Amino Acids Metabolism in Retinopathy: From Clinical and Basic Research Perspective
Abstract
:1. Introduction
2. Metabolomics-Based Amino Acid Metabolism in Retinopathy
2.1. Amino Acids in Retinopathy by Analysis of Metabolomics
2.2. Amino Acids- Related Metabolic Pathways in Retinopathy
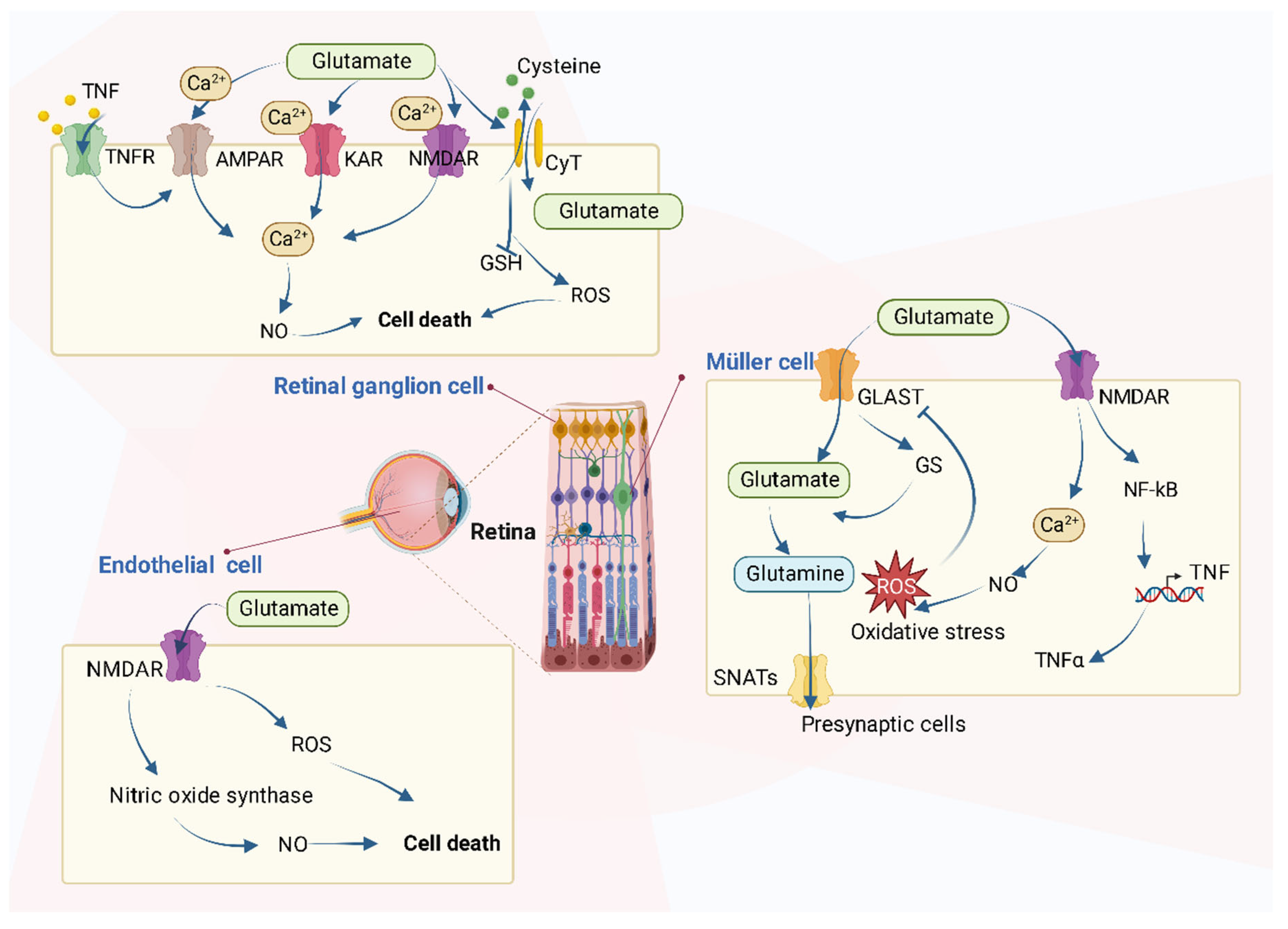
2.3. Potential Effects of Metabolites in Retina
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ramrattan, R.S.; Wolfs, R.C.; Panda-Jonas, S.; Jonas, J.B.; Bakker, D.; Pols, H.A.; Hofman, A.; de Jong, P.T. Prevalence and Causes of Visual Field Loss in the Elderly and Associations with Impairment in Daily Functioning: The Rotterdam Study. Arch. Ophthalmol. Chic. Ill 1960 2001, 119, 1788–1794. [Google Scholar] [CrossRef] [Green Version]
- Taylor, H.R.; Katala, S.; Muñoz, B.; Turner, V. Increase in Mortality Associated with Blindness in Rural Africa. Bull. World Health Organ. 1991, 69, 335–338. [Google Scholar] [PubMed]
- Stevens, G.A.; White, R.A.; Flaxman, S.R.; Price, H.; Jonas, J.B.; Keeffe, J.; Leasher, J.; Naidoo, K.; Pesudovs, K.; Resnikoff, S.; et al. Global Prevalence of Vision Impairment and Blindness: Magnitude and Temporal Trends, 1990-2010. Ophthalmology 2013, 120, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- GBD 2019 Blindness and Vision Impairment Collaborators; Vision Loss Expert Group of the Global Burden of Disease Study Trends in Prevalence of Blindness and Distance and near Vision Impairment over 30 Years: An Analysis for the Global Burden of Disease Study. Lancet Glob. Health 2021, 9, e130–e143. [CrossRef]
- Schmidt, C.H.; Volpe, N.J.; Bryar, P.J. Eye Disease in Medical Practice. Med. Clin. North Am. 2021, 105, 397–407. [Google Scholar] [CrossRef]
- Cheung, N.; Mitchell, P.; Wong, T.Y. Diabetic Retinopathy. The Lancet 2010, 376, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, N.; Arora, P.; Sandhir, R. Perturbed Biochemical Pathways and Associated Oxidative Stress Lead to Vascular Dysfunctions in Diabetic Retinopathy. Oxid. Med. Cell. Longev. 2019, 2019, 8458472. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson, C.P.; Ferris, F.L.; Klein, R.E.; Lee, P.P.; Agardh, C.D.; Davis, M.; Dills, D.; Kampik, A.; Pararajasegaram, R.; Verdaguer, J.T.; et al. Proposed International Clinical Diabetic Retinopathy and Diabetic Macular Edema Disease Severity Scales. Ophthalmology 2003, 110, 1677–1682. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.S.; Mitchell, P.; Seddon, J.M.; Holz, F.G.; Wong, T.Y. Age-Related Macular Degeneration. Lancet Lond. Engl. 2012, 379, 1728–1738. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, J.L. Age-Related Macular Degeneration. JAMA 2002, 288, 2233–2236. [Google Scholar] [CrossRef]
- Dogra, M.R.; Katoch, D.; Dogra, M. An Update on Retinopathy of Prematurity (ROP). Indian J. Pediatr. 2017, 84. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.-Y.; Hsih, W.-H.; Lin, Y.-B.; Wen, C.-Y.; Chang, T.-J. Update in the Epidemiology, Risk Factors, Screening, and Treatment of Diabetic Retinopathy. J. Diabetes Investig. 2021, 12, 1322–1325. [Google Scholar] [CrossRef] [PubMed]
- Jian, Q.; Wu, Y.; Zhang, F. Metabolomics in Diabetic Retinopathy: From Potential Biomarkers to Molecular Basis of Oxidative Stress. Cells 2022, 11, 3005. [Google Scholar] [CrossRef] [PubMed]
- Clish, C.B. Metabolomics: An Emerging but Powerful Tool for Precision Medicine. Cold Spring Harb. Mol. Case Stud. 2015, 1, a000588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laíns, I.; Gantner, M.; Murinello, S.; Lasky-Su, J.A.; Miller, J.W.; Friedlander, M.; Husain, D. Metabolomics in the Study of Retinal Health and Disease. Prog. Retin. Eye Res. 2019, 69, 57–79. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Miriel, V.; Bertun, E. Amino Acid Supplementation and Exercise Performance. Analysis of the Proposed Ergogenic Value. Sports Med. Auckl. NZ 1993, 16, 190–209. [Google Scholar] [CrossRef]
- Sun, Y.; Zou, H.; Li, X.; Xu, S.; Liu, C. Plasma Metabolomics Reveals Metabolic Profiling For Diabetic Retinopathy and Disease Progression. Front. Endocrinol. 2021, 12, 757088. [Google Scholar] [CrossRef]
