Fructose Alters Intermediary Metabolism of Glucose in Human Adipocytes and Diverts Glucose to Serine Oxidation in the One–Carbon Cycle Energy Producing Pathway
Abstract
:1. Introduction
2. Results
2.1. Fructose Dose-Dependently Increased the Oxidation of Glucose in Adipocytes
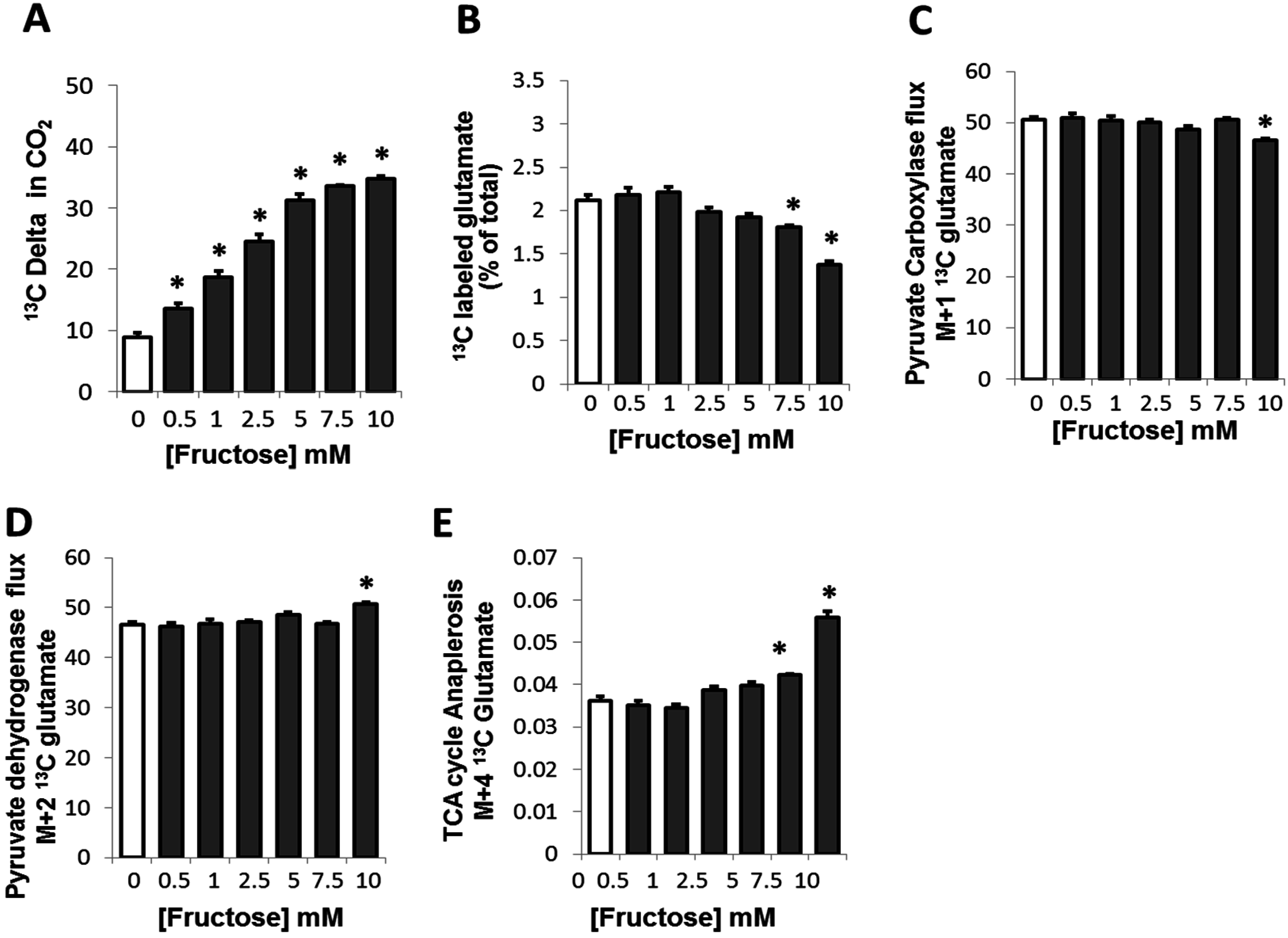
2.2. Fructose Altered Glucose Derived-Glutamate Output and Decreased the Cycling of Glucose Carbons through the TCA Cycle
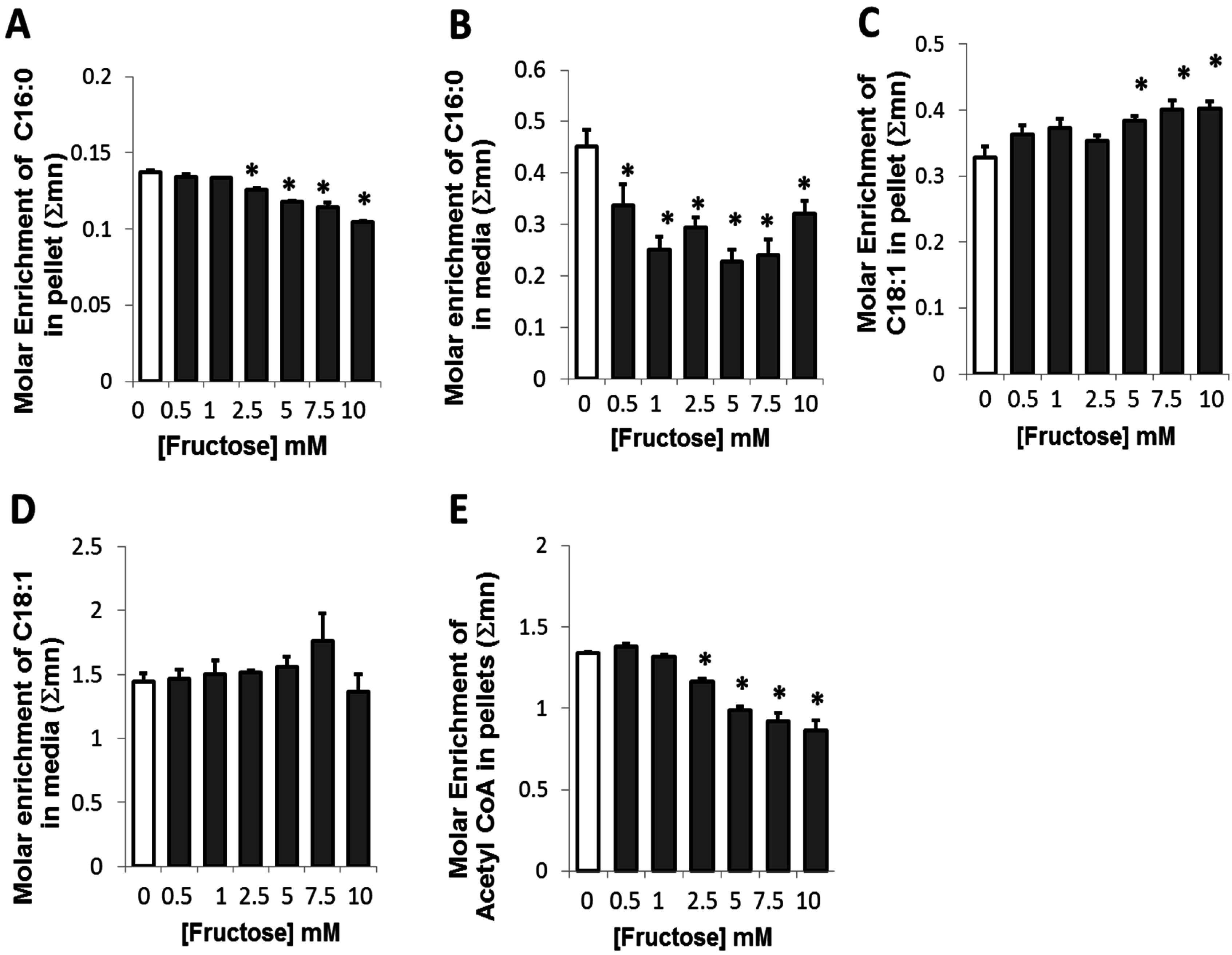
2.3. Presence of Fructose Decreased Glucose-Derived Palmitate Accumulation and Palmitate Release but Augmented Accumulation of Oleate in Adipocytes
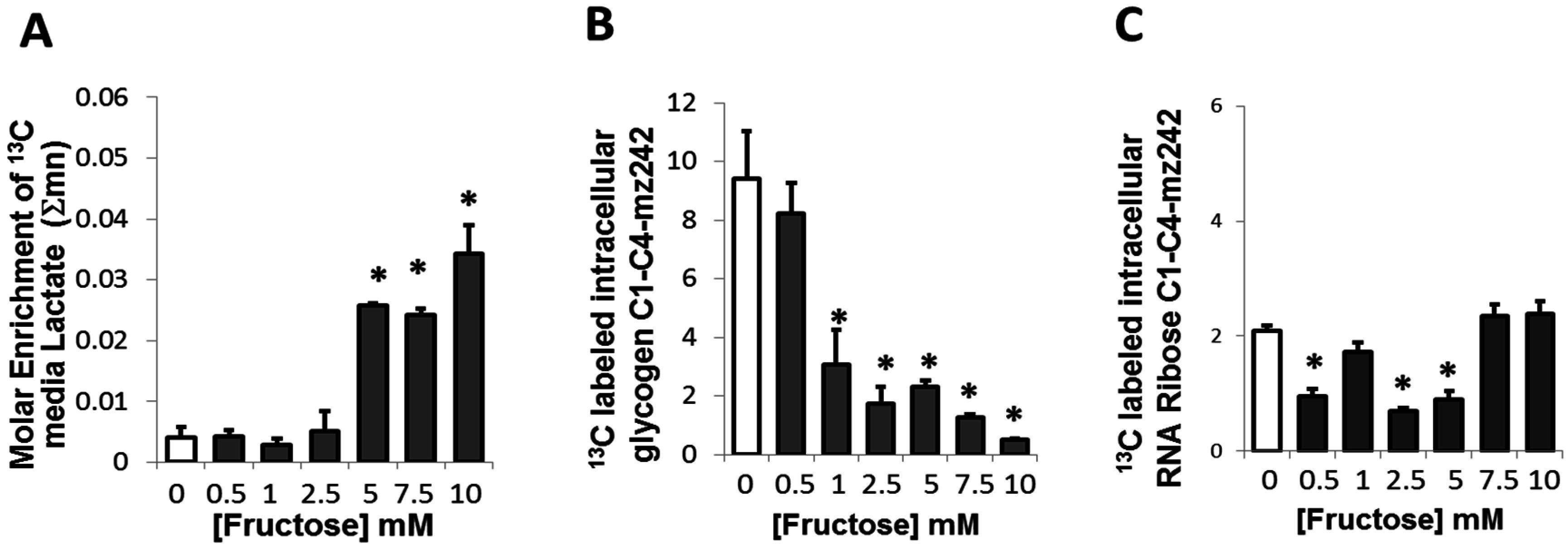
2.4. Presence of Fructose Triggered an Increased Conversion of Glucose to Lactate but Decreased the Conversion of Glucose to Glycogen or Ribose
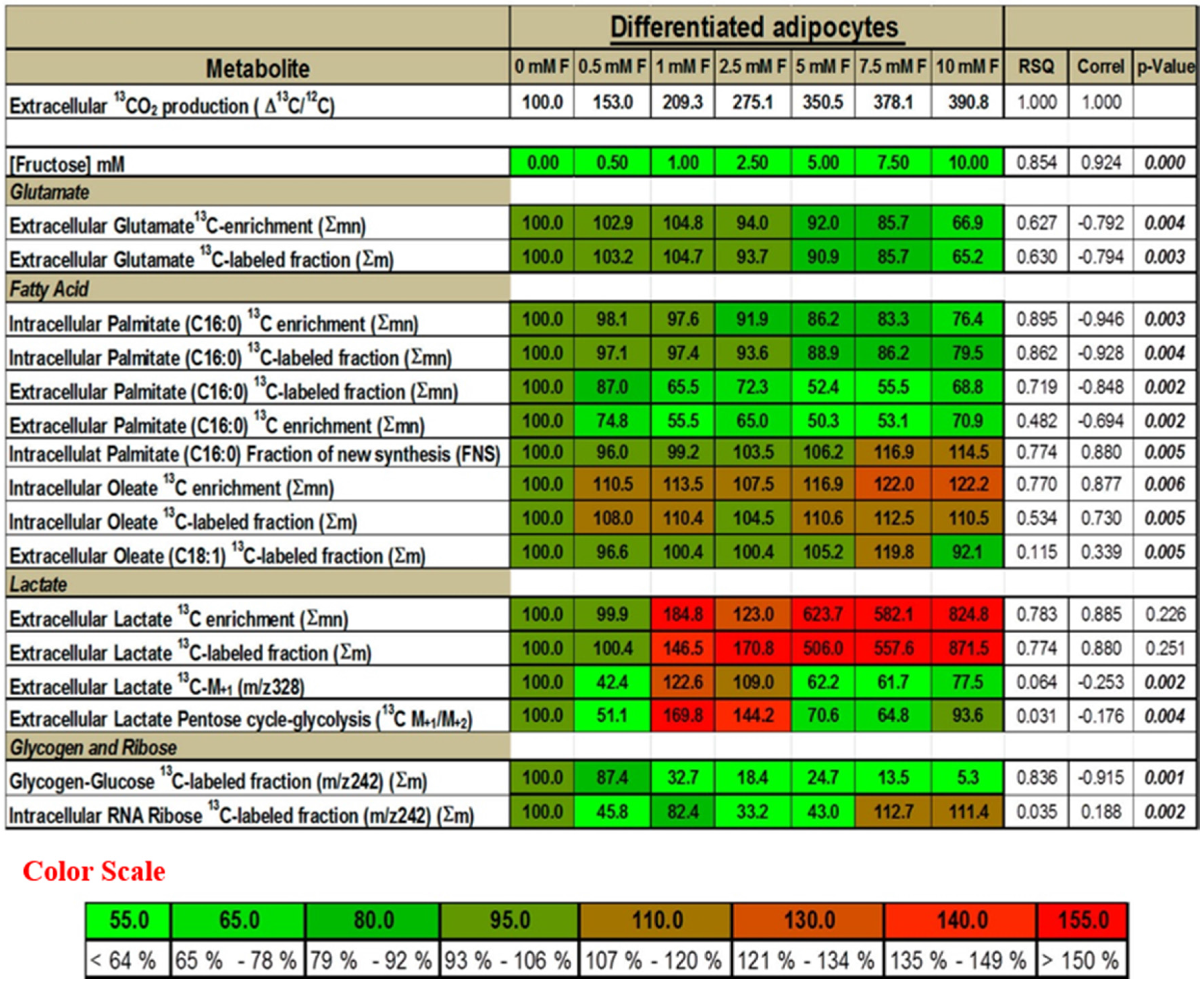
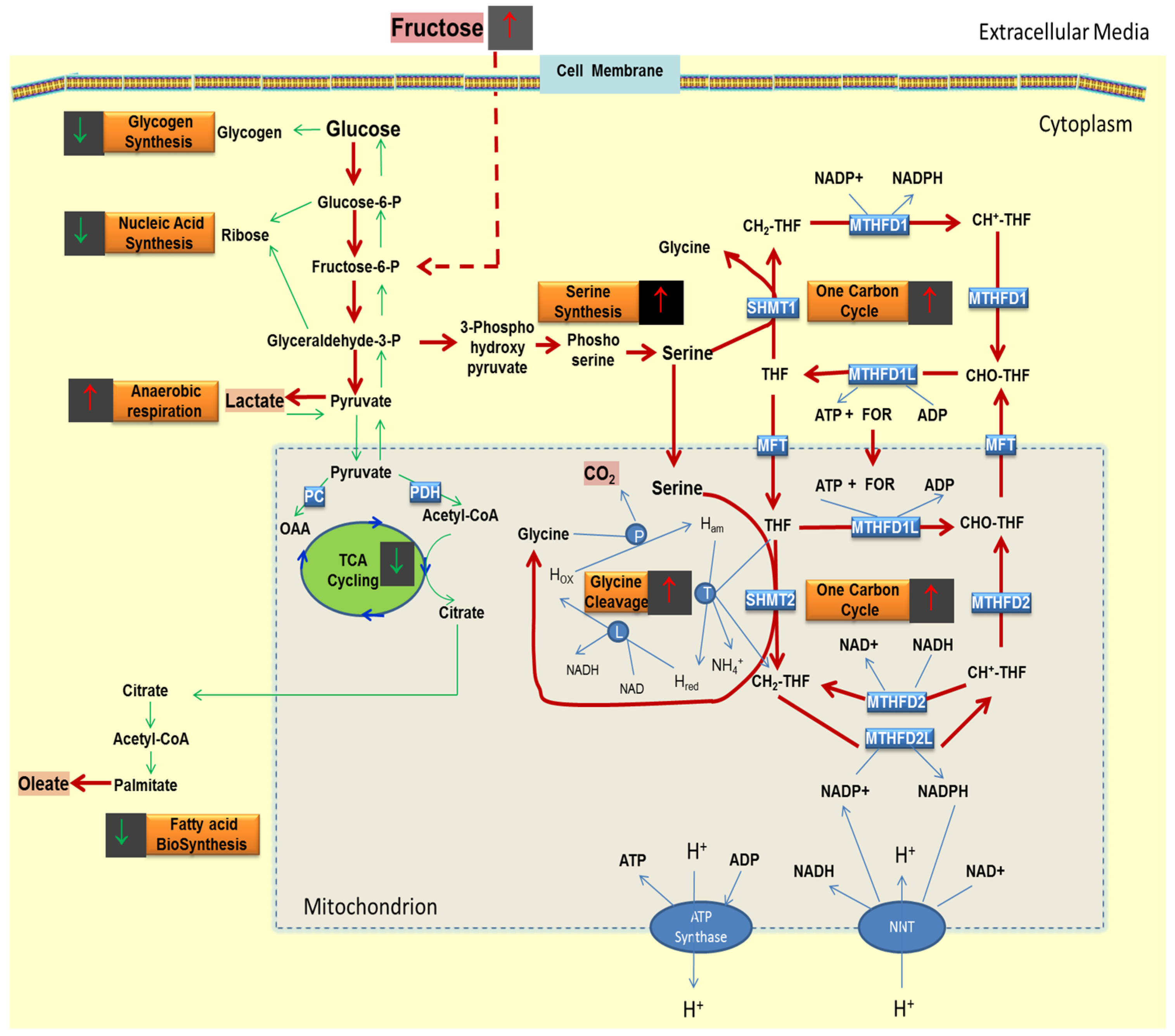
2.5. Presence of Fructose Increases the Flux of Glucose through the SOGC Pathway Resulting in Increased Oxidation of Glucose
3. Discussion
4. Experimental Section
4.1. Chemicals and Reagents
4.2. SGBS Cell Culture, Fructose Treatment and Labeling Using ([1, 2–13C2]-d-Glucose) Tracer
4.3. Targeted 13C Metabolite Studies and Metabolic Profiling Using ([1, 2–13C2]-d-Glucose)] Tracer
4.4. Quantitative Metabolite Analysis
4.5. Serine Oxidation Glycine Cleavage (SOGC) Pathway Analyses
4.6. Statistical Analysis
Disclaimer
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dekker, M.J.; Su, Q.; Baker, C.; Rutledge, A.C.; Adeli, K. Fructose: A highly lipogenic nutrient implicated in insulin resistance, hepatic steatosis, and the metabolic syndrome. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E685–E694. [Google Scholar] [CrossRef] [PubMed]
- Tappy, L.; Le, K.A. Metabolic effects of fructose and the worldwide increase in obesity. Physiol. Rev. 2010, 90, 23–46. [Google Scholar] [CrossRef] [PubMed]
