Vitamin B6 Inhibits High Glucose-Induced Islet β Cell Apoptosis by Upregulating Autophagy
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Antibodies
2.2. Cell Culture
2.3. Immunofluorescence Staining
2.4. Western Blotting
2.5. Cell Apoptosis Assay
2.6. Testing the Viability of RIN-m5F Cells
2.7. Test of Vitamin B6 Content
3. Results
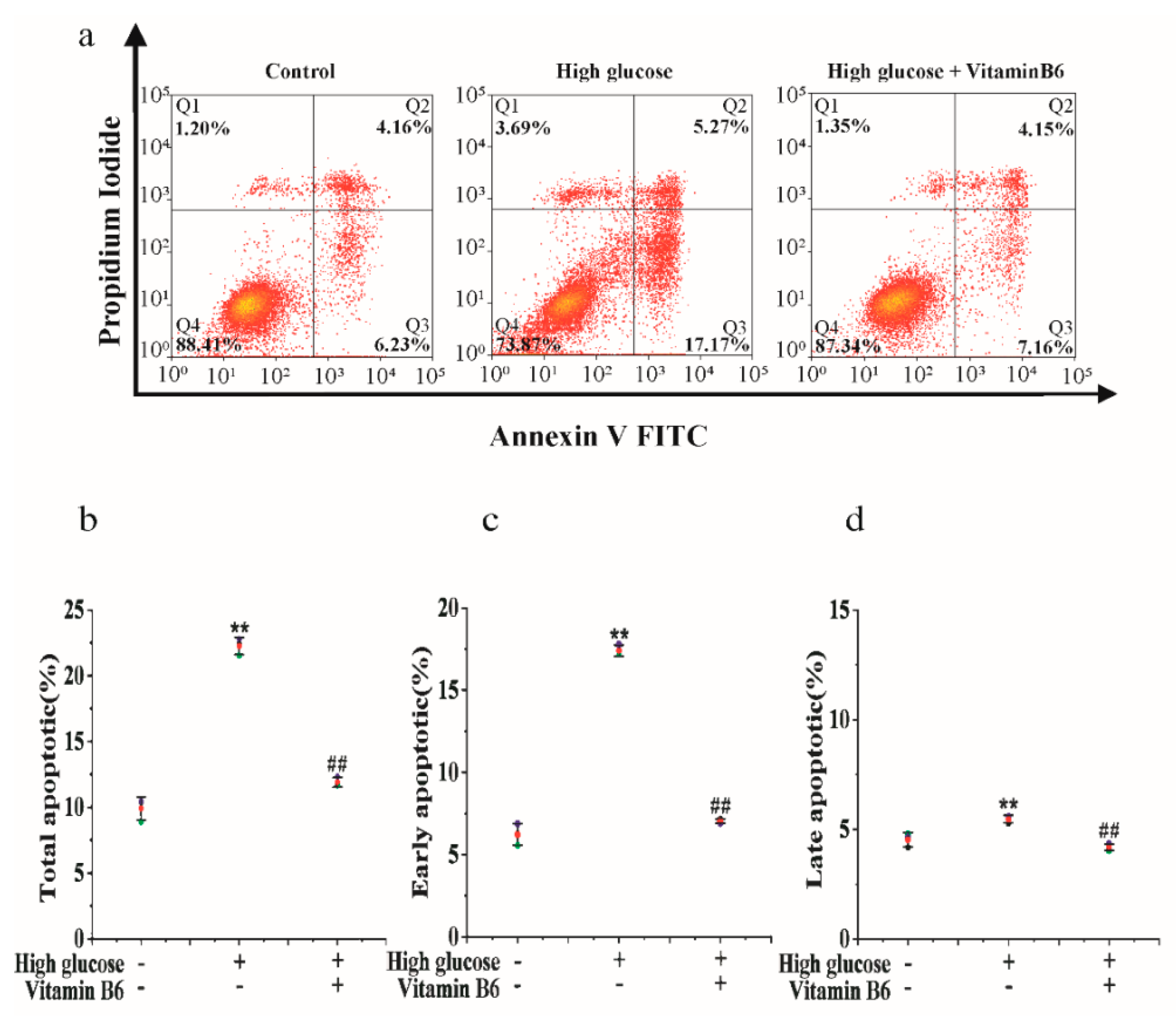
3.1. Vitamin B6 Inhibited Apoptosis Induced by High Glucose
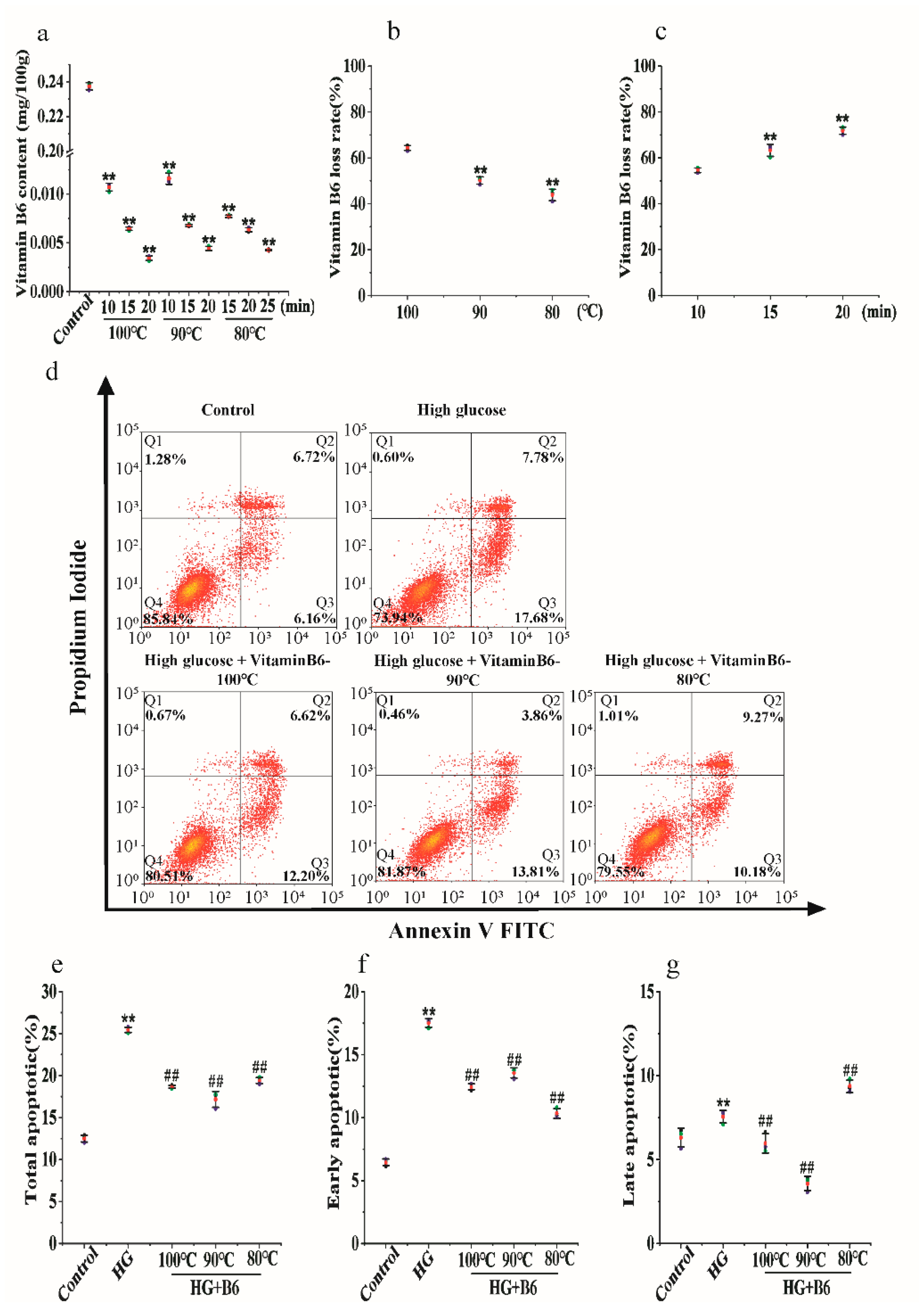
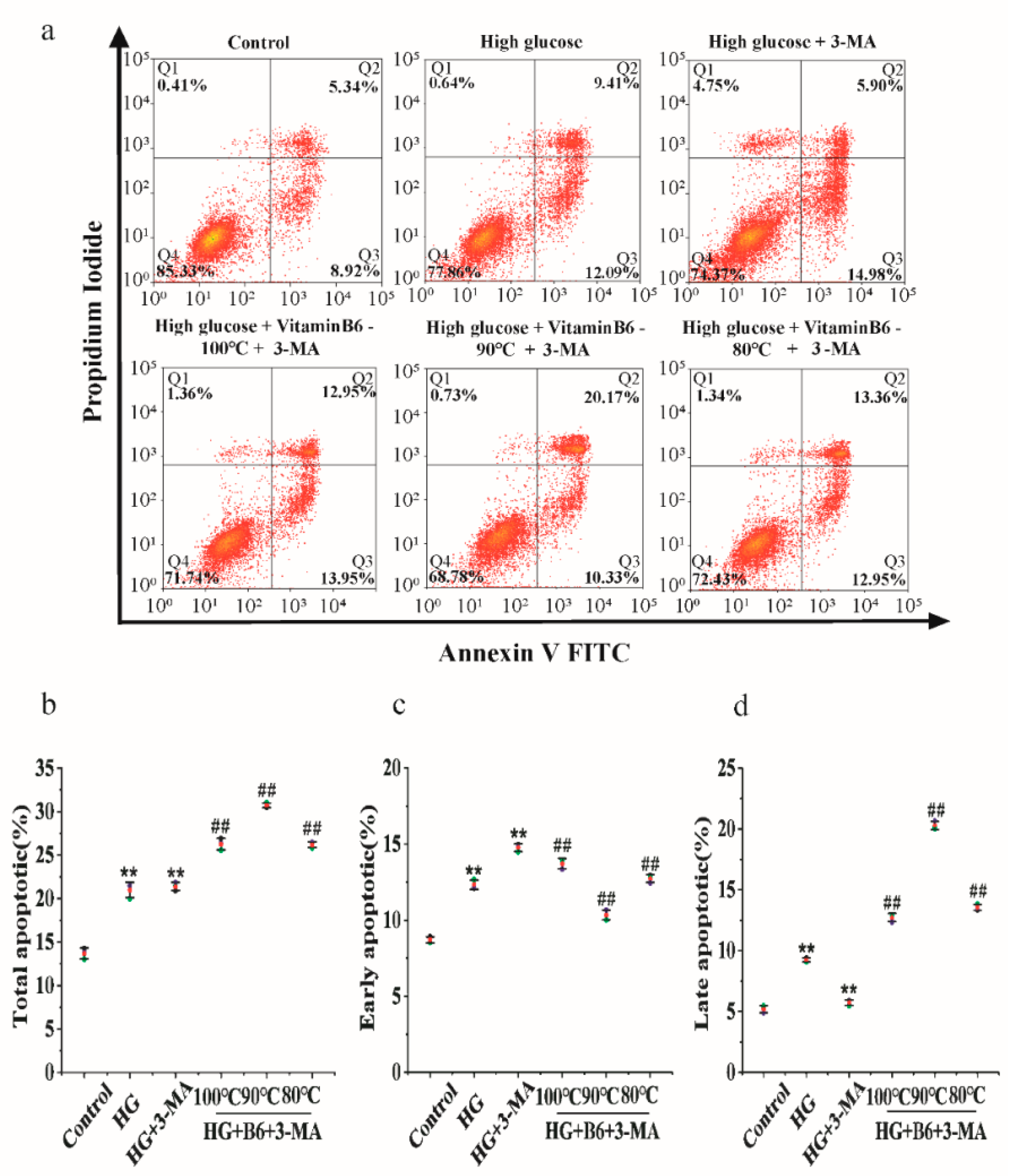
3.2. Effect of Hydrothermal Processing on the Biological Activity of Vitamin B6 in Eggs
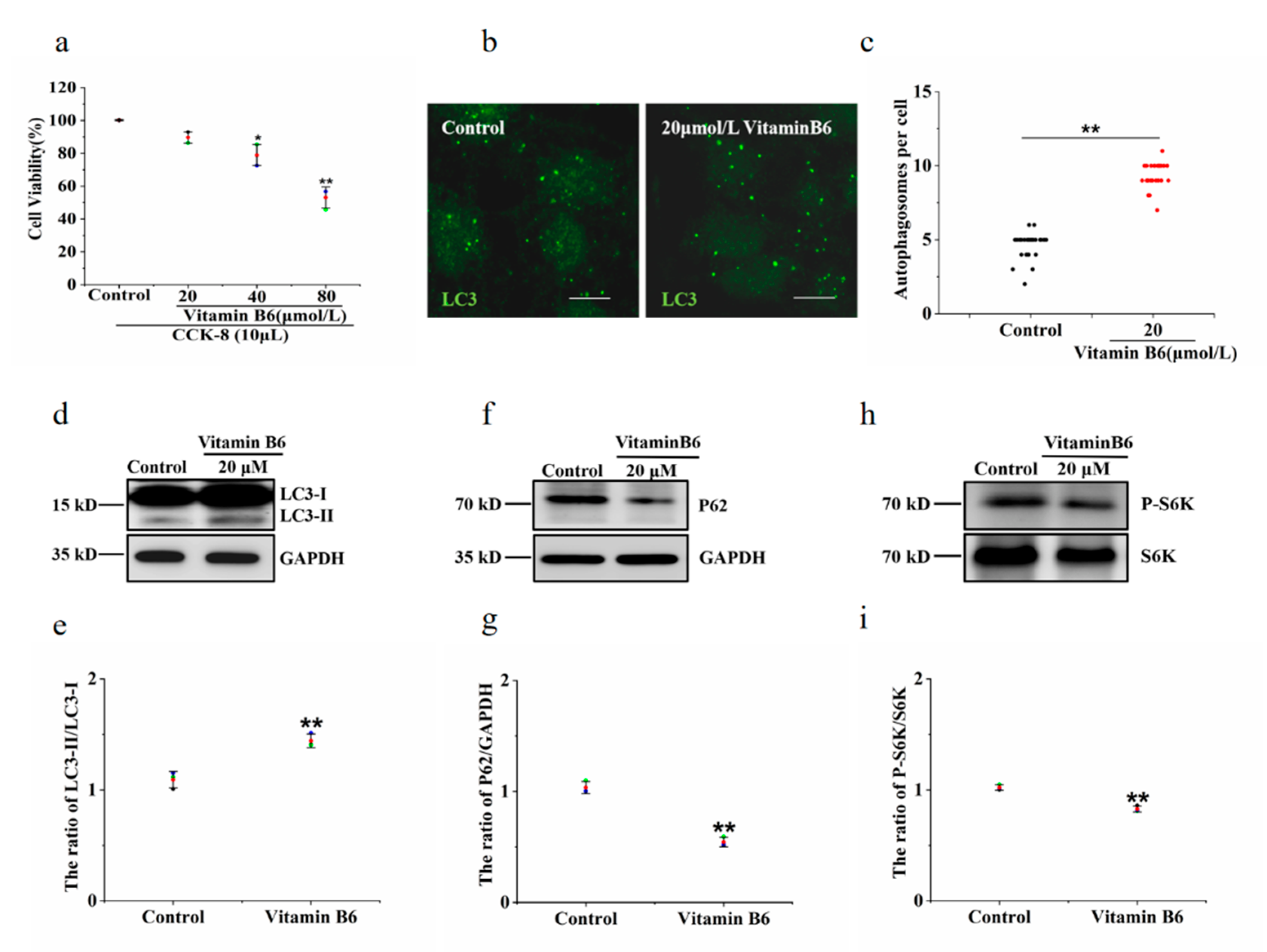
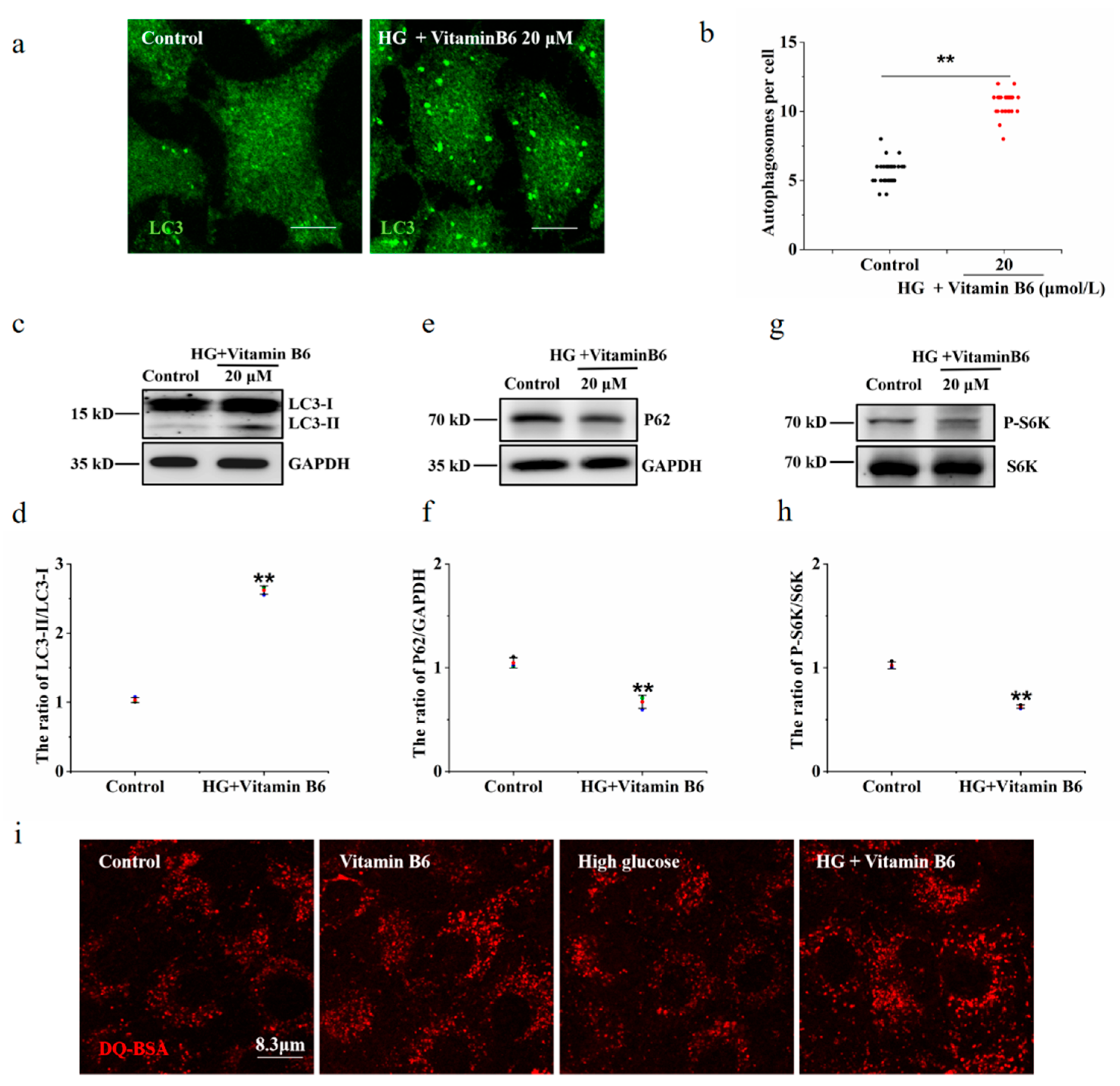
3.3. Vitamin B6 Induced Autophagy
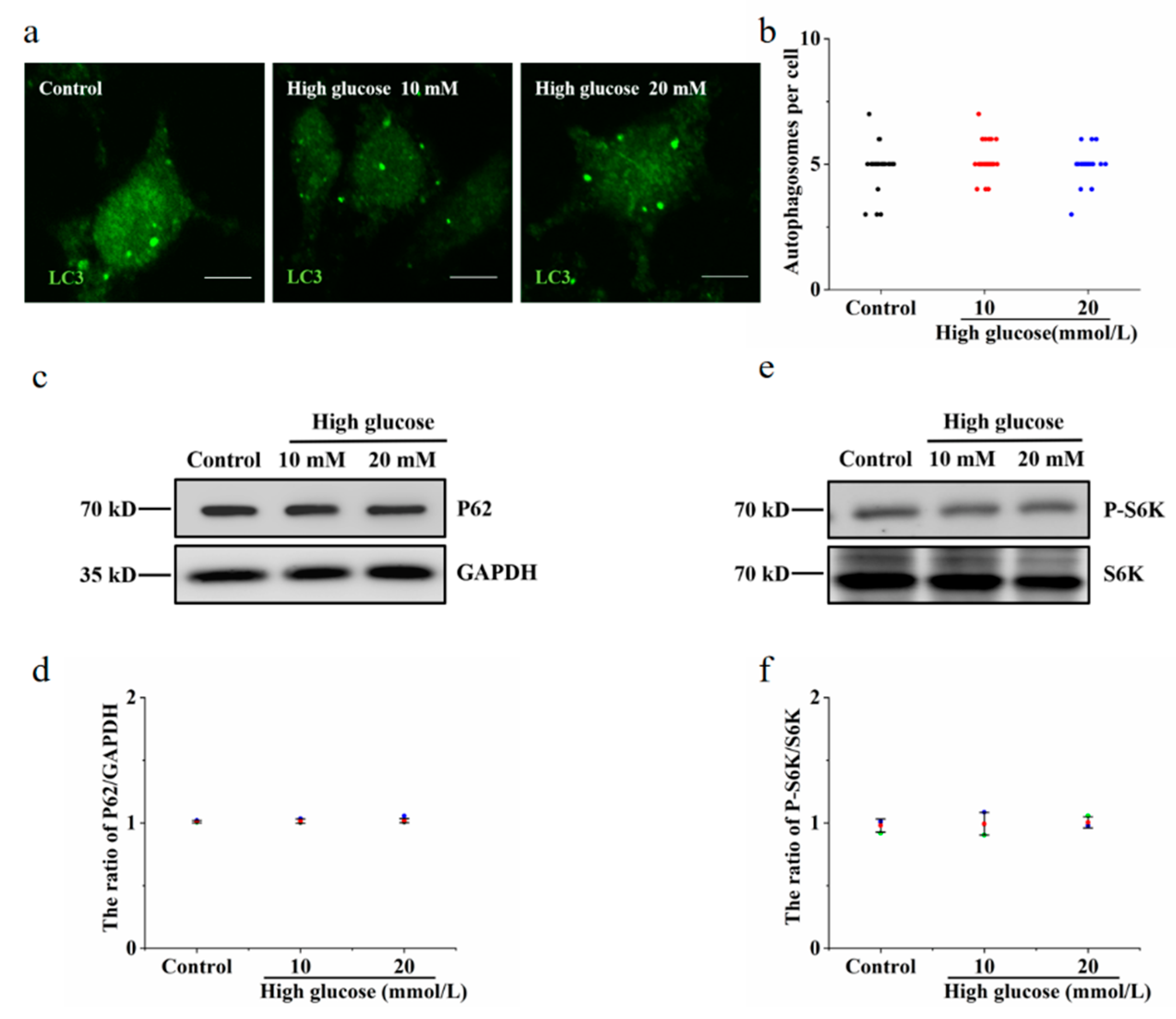
3.4. Vitamin B6 Induced Autophagy under High Glucose Stress
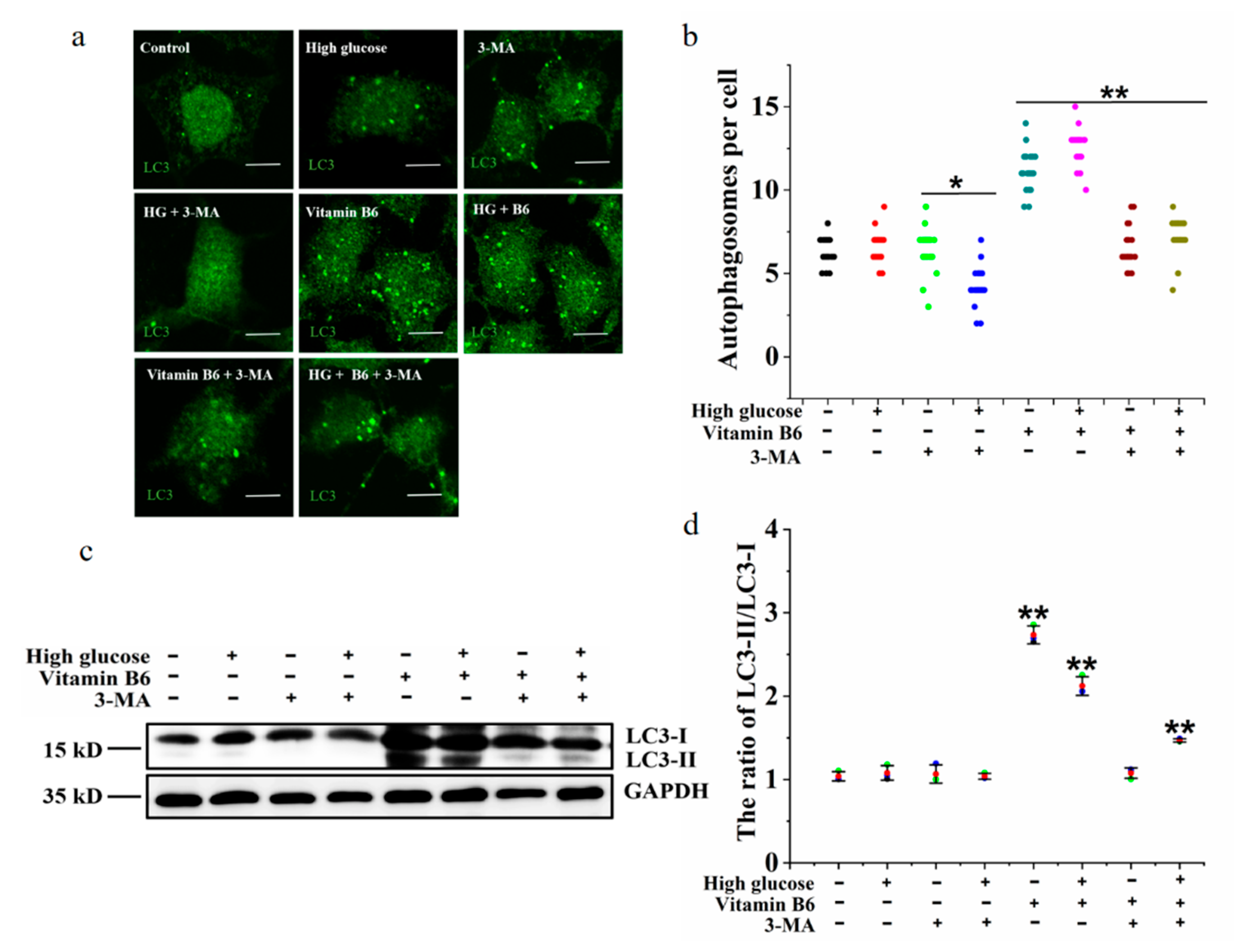
3.5. 3-MA Inhibited Vitamin B6-Induced Autophagy under High Glucose Stress
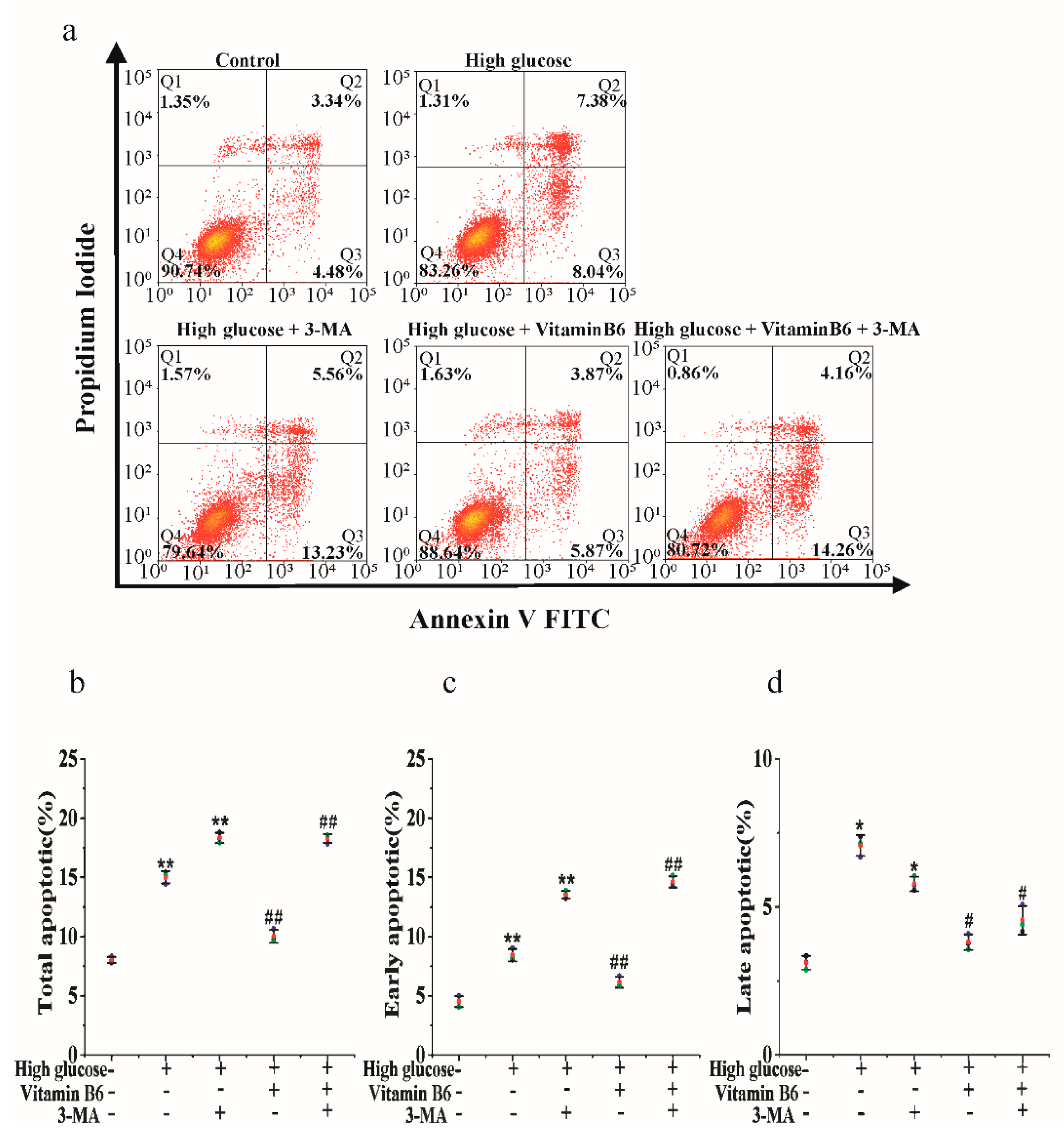
3.6. 3-MA Inhibits Vitamin B6-Induced Cytoprotective Autophagy against Apoptosis
3.7. Effect of Hydrothermal Processing on the Biological Activity of Vitamin B6
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abosamak, N.R.; Gupta, V. Vitamin B6 (Pyridoxine). In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2022. [Google Scholar]
- di Salvo, M.L.; Contestabile, R.; Safo, M.K. Vitamin B(6) salvage enzymes: Mechanism, structure and regulation. Biochim. Biophys. Acta BBA Proteins Proteom. 