, R.Z.; Ivanov, S.A.; Dentovskaya, S.V.; Titareva, G.M.; Knirel, Y.A. Characterization of a transposon Tn5-generated mutant of Yersinia pestis defective in lipooligosaccharide biosynthesis. Biochemistry 2019, 84, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Fukuto, H.S.; Vadyvaloo, V.; McPhee, J.B.; Poinar, H.N.; Holmes, E.C.; Bliska, J.B. A single amino acid change in the response regulator PhoP, acquired during Yersinia pestis evolution, affects PhoP target gene transcription and polymyxin B susceptibility. J. Bacteriol. 2018, 200, e00050-18. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, K.L.; Brooks, B.D.; Bearden, S.W.; Montenieri, J.A.; Gage, K.L.; Fisher, M.A. LPS modification promotes maintenance of Yersinia pestis in fleas. Microbiology 2015, 161, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Nair, M.K.; Galván, E.M.; Liu, S.L.; Schifferli, D.M. Tn5AraOut mutagenesis for the identification of Yersinia pestis genes involved in resistance towards cationic antimicrobial peptides. Microb. Pathog. 2011, 51, 121–132. [Google Scholar] [CrossRef]
- Felek, S.; Muszyński, A.; Carlson, R.W.; Tsang, T.M.; Hinnebusch, B.J.; Krukonis, E.S. Phosphoglucomutase of Yersinia pestis is required for autoaggregation and polymyxin B resistance. Infect. Immun. 2010, 78, 1163–1175. [Google Scholar] [CrossRef] [PubMed]
- Geng, J.; Song, Y.; Yang, L.; Feng, Y.; Qiu, Y.; Li, G.; Guo, J.; Bi, Y.; Qu, Y.; Wang, W.; et al. Involvement of the post-transcriptional regulator Hfq in Yersinia pestis virulence. PLoS ONE 2009, 4, e6213. [Google Scholar] [CrossRef]
- Nishino, K.; Hsu, F.F.; Turk, J.; Cromie, M.J.; Wösten, M.M.; Groisman, E.A. Identification of the lipopolysaccharide modifications controlled by the Salmonella PmrA/PmrB system mediating resistance to Fe(III) and Al(III). Mol. Microbiol. 2006, 61, 645–654. [Google Scholar] [CrossRef]
- Anisimov, A.P.; Dentovskaya, S.V.; Titareva, G.M.; Bakhteeva, I.V.; Shaikhutdinova, R.Z.; Balakhonov, S.V.; Lindner, B.; Kocharova, N.A.; Senchenkova, S.N.; Holst, O.; et al. Intraspecies and temperature-dependent variations in susceptibility of Yersinia pestis to the bactericidal action of serum and to polymyxin B. Infect. Immun. 2005, 73, 7324–7331. [Google Scholar] [CrossRef]
- Bengoechea, J.A.; Lindner, B.; Seydel, U.; Ramón, D.; Ignacio, M. Yersinia pseudotuberculosis and Yersinia pestis are more resistant to bactericidal cationic peptides than Yersinia enterocolitica. Microbiology 1998, 144, 1509–1515. [Google Scholar] [CrossRef] [PubMed]