- Jurgens, H.; Haass, W.; Castaneda, T.R.; Schurmann, A.; Koebnick, C.; Dombrowski, F.; Otto, B.; Nawrocki, A.R.; Scherer, P.E.; Spranger, J.; et al. Consuming fructose-sweetened beverages increases body adiposity in mice. Obes. Res. 2005, 13, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Sheludiakova, A.; Rooney, K.; Boakes, R.A. Metabolic and behavioural effects of sucrose and fructose/glucose drinks in the rat. Eur. J. Nutr. 2012, 51, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, K.; Wylie, A.T.; Tucker, K.L.; Hamp, T.J.; Gharaibeh, R.Z.; Fodor, A.A.; Cullen, J.M. Dietary fructose induces endotoxemia and hepatic injury in calorically controlled primates. Am. J. Clin. Nutr. 2013, 98, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Pollock, N.K.; Bundy, V.; Kanto, W.; Davis, C.L.; Bernard, P.J.; Zhu, H.; Gutin, B.; Dong, Y. Greater fructose consumption is associated with cardiometabolic risk markers and visceral adiposity in adolescents. J. Nutr. 2012, 142, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.H.; Wong, S.H.; Chen, Y.J.; Huang, Y.J.; Hsieh, S.S. Effect of glycemic index and fructose content in lunch on substrate utilization during subsequent brisk walking. Appl. Physiol. Nutr. Metab. 2011, 36, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, M.R. Normal roles for dietary fructose in carbohydrate metabolism. Nutrients 2014, 6, 3117–3129. [Google Scholar] [CrossRef] [PubMed]
- Boros, L.G.; Huang, D.; Heaney, A.P. Fructose drives glucose via direct oxidation and promotes palmitate/oleate co-release from HepG2 cells: Relevance with randle cycle. Metabolomics 2012, 2, 107. [Google Scholar]
- Liemburg-Apers, D.C.; Imamura, H.; Forkink, M.; Nooteboom, M.; Swarts, H.G.; Brock, R.; Smeitink, J.A.; Willems, P.H.; Koopman, W.J. Quantitative glucose and atp sensing in mammalian cells. Pharm. Res. 2011, 28, 2745–2757. [Google Scholar] [CrossRef] [PubMed]
- Mergenthaler, P.; Lindauer, U.; Dienel, G.A.; Meisel, A. Sugar for the brain: The role of glucose in physiological and pathological brain function. Trends Neurosci. 2013, 36, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Ye, J.; Kamphorst, J.J.; Shlomi, T.; Thompson, C.B.; Rabinowitz, J.D. Quantitative flux analysis reveals folate-dependent nadph production. Nature 2014, 510, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Brosnan, J.T. Glutamate, at the interface between amino acid and carbohydrate metabolism. J. Nutr. 2000, 130, 988S–990S. [Google Scholar] [PubMed]