2011, 1814, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- Looman, M.; van den Berg, C.; Geelen, A.; Samlal, R.A.K.; Heijligenberg, R.; Klein Gunnewiek, J.M.T.; Balvers, M.G.J.; Leendertz-Eggen, C.L.; Wijnberger, L.D.E.; Feskens, E.J.M.; et al. Supplement Use and Dietary Sources of Folate, Vitamin D, and n-3 Fatty Acids during Preconception: The GLIMP2 Study. Nutrients 2018, 10, 962. [Google Scholar] [CrossRef] [PubMed]
- Nix, W.A.; Zirwes, R.; Bangert, V.; Kaiser, R.P.; Schilling, M.; Hostalek, U.; Obeid, R. Vitamin B status in patients with type 2 diabetes mellitus with and without incipient nephropathy. Diabetes Res. Clin. Pract. 2015, 107, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.J.; Min, K.W.; Cho, Y.O. Assessment of vitamin B(6) status in Korean patients with newly diagnosed type 2 diabetes. Nutr. Res. Pract. 2011, 5, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Hu, Y.; Yuan, Y.; Luo, Y.; Lai, D.; Zhou, H.; Tong, Z.; Liu, D. Gymnemic acid I triggers mechanistic target of rapamycin-mediated beta cells cytoprotection through the promotion of autophagy under high glucose stress. J. Cell. Physiol. 2019, 234, 9370–9377. [Google Scholar] [CrossRef]
- Pitchika, A.; Vehik, K.; Hummel, S.; Norris, J.M.; Uusitalo, U.M.; Yang, J.; Virtanen, S.M.; Koletzko, S.; Andrén Aronsson, C.; Ziegler, A.G.; et al. Associations of Maternal Diabetes During Pregnancy with Overweight in Offspring: Results from the Prospective TEDDY Study. Obesity 2018, 26, 1457–1466. [Google Scholar] [CrossRef]
- Katsarou, A.; Gudbjornsdottir, S.; Rawshani, A.; Dabelea, D.; Bonifacio, E.; Anderson, B.J.; Jacobsen, L.M.; Schatz, D.A.; Lernmark, A. Type 1 diabetes mellitus. Nat. Rev. Dis. Prim. 2017, 3, 17016. [Google Scholar] [CrossRef]
