Biomarker Discovery for Cytochrome P450 1A2 Activity Assessment in Rats, Based on Metabolomics
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Animal Experiments and Sample Collection
2.3. Pharmacokinetic Studies
2.4. Determination of Cytochrome P450 1A2 mRNA Levels by Reverse Transcription Polymerase Chain Reaction
2.5. Untargeted Metabolomics Analysis
2.6. Data Quality Evaluation in Untargeted Metabolomics Analysis
2.7. Targeted Metabolomic Analysis of Branched-Chain Amino Acids, Phenylalanine, and Tyrosine
3. Results
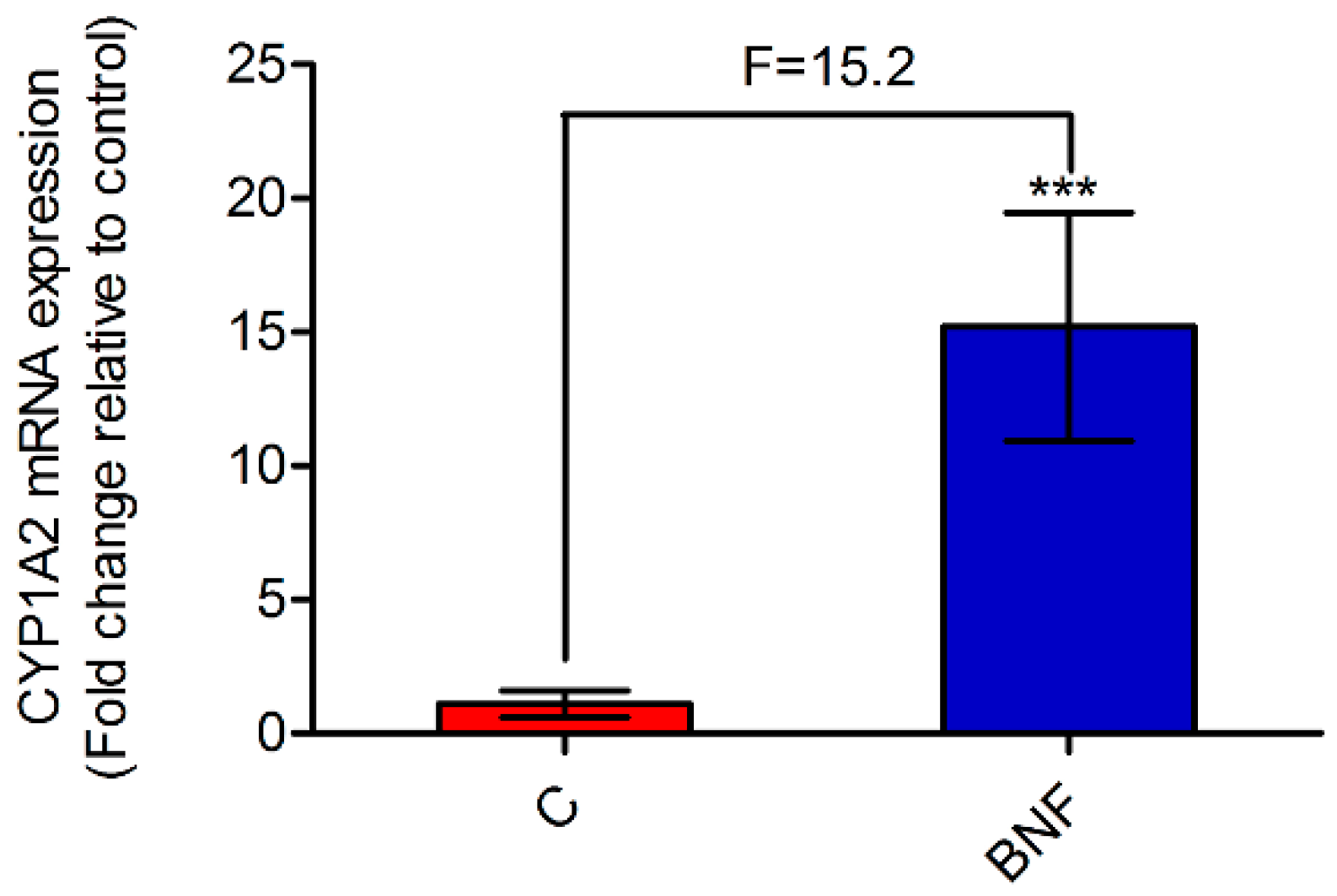
3.1. β-Naphthoflavone-Induced Cytochrome P450 1A2 Activity and mRNA Expression
3.2. Data Quality Evaluation in Untargeted Metabolomics
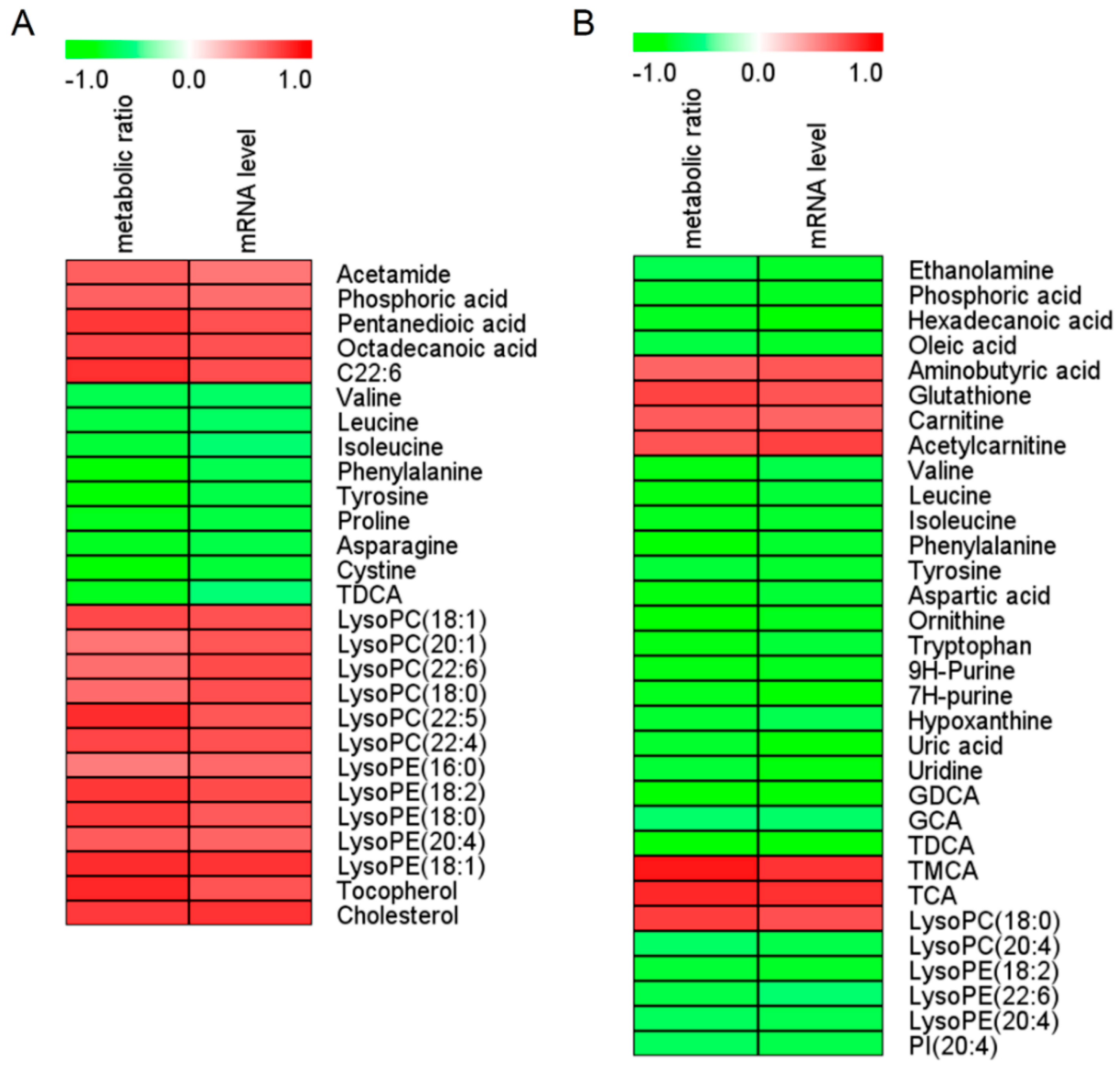
3.3. Biomarker Screening Based on Untargeted Metabolomic Analysis
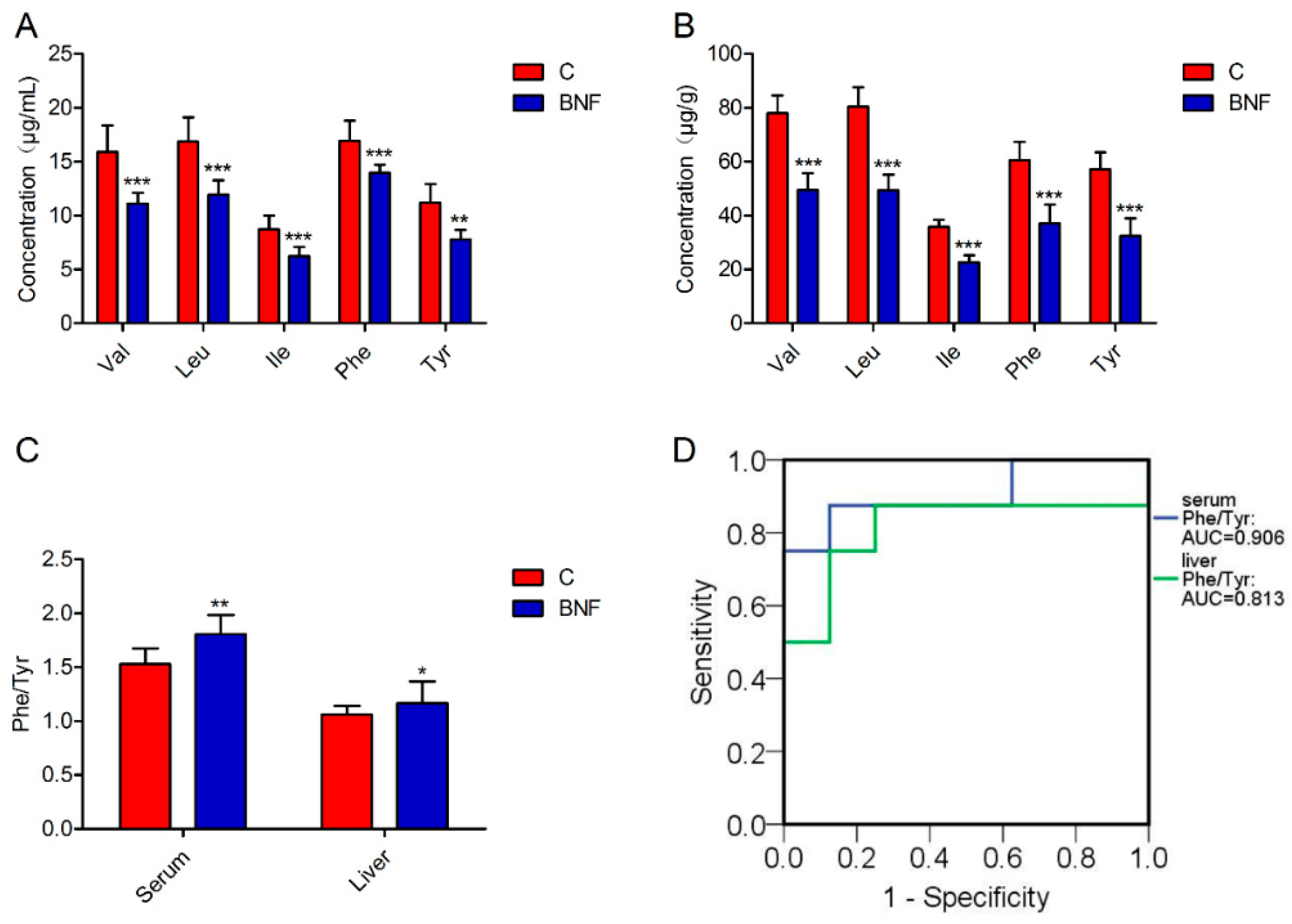
3.4. Targeted Quantification of Val, Leu, Ile, Phe and Tyr
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, J.F.; Chou, K.C. Molecular modeling of cytochrome P450 and drug metabolism. Curr. Drug Metab. 2010, 11, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Shimada, T.; Yamazaki, H.; Mimura, M.; Inui, Y.; Guengerich, F.P. Interindividual variations in human liver cytochrome P-450 enzymes involved in the oxidation of drugs, carcinogens and toxic chemicals: Studies with liver microsomes of 30 Japanese and 30 Caucasians. J. Pharmacol. Exp. Ther. 1994, 270, 414–423. [Google Scholar] [PubMed]
- Jiang, B.; Meng, L.; Zhang, F.; Jin, X.; Zhang, G. Enzyme-inducing effects of berberine on cytochrome P450 1A2 in vitro and in vivo. Life Sci. 2017, 189, 1–7. [Google Scholar] [CrossRef]
- Kim, D.; Guengerich, F.P. Cytochrome P450 activation of arylamines and heterocyclic amines. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 27. [Google Scholar] [CrossRef] [PubMed]
- Turesky, R.J.; Guengerich, F.P.; Guillouzo, A.; Langouët, S. Metabolism of heterocyclic aromatic amines by human hepatocytes and cytochrome P4501A2. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2002, 506, 187–195. [Google Scholar] [CrossRef]
