A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Disease Category
2.2.1. Hemato-Oncologic Disease
2.2.2. Rheumatoid Disease
2.2.3. Gastrointestinal Disease
2.2.4. Endocrine Disease
2.3. Data Collection
2.3.1. Anthropometric Measurement
2.3.2. BMD Assessment
2.3.3. Thyroid Function Test
2.4. Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics
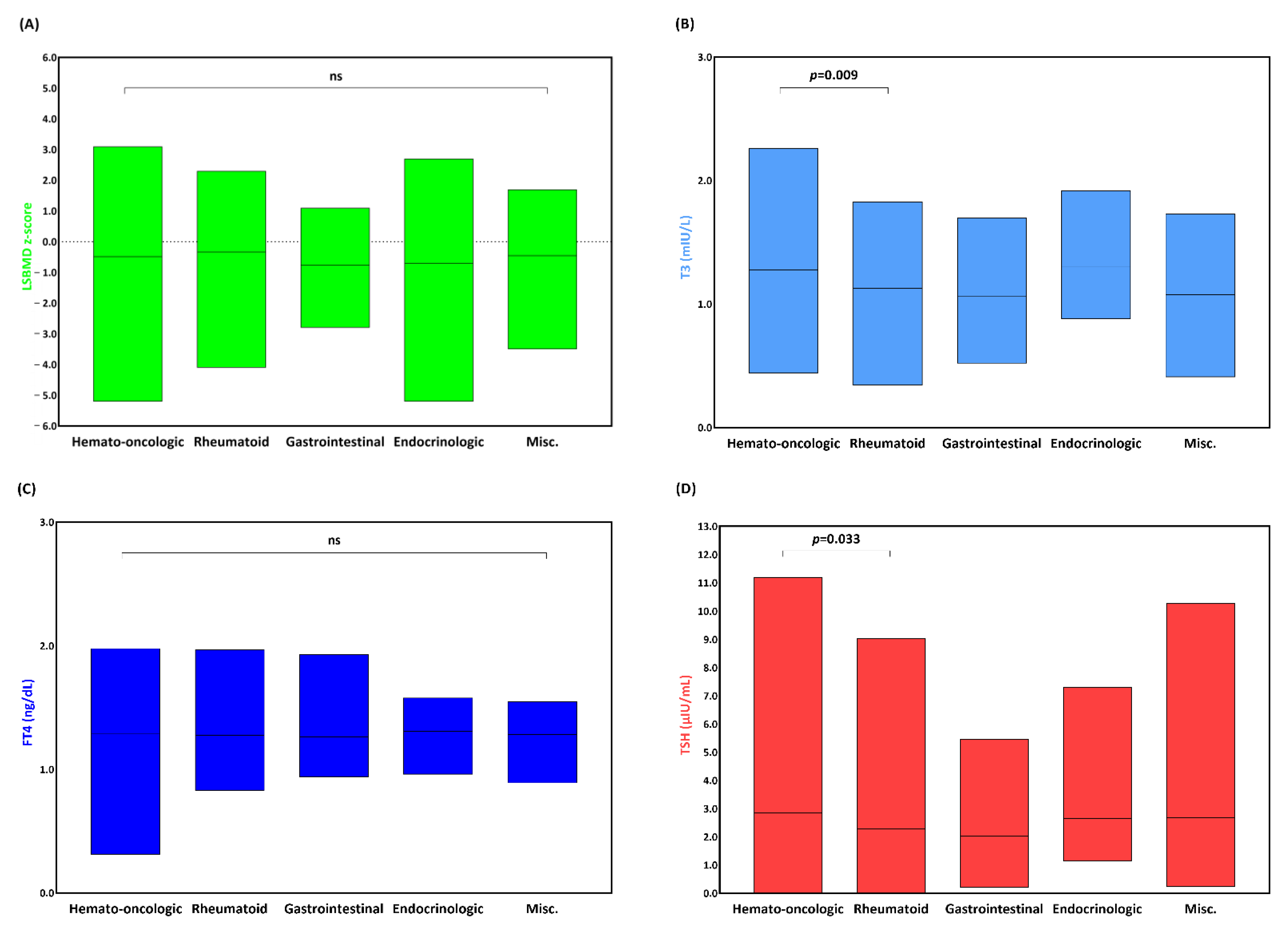
3.2. LSBMD and Thyroid Function of Patients with an Underlying Condition
3.3. BMD Status under Abnormal Thyroid Function
3.4. Causal Association between BMD and Thyroid Function
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kim, H.Y.; Mohan, S. Role and Mechanisms of Actions of Thyroid Hormone on the Skeletal Development. Bone Res. 2013, 1, 146–161. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Pang, Y.; Xu, J.; Chen, X.; Zhang, C.; Wu, B.; Gao, J. Endocrine Regulation on Bone by Thyroid. Front. Endocrinol. 2022, 13, 873820. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Ryu, V.; Miyashita, S.; Korkmaz, F.; Lizneva, D.; Gera, S.; Latif, R.; Davies, T.F.; Iqbal, J.; Yuen, T.; et al. Thyrotropin, Hyperthyroidism, and Bone Mass. J. Clin. Endocrinol. Metab. 2021, 106, e4809–e4821. [Google Scholar] [CrossRef] [PubMed]
- Ko, A.; Kong, J.; Samadov, F.; Mukhamedov, A.; Kim, Y.M.; Lee, Y.J.; Nam, S.O. Bone health in pediatric patients with neurological disorders. Ann. Pediatr. Endocrinol. Metab. 2020, 25, 15–23. [Google Scholar] [CrossRef]
- Ahn, M.B.; Suh, B.K. Bone morbidity in pediatric acute lymphoblastic leukemia. Ann. Pediatr. Endocrinol. Metab. 2020, 25, 1–9. [Google Scholar] [CrossRef]
- Jin, H.Y.; Lee, J.A. Low bone mineral density in children and adolescents with cancer. Ann. Pediatr. Endocrinol. Metab. 2020, 25, 137–144. [Google Scholar] [CrossRef]
- Yang, H.R. Updates on bone health in children with gastrointestinal diseases. Ann. Pediatr. Endocrinol. Metab. 2020, 25, 10–14. [Google Scholar] [CrossRef]
- Simm, P.J.; Biggin, A.; Zacharin, M.R.; Rodda, C.P.; Tham, E.; Siafarikas, A.; Jefferies, C.; Hofman, P.L.; Jensen, D.E.; Woodhead, H.; et al. Consensus guidelines on the use of bisphosphonate therapy in children and adolescents. J. Paediatr. Child Health 2018, 54, 223–233. [Google Scholar] [CrossRef]
- Lee, Y.A.; Kwon, A.; Kim, J.H.; Nam, H.K.; Yoo, J.H.; Lim, J.S.; Cho, S.Y.; Cho, W.K.; Shim, K.S. Clinical practice guidelines for optimizing bone health in Korean children and adolescents. Ann. Pediatr. Endocrinol. Metab. 2022, 27, 5–14. [Google Scholar] [CrossRef]