- Randle, P.J.; Garland, P.B.; Hales, C.N.; Newsholme, E.A. The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet 1963, 1, 785–789. [Google Scholar] [CrossRef]
- Frayn, K.N. The glucose-fatty acid cycle: A physiological perspective. Biochem. Soc. Trans. 2003, 31, 1115–1119. [Google Scholar] [CrossRef] [PubMed]
- Dirlewanger, M.; Schneiter, P.; Jequier, E.; Tappy, L. Effects of fructose on hepatic glucose metabolism in humans. Am. J. Physiol. Endocrinol. Metab. 2000, 279, E907–E911. [Google Scholar] [PubMed]
- Cermak, N.M.; van Loon, L.J. The use of carbohydrates during exercise as an ergogenic aid. Sports Med. 2013, 43, 1139–1155. [Google Scholar] [CrossRef] [PubMed]
- Rosen, E.D.; Spiegelman, B.M. Adipocytes as regulators of energy balance and glucose homeostasis. Nature 2006, 444, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Bellido, J.; Herrera, E. Effects of glucose on the metabolization of fructose and glycerol by isolated adipocytes from rat. Rev. Esp. Fisiol. 1978, 34, 437–441. [Google Scholar] [PubMed]
- Metallo, C.M.; Walther, J.L.; Stephanopoulos, G. Evaluation of 13c isotopic tracers for metabolic flux analysis in mammalian cells. J. Biotechnol. 2009, 144, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Boros, L.G.; Beger, R.D.; Meuillet, E.J.; Colca, J.R.; Szalma, S.; Thompson, P.A.; Dux, L.; Farkas, J., Jr.; Somlyai, G. Targeted 13C-labeled tracer fate associations for drug efficacy testing in cancer. In Tumor Cell Metabolism—Pathways, Regulation and Biology; Mazurek, S., Shoshan, M., Eds.; Springer-Verlag GmbH: Heidelberg, Germany, 2015. [Google Scholar]
- Buescher, J.M.; Antoniewicz, M.R.; Boros, L.G.; Burgess, S.C.; Brunengraber, H.; Clish, C.B.; DeBerardinis, R.J.; Feron, O.; Frezza, C.; Ghesquiere, B.; et al. A roadmap for interpreting c metabolite labeling patterns from cells. Curr. Opin. Biotechnol. 2015, 34, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Melo, T.M.; Haberg, A.K.; Risa, O.; Kondziella, D.; Henry, P.G.; Sonnewald, U. Tricarboxylic acid cycle activity measured by 13c magnetic resonance spectroscopy in rats subjected to the kaolin model of obstructed hydrocephalus. Neurochem. Res. 2011, 36, 1801–1808. [Google Scholar] [CrossRef] [PubMed]