- Rhee, S.Y.; Jung, E.S.; Park, H.M.; Jeong, S.J.; Kim, K.; Chon, S.; Yu, S.-Y.; Woo, J.-T.; Lee, C.H. Plasma Glutamine and Glutamic Acid Are Potential Biomarkers for Predicting Diabetic Retinopathy. Metabolomics Off. J. Metabolomic Soc. 2018, 14, 89. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.-R.; Yang, F.-Y.; Lu, J.; Zhang, H.-R.; Sun, R.; Zhou, J.-B.; Yang, J.-K. Plasma Metabolomic Profiling of Proliferative Diabetic Retinopathy. Nutr. Metab. 2019, 16, 37. [Google Scholar] [CrossRef] [Green Version]
- Sumarriva, K.; Uppal, K.; Ma, C.; Herren, D.J.; Wang, Y.; Chocron, I.M.; Warden, C.; Mitchell, S.L.; Burgess, L.G.; Goodale, M.P.; et al. Arginine and Carnitine Metabolites Are Altered in Diabetic Retinopathy. Invest. Ophthalmol. Vis. Sci. 2019, 60, 3119–3126. [Google Scholar] [CrossRef]
- Peters, K.S.; Rivera, E.; Warden, C.; Harlow, P.A.; Mitchell, S.L.; Calcutt, M.W.; Samuels, D.C.; Brantley, M.A. Plasma Arginine and Citrulline Are Elevated in Diabetic Retinopathy. Am. J. Ophthalmol. 2022, 235, 154–162. [Google Scholar] [CrossRef]
- Lin, H.-T.; Cheng, M.-L.; Lo, C.-J.; Lin, G.; Lin, S.-F.; Yeh, J.-T.; Ho, H.-Y.; Lin, J.-R.; Liu, F.-C. 1H Nuclear Magnetic Resonance (NMR)-Based Cerebrospinal Fluid and Plasma Metabolomic Analysis in Type 2 Diabetic Patients and Risk Prediction for Diabetic Microangiopathy. J. Clin. Med. 2019, 8, 874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Li, S.; Wang, C.; Wang, Y.; Fang, J.; Liu, K. Plasma and Vitreous Metabolomics Profiling of Proliferative Diabetic Retinopathy. Invest. Ophthalmol. Vis. Sci. 2022, 63, 17. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Luo, X.; Lu, X.; Duan, J.; Xu, G. Metabolomics Study of Diabetic Retinopathy Using Gas Chromatography-Mass Spectrometry: A Comparison of Stages and Subtypes Diagnosed by Western and Chinese Medicine. Mol. Biosyst. 2011, 7, 2228–2237. [Google Scholar] [CrossRef] [PubMed]
- Abhary, S.; Kasmeridis, N.; Burdon, K.P.; Kuot, A.; Whiting, M.J.; Yew, W.P.; Petrovsky, N.; Craig, J.E. Diabetic Retinopathy Is Associated with Elevated Serum Asymmetric and Symmetric Dimethylarginines. Diabetes Care 2009, 32, 2084–2086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xuan, Q.; Ouyang, Y.; Wang, Y.; Wu, L.; Li, H.; Luo, Y.; Zhao, X.; Feng, D.; Qin, W.; Hu, C.; et al. Multiplatform Metabolomics Reveals Novel Serum Metabolite Biomarkers in Diabetic Retinopathy Subjects. Adv. Sci. Weinh. Baden-Wurtt. Ger. 2020, 7, 2001714. [Google Scholar] [CrossRef]
- Yun, J.H.; Kim, J.-M.; Jeon, H.J.; Oh, T.; Choi, H.J.; Kim, B.-J. Metabolomics Profiles Associated with Diabetic Retinopathy in Type 2 Diabetes Patients. PLoS ONE 2020, 15, e0241365. [Google Scholar] [CrossRef]
- Zuo, J.; Lan, Y.; Hu, H.; Hou, X.; Li, J.; Wang, T.; Zhang, H.; Zhang, N.; Guo, C.; Peng, F.; et al. Metabolomics-Based Multidimensional Network Biomarkers for Diabetic Retinopathy Identification in Patients with Type 2 Diabetes Mellitus. BMJ Open Diabetes Res. Care 2021, 9, e001443. [Google Scholar] [CrossRef]
- Yousri, N.A.; Suhre, K.; Yassin, E.; Al-Shakaki, A.; Robay, A.; Elshafei, M.; Chidiac, O.; Hunt, S.C.; Crystal, R.G.; Fakhro, K.A. Metabolic and Metabo-Clinical Signatures of Type 2 Diabetes, Obesity, Retinopathy, and Dyslipidemia. Diabetes 2022, 71, 184–205. [Google Scholar] [CrossRef]
- Curovic, V.R.; Suvitaival, T.; Mattila, I.; Ahonen, L.; Trošt, K.; Theilade, S.; Hansen, T.W.; Legido-Quigley, C.; Rossing, P. Circulating Metabolites and Lipids Are Associated to Diabetic Retinopathy in Individuals With Type 1 Diabetes. Diabetes 2020, 69, 2217–2226. [Google Scholar] [CrossRef]
- Quek, D.Q.Y.; He, F.; Sultana, R.; Banu, R.; Chee, M.L.; Nusinovici, S.; Thakur, S.; Qian, C.; Cheng, C.-Y.; Wong, T.Y.; et al. Novel Serum and Urinary Metabolites Associated with Diabetic Retinopathy in Three Asian Cohorts. Metabolites 2021, 11, 614. [Google Scholar] [CrossRef]
- Wang, H.; Fang, J.; Chen, F.; Sun, Q.; Xu, X.; Lin, S.-H.; Liu, K. Metabolomic Profile of Diabetic Retinopathy: A GC-TOFMS-Based Approach Using Vitreous and Aqueous Humor. Acta Diabetol. 2020, 57, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Barba, I.; Garcia-Ramírez, M.; Hernández, C.; Alonso, M.A.; Masmiquel, L.; García-Dorado, D.; Simó, R. Metabolic Fingerprints of Proliferative Diabetic Retinopathy: An 1H-NMR-Based Metabonomic Approach Using Vitreous Humor. Invest. Ophthalmol. Vis. Sci. 2010, 51, 4416–4421. [Google Scholar] [CrossRef] [PubMed]
