No Indices of Increased Type 2 Diabetes Risk in Individuals with Reactive Postprandial Hypoglycemia
Abstract
:1. Introduction
2. Methods
2.1. Definition of Clinical and Laboratory Characteristics
2.2. Statistical Analysis
3. Results
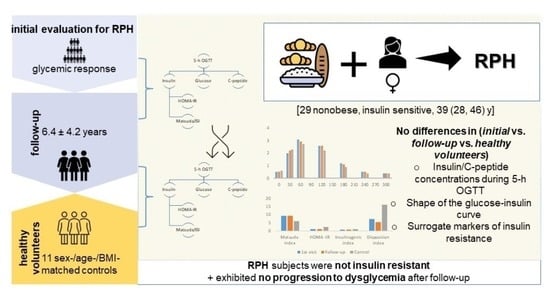
3.1. Characteristics of the Cohort of Individuals with RPH
3.2. Glycemic Response upon Glucose Stimulation in Individuals with RPH at Baseline and Follow-Up
3.3. Presentation of Symptoms in Subjects with RPH
3.4. New-Onset Diabetes Occurrence
3.5. Characteristics of Subjects with the RPH and Control Group
3.6. Comparison of Biochemical Characteristics of Subjects with RPH vs. the Control Group
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cryer, P.E.; Axelrod, L.; Grossman, A.B.; Heller, S.R.; Montori, V.M.; Seaquist, E.R.; Service, F.J. Endocrine Society Evaluation and Management of Adult Hypoglycemic Disorders: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2009, 94, 709–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brun, J.F.; Fedou, C.; Mercier, J. Postprandial Reactive Hypoglycemia. Diabetes Metab. 2000, 26, 337–351. [Google Scholar] [PubMed]
- Altuntaş, Y. Postprandial Reactive Hypoglycemia. Sisli Etfal Hastan. Tip Bul. 2019, 53, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Galati, S.-J.; Rayfield, E.J. Approach to the Patient with Postprandial Hypoglycemia. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2014, 20, 331–340. [Google Scholar] [CrossRef]
- Hall, M.; Walicka, M.; Panczyk, M.; Traczyk, I. Metabolic Parameters in Patients with Suspected Reactive Hypoglycemia. J. Pers. Med. 2021, 11, 276. [Google Scholar] [CrossRef]
- Mongraw-Chaffin, M.; Beavers, D.P.; McClain, D.A. Hypoglycemic Symptoms in the Absence of Diabetes: Pilot Evidence of Clinical Hypoglycemia in Young Women. J. Clin. Transl. Endocrinol. 2019, 18, 100202. [Google Scholar] [CrossRef]
- Simpson, E.J.; Holdsworth, M.; Macdonald, I.A. Prevalence of Self-Reported Symptoms Attributed to Hypoglycaemia within a General Female Population of the UK. J. Psychosom. Res. 2006, 60, 403–406. [Google Scholar] [CrossRef]
- Hall, M.; Walicka, M.; Panczyk, M.; Traczyk, I. Assessing Long-Term Impact of Dietary Interventions on Occurrence of Symptoms Consistent with Hypoglycemia in Patients without Diabetes: A One-Year Follow-Up Study. Nutrients 2022, 14, 497. [Google Scholar] [CrossRef]
- Martens, P.; Tits, J. Approach to the Patient with Spontaneous Hypoglycemia. Eur. J. Intern. Med. 2014, 25, 415–421. [Google Scholar] [CrossRef]
- Kandaswamy, L.; Raghavan, R.; Pappachan, J.M. Spontaneous Hypoglycemia: Diagnostic Evaluation and Management. Endocrine 2016, 53, 47–57. [Google Scholar] [CrossRef]
- Lupoli, R.; Cotugno, M.; Griffo, E.; Nosso, G.; Riccardi, G.; Capaldo, B. Role of the Entero-Insular Axis in the Pathogenesis of Idiopathic Reactive Hypoglycemia: A Pilot Study. J. Clin. Endocrinol. Metab. 2015, 100, 4441–4446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berlin, I.; Grimaldi, A.; Landault, C.; Cesselin, F.; Puech, A.J. Suspected Postprandial Hypoglycemia Is Associated with Beta-Adrenergic Hypersensitivity and Emotional Distress. J. Clin. Endocrinol. Metab. 1994, 79, 1428–1433. [Google Scholar] [CrossRef] [PubMed]
- Hadji-Georgopoulos, A.; Schmidt, M.I.; Margolis, S.; Kowarski, A.A. Elevated Hypoglycemic Index and Late Hyperinsulinism in Symptomatic Postprandial Hypoglycemia. J. Clin. Endocrinol. Metab. 1980, 50, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Tamburrano, G.; Leonetti, F.; Sbraccia, P.; Giaccari, A.; Locuratolo, N.; Lala, L. Increased Insulin Sensitivity in Patients with Idiopathic Reactive Hypoglycemia. J. Clin. Endocrinol. Metab. 1989, 69, 875–880. [Google Scholar] [CrossRef]
- IDF Diabetes Atlas, 10th Ed. Available online: https://diabetesatlas.org/data/ (accessed on 14 October 2022).