- Boros, L.G.; Cascante, M.; Lee, W.N. Metabolic profiling of cell growth and death in cancer: Applications in drug discovery. Drug Discov. Today 2002, 7, 364–372. [Google Scholar] [CrossRef]
- Leimer, K.R.; Rice, R.H.; Gehrke, C.W. Complete mass spectra of n-trifluoroacetyl-n-butyl esters of amino acids. J. Chromatogr. 1977, 141, 121–144. [Google Scholar] [CrossRef]
- Gladden, L.B. Lactate metabolism: A new paradigm for the third millennium. J. Physiol. 2004, 558, 5–30. [Google Scholar] [CrossRef] [PubMed]
- Markan, K.R.; Jurczak, M.J.; Brady, M.J. Stranger in a strange land: Roles of glycogen turnover in adipose tissue metabolism. Mol. Cell. Endocrinol. 2010, 318, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Tedeschi, P.M.; Markert, E.K.; Gounder, M.; Lin, H.; Dvorzhinski, D.; Dolfi, S.C.; Chan, L.L.; Qiu, J.; DiPaola, R.S.; Hirshfield, K.M.; et al. Contribution of serine, folate and glycine metabolism to the atp, nadph and purine requirements of cancer cells. Cell. Death Dis. 2013, 4, e877. [Google Scholar] [CrossRef] [PubMed]
- Brosnan, J.T. Comments on metabolic needs for glucose and the role of gluconeogenesis. Eur. J. Clin. Nutr. 1999, 53, S107–S111. [Google Scholar] [CrossRef] [PubMed]
- Grayson, B.E.; Seeley, R.J.; Sandoval, D.A. Wired on sugar: The role of the cns in the regulation of glucose homeostasis. Nat. Rev. Neurosci. 2013, 14, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Chen, X.; Ruiz, J.; White, J.V.; Rossetti, L. Mechanisms of fatty acid-induced inhibition of glucose uptake. J. Clin. Investig. 1994, 93, 2438–2446. [Google Scholar] [CrossRef] [PubMed]
- Froesch, E.R.; Ginsberg, J.L. Fructose metabolism of adipose tissue. I. Comparison of fructose and glucose metabolism in epididymal adipose tissue of normal rats. J. Biol. Chem. 1962, 237, 3317–3324. [Google Scholar] [PubMed]
- Varma, V.; Boros, L.G.; Nolen, G.T.; Chang, C.W.; Wabitsch, M.; Beger, R.D.; Kaput, J. Metabolic fate of fructose in human adipocytes: A targeted 13c tracer fate association study. Metabolomics: Off. J. Metab. Soc. 2014, 11, 529–544. [Google Scholar] [CrossRef] [PubMed]