- Tomita, Y.; Cagnone, G.; Fu, Z.; Cakir, B.; Kotoda, Y.; Asakage, M.; Wakabayashi, Y.; Hellström, A.; Joyal, J.-S.; Talukdar, S.; et al. Vitreous Metabolomics Profiling of Proliferative Diabetic Retinopathy. Diabetologia 2021, 64, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Paris, L.P.; Johnson, C.H.; Aguilar, E.; Usui, Y.; Cho, K.; Hoang, L.T.; Feitelberg, D.; Benton, H.P.; Westenskow, P.D.; Kurihara, T.; et al. Global Metabolomics Reveals Metabolic Dysregulation in Ischemic Retinopathy. Metabolomics Off. J. Metabolomic Soc. 2016, 12, 15. [Google Scholar] [CrossRef] [Green Version]
- Haines, N.R.; Manoharan, N.; Olson, J.L.; D’Alessandro, A.; Reisz, J.A. Metabolomics Analysis of Human Vitreous in Diabetic Retinopathy and Rhegmatogenous Retinal Detachment. J. Proteome Res. 2018, 17, 2421–2427. [Google Scholar] [CrossRef]
- Jin, H.; Zhu, B.; Liu, X.; Jin, J.; Zou, H. Metabolic Characterization of Diabetic Retinopathy: An 1H-NMR-Based Metabolomic Approach Using Human Aqueous Humor. J. Pharm. Biomed. Anal. 2019, 174, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Kunikata, H.; Ida, T.; Sato, K.; Aizawa, N.; Sawa, T.; Tawarayama, H.; Murayama, N.; Fujii, S.; Akaike, T.; Nakazawa, T. Metabolomic Profiling of Reactive Persulfides and Polysulfides in the Aqueous and Vitreous Humors. Sci. Rep. 2017, 7, 41984. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; Zheng, Z.; Xiong, X.; Chen, X.; Peng, J.; Yao, H.; Pu, J.; Chen, Q.; Zheng, M. Gut Microbiota Composition and Fecal Metabolic Profiling in Patients With Diabetic Retinopathy. Front. Cell Dev. Biol. 2021, 9, 732204. [Google Scholar] [CrossRef] [PubMed]
- Ye, P.; Zhang, X.; Xu, Y.; Xu, J.; Song, X.; Yao, K. Alterations of the Gut Microbiome and Metabolome in Patients With Proliferative Diabetic Retinopathy. Front. Microbiol. 2021, 12, 667632. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; Xie, M.; Deng, L.; Zhang, M.; Xie, X. Urine Metabolomics Study of Bushen Huoxue Prescription on Diabetic Retinopathy Rats by UPLC-Q-Exactive Orbitrap-MS. Biomed. Chromatogr. BMC 2020, 34, e4792. [Google Scholar] [CrossRef] [PubMed]
- Wiggenhauser, L.M.; Qi, H.; Stoll, S.J.; Metzger, L.; Bennewitz, K.; Poschet, G.; Krenning, G.; Hillebrands, J.-L.; Hammes, H.-P.; Kroll, J. Activation of Retinal Angiogenesis in Hyperglycemic Pdx1 -/- Zebrafish Mutants. Diabetes 2020, 69, 1020–1031. [Google Scholar] [CrossRef] [PubMed]
- Laíns, I.; Duarte, D.; Barros, A.S.; Martins, A.S.; Gil, J.; Miller, J.B.; Marques, M.; Mesquita, T.; Kim, I.K.; Cachulo, M.d.L.; et al. Human Plasma Metabolomics in Age-Related Macular Degeneration (AMD) Using Nuclear Magnetic Resonance Spectroscopy. PLoS ONE 2017, 12, e0177749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, S.L.; Ma, C.; Scott, W.K.; Agarwal, A.; Pericak-Vance, M.A.; Haines, J.L.; Jones, D.P.; Uppal, K.; Brantley, M.A. Plasma Metabolomics of Intermediate and Neovascular Age-Related Macular Degeneration Patients. Cells 2021, 10, 3141. [Google Scholar] [CrossRef]
- Luo, D.; Deng, T.; Yuan, W.; Deng, H.; Jin, M. Plasma Metabolomic Study in Chinese Patients with Wet Age-Related Macular Degeneration. BMC Ophthalmol. 2017, 17, 165. [Google Scholar] [CrossRef] [Green Version]
- Laíns, I.; Chung, W.; Kelly, R.S.; Gil, J.; Marques, M.; Barreto, P.; Murta, J.N.; Kim, I.K.; Vavvas, D.G.; Miller, J.B.; et al. Human Plasma Metabolomics in Age-Related Macular Degeneration: Meta-Analysis of Two Cohorts. Metabolites 2019, 9, 127. [Google Scholar] [CrossRef] [Green Version]
- Osborn, M.P.; Park, Y.; Parks, M.B.; Burgess, L.G.; Uppal, K.; Lee, K.; Jones, D.P.; Brantley, M.A. Metabolome-Wide Association Study of Neovascular Age-Related Macular Degeneration. PLoS ONE 2013, 8, e72737. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, S.; Saha, M.; Das, D. A Study on Plasma Homocysteine Level in Age-Related Macular Degeneration. Nepal. J. Ophthalmol. Biannu. Peer-Rev. Acad. J. Nepal Ophthalmic Soc. NEPJOPH 2013, 5, 195–200. [Google Scholar] [CrossRef] [Green Version]