- Dhanwal, D.K. Thyroid disorders and bone mineral metabolism. Indian J. Endocrinol. Metab. 2011, 15, S107–S112. [Google Scholar] [CrossRef]
- Tuchendler, D.; Bolanowski, M. The influence of thyroid dysfunction on bone metabolism. Thyroid. Res. 2014, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Bassett, J.H.; Williams, G.R. Role of Thyroid Hormones in Skeletal Development and Bone Maintenance. Endocr. Rev. 2016, 37, 135–187. [Google Scholar] [CrossRef] [PubMed]
- Veldscholte, K.; Barjaktarovic, M.; Trajanoska, K.; Jaddoe, V.W.V.; Visser, T.J.; de Rijke, Y.B.; Peeters, R.P.; Rivadeneira, F.; Korevaar, T.I.M. The Association of Thyroid Function With Bone Density During Childhood. J. Clin. Endocrinol. Metab. 2018, 103, 4125–4134. [Google Scholar] [CrossRef]
- Kim, J.H.; Yun, S.; Hwang, S.S.; Shim, J.O.; Chae, H.W.; Lee, Y.J.; Lee, J.H.; Kim, S.C.; Lim, D.; Yang, S.W.; et al. The 2017 Korean National Growth Charts for children and adolescents: Development, improvement, and prospects. Korean J. Pediatr. 2018, 61, 135–149. [Google Scholar] [CrossRef]
- Yi, K.H.; Hwang, J.S.; Kim, E.Y.; Lee, J.A.; Kim, D.H.; Lim, J.S. Reference values for bone mineral density according to age with body size adjustment in Korean children and adolescents. J. Bone Miner. Metab. 2014, 32, 281–289. [Google Scholar] [CrossRef]
- Rivkees, S.; Bauer, A.J. Thyroid Disorders in Children and Adolescents. In Sperling Pediatric Endocrinology; Sperling, M.A., Ed.; Elsevier: Philadelphia, PA, USA, 2021; pp. 395–424. [Google Scholar] [CrossRef]
- Galindo-Zavala, R.; Bou-Torrent, R.; Magallares-Lopez, B.; Mir-Perello, C.; Palmou-Fontana, N.; Sevilla-Perez, B.; Medrano-San Ildefonso, M.; Gonzalez-Fernandez, M.I.; Roman-Pascual, A.; Alcaniz-Rodriguez, P.; et al. Expert panel consensus recommendations for diagnosis and treatment of secondary osteoporosis in children. Pediatr. Rheumatol. Online J. 2020, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Hong, A.R.; Ahn, H.Y.; Kim, B.K.; Ahn, S.H.; Park, S.Y.; Kim, M.-H.; Lee, J.; Cho, S.W.; Kang, H.-C. Evaluation and Management of Bone Health in Patients with Thyroid Diseases: A Position Statement from the Korean Thyroid Association. Int. J. Thyroidol. 2022, 15, 1–16. [Google Scholar] [CrossRef]
- Delitala, A.P.; Scuteri, A.; Doria, C. Thyroid Hormone Diseases and Osteoporosis. J. Clin. Med. 2020, 9, 1034. [Google Scholar] [CrossRef] [PubMed]