- Ravnic, D.J.; Leberfinger, A.N.; Ozbolat, I.T. Bioprinting and Cellular Therapies for Type 1 Diabetes. Trends Biotechnol. 2017, 35, 1025–1034. [Google Scholar] [CrossRef]
- Marasco, M.R.; Linnemann, A.K. beta-Cell Autophagy in Diabetes Pathogenesis. Endocrinology 2018, 159, 2127–2141. [Google Scholar] [CrossRef]
- Kolodgie, F.D.; Narula, J.; Burke, A.P.; Haider, N.; Farb, A.; Hui-Liang, Y.; Smialek, J.; Virmani, R. Localization of apoptotic macrophages at the site of plaque rupture in sudden coronary death. Am. J. Pathol. 2000, 157, 1259–1268. [Google Scholar] [CrossRef]
- Abe, H.; Uchida, T.; Hara, A.; Mizukami, H.; Komiya, K.; Koike, M.; Shigihara, N.; Toyofuku, Y.; Ogihara, T.; Uchiyama, Y.; et al. Exendin-4 improves β-cell function in autophagy-deficient β-cells. Endocrinology 2013, 154, 4512–4524. [Google Scholar] [CrossRef] [PubMed]
- Steneberg, P.; Bernardo, L.; Edfalk, S.; Lundberg, L.; Backlund, F.; Ostenson, C.G.; Edlund, H. The type 2 diabetes-associated gene ide is required for insulin secretion and suppression of α-synuclein levels in β-cells. Diabetes 2013, 62, 2004–2014. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Chen, Q.; Zhao, Q.; Luo, Y.; Xu, Y.; Du, W.; Wang, H.; Li, H.; Yang, L.; Hu, C.; et al. Inhibition of COX2/PGD2-Related Autophagy Is Involved in the Mechanism of Brain Injury in T2DM Rat. Front. Cell. Neurosci. 2019, 13, 68. [Google Scholar] [CrossRef] [PubMed]
- Weir, G.C.; Bonner-Weir, S. Five stages of evolving beta-cell dysfunction during progression to diabetes. Diabetes 2004, 53 (Suppl. 3), S16–S21. [Google Scholar] [CrossRef]