- Chao de la Barca, J.M.; Rondet-Courbis, B.; Ferré, M.; Muller, J.; Buisset, A.; Leruez, S.; Plubeau, G.; Macé, T.; Moureauzeau, L.; Chupin, S.; et al. A Plasma Metabolomic Profiling of Exudative Age-Related Macular Degeneration Showing Carnosine and Mitochondrial Deficiencies. J. Clin. Med. 2020, 9, 631. [Google Scholar] [CrossRef] [Green Version]
- Deng, Y.; Shuai, P.; Wang, H.; Zhang, S.; Li, J.; Du, M.; Huang, P.; Qu, C.; Huang, L. Untargeted Metabolomics for Uncovering Plasma Biological Markers of Wet Age-Related Macular Degeneration. Aging 2021, 13, 13968–14000. [Google Scholar] [CrossRef]
- Mendez, K.M.; Kim, J.; Laíns, I.; Nigalye, A.; Katz, R.; Pundik, S.; Kim, I.K.; Liang, L.; Vavvas, D.G.; Miller, J.B.; et al. Association of Human Plasma Metabolomics with Delayed Dark Adaptation in Age-Related Macular Degeneration. Metabolites 2021, 11, 183. [Google Scholar] [CrossRef] [PubMed]
- Kersten, E.; Dammeier, S.; Ajana, S.; Groenewoud, J.M.M.; Codrea, M.; Klose, F.; Lechanteur, Y.T.; Fauser, S.; Ueffing, M.; Delcourt, C.; et al. Metabolomics in Serum of Patients with Non-Advanced Age-Related Macular Degeneration Reveals Aberrations in the Glutamine Pathway. PLoS ONE 2019, 14, e0218457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, G.; Wei, P.; He, M.; Teng, H.; Chu, Y. Metabolomic Profiling of the Aqueous Humor in Patients with Wet Age-Related Macular Degeneration Using UHPLC-MS/MS. J. Proteome Res. 2020, 19, 2358–2366. [Google Scholar] [CrossRef] [PubMed]
- Laíns, I.; Duarte, D.; Barros, A.S.; Martins, A.S.; Carneiro, T.J.; Gil, J.Q.; Miller, J.B.; Marques, M.; Mesquita, T.S.; Barreto, P.; et al. Urine Nuclear Magnetic Resonance (NMR) Metabolomics in Age-Related Macular Degeneration. J. Proteome Res. 2019, 18, 1278–1288. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, Y.; Zhang, X.; Huang, Q.; Tan, W.; Yang, Y.; He, X.; Yoshida, S.; Zhao, P.; Li, Y. Plasma Levels of Amino Acids and Derivatives in Retinopathy of Prematurity. Int. J. Med. Sci. 2021, 18, 3581–3587. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, Y.; Zhang, X.; Zhao, P.; Gong, X.; He, M.; Cao, J.; Jiang, B.; Yoshida, S.; Li, Y. Plasma Metabolites in Treatment-Requiring Retinopathy of Prematurity: Potential Biomarkers Identified by Metabolomics. Exp. Eye Res. 2020, 199, 108198. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, Q.; Luo, S.; Zhang, Y.; Lian, C.; He, H.; Zeng, J.; Zhang, G. Comparative Analysis Reveals Novel Changes in Plasma Metabolites and Metabolomic Networks of Infants With Retinopathy of Prematurity. Investig. Ophthalmol. Vis. Sci. 2022, 63, 28. [Google Scholar] [CrossRef]
- Ozcan, Y.; Huseyin, G.; Sonmez, K. Evaluation of Plasma Amino Acid Levels in Preterm Infants and Their Potential Correlation with Retinopathy of Prematurity. J. Ophthalmol. 2020, 2020, 8026547. [Google Scholar] [CrossRef]
- Nilsson, A.K.; Tebani, A.; Malmodin, D.; Pedersen, A.; Hellgren, G.; Löfqvist, C.; Hansen-Pupp, I.; Uhlén, M.; Hellström, A. Longitudinal Serum Metabolomics in Extremely Premature Infants: Relationships With Gestational Age, Nutrition, and Morbidities. Front. Neurosci. 2022, 16, 830884. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, Z.; Li, S.; Yang, M.; Xiao, X.; Lian, C.; Wen, W.; He, H.; Zeng, J.; Wang, J.; et al. Targeted Blood Metabolomic Study on Retinopathy of Prematurity. Invest. Ophthalmol. Vis. Sci. 2020, 61, 12. [Google Scholar] [CrossRef]
- Zhou, Y.; Tan, W.; Zou, J.; Cao, J.; Huang, Q.; Jiang, B.; Yoshida, S.; Li, Y. Metabolomics Analyses of Mouse Retinas in Oxygen-Induced Retinopathy. Investig. Ophthalmol. Vis. Sci. 2021, 62, 9. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Liu, Y.; Guo, Y.; Gao, Y.; Piao, Y.; Tan, S.; Tang, Y. Metabolomic Changes of Blood Plasma Associated with Two Phases of Rat OIR. Exp. Eye Res. 2020, 190, 107855. [Google Scholar] [CrossRef] [PubMed]
- Chetwynd, A.J.; Dunn, W.B.; Rodriguez-Blanco, G. Collection and Preparation of Clinical Samples for Metabolomics. Adv. Exp. Med. Biol. 2017, 965, 19–44. [Google Scholar] [CrossRef] [PubMed]