- Zhou, S.F.; Liu, J.P.; Chowbay, B. Polymorphism of human cytochrome P450 enzymes and its clinical impact. Drug Metab. Rev. 2009, 41, 689–723. [Google Scholar] [CrossRef]
- Bozina, N.; Bradamante, V.; Lovrić, M. Genetic polymorphism of metabolic enzymes P450 (CYP) as a susceptibility factor for drug response, toxicity, and cancer risk. Arh. Hig. Rada Toksikol. 2009, 60, 217–242. [Google Scholar] [CrossRef] [PubMed]
- Peterson, S.; Schwarz, Y.; Li, S.S.; Li, L.; King, I.B.; Chen, C.; Eaton, D.L.; Potter, J.D.; Lampe, J.W. CYP1A2, GSTM1, and GSTT1 polymorphisms and diet effects on CYP1A2 activity in a crossover feeding trial. Cancer Epidemiol. Biomark. Prev. 2009, 18, 3118–3125. [Google Scholar] [CrossRef] [PubMed]
- Roza, G.; Magnus, C.; Hyung-Keun, R.; Magnus, I.S.; Eleni, A.; Leif, B. Comparisons of CYP1A2 genetic polymorphisms, enzyme activity and the genotype-phenotype relationship in Swedes and Koreans. Eur. J. Clin. Pharmacol. 2007, 63, 537–546. [Google Scholar]
- Gunes, A.; Ozbey, G.; Vural, E.H.; Uluoglu, C.; Scordo, M.G.; Zengil, H.; Dahl, M.L. Influence of genetic polymorphisms, smoking, gender and age on CYP1A2 activity in a Turkish population. Pharmacogenomics 2009, 10, 769–778. [Google Scholar] [CrossRef]
- B’Chir, F.; Pavanello, S.; Knani, J.; Boughattas, S.; Arnaud, M.J.; Saguem, S. CYP1A2 genetic polymorphisms and adenocarcinoma lung cancer risk in the Tunisian population. Life Sci. 2009, 84, 779–784. [Google Scholar] [CrossRef]
- Pavanello, S.; Fedeli, U.; Mastrangelo, G.; Rota, F.; Overvad, K.; Raaschou-Nielsen, O.; Tjønneland, A.; Vogel, U. Role of CYP1A2 polymorphisms on lung cancer risk in a prospective study. Cancer Genet. 2012, 205, 278–284. [Google Scholar] [CrossRef]
- Pavanello, S.; Mastrangelo, G.; Placidi, D.; Campagna, M.; Pulliero, A.; Carta, A.; Arici, C.; Porru, S. CYP1A2 polymorphisms, occupational and environmental exposures and risk of bladder cancer. Eur. J. Epidemiol. 2010, 25, 491–500. [Google Scholar] [CrossRef]