- Fischer-Posovszky, P.; Newell, F.S.; Wabitsch, M.; Tornqvist, H.E. Human sgbs cells—A unique tool for studies of human fat cell biology. Obes. Facts 2008, 1, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Wood, I.S.; Wang, B.; Lorente-Cebrian, S.; Trayhurn, P. Hypoxia increases expression of selective facilitative glucose transporters (glut) and 2-deoxy-d-glucose uptake in human adipocytes. Biochem. Biophys. Res. Commun. 2007, 361, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Diggle, C.P.; Shires, M.; Leitch, D.; Brooke, D.; Carr, I.M.; Markham, A.F.; Hayward, B.E.; Asipu, A.; Bonthron, D.T. Ketohexokinase: Expression and localization of the principal fructose-metabolizing enzyme. J. Histochem. Cytochem.: Off. J. Histochem. Soc. 2009, 57, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Hui, H.; Huang, D.; McArthur, D.; Nissen, N.; Boros, L.G.; Heaney, A.P. Direct spectrophotometric determination of serum fructose in pancreatic cancer patients. Pancreas 2009, 38, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, I.; Keyser, A.; Pacy, D. Some effects, in man, of varying the load of glucose, sucrose, fructose, or sorbitol on various metabolites in blood. Am. J. Clin. Nutr. 1978, 31, 1305–1311. [Google Scholar] [PubMed]
- Munstedt, K.; Bohme, M.; Hauenschild, A.; Hrgovic, I. Consumption of rapeseed honey leads to higher serum fructose levels compared with analogue glucose/fructose solutions. Eur. J. Clin. Nutr. 2011, 65, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Metallo, C.M.; Vander Heiden, M.G. Understanding metabolic regulation and its influence on cell physiology. Mol. Cell 2013, 49, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Lunt, S.Y.; Vander Heiden, M.G. Aerobic glycolysis: Meeting the metabolic requirements of cell proliferation. Annu. Rev. Cell Dev. Biol. 2011, 27, 441–464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gray, L.R.; Tompkins, S.C.; Taylor, E.B. Regulation of pyruvate metabolism and human disease. Cell. Mol. Life Sci.: CMLS 2014, 71, 2577–2604. [Google Scholar] [CrossRef] [PubMed]