- Vignoli, A.; Tenori, L.; Morsiani, C.; Turano, P.; Capri, M.; Luchinat, C. Serum or Plasma (and Which Plasma), That Is the Question. J. Proteome Res. 2022, 21, 1061–1072. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Kastenmüller, G.; He, Y.; Belcredi, P.; Möller, G.; Prehn, C.; Mendes, J.; Wahl, S.; Roemisch-Margl, W.; Ceglarek, U.; et al. Differences between Human Plasma and Serum Metabolite Profiles. PLoS ONE 2011, 6, e21230. [Google Scholar] [CrossRef] [PubMed]
- Floyd, J.L.; Grant, M.B. The Gut-Eye Axis: Lessons Learned from Murine Models. Ophthalmol. Ther. 2020, 9, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Emwas, A.-H.M. The Strengths and Weaknesses of NMR Spectroscopy and Mass Spectrometry with Particular Focus on Metabolomics Research. Methods Mol. Biol. Clifton NJ 2015, 1277, 161–193. [Google Scholar] [CrossRef]
- Barnes, S.; Benton, H.P.; Casazza, K.; Cooper, S.J.; Cui, X.; Du, X.; Engler, J.A.; Kabarowski, J.H.; Li, S.; Pathmasiri, W.; et al. Training in Metabolomics Research. I. Designing the Experiment, Collecting and Extracting Samples and Generating Metabolomics Data. J. Mass Spectrom. JMS 2016, 51, ii–iii. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Connor, K.M.; Krah, N.M.; Dennison, R.J.; Aderman, C.M.; Chen, J.; Guerin, K.I.; Sapieha, P.; Stahl, A.; Willett, K.L.; Smith, L.E.H. Quantification of Oxygen-Induced Retinopathy in the Mouse: A Model of Vessel Loss, Vessel Regrowth and Pathological Angiogenesis. Nat. Protoc. 2009, 4, 1565–1573. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Fu, X.; Zeng, H.; Wang, J.-H.; Peng, Y.; Zhao, H.; Zou, J.; Zhang, L.; Li, Y.; Yoshida, S.; et al. Microarray Analysis of Long Non-Coding RNAs and Messenger RNAs in a Mouse Model of Oxygen-Induced Retinopathy. Int. J. Med. Sci. 2019, 16, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Amin, S.M.; Gonzalez, A.; Guevara, J.; Bolch, C.; Andersen, L.; Smith, W.C.; Agarwal-Sinha, S. Efficacy of Aflibercept Treatment and Its Effect on the Retinal Perfusion in the Oxygen-Induced Retinopathy Mouse Model of Retinopathy of Prematurity. Ophthalmic Res. 2021, 64, 91–98. [Google Scholar] [CrossRef]
- Becker, K.; Klein, H.; Simon, E.; Viollet, C.; Haslinger, C.; Leparc, G.; Schultheis, C.; Chong, V.; Kuehn, M.H.; Fernandez-Albert, F.; et al. In-Depth Transcriptomic Analysis of Human Retina Reveals Molecular Mechanisms Underlying Diabetic Retinopathy. Sci. Rep. 2021, 11, 10494. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, P.; Corraliza, L.; Villena, J.A.; Carvalho, A.R.; Garcia-Arumí, J.; Ramos, D.; Ruberte, J.; Simó, R.; Hernández, C. The Db/Db Mouse: A Useful Model for the Study of Diabetic Retinal Neurodegeneration. PLoS ONE 2014, 9, e97302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ng, Y.-K.; Zeng, X.-X.; Ling, E.-A. Expression of Glutamate Receptors and Calcium-Binding Proteins in the Retina of Streptozotocin-Induced Diabetic Rats. Brain Res. 2004, 1018, 66–72. [Google Scholar] [CrossRef]
- Santiago, A.R.; Gaspar, J.M.; Baptista, F.I.; Cristóvão, A.J.; Santos, P.F.; Kamphuis, W.; Ambrósio, A.F. Diabetes Changes the Levels of Ionotropic Glutamate Receptors in the Rat Retina. Mol. Vis. 2009, 15, 1620–1630. [Google Scholar]
- Hartwick, A.T.E.; Hamilton, C.M.; Baldridge, W.H. Glutamatergic Calcium Dynamics and Deregulation of Rat Retinal Ganglion Cells. J. Physiol. 2008, 586, 3425–3446. [Google Scholar] [CrossRef]
- Thoreson, W.B.; Witkovsky, P. Glutamate Receptors and Circuits in the Vertebrate Retina. Prog. Retin. Eye Res. 1999, 18, 765–810. [Google Scholar] [CrossRef] [PubMed]
- Kritis, A.A.; Stamoula, E.G.; Paniskaki, K.A.; Vavilis, T.D. Researching Glutamate—Induced Cytotoxicity in Different Cell Lines: A Comparative/Collective Analysis/Study. Front. Cell. Neurosci. 2015, 9, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schrammel, A.; Gorren, A.C.F.; Schmidt, K.; Pfeiffer, S.; Mayer, B. S-Nitrosation of Glutathione by Nitric Oxide, Peroxynitrite, and (*)NO/O(2)(*-). Free Radic. Biol. Med. 2003, 34, 1078–1088. [Google Scholar] [CrossRef]
- Brown, G.C. Nitric Oxide and Neuronal Death. Nitric Oxide Biol. Chem. 2010, 23, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Almasieh, M.; Wilson, A.M.; Morquette, B.; Cueva Vargas, J.L.; Di Polo, A. The Molecular Basis of Retinal Ganglion Cell Death in Glaucoma. Prog. Retin. Eye Res. 2012, 31, 152–181. [Google Scholar] [CrossRef] [PubMed]
