Differentiation of Cystic Fibrosis-Related Pathogens by Volatile Organic Compound Analysis with Secondary Electrospray Ionization Mass Spectrometry
Abstract
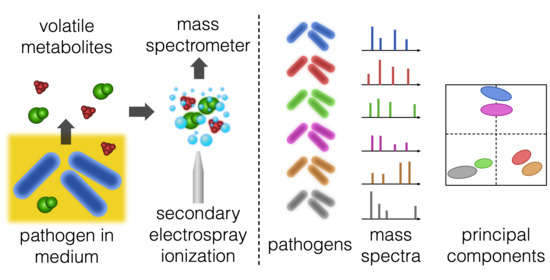
:1. Introduction
2. Results
2.1. Pathogen Separation and Predictive Analysis
2.2. Volatile Compounds Associated with Pathogen Strains
3. Discussion
4. Materials and Methods
4.1. Pathogen Strains and Sample Preparation
4.2. Continuous Headspace Analysis with SESI-HRMS
4.3. Data Preprocessing
4.4. Statistical Analysis
4.5. Putative Compound Identification with SESI-HRMS2
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATCC | American Type Culture Collection |
| BHI | Brain Heart Infusion |
| CF | Cystic fibrosis |
| CID | Collision-induced dissociation |
| GC | Gas chromatography |
| HCA | Hierarchical clustering analysis |
| HMDB | Human Metabolome Database |
| HRMS | High-resolution mass spectrometry |
| KEGG | Kyoto Encyclopedia of Genes and Genome |
| LC-MS/MS | Liquid chromatography–tandem mass spectrometry analysis |
| LOOCV | leave-one-out cross-validation |
| MS | Mass Spectrometry |
| PC | Principal component |
| PCA | Principal component analysis |
| RFE | Recursive feature elimination |
| SESI | Secondary electrospray ionization |
| SESI-MS | Secondary electrospray ionization-mass spectrometry |
| SESI-HRMS | Secondary electrospray ionization-high resolution mass spectrometry |
| SVM | Support vector machine algorithm |
| SVM-RFE | Recursive feature elimination with SVM |
| VOC | Volatile organic compound |
| TIC | Total ion chromatogram |
| TOF | Time of flight |
References
- Liu, D. Molecular Detection of Human Bacterial Pathogens; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Schivo, M.; Mccartney, M.; Yamaguchi, M.; Borràs, E.; Davis, C. Bottom-Up Cell Culture Models to Elucidate Human In Vitro Biomarkers of Infection. In Volatile Organic Compound Analysis in Biomedical Diagnosis Applications; Apple Academic Press: Palm Bay, FL, USA, 2018; pp. 105–122. [Google Scholar]
- Sethi, S.; Nanda, R.; Chakraborty, T. Clinical Application of Volatile Organic Compound Analysis for Detecting Infectious Diseases. Clin. Microbiol. Rev. 2013, 26, 462–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thorn, R.M.S.; Greenman, J. Microbial volatile compounds in health and disease conditions. J. Breath Res. 2012, 6, 024001. [Google Scholar] [CrossRef] [PubMed]
- Ratiu, I.A.; Ligor, T.; Bocos-Bintintan, V.; Buszewski, B. Mass spectrometric techniques for the analysis of volatile organic compounds emitted from bacteria. Bioanalysis 2017, 9, 1069–1092. [Google Scholar] [CrossRef]
- Labows, J.N.; Mcginley, K.J.; Webster, G.; Leyden, J. Headspace analysis of volatile metabolites of Pseudomonas aeruginosa and related species by gas chromatography-mass spectrometry. J. Clin. Microbiol. 1980, 12, 521–526. [Google Scholar] [CrossRef] [Green Version]
- Pavlou, A.; Turner, A.; Magan, N. Recognition of anaerobic bacterial isolates in vitro using electronic nose technology. Lett. Appl. Microbiol. 2002, 35, 366–369. [Google Scholar] [CrossRef] [PubMed]
- Jünger, M.; Vautz, W.; Kuhns, M.; Hofmann, L.; Ulbricht, S.; Baumbach, J.I.; Quintel, M.; Perl, T. Ion mobility spectrometry for microbial volatile organic compounds: A new identification tool for human pathogenic bacteria. Appl. Microbiol. Biotechnol. 2012, 93, 2603–2614. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bunge, M.; Araghipour, N.; Mikoviny, T.; Dunkl, J.; Schnitzhofer, R.; Hansel, A.; Schinner, F.; Wisthaler, A.; Margesin, R.; Märk, T.D. On-line monitoring of microbial volatile metabolites by proton transfer reaction-mass spectrometry. Appl. Environ. Microbiol. 2008, 74, 2179–2186. [Google Scholar] [CrossRef] [Green Version]