- Di, Y.Q.; Han, X.L.; Kang, X.L.; Wang, D.; Zhao, X.F. Autophagy triggers CTSD (cathepsin D) maturation and localization inside cells to promote apoptosis. Autophagy 2020, 17, 1170–1192. [Google Scholar] [CrossRef]
- Wu, D.J.; Adamopoulos, I.E. Autophagy and autoimmunity. Clin. Immunol. 2017, 176, 55–62. [Google Scholar] [CrossRef]
- Kamesaki, H.; Nishizawa, K.; Michaud, G.Y.; Cossman, J.; Kiyono, T. TGF-b1 Induces the Cyclin-Dependent Kinase Inhibitor p27 mRNA and Protein in Murine B Cells. J. Immunol. 1998, 160, 770–777. [Google Scholar]
- Kessel, D.; Reiners, J.J., Jr. Apoptosis and Autophagy After Mitochondrial or Endoplasmic Reticulum Photodamage. Photochem. Photobiol. 2007, 83, 1024–1028. [Google Scholar] [CrossRef]
- Ley, S.H.; Hamdy, O.; Mohan, V.; Hu, F.B. Prevention and management of type 2 diabetes: Dietary components and nutritional strategies. Lancet 2014, 383, 1999–2007. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Misra, A.; Mohan, V.; Taylor, R.; Yancy, W. Dietary and nutritional approaches for prevention and management of type 2 diabetes. BMJ 2018, 361, k2234. [Google Scholar] [CrossRef]
- Condon, K.J.; Sabatini, D.M. Nutrient regulation of mTORC1 at a glance. J. Cell Sci. 2019, 132, jcs222570. [Google Scholar] [CrossRef] [PubMed]
- Losiewicz, M.K.; Elghazi, L.; Fingar, D.C.; Rajala, R.; Gardner, T.W. mTORC1 and mTORC2 expression in inner retinal neurons and glial cells. Exp. Eye Res. 2020, 108131. [Google Scholar] [CrossRef] [PubMed]