- Ghoshal, U.; Tripathi, S.; Kumar, S.; Mittal, B.; Chourasia, D.; Kumari, N.; Krishnani, N.; Ghoshal, U.C. Genetic polymorphism of cytochrome P450 (CYP) 1A1, CYP1A2, and CYP2E1 genes modulate susceptibility to gastric cancer in patients with Helicobacter pylori infection. Gastric Cancer 2014, 17, 226–234. [Google Scholar] [CrossRef]
- Moonen, H.; Engels, L.; Kleinjans, J.; Td, K. The CYP1A2-164A-->C polymorphism (CYP1A2*1F) is associated with the risk for colorectal adenomas in humans. Cancer Lett. 2005, 229, 25–31. [Google Scholar] [CrossRef]
- Bethke, L.; Webb, E.; Sellick, G.; Rudd, M.; Penegar, S.; Withey, L.; Qureshi, M.; Houlston, R. Polymorphisms in the cytochrome P450 genes CYP1A2, CYP1B1, CYP3A4, CYP3A5, CYP11A1, CYP17A1, CYP19A1 and colorectal cancer risk. BMC Cancer 2007, 7, 123. [Google Scholar] [CrossRef]
- De, A.F.; Llerena, A. Simultaneous determination of cytochrome P450 oxidation capacity in humans: A review on the phenotyping cocktail approach. Curr. Pharm. Biotechnol. 2016, 17, 1159–1180. [Google Scholar]
- Faber, M.S.; Jetter, A.U. Assessment of CYP1A2 activity in clinical practice: Why, how, and when? Basic Clin. Pharmacol Toxicol. 2010, 97, 125–134. [Google Scholar] [CrossRef]
- Diczfalusy, U.; Bertilsson, L.; Nylén, H.; Elander, P. 4β-hydroxycholesterol, an endogenous marker of CYP3A4/5 activity in humans. Br. J. Clin. Pharmacol. 2011, 71, 183–189. [Google Scholar] [CrossRef]
- Shin, K.H.; Li, Y.A.; Man, H.C.; Moon, J.Y.; Lee, J.; Jang, I.J.; Yu, K.S.; Cho, J.Y. Urinary 6β-hydroxycortisol/cortisol ratio most highly correlates with midazolam clearance under hepatic CYP3A inhibition and induction in females: A pharmacometabolomics approach. AAPS J. 2016, 18, 1–8. [Google Scholar] [CrossRef]
- Zhongmei, T.; Martin, M.V.; Guengerich, F.P. Elucidation of functions of human cytochrome P450 enzymes: Identification of endogenous substrates in tissue extracts using metabolomic and isotopic labeling approaches. Anal. Chem. 2009, 81, 3071–3078. [Google Scholar]
