Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects
Abstract
1. Introduction
2. Materials and Methods
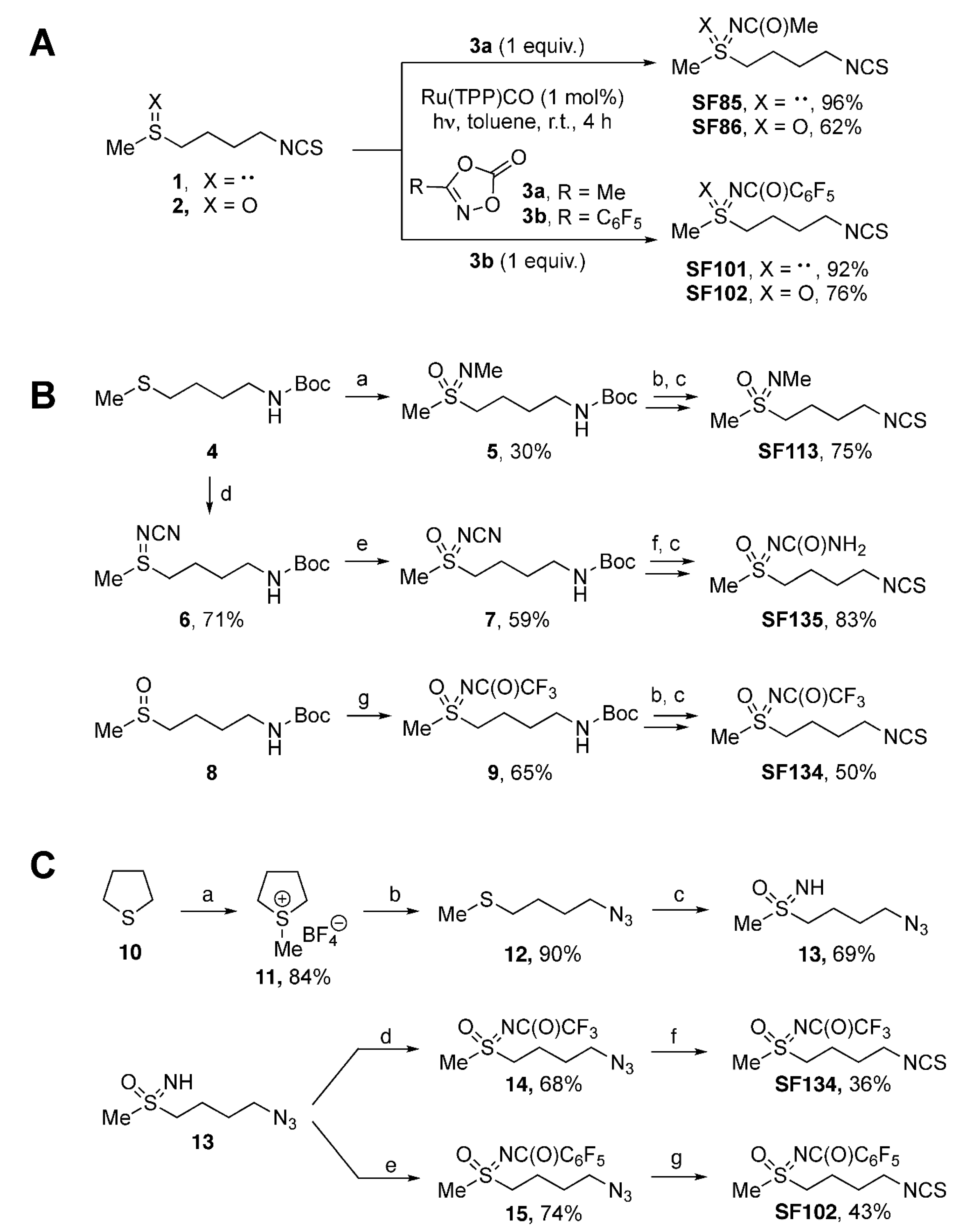
2.1. Chemical Syntheses of Sulforaphane Analogues
2.2. Human Tumor Cell Lines
2.3. Reagents
2.4. Cell Viability Assay
2.5. Colony Forming Assay
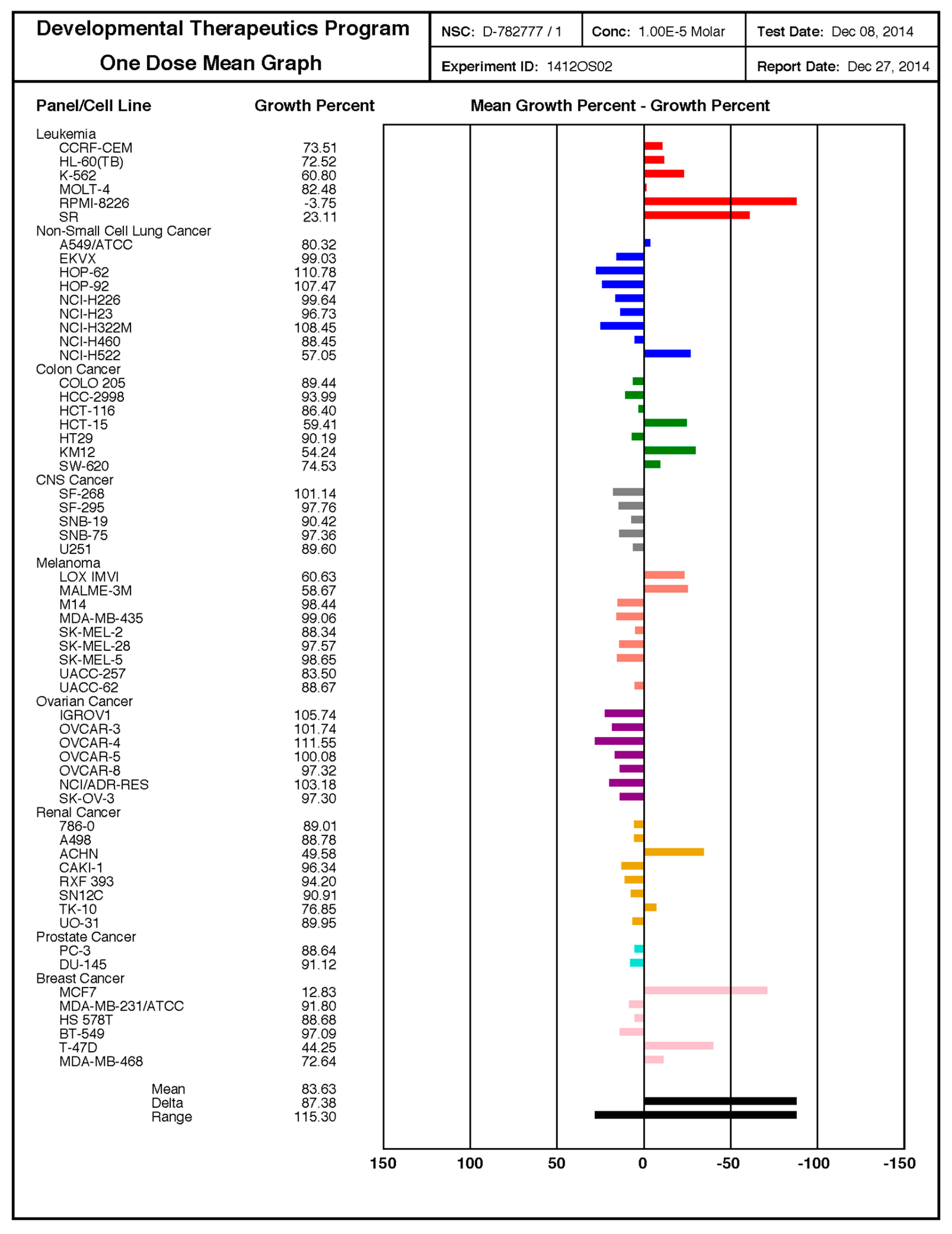
2.6. NCI-60 Human Tumor Cell Lines Screen
2.7. Tumor Xenotransplantation on Fertilized Chicken Eggs
2.8. Immunohistochemical Staining
2.9. Caenorhabditis Elegans Survival
2.10. miRNA Microarray Profiling
2.11. miRNA In Silico Analysis
2.12. Statistical Analysis
3. Results
3.1. Chemical Synthesis of Sulforaphane Derivatives
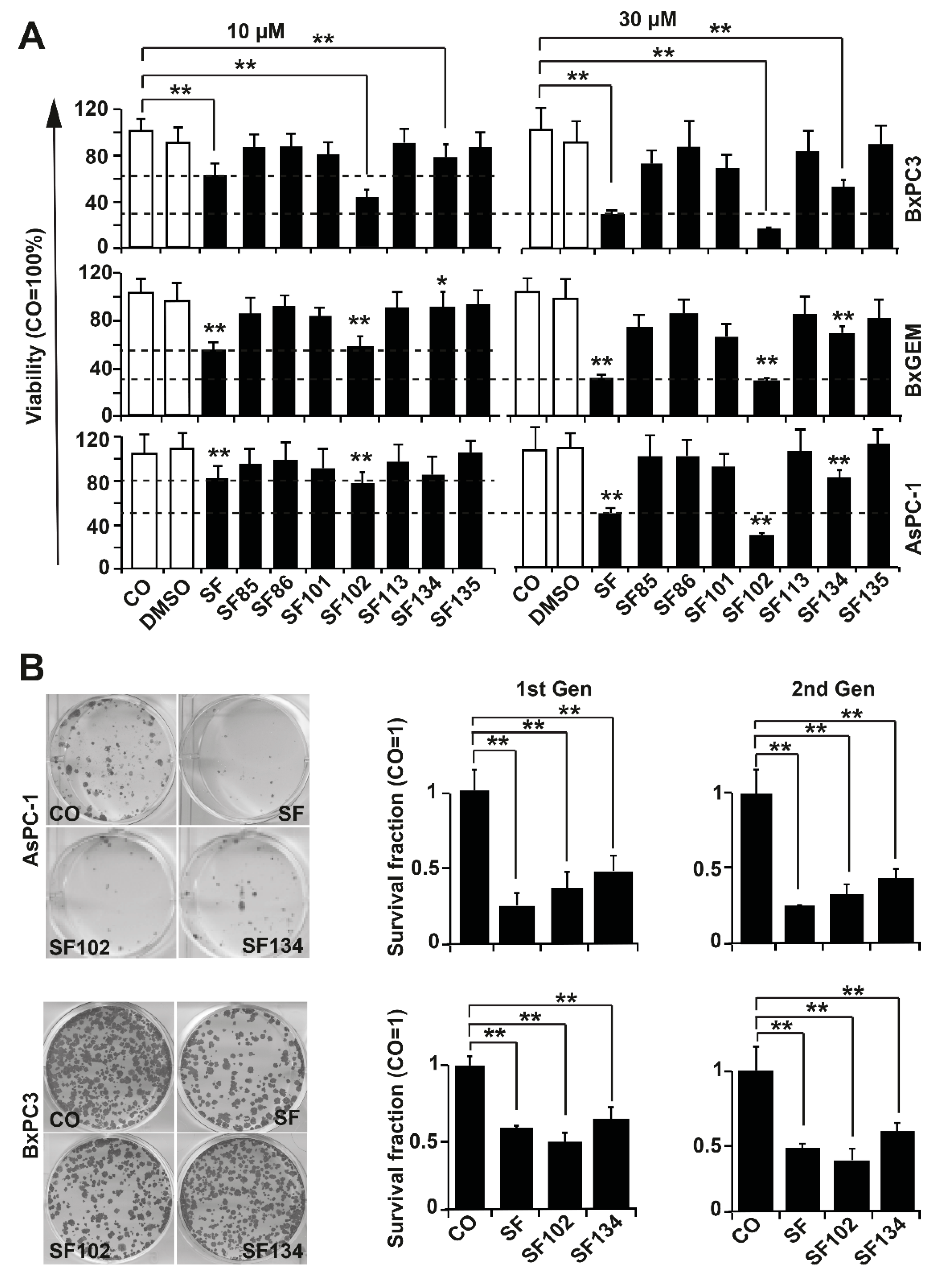
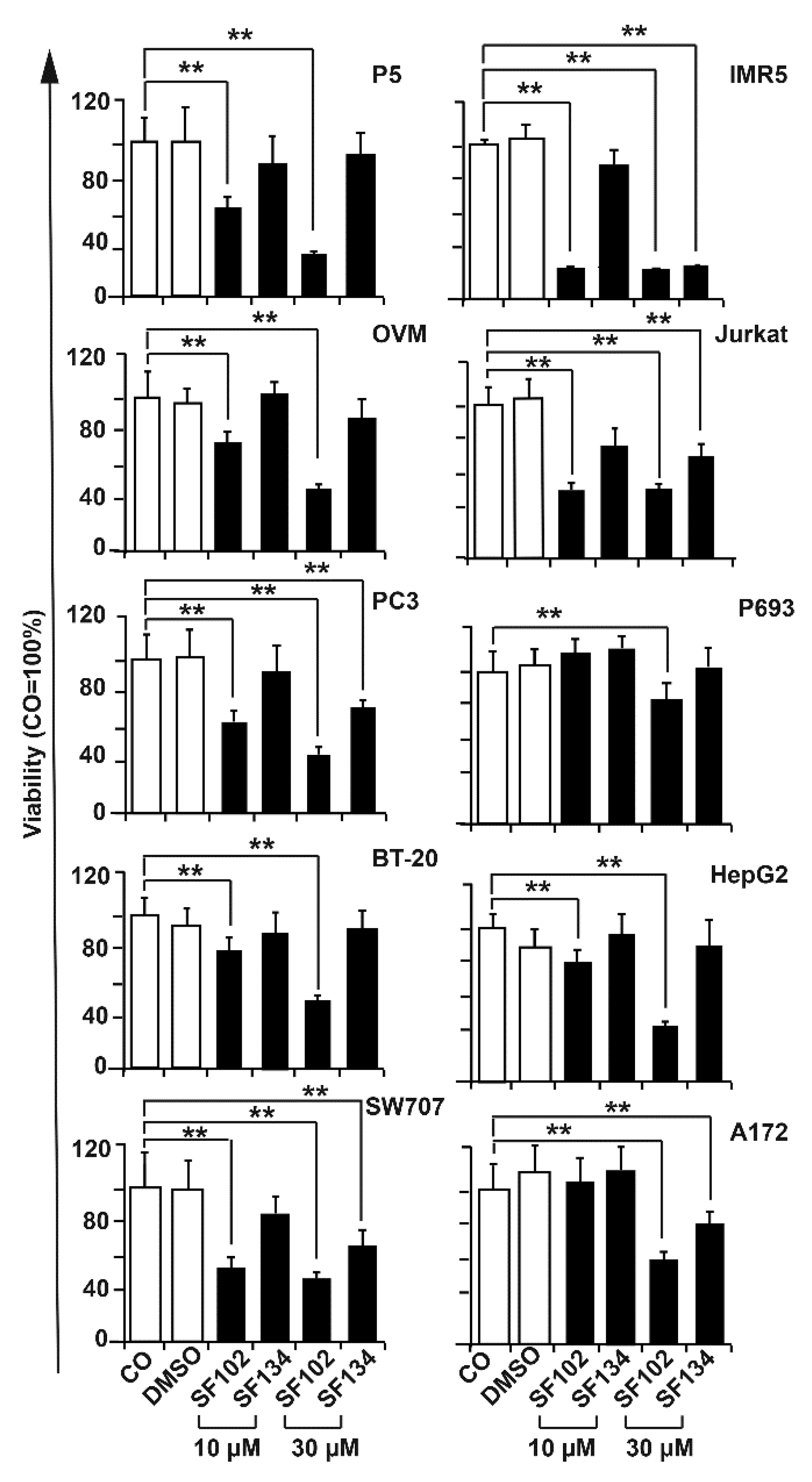
3.2. SF102 and SF134 Are Cytotoxic in All the Several Evaluated Tumor Cells
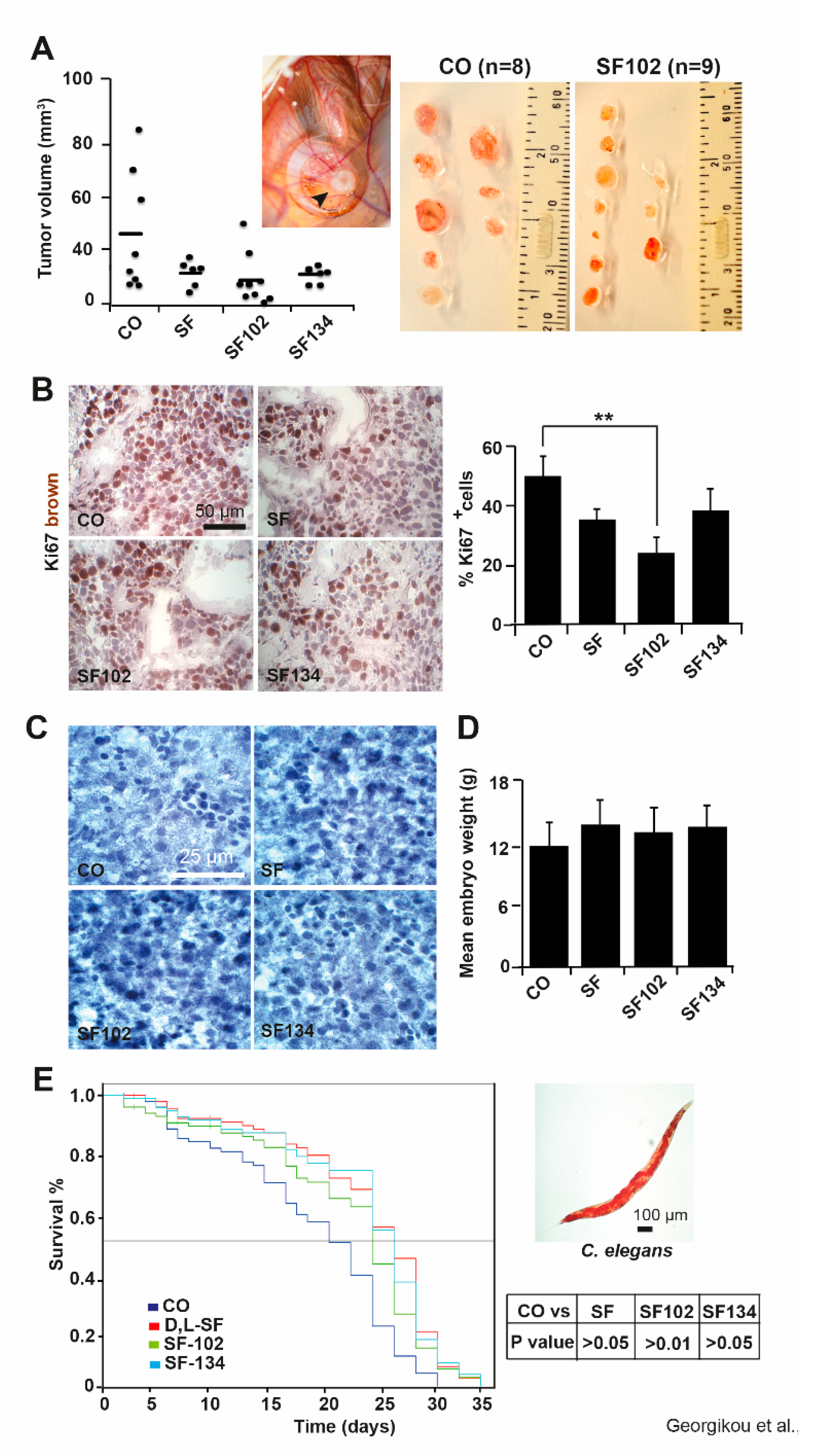
3.3. SF102 and SF134 Inhibit Tumor Growth without Obvious Side Effects
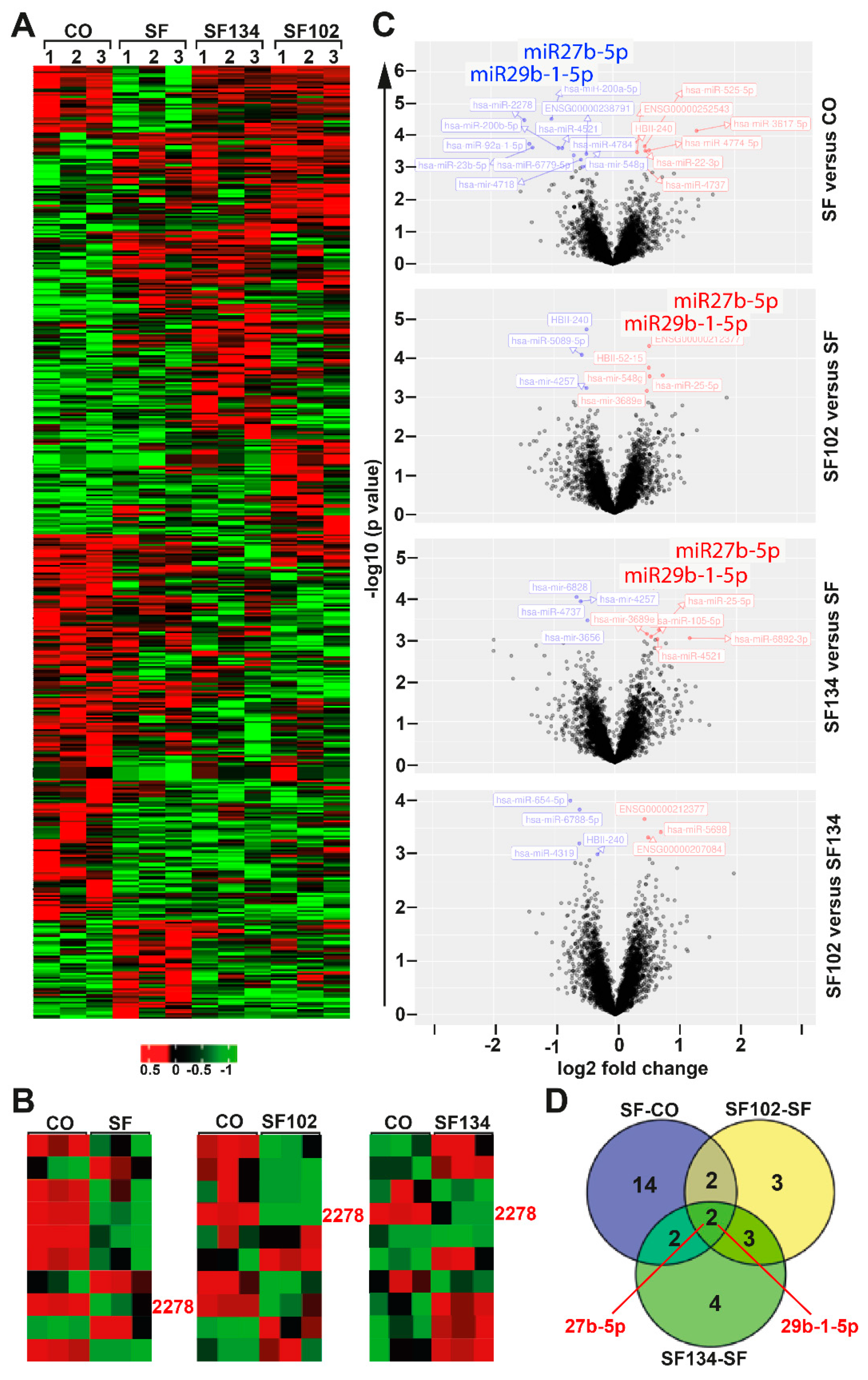
3.4. Sulforaphane, SF102 and SF134 Induce Differential microRNA Expression and NF-κB Signaling
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Neoptolemos, J.P.; Kleeff, J.; Michl, P.; Costello, E.; Greenhalf, W.; Palmer, D.H. Therapeutic developments in pancreatic cancer: Current and future perspectives. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Herr, I.; Lozanovski, V.; Houben, P.; Schemmer, P.; Buchler, M.W. Sulforaphane and related mustard oils in focus of cancer prevention and therapy. Wien. Med. Wochenschr. 