- Ruxton, C.; Derbyshire, E.; Gibson, S. The nutritional properties and health benefits of eggs. Nutr. Food Sci. 2010, 40, 263–279. [Google Scholar] [CrossRef]
- Flint, A.; Møller, B.K.; Raben, A.; Sloth, B.; Pedersen, D.; Tetens, I.; Holst, J.J.; Astrup, A. Glycemic and insulinemic responses as determinants of appetite in humans. Am. J. Clin. Nutr. 2006, 84, 1365–1373. [Google Scholar] [CrossRef]
- Ludwig, D.S. The Glycemic Index: Physiological Mechanisms Relating to Obesity, Diabetes, and Cardiovascular Disease. JAMA 2002, 287, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.B. High-glycemic index foods, hunger, and obesity: Is there a connection? Nutr. Rev. 2000, 58, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Ratliff, J.; Leite, J.O.; Ogburn, R.D.; Puglisi, M.J.; Fernandez, M.L. Consuming eggs for breakfast influences plasma glucose and ghrelin, while reducing energy intake during the next 24 hours in adult men. Nutr. Res. 2010, 30, 96–103. [Google Scholar] [CrossRef]
- Siega-Riz, A.M.; Popkin, B.M.; Carson, T. Differences in Food Patterns at Breakfast by Sociodemographic Characteristics among a Nationally Representative Sample of Adults in the United States. Prev. Med. 2000, 30, 415–424. [Google Scholar] [CrossRef]
- Reeves, S.; Halsey, L.G.; Mcmeel, Y.; Huber, J.W. Breakfast habits, beliefs and measures of health and wellbeing in a nationally representative UK sample. Appetite 2013, 60, 51–57. [Google Scholar] [CrossRef]
- Mascolo, E.; Verni, F. Vitamin B6 and Diabetes: Relationship and Molecular Mechanisms. Int. J. Mol. Sci. 2020, 21, 3669. [Google Scholar] [CrossRef]