- Zurabyan, V.A.; Pavlovich, N.V.; Makarovskaya, L.N.; Ryzhkova, V.V.; Bugaeva, O.K. In vitro influence of biologically active factors of microorganisms on antibiotic susceptibility of Yersinia pestis. Antibiot. Khimioter. 1997, 42, 22–25. [Google Scholar] [PubMed]
- Abdelbaqi, S.; Deslouches, B.; Steckbeck, J.; Montelaro, R.; Reed, D.S. Novel engineered cationic antimicrobial peptides display broad-spectrum activity against Francisella tularensis, Yersinia pestis and Burkholderia pseudomallei. J. Med. Microbiol. 2016, 65, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Pouillot, F.; Derbise, A.; Kukkonen, M.; Foulon, J.; Korhonen, T.K.; Carniel, E. Evaluation of O-antigen inactivation on Pla activity and virulence of Yersinia pseudotuberculosis harbouring the pPla plasmid. Microbiology 2005, 151, 3759–3768. [Google Scholar] [CrossRef]
- Chandler, C.E.; Harberts, E.M.; Pelletier, M.R.; Thaipisuttikul, I.; Jones, J.W.; Hajjar, A.M.; Sahl, J.W.; Goodlett, D.R.; Pride, A.C.; Rasko, D.A.; et al. Early evolutionary loss of the lipid A modifying enzyme PagP resulting in innate immune evasion in Yersinia pestis. Proc. Natl. Acad. Sci. USA 2020, 117, 22984–22991. [Google Scholar] [CrossRef] [PubMed]
- Telepnev, M.V.; Klimpel, G.R.; Haithcoat, J.; Knirel, Y.A.; Anisimov, A.P.; Motin, V.L. Tetraacylated lipopolysaccharide of Yersinia pestis can inhibit multiple Toll-like receptor-mediated signaling pathways in human dendritic cells. J. Infect. Dis. 2009, 200, 1694–1702, Erratum in J. Infect. Dis. 2010, 201, 314. [Google Scholar] [CrossRef]
- Skurnik, M. Molecular genetics, biochemistry and biological role of Yersinia lipopolysaccharide. Adv. Exp. Med. Biol. 2003, 529, 187–197. [Google Scholar] [CrossRef]
- Mikula, K.M.; Kolodziejczyk, R.; Goldman, A. Yersinia infection tools-characterization of structure and function of adhesins. Front. Cell. Infect. Microbiol. 2013, 2, 169. [Google Scholar] [CrossRef]
- Eren, E.; van den Berg, B. Structural basis for activation of an integral membrane protease by lipopolysaccharide. J. Biol. Chem. 2012, 287, 23971–23976. [Google Scholar] [CrossRef]
- Yang, K.; He, Y.; Park, C.G.; Kang, Y.S.; Zhang, P.; Han, Y.; Cui, Y.; Bulgheresi, S.; Anisimov, A.P.; Dentovskaya, S.V.; et al. Yersinia pestis Interacts With SIGNR1 (CD209b) for Promoting Host Dissemination and Infection. Front. Immunol. 2019, 10, 96. [Google Scholar] [CrossRef]