2013, 163, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Lozanovski, V.J.; Polychronidis, G.; Gross, W.; Gharabaghi, N.; Mehrabi, A.; Hackert, T.; Schemmer, P.; Herr, I. Broccoli sprout supplementation in patients with advanced pancreatic cancer is difficult despite positive effects-results from the POUDER pilot study. Investig. New Drugs 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Alumkal, J.J.; Slottke, R.; Schwartzman, J.; Cherala, G.; Munar, M.; Graff, J.N.; Beer, T.M.; Ryan, C.W.; Koop, D.R.; Gibbs, A.; et al. A phase II study of sulforaphane-rich broccoli sprout extracts in men with recurrent prostate cancer. Investig. New Drugs 2015, 33, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Bayat Mokhtari, R.; Baluch, N.; Homayouni, T.S.; Morgatskaya, E.; Kumar, S.; Kazemi, P.; Yeger, H. The role of Sulforaphane in cancer chemoprevention and health benefits: A mini-review. J. Cell Commun. Signal. 2018, 12, 91–101. [Google Scholar] [CrossRef]
- Liou, C.S.; Sirk, S.J.; Diaz, C.A.C.; Klein, A.P.; Fischer, C.R.; Higginbottom, S.K.; Erez, A.; Donia, M.S.; Sonnenburg, J.L.; Sattely, E.S. A metabolic pathway for activation of dietary glucosinolates by a human gut symbiont. Cell 2020, 180, 717–728.e719. [Google Scholar] [CrossRef]
- Yin, L.; Xiao, X.; Georgikou, C.; Yin, Y.; Liu, L.; Karakhanova, S.; Luo, Y.; Gladkich, J.; Fellenberg, J.; Sticht, C.; et al. MicroRNA-365a-3p inhibits c-Rel-mediated NF-kappaB signaling and the progression of pancreatic cancer. Cancer Lett. 2019, 452, 203–212. [Google Scholar] [CrossRef]