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis Model Assessment: Insulin Resistance and Beta-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuda, M.; DeFronzo, R.A. Insulin Sensitivity Indices Obtained from Oral Glucose Tolerance Testing: Comparison with the Euglycemic Insulin Clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef]
- Goedecke, J.H.; Dave, J.A.; Faulenbach, M.V.; Utzschneider, K.M.; Lambert, E.V.; West, S.; Collins, M.; Olsson, T.; Walker, B.R.; Seckl, J.R.; et al. Insulin Response in Relation to Insulin Sensitivity. Diabetes Care 2009, 32, 860–865. [Google Scholar] [CrossRef] [Green Version]
- Utzschneider, K.M.; Prigeon, R.L.; Faulenbach, M.V.; Tong, J.; Carr, D.B.; Boyko, E.J.; Leonetti, D.L.; McNeely, M.J.; Fujimoto, W.Y.; Kahn, S.E. Oral Disposition Index Predicts the Development of Future Diabetes Above and Beyond Fasting and 2-h Glucose Levels. Diabetes Care 2009, 32, 335–341. [Google Scholar] [CrossRef] [Green Version]
- Stumvoll, M.; Mitrakou, A.; Pimenta, W.; Jenssen, T.; Yki-Järvinen, H.; Van Haeften, T.; Renn, W.; Gerich, J. Use of the Oral Glucose Tolerance Test to Assess Insulin Release and Insulin Sensitivity. Diabetes Care 2000, 23, 295–301. [Google Scholar] [CrossRef] [Green Version]
- Crofts, C.; Schofield, G.; Zinn, C.; Wheldon, M.; Kraft, J. Identifying Hyperinsulinaemia in the Absence of Impaired Glucose Tolerance: An Examination of the Kraft Database. Diabetes Res. Clin. Pract. 2016, 118, 50–57. [Google Scholar] [CrossRef]
- Lv, X.; Fang, K.; Hao, W.; Han, Y.; Yang, N.; Yu, Q. Identification of Reactive Hypoglycemia with Different Basic BMI and Its Causes by Prolonged Oral Glucose Tolerance Test. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 4717–4726. [Google Scholar] [CrossRef] [PubMed]
- Mumm, H.; Altinok, M.L.; Henriksen, J.E.; Ravn, P.; Glintborg, D.; Andersen, M. Prevalence and Possible Mechanisms of Reactive Hypoglycemia in Polycystic Ovary Syndrome. Hum. Reprod. Oxf. Engl. 2016, 31, 1105–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, X.; Han, X.; Zhou, X.; Zhou, L.; Zhang, S.; Ji, L. Associated Factors with Biochemical Hypoglycemia during an Oral Glucose Tolerance Test in a Chinese Population. J. Diabetes Res. 2017, 2017, 3212814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parekh, S.; Bodicoat, D.H.; Brady, E.; Webb, D.; Mani, H.; Mostafa, S.; Levy, M.J.; Khunti, K.; Davies, M.J. Clinical Characteristics of People Experiencing Biochemical Hypoglycaemia during an Oral Glucose Tolerance Test: Cross-Sectional Analyses from a UK Multi-Ethnic Population. Diabetes Res. Clin. Pract. 2014, 104, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Charles, M.A.; Hofeldt, F.; Shackelford, A.; Waldeck, N.; Dodson, L.E.; Bunker, D.; Coggins, J.T.; Eichner, H. Comparison of Oral Glucose Tolerance Tests and Mixed Meals in Patients with Apparent Idiopathic Postabsorptive Hypoglycemia: Absence of Hypoglycemia after Meals. Diabetes 1981, 30, 465–470. [Google Scholar] [CrossRef]
- Esser, N.; Utzschneider, K.M.; Kahn, S.E. Early Beta Cell Dysfunction vs Insulin Hypersecretion as the Primary Event in the Pathogenesis of Dysglycaemia. Diabetologia 2020, 63, 2007–2021. [Google Scholar] [CrossRef] [PubMed]