- Paragliola, R.M.; Corsello, A.; Papi, G.; Pontecorvi, A.; Corsello, S.M. Cushing’s Syndrome Effects on the Thyroid. Int. J. Mol. Sci. 2021, 22, 3131. [Google Scholar] [CrossRef]
- Deng, T.; Zhang, W.; Zhang, Y.; Zhang, M.; Huan, Z.; Yu, C.; Zhang, X.; Wang, Y.; Xu, J. Thyroid-stimulating hormone decreases the risk of osteoporosis by regulating osteoblast proliferation and differentiation. BMC Endocr. Disord. 2021, 21, 49. [Google Scholar] [CrossRef]
- Noh, H.M.; Park, Y.S.; Lee, J.; Lee, W. A cross-sectional study to examine the correlation between serum TSH levels and the osteoporosis of the lumbar spine in healthy women with normal thyroid function. Osteoporos. Int. 2015, 26, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Sultana, F.; Korkmaz, F.; Lizneva, D.; Yuen, T.; Zaidi, M. Independent Skeletal Actions of Pituitary Hormones. Endocrinol. Metab. 2022, 37, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.P. Thyroid Function across the Lifespan: Do Age-Related Changes Matter? Endocrinol. Metab. 2022, 37, 208–219. [Google Scholar] [CrossRef]
- Kaplowitz, P.B. Subclinical hypothyroidism in children: Normal variation or sign of a failing thyroid gland? Int. J. Pediatr. Endocrinol. 2010, 2010, 281453. [Google Scholar] [CrossRef] [PubMed]
- Aeberli, I.; Jung, A.; Murer, S.B.; Wildhaber, J.; Wildhaber-Brooks, J.; Knopfli, B.H.; Zimmermann, M.B. During rapid weight loss in obese children, reductions in TSH predict improvements in insulin sensitivity independent of changes in body weight or fat. J. Clin. Endocrinol. Metab. 2010, 95, 5412–5418. [Google Scholar] [CrossRef]
- Metwalley, K.A.; Farghaly, H.S. Subclinical hyperthyroidism in children. J. Pediatr. Endocrinol. Metab. 2022. [CrossRef]
- Premawardhana, L.D. Thyroid testing in acutely ill patients may be an expensive distraction. Biochem. Med. 2017, 27, 300–307. [Google Scholar] [CrossRef]
- Sheng, N.; Xing, F.; Wang, J.; Duan, X.; Xiang, Z. T4 rather than TSH correlates with BMD among euthyroid adults. Front. Endocrinol. 2022, 13, 1039079. [Google Scholar] [CrossRef]
- Lin, J.D.; Pei, D.; Hsia, T.L.; Wu, C.Z.; Wang, K.; Chang, Y.L.; Hsu, C.H.; Chen, Y.L.; Chen, K.W.; Tang, S.H. The Relationship between Thyroid Function and Bone Mineral Density in Euthyroid Healthy Subjects in Taiwan. Endocr. Res. 2011, 36, 1–8. [Google Scholar] [CrossRef]