- Sobrecases, H.; Le, K.A.; Bortolotti, M.; Schneiter, P.; Ith, M.; Kreis, R.; Boesch, C.; Tappy, L. Effects of short-term overfeeding with fructose, fat and fructose plus fat on plasma and hepatic lipids in healthy men. Diabetes Metab. 2010, 36, 244–246. [Google Scholar] [CrossRef] [PubMed]
- Kruszynska, Y.T.; Harry, D.S.; Fryer, L.G.; McIntyre, N. Lipid metabolism and substrate oxidation during intravenous fructose administration in cirrhosis. Metab.: Clin. Exp. 1994, 43, 1171–1181. [Google Scholar] [CrossRef]
- Moore, M.C.; Cherrington, A.D.; Mann, S.L.; Davis, S.N. Acute fructose administration decreases the glycemic response to an oral glucose tolerance test in normal adults. J. Clin. Endocrinol. Metab. 2000, 85, 4515–4519. [Google Scholar] [CrossRef] [PubMed]
- DiGirolamo, M.; Newby, F.D.; Lovejoy, J. Lactate production in adipose tissue: A regulated function with extra-adipose implications. FASEB J.: Off. Publ. Fed. Am. Soc. Exp. Biol. 1992, 6, 2405–2412. [Google Scholar]
- Munoz, S.; Franckhauser, S.; Elias, I.; Ferre, T.; Hidalgo, A.; Monteys, A.M.; Molas, M.; Cerdan, S.; Pujol, A.; Ruberte, J.; et al. Chronically increased glucose uptake by adipose tissue leads to lactate production and improved insulin sensitivity rather than obesity in the mouse. Diabetologia 2010, 53, 2417–2430. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, A.; Beg, Q.K.; Demenezes, M.A.; Ernst, J.; Bar-Joseph, Z.; Barabasi, A.L.; Boros, L.G.; Oltvai, Z.N. Impact of the solvent capacity constraint on E. coli metabolism. BMC Syst. Biol. 2008. [Google Scholar] [CrossRef] [PubMed]
- Leonards, J.R.; Landau, B.R. A study of the equivalence of metabolic patterns in rat adipose tissue: Insulin versus glucose concentration. Arch. Biochem. Biophys. 1960, 91, 194–200. [Google Scholar] [CrossRef]
- Moody, A.J.; Gliemann, J. The effect of insulin on the glycogen metabolism of isolated fat cells. Experientia 1968, 24, 628–630. [Google Scholar] [CrossRef] [PubMed]
- Robubi, A.; Huber, K.R.; Krugluger, W. Extra fructose in the growth medium fuels lipogenesis of adipocytes. J. Obes. 2014, 2014, 647034. [Google Scholar] [CrossRef] [PubMed]