- Shanik, M.H.; Xu, Y.; Skrha, J.; Dankner, R.; Zick, Y.; Roth, J. Insulin Resistance and Hyperinsulinemia: Is Hyperinsulinemia the Cart or the Horse? Diabetes Care 2008, 31 (Suppl. S2), S262–S268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdul-Ghani, M.A.; Lyssenko, V.; Tuomi, T.; Defronzo, R.A.; Groop, L. The Shape of Plasma Glucose Concentration Curve during OGTT Predicts Future Risk of Type 2 Diabetes. Diabetes Metab. Res. Rev. 2010, 26, 280–286. [Google Scholar] [CrossRef]
- Kahn, S.E.; Prigeon, R.L.; McCulloch, D.K.; Boyko, E.J.; Bergman, R.N.; Schwartz, M.W.; Neifing, J.L.; Ward, W.K.; Beard, J.C.; Palmer, J.P. Quantification of the Relationship between Insulin Sensitivity and Beta-Cell Function in Human Subjects. Evidence for a Hyperbolic Function. Diabetes 1993, 42, 1663–1672. [Google Scholar] [CrossRef] [PubMed]
- Mooy, J.M.; Grootenhuis, P.A.; de Vries, H.; Kostense, P.J.; Popp-Snijders, C.; Bouter, L.M.; Heine, R.J. Intra-Individual Variation of Glucose, Specific Insulin and Proinsulin Concentrations Measured by Two Oral Glucose Tolerance Tests in a General Caucasian Population: The Hoorn Study. Diabetologia 1996, 39, 298–305. [Google Scholar] [CrossRef]
- Tschritter, O.; Fritsche, A.; Shirkavand, F.; Machicao, F.; Häring, H.; Stumvoll, M. Assessing the Shape of the Glucose Curve during an Oral Glucose Tolerance Test. Diabetes Care 2003, 26, 1026–1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hulman, A.; Vistisen, D.; Glümer, C.; Bergman, M.; Witte, D.R.; Færch, K. Glucose Patterns during an Oral Glucose Tolerance Test and Associations with Future Diabetes, Cardiovascular Disease and All-Cause Mortality Rate. Diabetologia 2018, 61, 101–107. [Google Scholar] [CrossRef]
- Bergman, M.; Chetrit, A.; Roth, J.; Dankner, R. One-Hour Post-Load Plasma Glucose Level during the OGTT Predicts Mortality: Observations from the Israel Study of Glucose Intolerance, Obesity and Hypertension. Diabet. Med. J. Br. Diabet. Assoc. 2016, 33, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Michaliszyn, S.F.; Nasr, A.; Lee, S.; Tfayli, H.; Hannon, T.; Hughan, K.S.; Bacha, F.; Arslanian, S. The Shape of the Glucose Response Curve During an Oral Glucose Tolerance Test Heralds Biomarkers of Type 2 Diabetes Risk in Obese Youth. Diabetes Care 2016, 39, 1431–1439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, T.; Boyko, E.J.; Sato, K.K.; McNeely, M.J.; Leonetti, D.L.; Kahn, S.E.; Fujimoto, W.Y. Patterns of Insulin Concentration during the OGTT Predict the Risk of Type 2 Diabetes in Japanese Americans. Diabetes Care 2013, 36, 1229–1235. [Google Scholar] [CrossRef] [Green Version]
- Alyass, A.; Almgren, P.; Akerlund, M.; Dushoff, J.; Isomaa, B.; Nilsson, P.; Tuomi, T.; Lyssenko, V.; Groop, L.; Meyre, D. Modelling of OGTT Curve Identifies 1 h Plasma Glucose Level as a Strong Predictor of Incident Type 2 Diabetes: Results from Two Prospective Cohorts. Diabetologia 2015, 58, 87–97. [Google Scholar] [CrossRef]
- Crofts, C.A.P.; Brookler, K.; Henderson, G. Can Insulin Response Patterns Predict Metabolic Disease Risk in Individuals with Normal Glucose Tolerance? Diabetologia 2018, 61, 1233. [Google Scholar] [CrossRef] [Green Version]