- Shuhart, C.R.; Yeap, S.S.; Anderson, P.A.; Jankowski, L.G.; Lewiecki, E.M.; Morse, L.R.; Rosen, H.N.; Weber, D.R.; Zemel, B.S.; Shepherd, J.A. Executive Summary of the 2019 ISCD Position Development Conference on Monitoring Treatment, DXA Cross-calibration and Least Significant Change, Spinal Cord Injury, Peri-prosthetic and Orthopedic Bone Health, Transgender Medicine, and Pediatrics. J. Clin. Densitom. 2019, 22, 453–471. [Google Scholar] [CrossRef]



| Total (n = 619) | |
|---|---|
| Male, n (%) | 312 (50.9) |
| Age, y | 13.22 ± 3.23 |
| Anthropometry, z-score | |
| Height | −0.51 ± 1.27 |
| Weight | −0.22 ± 1.48 |
| Body mass index | 0.03 ± 1.52 |
| Underlying condition, n (%) | |
| Hemato-oncologic | 493 (79.6) |
| Rheumatoid | 64 (10.3) |
| Gastrointestinal | 23 (3.7) |
| Endocrinologic | 19 (3.1) |
| Miscellaneous | 20 (3.2) |
| Age at diagnosis, years | 9.26 ± 11.86 |
| Bone mineral density | |
| Lumbar spine, g/cm2 | 0.81 ± 0.19 |
| Lumbar spine, z-score | 0.49 ± 1.28 |
| Low bone mass, n (%) | |
| ≤0 | 432 (70.5) |
| ≤−1.0 | 231 (37.7) |
| ≤−2.0 | 72 (11.7) |
| ≤−3.0 | 18 (2.9) |
| Right femur, g/cm2 | 0.76 ± 0.16 |
| Right femur, z-score | −1.04 ± 1.32 |
| Left femur, g/cm2 | 0.77 ± 0.16 |
| Left femur, z-score | −1.03 ± 1.34 |
| Thyroid function | |
| T3, ng/mL | 1.25 ± 0.29 |
| Interpretation, n (%) | |
| Normal | 505 (92.7) |
| Low | 39 (7.2) |
| High | 1 (0.2) |
| FT4, ng/dL | 1.28 ± 0.19 |
| Interpretation, n (%) | |
| Normal | 614 (99.5) |
| Low | 3 (0.5) |
| High | 0 (0) |
| TSH, µU/mL | 2.76 ± 1.87 |
| Interpretation, n (%) | |
| Normal | 560 (90.5) |
| Low | 36 (5.8) |
| High | 23 (3.7) |
| z-Scores | T3 | FT4 | TSH | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Normal | Abnormal | p | Normal | Abnormal | p | Normal | Abnormal | p | |
| LSBMD | −0.47 ± 1.26 | −0.67 ± 1.4 | 0.357 | −0.48 ± 1.28 | −1.23 ± 1.53 | 0.31 | −0.45 ± 1.25 | −0.86±1.46 | 0.018 |
| RFBMD | −1.03 ± 1.28 | −1.17 ± 1.7 | 0.537 | −1.04 ± 1.33 | −0.8 ± 1.4 | 0.75 | −1.01 ± 1.32 | −1.39±1.35 | 0.036 |
| LFBMD | −1.01 ± 1.3 | −1.31 ± 1.73 | 0.211 | −1.03 ± 1.34 | −1.0 ± 1.56 | 0.969 | −0.98 ± 1.32 | −1.49±1.48 | 0.007 |
| Clinical Parameter | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| β (95% CI) | SE | p | β (95% CI) | SE | p | |
| Underlying condition | ||||||
| Hemato-oncologic | −0.15 (−0.18–0.49) | 0.17 | 0.371 | 0.18 (−0.15–0.51) | 0.17 | 0.291 |
| Rheumatoid | −0.27 (−0.81–0.27) | 0.27 | 0.324 | 0.27 (−0.31–0.86) | 0.29 | 0.359 |