- de Ferranti, S.; Mozaffarian, D. The perfect storm: Obesity, adipocyte dysfunction, and metabolic consequences. Clin. Chem. 2008, 54, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Wabitsch, M.; Brenner, R.E.; Melzner, I.; Braun, M.; Moller, P.; Heinze, E.; Debatin, K.M.; Hauner, H. Characterization of a human preadipocyte cell strain with high capacity for adipose differentiation. Int. J. Obes. Relat. Metab. Disord.: J. Int. Assoc. Study Obes. 2001, 25, 8–15. [Google Scholar] [CrossRef]
- Lee, W.N.; Byerley, L.O.; Bergner, E.A.; Edmond, J. Mass isotopomer analysis: Theoretical and practical considerations. Biol. Mass Spectrometry 1991, 20, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Marin, S.; Lee, W.N.; Bassilian, S.; Lim, S.; Boros, L.G.; Centelles, J.J.; FernAndez-Novell, J.M.; Guinovart, J.J.; Cascante, M. Dynamic profiling of the glucose metabolic network in fasted rat hepatocytes using [1,2–13C2] glucose. Biochem. J. 2004, 381, 287–294. [Google Scholar] [PubMed]
- Vizan, P.; Boros, L.G.; Figueras, A.; Capella, G.; Mangues, R.; Bassilian, S.; Lim, S.; Lee, W.N.; Cascante, M. K-ras codon-specific mutations produce distinctive metabolic phenotypes in nih3t3 mice [corrected] fibroblasts. Cancer Res. 2005, 65, 5512–5515. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.N.; Boros, L.G.; Puigjaner, J.; Bassilian, S.; Lim, S.; Cascante, M. Mass isotopomer study of the nonoxidative pathways of the pentose cycle with [1,2–13C2] glucose. Am. J. Physiol. 1998, 274, E843–E851. [Google Scholar] [PubMed]
- Hellerstein, M.K.; Neese, R.A. Mass isotopomer distribution analysis: A technique for measuring biosynthesis and turnover of polymers. Am. J. Physiol. 1992, 263, E988–1001. [Google Scholar] [PubMed]
- Lee, W.N. Stable isotopes and mass isotopomer study of fatty acid and cholesterol synthesis. A review of the mida approach. Adv. Exp. Med. Biol. 1996, 399, 95–114. [Google Scholar] [PubMed]
- Mizumori, M.; Meyerowitz, J.; Takeuchi, T.; Lim, S.; Lee, P.; Supuran, C.T.; Guth, P.H.; Engel, E.; Kaunitz, J.D.; Akiba, Y. Epithelial carbonic anhydrases facilitate pco2 and ph regulation in rat duodenal mucosa. J. Physiol. 2006, 573, 827–842. [Google Scholar] [CrossRef] [PubMed]