- Kallifatidis, G.; Labsch, S.; Rausch, V.; Mattern, J.; Gladkich, J.; Moldenhauer, G.; Buchler, M.W.; Salnikov, A.V.; Herr, I. Sulforaphane increases drug-mediated cytotoxicity toward cancer stem-like cells of pancreas and prostate. Mol. Ther. 2011, 19, 188–195. [Google Scholar] [CrossRef]
- Posner, G.H.; Cho, C.G.; Green, J.V.; Zhang, Y.; Talalay, P. Design and synthesis of bifunctional isothiocyanate analogs of sulforaphane: Correlation between structure and potency as inducers of anticarcinogenic detoxication enzymes. J. Med. Chem. 1994, 37, 170–176. [Google Scholar] [CrossRef]
- Ahn, Y.H.; Hwang, Y.; Liu, H.; Wang, X.J.; Zhang, Y.; Stephenson, K.K.; Boronina, T.N.; Cole, R.N.; Dinkova-Kostova, A.T.; Talalay, P.; et al. Electrophilic tuning of the chemoprotective natural product sulforaphane. Proc. Natl. Acad. Sci. USA 2010, 107, 9590–9595. [Google Scholar] [CrossRef] [PubMed]
- Khiar, N.; Werner, S.; Mallouk, S.; Lieder, F.; Alcudia, A.; Fernandez, I. Enantiopure sulforaphane analogues with various substituents at the sulfinyl sulfur: Asymmetric synthesis and biological activities. J. Org. Chem. 2009, 74, 6002–6009. [Google Scholar] [CrossRef] [PubMed]
- Kielbasinski, P.; Luczak, J.; Cierpial, T.; Blaszczyk, J.; Sieron, L.; Wiktorska, K.; Lubelska, K.; Milczarek, M.; Chilmonczyk, Z. New enantiomeric fluorine-containing derivatives of sulforaphane: Synthesis, absolute configurations and biological activity. Eur. J. Med. Chem. 2014, 76, 332–342. [Google Scholar] [CrossRef]
- Lücking, U. Sulfoximines: A neglected opportunity in medicinal chemistry. Angew. Chem. Int. Ed. Engl. 2013, 52, 9399–9408. [Google Scholar] [CrossRef] [PubMed]