- Jie, C.; Chi, C.; Kristopher, K.W.; Manna, S.K.; Mike, S.; Friedman, F.K.; Hans, L.; Idle, J.R.; Gonzalez, F.J. Identification of 2-piperidone as a biomarker of CYP2E1 activity through metabolomic phenotyping. Toxicol. Sci. 2013, 135, 37–47. [Google Scholar]
- Tay-Sontheimer, J.; Shireman, L.M.; Beyer, R.P.; Senn, T.; Witten, D.; Pearce, R.E.; Gaedigk, A.; Fomban, C.L.G.; Lutz, J.D.; Isoherranen, N.; et al. Detection of an endogenous urinary biomarker associated with CYP2D6 activity using global metabolomics. Pharmacogenomics 2014, 15, 1947–1962. [Google Scholar]
- Deng, Y.; Bi, H.C.; Zhao, L.Z.; He, F.; Liu, Y.Q.; Yu, J.J.; Ou, Z.M.; Ding, L.; Chen, X.; Huang, Z.Y. Induction of cytochrome P450s by terpene trilactones and flavonoids of the Ginkgo biloba extract EGb 761 in rats. Xenobiotica 2008, 38, 465–481. [Google Scholar] [CrossRef]
- Geng, T.; Si, H.; Kang, D.; Li, Y.; Huang, W.; Gang, D.; Wang, Z.; Bi, Y.A.; Hong, Z.; Wei, X. Influences of Re Du Ning Injection, a traditional Chinese medicine injection, on the CYP450 activities in rats using a cocktail method. J. Ethnopharmacol. 2015, 174, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.M.; Temple, R.; Throckmorton, D.C.; Lesko, L.J. Drug interaction studies: Study design, data analysis, and implications for dosing and labeling. Clin. Pharmacol. Ther. 2007, 81, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C (T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Dai, D.; Tian, Y.; Guo, H.; Zhang, P.; Huang, Y.; Zhang, W.; Xu, F.; Zhang, Z. A pharmacometabonomic approach using predose serum metabolite profiles reveals differences in lipid metabolism in survival and non-survival rats treated with lipopolysaccharide. Metabolomics 2016, 12, 2. [Google Scholar] [CrossRef]
- Zhang, P.; Chen, J.Q.; Huang, W.Q.; Li, W.; Huang, Y.; Zhang, Z.J.; Xu, F.G. Renal medulla is more sensitive to cisplatin than cortex revealed by untargeted mass spectrometry-based metabolomics in rats. Sci. Rep. 2017, 7, 44804. [Google Scholar] [CrossRef]