- He, Y.X.; Ye, C.L.; Zhang, P.; Li, Q.; Park, C.G.; Yang, K.; Jiang, L.Y.; Lv, Y.; Ying, X.L.; Ding, H.H.; et al. Yersinia pseudotuberculosis Exploits CD209 Receptors for Promoting Host Dissemination and Infection. Infect. Immun. 2018, 87, e00654-18. [Google Scholar] [CrossRef]
- Zhang, P.; Skurnik, M.; Zhang, S.S.; Schwartz, O.; Kalyanasundaram, R.; Bulgheresi, S.; He, J.J.; Klena, J.D.; Hinnebusch, B.J.; Chen, T. Human dendritic cell-specific intercellular adhesion molecule-grabbing nonintegrin (CD209) is a receptor for Yersinia pestis that promotes phagocytosis by dendritic cells. Infect. Immun. 2008, 76, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- Strittmatter, W.; Galanos, C. Characterization of protein co-extracted together with LPS in Escherichia coli, Salmonella minnesota and Yersinia enterocolitica. Microb. Pathog. 1987, 2, 29–36. [Google Scholar] [CrossRef]
- Vakorina, T.I.; Novikova, O.D.; Krasikova, I.N.; Naberezhnykh, G.N.; Solov’eva, T.F.; Ovodov, Y.S. Interaction of porin from Yersinia pseudotuberculosis with different structural forms of endogenous lipopolysaccharide. Biochemistry 2003, 68, 976–983. [Google Scholar] [CrossRef]
- Confer, A.W.; Ayalew, S. The OmpA family of proteins: Roles in bacterial pathogenesis and immunity. Vet. Microbiol. 2013, 163, 207–222. [Google Scholar] [CrossRef]
- Bartra, S.S.; Gong, X.; Lorica, C.D.; Jain, C.; Nair, M.K.; Schifferli, D.; Qian, L.; Li, Z.; Plano, G.V.; Schesser, K. The outer membrane protein A (OmpA) of Yersinia pestis promotes intracellular survival and virulence in mice. Microb. Pathog. 2012, 52, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Krukonis, E.S.; Thomson, J.J. Complement evasion mechanisms of the systemic pathogens Yersiniae and Salmonellae. FEBS Lett. 2020, 594, 2598–2620. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, T.K.; Haiko, J.; Laakkonen, L.; Järvinen, H.M.; Westerlund-Wikström, B. Fibrinolytic and coagulative activities of Yersinia pestis. Front. Cell Infect. Microbiol. 2013, 3, 35. [Google Scholar] [CrossRef] [PubMed]
- Suomalainen, M.; Lobo, L.A.; Brandenburg, K.; Lindner, B.; Virkola, R.; Knirel, Y.A.; Anisimov, A.P.; Holst, O.; Korhonen, T.K. Temperature-induced changes in the lipopolysaccharide of Yersinia pestis affect plasminogen activation by the Pla surface protease. Infect. Immun. 2010, 78, 2644–2652. [Google Scholar] [CrossRef]