- Frings, M.; Bolm, C.; Blum, A.; Gnamm, C. Sulfoximines from a Medicinal Chemist’s Perspective: Physicochemical and In Vitro parameters relevant for drug discovery. Eur. J. Med. Chem. 2017, 126, 225–245. [Google Scholar] [CrossRef]
- Chinthakindi, P.K.; Naicker, T.; Thota, N.; Govender, T.; Kruger, H.G.; Arvidsson, P.I. Sulfonimidamides in medicinal and agricultural chemistry. Angew. Chem. Int. Ed. Engl. 2017, 56, 4100–4109. [Google Scholar] [CrossRef]
- Park, S.J.; Baars, H.; Mersmann, S.; Buschmann, H.; Baron, J.M.; Amann, P.M.; Czaja, K.; Hollert, H.; Bluhm, K.; Redelstein, R.; et al. N-cyano sulfoximines: COX inhibition, anticancer activity, cellular toxicity, and mutagenicity. ChemMedChem 2013, 8, 217–220. [Google Scholar] [CrossRef]
- Hendriks, C.M.M.; Hartkamp, J.; Wiezorek, S.; Steinkamp, A.D.; Rossetti, G.; Luscher, B.; Bolm, C. Sulfoximines as ATR inhibitors: Analogs of VE-821. Bioorg. Med. Chem. Lett. 2017, 27, 2659–2662. [Google Scholar] [CrossRef]
- Steinkamp, A.D.; Schmitt, L.; Chen, X.; Fietkau, K.; Heise, R.; Baron, J.M.; Bolm, C. Synthesis of a sulfonimidamide-based analog of tasisulam and its biological evaluation in the melanoma cell lines SKMel23 and A375. Ski. Pharm. Physiol. 2016, 29, 281–290. [Google Scholar] [CrossRef]
- Karpel-Massler, G.; Kast, R.E.; Siegelin, M.D.; Dwucet, A.; Schneider, E.; Westhoff, M.A.; Wirtz, C.R.; Chen, X.Y.; Halatsch, M.E.; Bolm, C. Anti-glioma activity of dapsone and its enhancement by synthetic chemical modification. Neurochem. Res. 2017, 42, 3382–3389. [Google Scholar] [CrossRef]