- Gao, Y.Q.; Li, W.; Chen, J.Q.; Wang, X.; Lv, Y.T.; Huang, Y.; Zhang, Z.J.; Xu, F.G. Pharmacometabolomic prediction of individual differences of gastrointestinal toxicity complicating myelosuppression in rats induced by irinotecan. Acta Pharm. Sin. B 2019, 9, 157–166. [Google Scholar] [CrossRef]
- Broadhurst, D.I.; Kell, D.B. Statistical strategies for avoiding false discoveries in metabolomics and related experiments. Metabolomics 2006, 2, 171–196. [Google Scholar] [CrossRef]
- Mahadevan, S.; Shah, S.L.; Marrie, T.J.; Slupsky, C.M. Analysis of metabolomic data using support vector machines. Anal. Chem. 2008, 80, 7562–7570. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Liu, P.; Liu, P.; Yuan, T.; Hua, Y.; Gao, Y.; Hua, H.; Chen, J.; Zhang, Z.; Yin, H. A novel liquid chromatography tandem mass spectrometry method for simultaneous determination of branched-chain amino acids and branched-chain α-keto acids in human plasma. Amino Acids 2016, 48, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Angel, M.J.; García-Alvarez-Coque, M.C.; Berthod, A.; Carda-Broch, S. Are analysts doing method validation in liquid chromatography? J. Chromatogr. A 2014, 1353, 2–9. [Google Scholar] [CrossRef]
- González, O.; Blanco, M.E.; Iriarte, G.; Bartolomé, L.; Maguregui, M.I.; Alonso, R.M. Bioanalytical chromatographic method validation according to current regulations, with a special focus on the non-well defined parameters limit of quantification, robustness and matrix effect. J. Chromatogr. A 2014, 1353, 10–27. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.N.; Wang, X.; Chen, J.Q.; Lv, B.; Zhang, P.; Zhang, W.; Zhang, Z.J.; Xu, F.G. Quantitative evaluation of the compatibility effects of huangqin decoction on the treatment of irinotecan-induced gastrointestinal toxicity using untargeted metabolomics. Front. Pharmacol. 2017, 8, 211. [Google Scholar] [CrossRef]
- Marcella, M.; Groothuis, G.M.M.; Ruben, D.K. Species differences between mouse, rat, dog, monkey and human CYP-mediated drug metabolism, inhibition and induction. Expert Opin. Drug Metab. Toxicol. 2006, 2, 875–894. [Google Scholar]