- Lebrun-Julien, F.; Duplan, L.; Pernet, V.; Osswald, I.; Sapieha, P.; Bourgeois, P.; Dickson, K.; Bowie, D.; Barker, P.A.; Di Polo, A. Excitotoxic Death of Retinal Neurons in Vivo Occurs via a Non-Cell-Autonomous Mechanism. J. Neurosci. Off. J. Soc. Neurosci. 2009, 29, 5536–5545. [Google Scholar] [CrossRef] [Green Version]
- Gunasekar, P.G.; Kanthasamy, A.G.; Borowitz, J.L.; Isom, G.E. NMDA Receptor Activation Produces Concurrent Generation of Nitric Oxide and Reactive Oxygen Species: Implication for Cell Death. J. Neurochem. 1995, 65, 2016–2021. [Google Scholar] [CrossRef]
- Kashii, S.; Mandai, M.; Kikuchi, M.; Honda, Y.; Tamura, Y.; Kaneda, K.; Akaike, A. Dual Actions of Nitric Oxide in N-Methyl-D-Aspartate Receptor-Mediated Neurotoxicity in Cultured Retinal Neurons. Brain Res. 1996, 711, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Engerman, R.L.; Case, G.L.; Kern, T.S. Retinal Glutamate in Diabetes and Effect of Antioxidants. Neurochem. Int. 2001, 38, 385–390. [Google Scholar] [CrossRef]
- Lieth, E.; Barber, A.J.; Xu, B.; Dice, C.; Ratz, M.J.; Tanase, D.; Strother, J.M. Glial Reactivity and Impaired Glutamate Metabolism in Short-Term Experimental Diabetic Retinopathy. Penn State Retina Research Group. Diabetes 1998, 47, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Puro, D.G. Diabetes-Induced Dysfunction of the Glutamate Transporter in Retinal Müller Cells. Investig. Ophthalmol. Vis. Sci. 2002, 43, 3109–3116. [Google Scholar]
- Fan, W.; Cooper, N.G.F. Glutamate-Induced NFκB Activation in the Retina. Investig. Ophthalmol. Vis. Sci. 2009, 50, 917–925. [Google Scholar] [CrossRef] [Green Version]
- Elsherbiny, N.M.; Sharma, I.; Kira, D.; Alhusban, S.; Samra, Y.A.; Jadeja, R.; Martin, P.; Al-Shabrawey, M.; Tawfik, A. Homocysteine Induces Inflammation in Retina and Brain. Biomolecules 2020, 10, 393. [Google Scholar] [CrossRef] [Green Version]
- Singh, M.; Tyagi, S.C. Homocysteine Mediates Transcriptional Changes of the Inflammatory Pathway Signature Genes in Human Retinal Pigment Epithelial Cells. Int. J. Ophthalmol. 2017, 10, 696–704. [Google Scholar] [CrossRef]


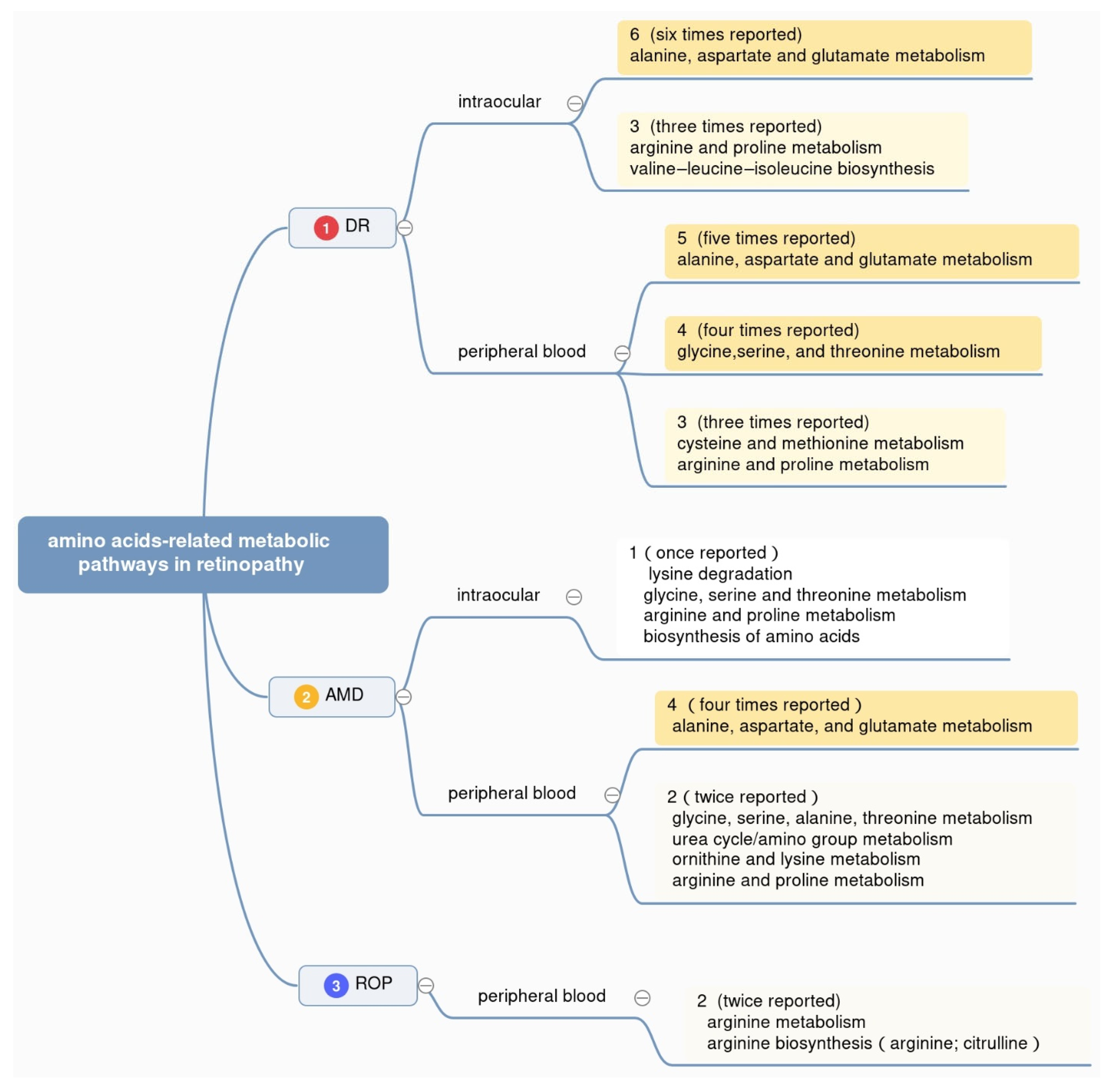
| Species | Samples | Subjects | Platforms | Criteria | Differential Metabolites | Study |
|---|---|---|---|---|---|---|
| Human | Plasma | 42 DR/ 32 NDR | UPLC-MS | Fold change (FC) > 1.2 and p < 0.05 | N-Fructosyl isoleucine | Sun et al. (2021) [17] |
| N-acetyltryptophan | ||||||
| Leucylleucine | ||||||
| Kynurenic acid | ||||||
| 3-Methylhistidine | ||||||
| Phenylacetylglutamine | ||||||
| 21 PDR/ 21 NPDR | Glutamine | |||||
| 52 PDR/ 72 NPDR/ 59 NDR | UPLC-MS, GC-MS | Variable important in the projection (VIP) > 0.7 | asparagine, aspartic acid, glutamic acid, glutamine, glycine, methionine, pyroglutamic acid | Rhee et al. (2018) [18] | ||
| 21 PDR/ 21 NDR | UPLC-MS | p < 10 × 10−5, Area under the curve (AUC) ≥ 0.95, VIP > 1 | L-serine, β-alanine, L-Proline, L-Homoserine,4-Hydroxyproline, Ornithine, L-Aspartic acid, L-Glutamine, L-Glutamic acid, L-Methionine, N-Acetylornithine, L-Arginine, N-Acetyl-L-aspartic acid, Citrulline, Phosphoserine | Zhu et al. (2019) [19] | ||
| 83 DR/ 90 NDR | LC-MS | VIP ≥ 2.0 | arginine, citrulline | Sumarriva et al. (2019) [20] | ||
| 64 PDR/ 92 NPDR/ 159 NDR | LC-MS | FDR (false-discovery rate) < 0.05 | ↑: arginine, citrulline (DR/ NDR) | Peters et al. (2021) [21] | ||
| 19 DR/ 14 controls | NMR | p < 0.05 | ↑: leucine, isoleucine, tyrosine, and valine; ↓: histidine, Alanine | Lin et al. (2019) [22] | ||
| 88 PDR/ 51 controls | UPLC-MS | VIP > 1, FC > 1.2 and < 0.83, FDR = 0.05 | ↑: Pyroglutamic acid, Alpha-N-phenylacetyl-L glutamine | Wang et al. (2022) [23] | ||
| Serum | 25 NDR/ 39 DR/ 25 PDR | GC-MS | largest VIP | L-aspartic acid | Li et al. (2011) [24] | |
| 176 DR/ 329 NDR | LC-MS | p < 0.05 | ↑: asymmetric dimethylarginine (ADMA), L-arginine, symmetric dimethylarginine (SDMA) | Abhary et al. (2009) [25] | ||