- Herr, I.; Ucur, E.; Herzer, K.; Okouoyo, S.; Ridder, R.; Krammer, P.H.; von Knebel Doeberitz, M.; Debatin, K.M. Glucocorticoid cotreatment induces apoptosis resistance toward cancer therapy in carcinomas. Cancer Res. 2003, 63, 3112–3120. [Google Scholar] [PubMed]
- Fan, P.; Zhang, Y.; Liu, L.; Zhao, Z.; Yin, Y.; Xiao, X.; Bauer, N.; Gladkich, J.; Mattern, J.; Gao, C.; et al. Continuous exposure of pancreatic cancer cells to dietary bioactive agents does not induce drug resistance unlike chemotherapy. Cell Death Dis. 2016, 7, e2246. [Google Scholar] [CrossRef] [PubMed]
- Kallifatidis, G.; Rausch, V.; Baumann, B.; Apel, A.; Beckermann, B.M.; Groth, A.; Mattern, J.; Li, Z.; Kolb, A.; Moldenhauer, G.; et al. Sulforaphane targets pancreatic tumour-initiating cells by NF-kappaB-induced antiapoptotic signalling. Gut 2009, 58, 949–963. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Bauer, N.; Aleksandrowicz, E.; Yin, L.; Gladkich, J.; Gross, W.; Kaiser, J.; Hackert, T.; Strobel, O.; Herr, I. Intraductal papillary mucinous neoplasm of the pancreas rapidly xenografts in chicken eggs and predicts aggressiveness. Int. J. Cancer 2018, 142, 1440–1452. [Google Scholar] [CrossRef] [PubMed]
- Stiernagle, T. Maintenance of C. elegans. WormBook 2006, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Buglioni, L.; Bizet, V.; Bolm, C. Methionine and buthionine sulfoximines: Syntheses under mild and safe imidation/oxidation conditions. Adv. Synth. Catal. 2014, 356, 2209–2213. [Google Scholar] [CrossRef]
- Bizet, V.; Buglioni, L.; Bolm, C. Light-induced ruthenium-catalyzed nitrene transfer reactions: A photochemical approach towards N-acyl sulfimides and sulfoximines. Angew. Chem. Int. Ed. Engl. 2014, 53, 5639–5642. [Google Scholar] [CrossRef]