- Meibohm, B.; Beierle, I.; Derendorf, H. How important are gender differences in pharmacokinetics? Clin. Pharmacokinet. 2002, 41, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Gunes, A.; Dahl, M.L. Variation in CYP1A2 activity and its clinical implications: Influence of environmental factors and genetic polymorphisms. Pharmacogenomics 2008, 9, 625–637. [Google Scholar] [CrossRef]
- Kaoru, K.; Kikuko, U.; Noriaki, S.; Kan, C. Substrate specificity for rat cytochrome P450 (CYP) isoforms: Screening with cDNA-expressed systems of the rat. Biochem. Pharmacol. 2002, 63, 889–896. [Google Scholar]
- Gutiérrez-Vázquez, C.; Quintana, F.J. Regulation of the immune response by the aryl hydrocarbon receptor. Immunity 2018, 48, 19–33. [Google Scholar] [CrossRef]
- Chen, E.P.; Chen, L.; Ji, Y.; Tai, G.; Wen, Y.H.; Ellens, H. A mechanism-based mathematical model of aryl hydrocarbon receptor-mediated CYP1A induction in rats using beta-naphthoflavone as a tool compound. Drug Metab. Dispos. 2010, 38, 2278–2285. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.F.; Yang, L.P.; Zhou, Z.W.; Liu, Y.H.; Chan, E. Insights into the substrate specificity, inhibitors, regulation, and polymorphisms and the clinical impact of human cytochrome P450 1A2. AAPS J. 2009, 11, 481–494. [Google Scholar] [CrossRef] [PubMed]
- Mayers, J.R.; Wu, C.; Clish, C.B.; Kraft, P.; Torrence, M.E.; Fiske, B.P.; Yuan, C.; Bao, Y.; Townsend, M.K.; Tworoger, S.S.; et al. Elevation of circulating branched-chain amino acids is an early event in human pancreatic adenocarcinoma development. Nat. Med. 2014, 20, 1193–1198. [Google Scholar] [CrossRef] [PubMed]
- Mayers, J.R.; Torrence, M.E.; Danai, L.V.; Papagiannakopoulos, T.; Davidson, S.M.; Bauer, M.R.; Lau, A.N.; Ji, B.W.; Dixit, P.D.; Hosios, A.M. Tissue-of-origin dictates branched-chain amino acid metabolism in mutant kras-driven cancers. Science 2016, 353, 1161. [Google Scholar] [CrossRef]