- Zhu, J.; Chen, C.; Lu, L.; Yang, K.; Reis, J.; He, K. Intakes of Folate, Vitamin B(6), and Vitamin B(12) in Relation to Diabetes Incidence Among American Young Adults: A 30-Year Follow-up Study. Diabetes Care 2020, 43, 2426–2434. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Hjorth, M.F.; Astrup, A. Diet and exercise in the prevention and treatment of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2020, 16, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Martin-Pelaez, S.; Fito, M.; Castaner, O. Mediterranean Diet Effects on Type 2 Diabetes Prevention, Disease Progression, and Related Mechanisms. A Review. Nutrients 2020, 12, 2236. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S. Sleep and diabetes. Curr. Opin. Pulm. Med. 2018, 24, 555–560. [Google Scholar] [CrossRef]
- Petroni, M.L.; Brodosi, L.; Marchignoli, F.; Sasdelli, A.S.; Caraceni, P.; Marchesini, G.; Ravaioli, F. Nutrition in Patients with Type 2 Diabetes: Present Knowledge and Remaining Challenges. Nutrients 2021, 13, 2748. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Q.; Liu, Y.; Jiang, H.; Chen, W. Intermittent fasting versus continuous energy-restricted diet for patients with type 2 diabetes mellitus and metabolic syndrome for glycemic control: A systematic review and meta-analysis of randomized controlled trials. Diabetes Res. Clin. Pract. 2021, 179, 109003. [Google Scholar] [CrossRef]
- Casanueva, F.F.; Castellana, M.; Bellido, D.; Trimboli, P.; Castro, A.I.; Sajoux, I.; Rodriguez-Carnero, G.; Gomez-Arbelaez, D.; Crujeiras, A.B.; Martinez-Olmos, M.A. Ketogenic diets as treatment of obesity and type 2 diabetes mellitus. Rev. Endocr. Metab. Dis. 2020, 21, 381–397. [Google Scholar] [CrossRef]