- Kolodziejek, A.M.; Schnider, D.R.; Rohde, H.N.; Wojtowicz, A.J.; Bohach, G.A.; Minnich, S.A.; Hovde, C.J. Outer membrane protein X (Ail) contributes to Yersinia pestis virulence in pneumonic plague and its activity is dependent on the lipopolysaccharide core length. Infect. Immun. 2010, 78, 5233–5243. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.; Lee, H.; Tian, Y.; Schesser Bartra, S.; Hower, S.; Fujimoto, L.M.; Yao, Y.; Ivanov, S.A.; Shaikhutdinova, R.Z.; Anisimov, A.P.; et al. Mutually constructive roles of Ail and LPS in Yersinia pestis serum survival. Mol. Microbiol. 2020, 114, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Tsang, T.M.; Wiese, J.S.; Felek, S.; Kronshage, M.; Krukonis, E.S. Ail proteins of Yersinia pestis and Y. pseudotuberculosis have different cell binding and invasion activities. PLoS ONE 2013, 8, e83621. [Google Scholar] [CrossRef]
- Davies, D.A.L. A specific polysaccharide of Pasteurella pestis. Biochem. J. 1956, 63, 105–116. [Google Scholar] [CrossRef]
- Larrabee, A.R.; Marshall, J.D.; Crozier, D. Isolation of antigens of Pasteurella pestis I. Lipopolysaccharide-protein complex and R and S Antigens. J. Bacteriol. 1965, 90, 116–119. [Google Scholar] [CrossRef]
- Byvalov, A.A.; Pautov, V.N.; Chicherin, L.V.; Lebedinskii, V.A.; Evstigneev, V.I. Effectiveness of revaccinating hamadryas baboons with NISS live dried plague vaccine and fraction I of the plague microbe. Zh. Mikrobiol. Epidemiol. Immunobiol. 1984, 4, 74–76. [Google Scholar]
- Montminy, S.W.; Khan, N.; McGrath, S.; Walkowicz, M.J.; Sharp, F.; Conlon, J.E.; Fukase, K.; Kusumoto, S.; Sweet, C.; Miyake, K.; et al. Virulence factors of Yersinia pestis are overcome by a strong lipopolysaccharide response. Nat. Immunol. 2006, 7, 1066–1073. [Google Scholar] [CrossRef]
- Anisimov, A.P.; Shaikhutdinova, R.Z.; Pan’kina, L.N.; Feodorova, V.A.; Savostina, E.P.; Bystrova, O.V.; Lindner, B.; Mokrievich, A.N.; Bakhteeva, I.V.; Titareva, G.M.; et al. Effect of deletion of the lpxM gene on virulence and vaccine potential of Yersinia pestis in mice. J. Med. Microbiol. 2007, 56, 443–453. [Google Scholar] [CrossRef]
- Airhart, C.L.; Rohde, H.N.; Bohach, G.A.; Hovde, C.J.; Deobald, C.F.; Lee, S.S.; Minnich, S.A. Induction of innate immunity by lipid A mimetics increases survival from pneumonic plague. Microbiology 2008, 154, 2131–2138. [Google Scholar] [CrossRef]
- Filippov, A.A.; Sergueev, K.V.; Nikolich, M.P. Can phage effectively treat multidrug-resistant plague? Bacteriophage 2012, 2, 186–189. [Google Scholar] [CrossRef]