- Bizet, V.; Hendriks, C.M.; Bolm, C. Sulfur imidations: Access to sulfimides and sulfoximines. Chem. Soc. Rev. 2015, 44, 3378–3390. [Google Scholar] [CrossRef]
- Chen, X.; Li, Z.; Sun, X.; Ma, H.; Chen, X.; Ren, J.; Hu, K. New method for the synthesis of sulforaphane and related isothiocyanates. Synthesis 2011, 24, 3991–3996. [Google Scholar] [CrossRef]
- Dannenberg, C.A.; Fritze, L.; Krauskopf, F.; Bolm, C. Access to N-cyanosulfoximines by transition metal-free iminations of sulfoxides. Org. Biomol. Chem. 2017, 15, 1086–1090. [Google Scholar] [CrossRef]
- Munch, H.; Hansen, J.S.; Pittelkow, M.; Christnsen, J.B.; Boas, U. A new efficient synthesis of isothiocyanates from amines using di-tert-butyl dicarbonate. Tetrahedron Lett. 2008, 49, 3117–3119. [Google Scholar] [CrossRef]
- Mancheño, O.G.; Bolm, C. Synthesis of N-(1H)-tetrazole sulfoximines. Org. Lett. 2007, 9, 2951–2954. [Google Scholar] [CrossRef] [PubMed]
- Barry, N.; Brondel, N.; Lawrence, S.E.; Maguire, A.R. Synthesis of aryl benzyl NH-sulfoximines. Tetrahedron 2009, 65, 10660–10670. [Google Scholar] [CrossRef]
- Mancheño, O.G.; Dallimore, J.; Plant, A.; Bolm, C. Synthesis of sulfoximines and sulfilimines with aryl and pyrazolylmethyl substituents. Adv. Synth. Catal. 2010, 352, 309–316. [Google Scholar] [CrossRef]
- Okamura, H.; Bolm, C. Rhodium-catalyzed imination of sulfoxides and sulfides: Efficient preparation of N-unsubstituted sulfoximines and sulfilimines. Org. Lett. 2004, 6, 1305–1307. [Google Scholar] [CrossRef]