- Barlow, A.D.; Thomas, D.C. Autophagy in diabetes: Beta-cell dysfunction, insulin resistance, and complications. DNA Cell Biol. 2015, 34, 252–260. [Google Scholar] [CrossRef]
- Packer, M. Autophagy-dependent and -independent modulation of oxidative and organellar stress in the diabetic heart by glucose-lowering drugs. Cardiovasc. Diabetol. 2020, 19, 62. [Google Scholar] [CrossRef]
- Lu, G.; Wu, Z.; Shang, J.; Xie, Z.; Chen, C.; Zhang, C. The effects of metformin on autophagy. Biomed. Pharmacother. 2021, 137, 111286. [Google Scholar] [CrossRef]
- Benchoula, K.; Parhar, I.S.; Wong, E.H. The crosstalk of hedgehog, PI3K and Wnt pathways in diabetes. Arch. Biochem. Biophys. 2021, 698, 108743. [Google Scholar] [CrossRef] [PubMed]
- Ho, F.M.; Liu, S.H.; Liau, C.S.; Huang, P.J.; Lin-Shiau, S.Y. High glucose-induced apoptosis in human endothelial cells is mediated by sequential activations of c-Jun NH(2)-terminal kinase and caspase-3. Circulation 2000, 101, 2618–2624. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.A.; Yaqoob, M.M.; Harwood, S.M. Mechanisms of high glucose-induced apoptosis and its relationship to diabetic complications. J. Nutr. Biochem. 2005, 16, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Su, L.; He, X.; Zhao, B.; Miao, J. Long noncoding RNA CA7-4 promotes autophagy and apoptosis via sponging MIR877-3P and MIR5680 in high glucose-induced vascular endothelial cells. Autophagy 2020, 16, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; He, N.; Xing, Y.; Lu, Y. Knockdown of GCN2 inhibits high glucose-induced oxidative stress and apoptosis in retinal pigment epithelial cells. Clin. Exp. Pharmacol. Physiol. 