- Zhao, X.; Cui, Y.; Yan, Y.; Du, Z.; Tan, Y.; Yang, H.; Bi, Y.; Zhang, P.; Zhou, L.; Zhou, D.; et al. Outer membrane proteins ail and OmpF of Yersinia pestis are involved in the adsorption of T7-related bacteriophage Yep-phi. J. Virol. 2013, 87, 2260–2269. [Google Scholar] [CrossRef] [PubMed]
- Derbise, A.; Carniel, E. YpfPhi: A filamentous phage acquired by Yersinia pestis. Front. Microbiol. 2014, 5, 701. [Google Scholar] [CrossRef]
- Garcia, E.; Elliott, J.M.; Ramanculov, E.; Chain, P.S.; Chu, M.C.; Molineux, I.J. The genome sequence of Yersinia pestis bacteriophage phiA1122 reveals an intimate history with the coliphage T3 and T7 genomes. J. Bacteriol. 2003, 185, 5248–5262. [Google Scholar] [CrossRef]
- Kiljunen, S.; Datta, N.; Dentovskaya, S.V.; Anisimov, A.P.; Knirel, Y.A.; Bengoechea, J.A.; Holst, O.; Skurnik, M. Identification of the lipopolysaccharide core of Yersinia pestis and Yersinia pseudotuberculosis as the receptor for bacteriophage phiA1122. J. Bacteriol. 2011, 193, 4963–4972. [Google Scholar] [CrossRef]
- Schofield, D.A.; Molineux, I.J.; Westwater, C. Diagnostic bioluminescent phage for detection of Yersinia pestis. J. Clin. Microbiol. 2009, 47, 3887–3894. [Google Scholar] [CrossRef]
- Schofield, D.A.; Molineux, I.J.; Westwater, C. Rapid identification and antibiotic susceptibility testing of Yersinia pestis using bioluminescent reporter phage. J. Microbiol Methods. 2012, 90, 80–82. [Google Scholar] [CrossRef]
- Filippov, A.A.; Sergueev, K.V.; He, Y.; Huang, X.Z.; Gnade, B.T.; Mueller, A.J.; Fernandez-Prada, C.M.; Nikolich, M.P. Bacteriophage-resistant mutants in Yersinia pestis: Identification of phage receptors and attenuation for mice. PLoS ONE 2011, 6, e25486. [Google Scholar] [CrossRef] [PubMed]
- Filippov, A.A.; Sergueev, K.V.; He, Y.; Nikolich, M.P. Bacteriophages capable of lysing Yersinia pestis and Yersinia pseudotuberculosis: Efficiency of plating tests and identification of receptors in Escherichia coli K-12. Adv. Exp. Med. Biol. 2012, 954, 123–134. [Google Scholar] [PubMed]
- Zhao, X.; Wu, W.; Qi, Z.; Cui, Y.; Yan, Y.; Guo, Z.; Wang, Z.; Wang, H.; Deng, H.; Xue, Y.; et al. The complete genome sequence and proteomics of Yersinia pestis phage Yep-phi. J. Gen. Virol. 