- Ruiz-Canela, M.; Toledo, E.; Clish, C.B.; Hruby, A.; Liang, L.; Salas-Salvadó, J.; Razquin, C.; Corella, D.; Estruch, R.; Ros, E. Plasma branched-chain amino acids and incident cardiovascular disease in the PREDIMED trial. Clin. Chem. 2016, 62, 582. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 2016, 535, 376. [Google Scholar] [CrossRef]
- Hirayama, M.; Tsunoda, M.; Yamamoto, M.; Tsuda, T.; Ohno, K. Serum tyrosine-to-phenylalanine ratio is low in Parkinson’s disease. J. Park. Dis. 2016, 6, 423–431. [Google Scholar] [CrossRef]
- Neurauter, G.; Grahmann, A.V.; Klieber, M.; Zeimet, A.; Ledochowski, M.; Sperner-Unterweger, B.; Fuchs, D. Serum phenylalanine concentrations in patients with ovarian carcinoma correlate with concentrations of immune activation markers and of isoprostane-8. Cancer Lett. 2008, 272, 141–147. [Google Scholar] [CrossRef]
- Ploder, M.; Neurauter, G.; Spittler, A.; Schroecksnadel, K.; Roth, E.; Fuchs, D. Serum phenylalanine in patients post trauma and with sepsis correlate to neopterin concentrations. Amino Acids 2008, 35, 303–307. [Google Scholar] [CrossRef]
- Kopple, J.D. Phenylalanine and tyrosine metabolism in chronic kidney failure. J. Nutr. 2007, 137. [Google Scholar] [CrossRef] [PubMed]
- Strasser, B.; Sperner-Unterweger, B.; Fuchs, D.; Gostner, J.M. Mechanisms of inflammation-associated depression: Immune influences on tryptophan and phenylalanine metabolisms. Curr. Top. Behav. Neurosci. 2016, 31, 95. [Google Scholar]
| Gene | Sequence (5′-3′) | Product Size (bp) |
|---|---|---|
| CYP1a2 | Forward: GCCATCTTCTGGAGCATTTTG | 208 |
| Reverse: TGTCCCTCGTTGTGCTGTG | ||
| β-actin | Forward: GGAGATTACTGCCCTGGCTCCTA | 150 |
| Reverse: GACTCATCGTACTCCTGCTTGCTG |
| Parameter | C Group | BNF Group |
|---|---|---|
| AUC0-12h (μg/L·h) | 3290.08 ± 302.66 | 105.63 ± 66.41 *** |
| Cmax (μg/L) | 4469.16 ± 331.91 | 313.84 ± 229.00 *** |
| T1/2 (h) | 0.47 ± 0.37 | 0.32 ± 0.19 |
| Metabolic ratio a | 0.77 ± 0.11 | 34.58 ± 22.02 ** |