| Gastrointestinal | −0.22 (−0.81–0.36) | 0.29 | 0.455 | 0.13 (−0.57–0.84) | 0.36 | 0.709 |
| Endocrinologic | 0.04 (−0.54–0.61) | 0.29 | 0.9 | 0.29 (−0.29–0.87) | 0.29 | 0.322 |
| † Height | 0.42 (0.35–0.49) | 0.04 | <0.001 | |||
| † Weight | 0.41 (0.35–0.47) | 0.03 | <0.001 | |||
| † BMI | 0.31 (0.25–0.37) | 0.03 | <0.001 | 0.32 (0.26–0.39) | 0.03 | <0.001 |
| † RFBMD | 0.73 (0.68–0.78) | 0.03 | <0.001 | |||
| † LFBMD | 0.74 (0.69–0.79) | 0.03 | <0.001 | |||
| T3 | 0.19 (−0.17–0.55) | 0.18 | 0.302 | −0.06 (−0.41–0.29) | 0.18 | 0.749 |
| FT4 | −0.04 (−0.55–0.48) | 0.26 | 0.889 | −0.05 (−0.44–0.55) | 0.25 | 0.833 |
| TSH | 0.02 (−0.03–0.07) | 0.03 | 0.455 | 0.01 (−0.04–0.07) | 0.03 | 0.637 |
| Clinical Parameter | LSBMD z-Score ≤ −1.0 | LSBMD z-Score ≤ −2.0 | LSBMD z-Score ≤ −3.0 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | SE | p | OR (95% CI) | SE | p | OR (95% CI) | SE | p | |
| Abnormal T3 | |||||||||
| Unadjusted | 1.02 (0.53–1.98) | 0.34 | 0.953 | 2.38 (1.07–5.25) | 0.41 | 0.033 | 0.97 (0.12–7.61) | 1.05 | 0.977 |
| † Adjusted | 0.93 (0.43–2.01) | 0.39 | 0.931 | 2.29 (0.92–5.69) | 0.47 | 0.075 | 1.05 (0.11–10.36) | 0.04 | 0.968 |
| Abnormal FT4 | |||||||||
| Unadjusted | 3.39 (0.31–37.55) | 1.23 | 0.32 | 3.82 (0.34–42.71) | 1.23 | 0.276 | <0.01 (<0.01–0.01) | 1385.38 | 0.992 |
| † Adjusted | 1.81 (0.14–23.24) | 1.3 | 0.649 | 2.43 (0.18–32.13) | 1.32 | 0.499 | <0.01 (<0.01–0.01) | <0.01 | 0.996 |
| Abnormal TSH | |||||||||
| Unadjusted | 1.85 (1.08–3.17) | 0.27 | 0.026 | 2.4 (1.23–4.7) | 0.34 | 0.011 | 3.89 (1.34–11.34) | 0.55 | 0.013 |
| † Adjusted | 2.25 (1.24–4.08) | 0.3 | 0.008 | 2.65 (1.29–5.46) | 0.37 | 0.008 | 6.21 (1.8–21.41) | 2.89 | 0.004 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.; Ahn, M.B. A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility. Metabolites 2023, 13, 372. https://doi.org/10.3390/metabo13030372
Lee D, Ahn MB. A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility. Metabolites. 2023; 13(3):372. https://doi.org/10.3390/metabo13030372
Chicago/Turabian StyleLee, Dongjin, and Moon Bae Ahn. 2023. "A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility" Metabolites 13, no. 3: 372. https://doi.org/10.3390/metabo13030372
APA StyleLee, D., & Ahn, M. B. (2023). A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility. Metabolites, 13(3), 372. https://doi.org/10.3390/metabo13030372