| 689 DR/ 216 control | GC-MS, LC-MS | p < 0.05, FDR < 0.05 | ↑: serine, glycine, arginine, ornithine, citrulline, proline, leucine, isoleucine, and valine | Xuan et al. (2020) [26] | ||
| 51 PDR/ 123 NPDR/ 143 NDR | LC–MS | p < 0.05 | DR/ NDR: proline, NPDR than NDR: alanine, aspartic acid, and glutamine, arginine, histidine, lysine, methionine, threonine, tryptophan, and tyrosine, PDR than NDR: lysine, methionine, serine, tryptophan, and tyrosine; PDR than NPDR: total dimethylarginine | Yun et al. (2020) [27] | ||
| 69 DR/ 69 NDR | UPLC-MS | VIP > 1, FC< 0.8 or > 1.2 and FDR < 0.05 | ornithine, phenylacetylglutamine | Zuo et al. (2021) [28] | ||
| 123 DR/ 116 NDR | Metabolon DiscoveryHD4 | p < 0.05 | cysteine-glutathione disulfide, phenylacetylglutamine, cys-gly (oxidized), N-acetylmethionine glycylvaline, phenylalanine, aspartate, tryptophan, glutamate | Yousri et al. (2022) [29] | ||
| Plasma and serum | 228 PDR/ 276 NPDR/ 141 NDR | GC-MS, UHPLC-MS | (Benjamini–Hochberg) BH- adjusted p < 0.05 | alanine and serine | Curovic et al. (2020) [30] | |
| 666 DR/ 2211 NDR | NMR | p < 0.05 | tyrosine | Quek et al. (2021) [31] | ||
| Vitreous humor | 28 PDR/ 22 NDM | GC-MS | VIP > 1 p < 0.05 | alanine, alloisoleucine, creatinine, glutamine, leucine, lysine, ornithine, pyroglutamic acid, phenylalanine, threonine, valine | Wang et al. (2020) [32] | |
| 51 PDR/ 23 controls | UPLC-MS | VIP > 1 FC >1.2 and < 0.83 FDR = 0.05 | alpha-N-phenylacetyl-L glutamine, pyroglutamic acid | Wang et al. (2022) [23] | ||
| 22 PDR/ 22 NDM | NMR | p ≤ 0.05 | alanine, valine, glutamine, leucine, isoleucine | Barba et al. (2010) [33] | ||
| 35 PDR/ 19 NDM | UHPLC-MS | p < 0.05 | citrulline, dimethylglycine, glycine, ornithine, proline, creatine | Tomita et al. (2021) [34] | ||
| 20 PDR/ 31 NDM | HPLC-MS | p < 0.05 | Methionine, arginine, proline, citrulline, ornithine | Paris et al. (2016) [35] | ||
| 9 PDR/ 8 controls | UHPLC-MS | p < 0.05 | citrulline, glutamine, N-amidino-L-aspartate, proline, 5-oxoproline | Haines et al. (2018) [36] | ||
| Aqueous and humor | 13 DR/ 14 NDR | NMR | VIP > 1.0 p < 0.05 | asparagine, DMA, glutamine, histidine, threonine | Jin et al. (2019) [37] | |
| 23 PDR/ 25 NDM | GC-MS | VIP > 1.0 p < 0.05 | citrulline | Wang et al. (2020) [32] | ||
| Aqueous and vitreous humor | 18 PDR/ 22 controls | LC-MS | p < 0.05 | Cysteine persulfides (CysSSH), cystine, oxidized glutathione trisulfide (GSSSG) | Kunikata et al. (2017) [38] | |
| CSF | 19 DR/ 14 controls | NMR | FC > 1.2 or < 0.8, FDR < 0.05 | alanine, leucine, isoleucine, tyrosine, and valine, histidine, | Lin et al. (2019) [22] | |
| Urine | 666 DR/ 2211 NDR | NMR | p < 0.05 | Alanine, glutamine | Quek et al. (2021) [31] | |
| Fecal samples | 21 PDR/ 14 NDR | UPLC-MS | VIP > 1, p < 0.05 | pyro-L-glutaminyl-L-glutamine, D-proline, N-gamma-L-glutamyl-D-alanine, N-acetyl-L-methionine, L-threo-3-phenylserine, | Zhou et al. (2021) [39] | |
| 45 PDR/ 90 NDR | UPLC-MS | p < 0.05, VIP> 1, and log2 FC > 1 | tyrosine | Ye et al. (2021) [40] | ||
| Rats | Urine | 6 DR rats/6 controls | UPLC-MS | VIP > 1, p < 0.05 | phenylacetylglycine, 5-l-glutamyl-taurine | Wang et al. (2020) [41] |
| Zebrafish | Whole body | 50 pdx1−/− zebrafish | UHPLC–MS | p < 0.05 | glutamate, proline, ornithine, tyrosine | Wiggenhauser et al. (2020) [42] |
| Species | Samples | Subjects | Platforms | Criteria | Differential Metabolites | Study |
|---|---|---|---|---|---|---|
| Human | Plasma | Coimbra: 201 AMD /42 controls | NMR | p < 0.05 | Early AMD vs. Controls: creatine; Late vs. Intermediate AMD: histidine | Laíns et al. (2017) [43] |
| Boston: 113 AMD /40 controls | p < 0.05 | Early AMD vs. Controls: glutamine; Intermediate vs. Early AMD: glutamine, histidine; Late vs. Intermediate AMD: histidine, alanine | ||||
| 91 IAMD /100 NVAMD /195 controls | LC-MS | VIP ≥ 2.0 and an p < 0.05 | AMD vs. controls: pyroglutamic acid; NVAMD vs. IAMD: kynurenine | Mitchell et al. (2021) [44] | ||
| 20 wetAMD /20 controls | UHPLC-MS, QTOF-MS | VIP > 1 and p < 0.05 or 0.05 < p < 0.1 | N-Acetyl-L-alanine, L-Tyrosine, L-Phenylalanine, L-Methionine, L-Arginine, | Luo et al. (2017) [45] | ||