- Vo, D.-V.; Truong, V.-D.; Tran, T.-D.; Do, V.-T.-N.; Pham, N.-T.-A.; Thai, K.-M. A new and effective approach to the synthesis of sulforaphane. Lett. Org. Chem. 2016, 13, 7–10. [Google Scholar] [CrossRef]
- Zenzola, M.; Doran, R.; Degennaro, L.; Luisi, R.; Bull, J.A. Transfer of electrophilic NH using convenient sources of ammonia: Direct synthesis of NH sulfoximines from sulfoxides. Angew. Chem. Int. Ed. Engl. 2016, 55, 7203–7207. [Google Scholar] [CrossRef]
- Hu, K.; Qi, Y.J.; Zhao, J.; Jiang, H.F.; Chen, X.; Ren, J. Synthesis and biological evaluation of sulforaphane derivatives as potential antitumor agents. Eur. J. Med. Chem. 2013, 64, 529–539. [Google Scholar] [CrossRef]
- Shi, Y.H.; Dai, D.F.; Li, J.; Dong, Y.W.; Jiang, Y.; Li, H.G.; Gao, Y.; Chong, C.K.; Li, H.Y.; Chu, X.Q.; et al. Sulforaphane analogues with heterocyclic moieties: Syntheses and inhibitory activities against cancer cell lines. Molecules 2016, 21, 514. [Google Scholar] [CrossRef]
- Kim, Y.J.; Hwang, K.C.; Kim, S.W.; Lee, Y.C. Potential miRNA-target interactions for the screening of gastric carcinoma development in gastric adenoma/dysplasia. Int. J. Med. Sci. 2018, 15, 610–616. [Google Scholar] [CrossRef]
- Kurihara-Shimomura, M.; Sasahira, T.; Shimomura, H.; Nakashima, C.; Kirita, T. The oncogenic activity of miR-29b-1-5p induces the epithelial-mesenchymal transition in oral squamous cell carcinoma. J. Clin. Med. 2019, 8, 273. [Google Scholar] [CrossRef] [PubMed]