- Ko, G.T.; Chan, J.C.; Woo, J.; Lau, E.; Yeung, V.T.; Chow, C.C.; Cockram, C.S. The Reproducibility and Usefulness of the Oral Glucose Tolerance Test in Screening for Diabetes and Other Cardiovascular Risk Factors. Ann. Clin. Biochem. 1998, 35 Pt 1, 62–67. [Google Scholar] [CrossRef]
- Bogdanet, D.; O’Shea, P.; Lyons, C.; Shafat, A.; Dunne, F. The Oral Glucose Tolerance Test—Is It Time for a Change?—A Literature Review with an Emphasis on Pregnancy. J. Clin. Med. 2020, 9, 3451. [Google Scholar] [CrossRef]
- Palmu, S.; Rehunen, S.; Kautiainen, H.; Eriksson, J.G.; Korhonen, P.E. Body Surface Area and Glucose Tolerance—The Smaller the Person, the Greater the 2-Hour Plasma Glucose. Diabetes Res. Clin. Pract. 2019, 157, 107877. [Google Scholar] [CrossRef]
- Hofeldt, F.D. Reactive Hypoglycemia. Endocrinol. Metab. Clin. N. Am. 1989, 18, 185–201. [Google Scholar] [CrossRef]
- Simpson, E.J.; Holdsworth, M.; Macdonald, I.A. Interstitial Glucose Profile Associated with Symptoms Attributed to Hypoglycemia by Otherwise Healthy Women. Am. J. Clin. Nutr. 2008, 87, 354–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Snorgaard, O.; Binder, C. Monitoring of Blood Glucose Concentration in Subjects with Hypoglycaemic Symptoms during Everyday Life. BMJ 1990, 300, 16–18. [Google Scholar] [CrossRef] [Green Version]
- Libman, I.M.; Barinas-Mitchell, E.; Bartucci, A.; Robertson, R.; Arslanian, S. Reproducibility of the Oral Glucose Tolerance Test in Overweight Children. J. Clin. Endocrinol. Metab. 2008, 93, 4231–4237. [Google Scholar] [CrossRef]
- Lages, M.; Barros, R.; Moreira, P.; Guarino, M.P. Metabolic Effects of an Oral Glucose Tolerance Test Compared to the Mixed Meal Tolerance Tests: A Narrative Review. Nutrients 2022, 14, 2032. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Saxena, A. Surrogate Markers of Insulin Resistance: A Review. World J. Diabetes 2010, 1, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Marathe, C.S.; Horowitz, M.; Trahair, L.G.; Wishart, J.M.; Bound, M.; Lange, K.; Rayner, C.K.; Jones, K.L. Relationships of Early And Late Glycemic Responses with Gastric Emptying During An Oral Glucose Tolerance Test. J. Clin. Endocrinol. Metab. 2015, 100, 3565–3571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macdonald, I.; Crossley, J.N. Glucose Tolerance During the Menstrual Cycle. Diabetes 1970, 19, 450–452. [Google Scholar] [CrossRef]
- Barata, D.S.; Adan, L.F.; Netto, E.M.; Ramalho, A.C. The Effect of the Menstrual Cycle on Glucose Control in Women with Type 1 Diabetes Evaluated Using a Continuous Glucose Monitoring System. Diabetes Care 2013, 36, e70. [Google Scholar] [CrossRef] [Green Version]
- Yan, H.; Yang, W.; Zhou, F.; Li, X.; Pan, Q.; Shen, Z.; Han, G.; Newell-Fugate, A.; Tian, Y.; Majeti, R.; et al. Estrogen Improves Insulin Sensitivity and Suppresses Gluconeogenesis via the Transcription Factor Foxo1. Diabetes 2019, 68, 291–304. [Google Scholar] [CrossRef]
- Moreira, R.O.; Moreira, R.B.M.; Machado, N.A.M.; Gonçalves, T.B.; Coutinho, W.F. Post-Prandial Hypoglycemia after Bariatric Surgery: Pharmacological Treatment with Verapamil and Acarbose. Obes. Surg. 2008, 18, 1618–1621. [Google Scholar] [CrossRef] [PubMed]