- Bartnik, B.L.; Hovda, D.A.; Lee, P.W. Glucose metabolism after traumatic brain injury: Estimation of pyruvate carboxylase and pyruvate dehydrogenase flux by mass isotopomer analysis. J. Neurotrauma 2007, 24, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.N.; Lim, S.; Bassilian, S.; Bergner, E.A.; Edmond, J. Fatty acid cycling in human hepatoma cells and the effects of troglitazone. J. Biol. Chem. 1998, 273, 20929–20934. [Google Scholar] [CrossRef] [PubMed]
- Harrigan, G.G.; Colca, J.; Szalma, S.; Boros, L.G. Pnu-91325 increases fatty acid synthesis from glucose and mitochondrial long chain fatty acid degradation: A comparative tracer-based metabolomics study with rosiglitazone and pioglitazone in hepg2 cells. Metabolomics: Off. J. Metab. Soc. 2006, 2, 21–29. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varma, V.; Boros, L.G.; Nolen, G.T.; Chang, C.-W.; Wabitsch, M.; Beger, R.D.; Kaput, J. Fructose Alters Intermediary Metabolism of Glucose in Human Adipocytes and Diverts Glucose to Serine Oxidation in the One–Carbon Cycle Energy Producing Pathway. Metabolites 2015, 5, 364-385. https://doi.org/10.3390/metabo5020364
Varma V, Boros LG, Nolen GT, Chang C-W, Wabitsch M, Beger RD, Kaput J. Fructose Alters Intermediary Metabolism of Glucose in Human Adipocytes and Diverts Glucose to Serine Oxidation in the One–Carbon Cycle Energy Producing Pathway. Metabolites. 2015; 5(2):364-385. https://doi.org/10.3390/metabo5020364
Chicago/Turabian StyleVarma, Vijayalakshmi, László G. Boros, Greg T. Nolen, Ching-Wei Chang, Martin Wabitsch, Richard D. Beger, and Jim Kaput. 2015. "Fructose Alters Intermediary Metabolism of Glucose in Human Adipocytes and Diverts Glucose to Serine Oxidation in the One–Carbon Cycle Energy Producing Pathway" Metabolites 5, no. 2: 364-385. https://doi.org/10.3390/metabo5020364
APA StyleVarma, V., Boros, L. G., Nolen, G. T., Chang, C.-W., Wabitsch, M., Beger, R. D., & Kaput, J. (2015). Fructose Alters Intermediary Metabolism of Glucose in Human Adipocytes and Diverts Glucose to Serine Oxidation in the One–Carbon Cycle Energy Producing Pathway. Metabolites, 5(2), 364-385. https://doi.org/10.3390/metabo5020364