2011, 92, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Rashid, M.H.; Revazishvili, T.; Dean, T.; Butani, A.; Verratti, K.; Bishop-Lilly, K.A.; Sozhamannan, S.; Sulakvelidze, A.; Rajanna, C. A Yersinia pestis-specific, lytic phage preparation significantly reduces viable Y. pestis on various hard surfaces experimentally contaminated with the bacterium. Bacteriophage 2012, 2, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Comeau, A.M.; Arbiol, C.; Krisch, H.M. Composite conserved promoter-terminator motifs (PeSLs) that mediate modular shuffling in the diverse T4-like myoviruses. Genome Biol. Evol. 2014, 6, 1611–1619. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yuan, Y.; Xi, H.; Dai, J.; Zhong, Y.; Lu, S.; Wang, T.; Yang, L.; Guan, Y.; Wang, P. The characteristics and genome analysis of the novel Y. pestis phage JC221. Virus Res. 2020, 283, 197982. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.; Pajunen, M.I.; Jun, J.W.; Skurnik, M. T4-like Bacteriophages Isolated from Pig Stools Infect Yersinia pseudotuberculosis and Yersinia pestis Using LPS and OmpF as Receptors. Viruses 2021, 13, 296. [Google Scholar] [CrossRef]
- Laird, M.W.; Kloser, A.W.; Misra, R. Assembly of LamB and OmpF in deep rough lipopolysaccharide mutants of Escherichia coli K-12. J. Bacteriol. 1994, 176, 2259–2264. [Google Scholar] [CrossRef][Green Version]
- de Cock, H.; Tommassen, J. Lipopolysaccharides and divalent cations are involved in the formation of an assembly competent intermediate of outer-membrane protein PhoE of E. coli. EMBO J. 1996, 15, 5567–5573. [Google Scholar] [CrossRef]
- de Cock, H.; Pasveer, M.; Tommassen, J.; Bouveret, E. Identification of phospholipids as new components that assist in the in vitro trimerization of a bacterial pore protein. Eur. J. Biochem. 2001, 268, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, M.; Dowhan, W. Lipid-assisted protein folding. J. Biol. Chem. 1999, 274, 36827–36830. [Google Scholar] [CrossRef]
- Bogdanov, M. Lipid-dependent membrane protein topogenesis. Annu. Rev. Biochem. 2009, 78, 515–540. [Google Scholar] [CrossRef]
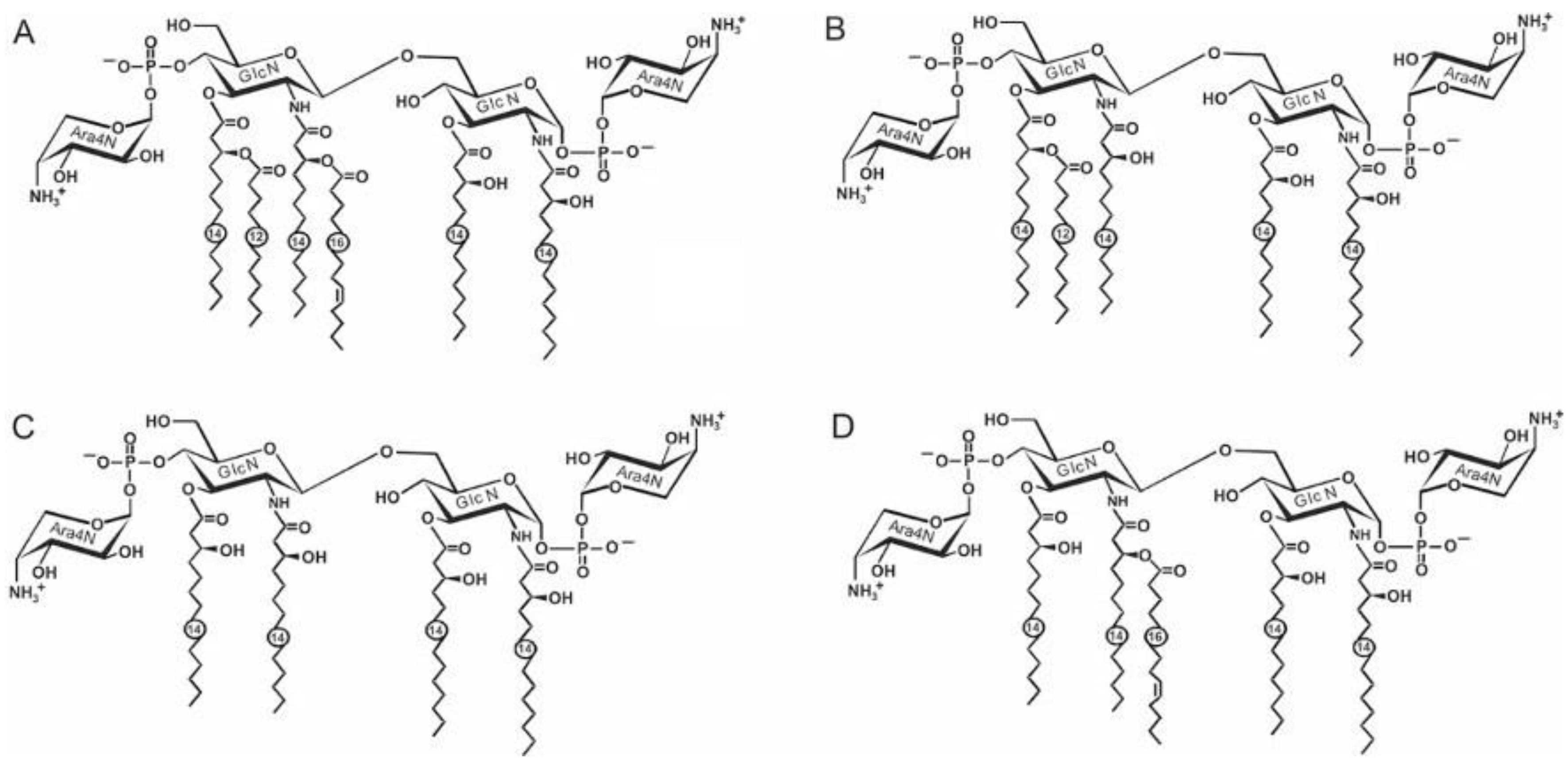

| Species, Growth Temperature [Reference] | Major Structure |
|---|---|
| Y. pseudotuberculosis/Y. pestis ssp. pestis 37 °C [28] LPS-37 |  |
| Y. pestis ssp. microti bvv. caucasica, altaica 37 °C [5] LPS-37 | 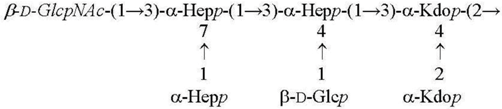 |
| Y. pseudotuberculosis/Y. pestis ssp. pestis 25 °C [28] LPS-25 | 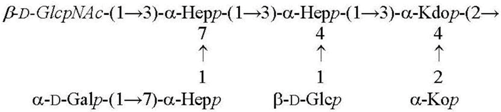 |
| Y. pestis ssp. pestis 6 °C [29] LPS-6 |  |
| Gene Y. pestis CO92 | Related Proteins (% identity Y. pseudotuberculosis/E. coli) | Proposed Function |
|---|---|---|