| NO. | Metabolites | VIP Value | pFDR | r Value (Metabolic Ratio) a | r Value (mRNA level) b | AUC-ROC | Change Trend c | Detected From | Detected By |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Valine | 1.21 | 0.012 | −0.63 | −0.56 | 0.89 | ↓ | serum | GC-MS |
| 1.57 | 0.000 | −0.75 | −0.64 | 1.00 | ↓ | liver | GC-MS | ||
| 2 | Leucine | 1.27 | 0.006 | −0.66 | −0.57 | 0.92 | ↓ | serum | GC-MS |
| 1.60 | 0.000 | −0.76 | −0.68 | 1.00 | ↓ | liver | GC-MS | ||
| 3 | Isoleucine | 1.12 | 0.009 | −0.68 | −0.52 | 0.91 | ↓ | serum | GC-MS |
| 1.60 | 0.000 | −0.74 | −0.71 | 1.00 | ↓ | liver | GC-MS | ||
| 4 | Phenylalanine | 1.07 | 0.007 | −0.77 | −0.62 | 0.94 | ↓ | serum | LC-MS |
| 1.52 | 0.000 | −0.78 | −0.71 | 1.00 | ↓ | liver | GC-MS | ||
| 5 | Tyrosine | 1.26 | 0.006 | −0.79 | −0.65 | 0.92 | ↓ | serum | GC-MS |
| 1.55 | 0.000 | −0.68 | −0.71 | 1.00 | ↓ | liver | GC-MS | ||
| 6 | TDCA | 1.98 | 0.028 | −0.74 | −0.51 | 0.84 | ↓ | serum | LC-MS |
| 4.56 | 0.000 | −0.77 | −0.77 | 0.98 | ↓ | liver | LC-MS | ||
| 7 | LysoPC(18:0) | 1.58 | 0.005 | 0.58 | 0.69 | 0.94 | ↑ | serum | LC-MS |
| 1.19 | 0.001 | 0.75 | 0.69 | 0.97 | ↑ | liver | LC-MS |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pu, X.; Gao, Y.; Li, R.; Li, W.; Tian, Y.; Zhang, Z.; Xu, F. Biomarker Discovery for Cytochrome P450 1A2 Activity Assessment in Rats, Based on Metabolomics. Metabolites 2019, 9, 77. https://doi.org/10.3390/metabo9040077
Pu X, Gao Y, Li R, Li W, Tian Y, Zhang Z, Xu F. Biomarker Discovery for Cytochrome P450 1A2 Activity Assessment in Rats, Based on Metabolomics. Metabolites. 2019; 9(4):77. https://doi.org/10.3390/metabo9040077
Chicago/Turabian StylePu, Xiao, Yiqiao Gao, Ruiting Li, Wei Li, Yuan Tian, Zunjian Zhang, and Fengguo Xu. 2019. "Biomarker Discovery for Cytochrome P450 1A2 Activity Assessment in Rats, Based on Metabolomics" Metabolites 9, no. 4: 77. https://doi.org/10.3390/metabo9040077
APA StylePu, X., Gao, Y., Li, R., Li, W., Tian, Y., Zhang, Z., & Xu, F. (2019). Biomarker Discovery for Cytochrome P450 1A2 Activity Assessment in Rats, Based on Metabolomics. Metabolites, 9(4), 77. https://doi.org/10.3390/metabo9040077