| Boston: 149 AMD /47 controls, Coimbra: 242 AMD /53 controls | UHPLC-MS | FDR < 0.05 | Beta-citrylglutamate, N-acetylmethionine, aspartate, N-acetylasparagine, S-adenosylhomocysteine (SAH), isoleucylglycine N-acetylasparagine, beta-citrylglutamate, N-acetylleucine | Laíns et al. (2019) [46] | ||
| 26 NVAMD /19 controls | LC-FTMS | FDR< 0.05 | Acetylphenylalanine | Osborn et al. (2013) [47] | ||
| 32 AMD /32 controls | HPLC-MS | p < 0.05 | Homocysteine | Ghosh et al. (2013) [48] | ||
| 40 AMD /40 controls | LC-MS | 0.6 < FC < 1.4 p < 0.1 | Valine, lysine, proline | Chao et al. (2020) [49] | ||
| 127 wAMD /50 controls | UHPLC-MS | FC ≥ 2 and FC ≤ 0.5, p < 0.05, VIP ≥ 1 | L-Tryptophan; L-Alanyl-L-Lysine | Deng et al. (2021) [50] | ||
| Plasma | 53 AMD /18 controls | UPLC-MS | p < 0.01 | N-acetylglutamine, N-acetylleucine | Mendez et al. (2021) [51] | |
| Serum | 72 AMD /72 controls | microLC-MS | sPLSda (to select the most predictive variables from dataset) | (Predictors for non-advanced AMD) glutamine, glutamate:glutamine ratio, glutaminolysis | Kersten et al. (2019) [52] | |
| Aqueous humor | 26 wetAMD /20 controls | UHPLC-MS | VIP > 1.0 and p < 0.05 | N6, N6, N6-trimethyl-L-lysine, norleucine, L-phenylalanine, γ-glutamylglutamine, N-acetylhistidine, creatine, N-fructosyl isoleucine, L-proline | Han et al. (2020) [53] | |
| Urine | Coimbra cohort: 252 AMD /53 controls | NMR | p < 0.05 | Late AMD vs. Intermediate AMD: valine | Laíns et al. (2019) [54] | |
| Boston cohort: 147 AMD /47 controls | p < 0.05 | Intermediate AMD vs. Early AMD: glycine, lysine, tyrosine |
| Species | Samples | Subjects | Platforms | Criteria | Differential Metabolites | Study |
|---|---|---|---|---|---|---|
| Human | Plasma | 58 ROP /25 controls | HPLC-MS/MS | p < 0.05 | Creatinine, citrulline, arginine, and aminoadipic acid, creatinine, and aminoadipic acid | Zhou et al. (2021) [55] |
| 38 treatment requiring-ROP /23 ROP | UHPLC-MS | FC > 1.5, p < 0.05 | L-Lysine, L-Citrulline, Pro-Thr, L-Glutamine, L-Pyroglutamic acid, L-Tryptophan, Cysteine-S-sulfate, | Zhou et al. (2020) [56] | ||
| 57 ROP /57 controls | UPLC-MS/MS | VIP > 0.5 | Glutamic acid gamma-methyl ester, Picolinoylglycine, Creatinine, Ornithine, 1-Carboxyethylisoleucine | Yang et al. (2022) [57] | ||
| 26 ROP /29 controls | LC-MS/MS | p < 0.05 | Arginine, Lysine, aspartic acid, glutamine | Ozcan et al. (2020) [58] | ||
| Serum | 87 ROP | NMR | p < 0.05 | Phenylalanine, Lysine | Nilsson et al. (2022) [59] | |
| Heel blood | 40 ROP /41 controls | UPLC-MS | VIP > 1 | Glycine, glutamate, leucine, serine, valine, tryptophan, citrulline and homocysteine | Yang et al. (2020) [60] | |
| OIR mice | Retina | 10 OIR /10 controls | HPLC-MS/MS | FC > 1.5, p < 0.05. | Dimethylglycine, L-Citrulline, N-Acetyl-L-aspartic acid, L-Alanine, Gly-Glu, D-Proline, Gamma-L-Glutamyl-L-glutamic acid, Creatine, L-Pyroglutamic acid, L-Leucine, L-Aspartate, L-Glutamate, Glycine, L-Citrulline, D-Proline, L-Valine, N-Acetyl-L-aspartic acid ↓, Gamma-L-Glutamyl-L-glutamic acid, Creatine, L-Alanine, L-Aspartate | Zhou et al. (2021) [61] |
| Whole eyes | 3 P12 mice /3 P14 mice /4 P17 mice /5 controls | HPLC-MS | p < 0.05 | Arginine, Proline, Citrulline, Ornithine, Lysine | Paris et al. (2016) [35] | |
| Rats | Plasma | Four groups, each with 12 individuals: (i) CT1; (ii) HO1; (iii) CT2; (iv) HO2 | GC-MS | p < 0.05, VIP ≥ 1 | HO1/CT1: L-Tyrosine, L-Proline, Ornithine, L-Tryptophan, L-Glutamine, L-Threonine, 4-Hydroxy-L-proline, Glycine, HO2/HO1: L-Proline, L-Alanine, L-Glutamine, L-Tyrosine, Ornithine, L-Threonine, L-Valine, L-Alloisoleucine, L-Lysine, L-Serine, L-Histidine, L-Arginine | Lu et al. (2020) [62] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, M.; Zhang, F. Amino Acids Metabolism in Retinopathy: From Clinical and Basic Research Perspective. Metabolites 2022, 12, 1244. https://doi.org/10.3390/metabo12121244
Xia M, Zhang F. Amino Acids Metabolism in Retinopathy: From Clinical and Basic Research Perspective. Metabolites. 2022; 12(12):1244. https://doi.org/10.3390/metabo12121244
Chicago/Turabian StyleXia, Mengxue, and Fang Zhang. 2022. "Amino Acids Metabolism in Retinopathy: From Clinical and Basic Research Perspective" Metabolites 12, no. 12: 1244. https://doi.org/10.3390/metabo12121244