| Biosynthesis | ||
| lpxA/YPO1056 | 100% Y. pseudotuberculosis CAH22229.1/82% E. coli BAA77856.2 | UDP-N-acetylglucosamine acetyltransferase |
| lpxC/YPO0561 | 100% Y. pseudotuberculosis CAH19934.1/93% E. coli NP_414638.1 | UDP-3-O-acyl-N-acetylglucosamine deacetylase |
| lpxD/YPO1054 | 100% Y. pseudotuberculosis CAH22231.1/83% E. coli NP_414721.1 | UDP-3-O-(3-hydroxymyristoyl)glucosamine N-acyltransferase |
| lpxH/YPO3075 | 100% Y. pseudotuberculosis CAH22231.1/70% E. coli NP_415057.1 | UDP-2,3-diacylglucosamine diphosphatase |
| lpxB/YPO1057 | 100% Y. pseudotuberculosis CAH20273.1/82% E. coli NP_414724.1 | Lipid A disaccharide synthase |
| lpxK/YPO1396 | 100% Y. pseudotuberculosis CAH20661.1/70% E. coli NP_415435.1 | Tetraacyldisaccharide 4′-kinase |
| waaA (kdtA)/YPO0055 | 100% Y. pseudotuberculosis CAH19292.1/80% E. coli NP_418090.1 | Kdo transferase |
| lpxM/YPO2063 | 100% Y. pseudotuberculosis CAH21284.1/65% E. coli NP_416369.1 | Lauroyl acyltransferase |
| lpxP/YPO3632 | 100% Y. pseudotuberculosis CAH22835.1/67% E. coli NP_416879.4 | Palmitoleoyl acyltransferase |
| Structural Modification | ||
| pagP/YPO1744 | 100% Y. pseudotuberculosis CAH20861.1/53% E. coli NP_415155.1 | Palmitoyltransferase acyltransferase |
| arnT/YPO2418 | 100% Y. pseudotuberculosis CAH21564.1/54% E. coli ArnT NP_416760.1 | Lipid IVA 4-amino-4-deoxy-L-arabinosyltransferase |
| lpxT (yeiU)/YPO1276 | 99% Y. pseudotuberculosis CAH20550.1/62% E. coli NP_416679.4 | Kdo2-lipid A phosphotransferase |
| Gene Y. pestis CO92 | Related Proteins (% Identity Y. pseudotuberculosis/E. coli) | Proposed Function |
|---|---|---|
| CORE BIOSYNTHESIS GENE CLUSTER waaI | ||
| gmhD/YPO0058 | 100% Y. pseudotuberculosis CAH19295.1/83% E. coli BAE77673.1 | ADP-l,d-Heptose epimerase |
| waaF/YPO0057 | 99% Y. pseudotuberculosis CAH19294.1/74% E. coli NP_418077.1 | Heptosyltransferase (HepI) |
| waaC/YPO0056 | 100% Y. pseudotuberculosis CAH19293.1/68% E. coli NP_418078.1 | Heptosyltransferase (HepII) |
| waaA/YPO0055 | 100% Y. pseudotuberculosis CAH19292.1/80% E. coli NP_418090.1 | Kdo-transferase (KdoI, KdoII) |
| waaE/YPO054 | 100% Y. pseudotuberculosis CAH19291.1/76% Serratia marcescens AAC44433.1 | Glycosyltransferase (Glc) |
| CORE BIOSYNTHESIS GENE CLUSTER wab | ||
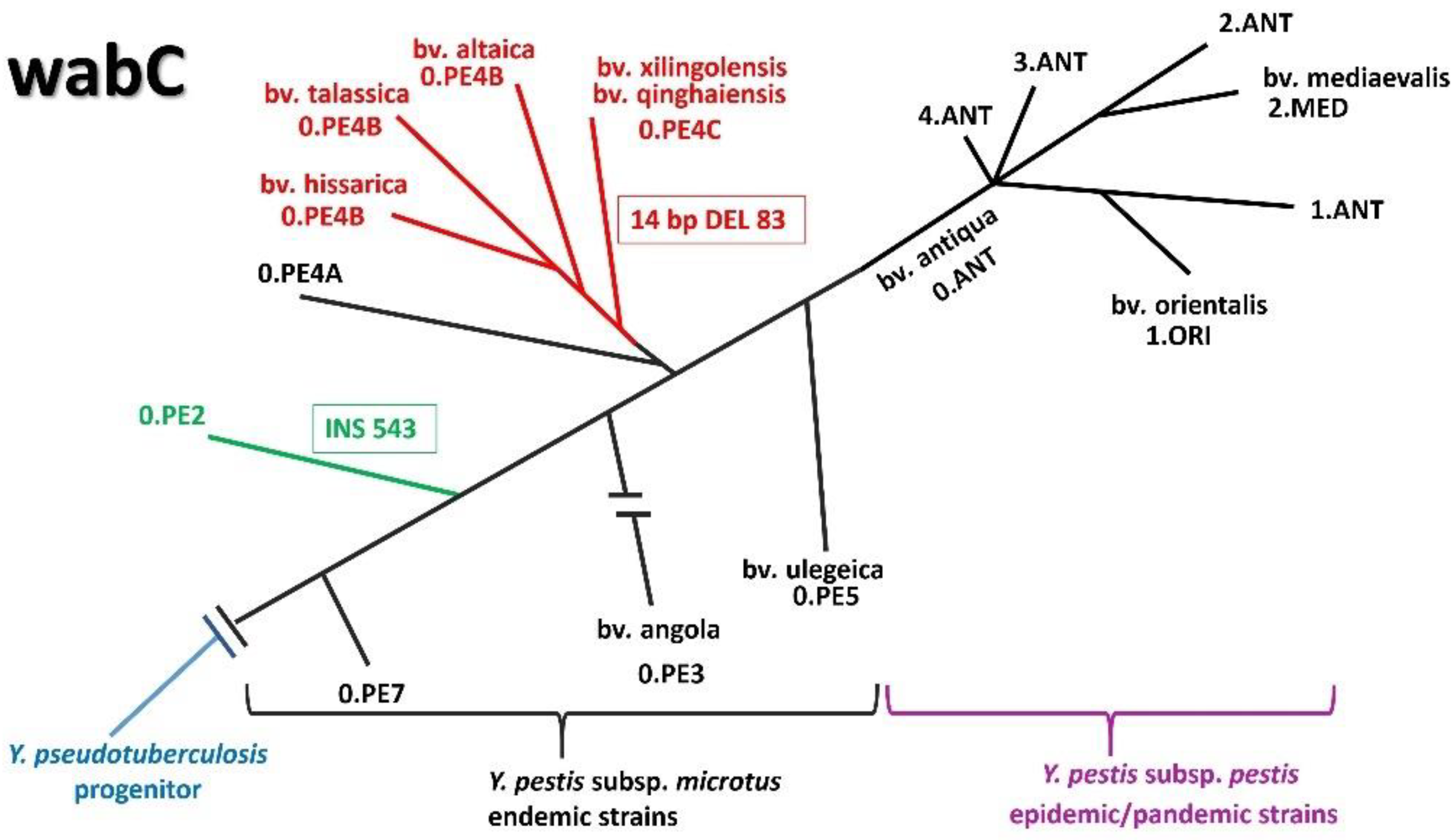
| wabC/YPO0186 | 99% Y. pseudotuberculosis CAH22956.1/61% Burkholderia pyrrocinia PXX29563.1 | Heptosyltransferase (HepIV) |