- Kaymaz, B.T.; Gunel, N.S.; Ceyhan, M.; Cetintas, V.B.; Ozel, B.; Yandim, M.K.; Kipcak, S.; Aktan, C.; Gokbulut, A.A.; Baran, Y.; et al. Revealing genome-wide mRNA and microRNA expression patterns in leukemic cells highlighted “hsa-miR-2278” as a tumor suppressor for regain of chemotherapeutic imatinib response due to targeting STAT5A. Tumour Biol. 2015, 36, 7915–7927. [Google Scholar] [CrossRef] [PubMed]
- Nwaeburu, C.C.; Aleksandrowicz, E.; Bauer, N.; Zhao, Z.; Herr, I. MicroRNA In Vivo delivery to human pancreas tumor xenografts on chicken eggs. Protoc. Exch. 2017, 2017. [Google Scholar] [CrossRef]
- Kyriakakis, E.; Markaki, M.; Tavernarakis, N. Caenorhabditis elegans as a model for cancer research. Mol. Cell Oncol. 2015, 2, e975027. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgikou, C.; Buglioni, L.; Bremerich, M.; Roubicek, N.; Yin, L.; Gross, W.; Sticht, C.; Bolm, C.; Herr, I. Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects. Biomolecules 2020, 10, 769. https://doi.org/10.3390/biom10050769
Georgikou C, Buglioni L, Bremerich M, Roubicek N, Yin L, Gross W, Sticht C, Bolm C, Herr I. Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects. Biomolecules. 2020; 10(5):769. https://doi.org/10.3390/biom10050769
Chicago/Turabian StyleGeorgikou, Christina, Laura Buglioni, Maximilian Bremerich, Nico Roubicek, Libo Yin, Wolfgang Gross, Carsten Sticht, Carsten Bolm, and Ingrid Herr. 2020. "Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects" Biomolecules 10, no. 5: 769. https://doi.org/10.3390/biom10050769
APA StyleGeorgikou, C., Buglioni, L., Bremerich, M., Roubicek, N., Yin, L., Gross, W., Sticht, C., Bolm, C., & Herr, I. (2020). Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects. Biomolecules, 10(5), 769. https://doi.org/10.3390/biom10050769