- Tura, A.; Morbiducci, U.; Sbrignadello, S.; Winhofer, Y.; Pacini, G.; Kautzky-Willer, A. Shape of Glucose, Insulin, C-Peptide Curves during a 3-h Oral Glucose Tolerance Test: Any Relationship with the Degree of Glucose Tolerance? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R941–R948. [Google Scholar] [CrossRef] [PubMed]
- Dalla Man, C.; Campioni, M.; Polonsky, K.S.; Basu, R.; Rizza, R.A.; Toffolo, G.; Cobelli, C. Two-Hour Seven-Sample Oral Glucose Tolerance Test and Meal Protocol: Minimal Model Assessment of Beta-Cell Responsivity and Insulin Sensitivity in Nondiabetic Individuals. Diabetes 2005, 54, 3265–3273. [Google Scholar] [CrossRef] [PubMed]



| Baseline Visit | Follow-Up Visit | ||
|---|---|---|---|
| Variable Value | p-Value | ||
| Body weight (kg) | 67.6 ± 11.3 | 69.3 ± 10.11 | 0.141 |
| BMI (kg/m2) | 23.7 (20.4, 26.2) | 24.5 (21.3, 26.8) | 0.215 |
| HbA1c (%/ mmol/mol)) | 5.3 (4.9, 5.6) * /34.4 (30.1, 41.0) | 5.0 (4.8, 5.3) * /31.8 (29.0, 34.4) | 0.004 |
| Matsuda index | 9.3 (5.3, 12.5) | 9.2 (3.6, 57.4) | 0.620 |
| HOMA -IR | 1.07 ± 0.74 | 1.08 ± 1.2 | 0.985 |
| Insulinogenic index | 0.84 (0.59, 1.7) | 0.73 (0.43, 1.5) | 0.069 |
| Disposition index | 7.4 (5.1, 19.4) | 5.4 (2.0, 29.7) | 0.283 |
| RPH Group | Control Group | p-Value | |
|---|---|---|---|
| Age (years) | 37.2 ± 10.0 | 39.7 ± 9.2 | 0.515 |
| Body weight (kg) | 68.6 ± 10.0 | 70.3 ± 11.1 | 0.640 |
| Waist circumference (cm) | 84.8 ± 12.0 | 84.2 ± 10.6 | 0.886 |
| Hip circumference (cm) | 102.8 ± 8.6 | 104.1 ± 7.9 | 0.755 |
| WHR | 0.8 ± 0.1 | 0.8 ± 0.1 | 0.675 |
| BMI (kg/m2) | 24.6 ± 3.5 | 24.8 ± 4.3 | 0.877 |
| Systolic blood pressure (mmHg) | 107 ± 10 | 108 ± 9 | 0.884 |
| Diastolic blood pressure (mmHg) | 70 ± 7 | 72 ± 8 | 0.618 |
| Heart rate (beats/min) | 61 ± 10 | 64 ± 11 | 0.423 |
| LDL-c (mmol/L) | 2.9 ± 0.61 | 2.66 ± 0.56 | 0.546 |
| TG (mmol/L) | 1.1 ± 0.41 | 0.84 ± 0.33 | 0.078 |
| HbA1c (%; mmol/mol) | 5.0 ± 0.3 31.1 ± 2.1 | 5.2 ± 0.6 33.3 ± 4.2 | 0.593 |
| Matsuda index | 9.2 (3.6, 57.4) | 6.0 (4.0, 76.0) | 0.804 |
| HOMA-IR | 1.08 ± 1.2 | 2.5 ± 4.2 | 0.136 |
| Insulinogenic index | 0.73 (0.43, 1.5) * | 1.0 (1.0, 3.0) * | 0.028 * |
| Disposition index | 5.4 (2.0, 29.7) | 16.0 (7.0, 42.0) | 0.217 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Volčanšek, Š.; Rahne Perc, U.; Lunder, M.; Pongrac Barlovič, D. No Indices of Increased Type 2 Diabetes Risk in Individuals with Reactive Postprandial Hypoglycemia. Metabolites 2022, 12, 1232. https://doi.org/10.3390/metabo12121232
Volčanšek Š, Rahne Perc U, Lunder M, Pongrac Barlovič D. No Indices of Increased Type 2 Diabetes Risk in Individuals with Reactive Postprandial Hypoglycemia. Metabolites. 2022; 12(12):1232. https://doi.org/10.3390/metabo12121232
Chicago/Turabian StyleVolčanšek, Špela, Urška Rahne Perc, Mojca Lunder, and Draženka Pongrac Barlovič. 2022. "No Indices of Increased Type 2 Diabetes Risk in Individuals with Reactive Postprandial Hypoglycemia" Metabolites 12, no. 12: 1232. https://doi.org/10.3390/metabo12121232
APA StyleVolčanšek, Š., Rahne Perc, U., Lunder, M., & Pongrac Barlovič, D. (2022). No Indices of Increased Type 2 Diabetes Risk in Individuals with Reactive Postprandial Hypoglycemia. Metabolites, 12(12), 1232. https://doi.org/10.3390/metabo12121232