| wabD/YPO0187 | 99% Y. pseudotuberculosis CAH22955.1/44% Proteus mirabilis MBG3081359.1 | Glycosyltransferase (Gal) |
| CORE BIOSYNTHESIS GENE CLUSTER waaII | ||
| waaQ/YPO0416 | 99% Y. pseudotuberculosis CAH19795.1/40% E. coli NP_418089.1 | Heptosyltransferase (HepIII) |
| waaL/YPO0417 | 100% Y. pseudotuberculosis CAH19796.1/59% Serratia fonticola WP_074031921.1 | O-antigen ligase |
| structural modification | ||
| eptB/YPO4013 | 99% Y. pseudotuberculosis CAH23086.1/63% E. coli NP_418002.2 | Kdo2-lipid A phosphoethanolamine 7″-transferase |
| kdoO/YPO1650 | 99% Y. pseudotuberculosis CAH21657.1/69% Serratia marcescens ASL83593.1 | Kdo-3-hydroxylase |
| Group | Bacteriophage | Receptor | References |
|---|---|---|---|
| T7 | PhiA1122 | Kdo/Ko of LPS | [115,116,117,118] |
| YpP-Y | Hep(I)/Glc of LPS | [112,119,120] | |
| YpP-R | Beyond the LPS core | [119,120] | |
| Yep-phi | LPS; OmpF; Ail | [113,121] | |
| Pokrovskaya (Yepe2,YpP-G) | Hep(II)/Hep(III) of LPS | [120,121,122] | |
| T7Yp | Hep(I)/Glc of LPS | [112] | |
| P2 | L-413C | GlcNAc of LPS | [112,119,120] |
| P2 vir1 | GlcNAc of LPS | [112] | |
| T4 | PhiJA1 | Kdo/Ko of LPS | [112,119,120] |
| YpsP-PST | Hep(II)/Hep(III) of LPS | [112,119,120,123] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knirel, Y.A.; Anisimov, A.P.; Kislichkina, A.A.; Kondakova, A.N.; Bystrova, O.V.; Vagaiskaya, A.S.; Shatalin, K.Y.; Shashkov, A.S.; Dentovskaya, S.V. Lipopolysaccharide of the Yersinia pseudotuberculosis Complex. Biomolecules 2021, 11, 1410. https://doi.org/10.3390/biom11101410
Knirel YA, Anisimov AP, Kislichkina AA, Kondakova AN, Bystrova OV, Vagaiskaya AS, Shatalin KY, Shashkov AS, Dentovskaya SV. Lipopolysaccharide of the Yersinia pseudotuberculosis Complex. Biomolecules. 2021; 11(10):1410. https://doi.org/10.3390/biom11101410
Chicago/Turabian StyleKnirel, Yuriy A., Andrey P. Anisimov, Angelina A. Kislichkina, Anna N. Kondakova, Olga V. Bystrova, Anastasia S. Vagaiskaya, Konstantin Y. Shatalin, Alexander S. Shashkov, and Svetlana V. Dentovskaya. 2021. "Lipopolysaccharide of the Yersinia pseudotuberculosis Complex" Biomolecules 11, no. 10: 1410. https://doi.org/10.3390/biom11101410
APA StyleKnirel, Y. A., Anisimov, A. P., Kislichkina, A. A., Kondakova, A. N., Bystrova, O. V., Vagaiskaya, A. S., Shatalin, K. Y., Shashkov, A. S., & Dentovskaya, S. V. (2021). Lipopolysaccharide of the Yersinia pseudotuberculosis Complex. Biomolecules, 11(10), 1410. https://doi.org/10.3390/biom11101410