2020, 47, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhao, K.; Li, Y. Gasdermin D Protects Mouse Podocytes Against High-Glucose-Induced Inflammation and Apoptosis via the C-Jun N-Terminal Kinase (JNK) Pathway. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2021, 27, e928411. [Google Scholar] [CrossRef]
- Ma, L.; Cao, Y.; Zhang, L.; Li, K.; Yan, L.; Pan, Y.; Zhu, J. Celastrol mitigates high glucose-induced inflammation and apoptosis in rat H9c2 cardiomyocytes via miR-345-5p/growth arrest-specific 6. J. Gene Med. 2020, 22, e3201. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhou, X.-a.; Liu, C.; Shen, Q.; Wu, Y. Vitamin B6 Inhibits High Glucose-Induced Islet β Cell Apoptosis by Upregulating Autophagy. Metabolites 2022, 12, 1048. https://doi.org/10.3390/metabo12111048
Zhang Y, Zhou X-a, Liu C, Shen Q, Wu Y. Vitamin B6 Inhibits High Glucose-Induced Islet β Cell Apoptosis by Upregulating Autophagy. Metabolites. 2022; 12(11):1048. https://doi.org/10.3390/metabo12111048
Chicago/Turabian StyleZhang, Yu, Xi-an Zhou, Chuxin Liu, Qingwu Shen, and Yanyang Wu. 2022. "Vitamin B6 Inhibits High Glucose-Induced Islet β Cell Apoptosis by Upregulating Autophagy" Metabolites 12, no. 11: 1048. https://doi.org/10.3390/metabo12111048
APA StyleZhang, Y., Zhou, X.-a., Liu, C., Shen, Q., & Wu, Y. (2022). Vitamin B6 Inhibits High Glucose-Induced Islet β Cell Apoptosis by Upregulating Autophagy. Metabolites, 12(11), 1048. https://doi.org/10.3390/metabo12111048