- Allardyce, R.; Langford, V.; Hill, A.; Murdoch, D. Detection of volatile metabolites produced by bacterial growth in blood culture media by selected ion flow tube mass spectrometry (SIFT-MS). J. Microbiol. Methods 2006, 65 2, 361–365. [Google Scholar] [CrossRef]
- Schulz, S.; Dickschat, J. Bacterial Volatiles: The Smell of Small Organisms. Nat. Prod. Rep. 2007, 24, 814–842. [Google Scholar] [CrossRef]
- Martínez-Lozano, P.; Rus, J.; Fernández de la Mora, G.; Hernández, M.; Fernández de la Mora, J. Secondary electrospray ionization (SESI) of ambient vapors for explosive detection at concentrations below parts per trillion. J. Am. Soc. Mass Spectrom. 2009, 20, 287–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaugg, M.T.; Gomez, D.G.; Barrios-Collado, C.; de Miguel, G.V.; Kohler, M.; Zenobi, R.; Sinues, P.M.L. Expanding metabolite coverage of real-time breath analysis by coupling a universal secondary electrospray ionization source and high resolution mass spectrometry—A pilot study on tobacco smokers. J. Breath Res. 2016, 10, 016010. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Bean, H.D.; Kuo, Y.M.; Hill, J.E. Fast Detection of Volatile Organic Compounds from Bacterial Cultures by Secondary Electrospray Ionization-Mass Spectrometry. J. Clin. Microbiol. 2010, 48, 4426–4431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ballabio, C.; Cristoni, S.; Puccio, G.; Kohler, M.; Sala, M.R.; Brambilla, P.; Martinez-Lozano Sinues, P. Rapid identification of bacteria in blood cultures by mass-spectrometric analysis of volatiles. J. Clin. Pathol. 2014, 67, 743–746. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Bean, H.D.; Jiménez-Díaz, J.; Hill, J.E. Secondary electrospray ionization-mass spectrometry (SESI-MS) breathprinting of multiple bacterial lung pathogens, a mouse model study. J. Appl. Physiol. 2013, 114, 1544–1549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Bean, H.D.; Wargo, M.J.; Leclair, L.W.; Hill, J.E. Detecting bacterial lung infections: In vivo evaluation of in vitro volatile fingerprints. J. Breath Res. 2013, 7, 016003. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhu, J. Differentiating antibiotic-resistant staphylococcus aureus using secondary electrospray ionization tandem mass spectrometry. Anal. Chem. 2018, 90, 12108–12115. [Google Scholar] [CrossRef] [PubMed]
- Bregy, L.; Müggler, A.R.; Sinues, P.M.L.; García-Gómez, D.; Suter, Y.; Belibasakis, G.N.; Kohler, M.; Schmidlin, P.R.; Zenobi, R. Differentiation of oral bacteria in in vitro cultures and human saliva by secondary electrospray ionization–mass spectrometry. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Zhu, J.; Jiménez-Díaz, J.; Bean, H.D.; Daphtary, N.A.; Aliyeva, M.I.; Lundblad, L.K.A.; Hill, J.E. Robust detection of P. aeruginosa and S. aureus acute lung infections by secondary electrospray ionization-mass spectrometry (SESI-MS) breathprinting: From initial infection to clearance. J. Breath Res. 2013, 7, 037106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Hill, J.E. Detection of Escherichia coli via VOC profiling using secondary electrospray ionization-mass spectrometry (SESI-MS). Food Microbiol. 2013, 34, 412–417. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Xu, M.; Zhu, J. Headspace Gas Monitoring of Gut Microbiota Using Targeted and Globally Optimized Targeted Secondary Electrospray Ionization Mass Spectrometry. Anal. Chem. 2019, 91, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.J.; Zhu, J. Optimizing Secondary Electrospray Ionization High-Resolution Mass Spectrometry (SESI-HRMS) for the Analysis of Volatile Fatty Acids from Gut Microbiome. Metabolites 2020, 10, 351. [Google Scholar] [CrossRef] [PubMed]
- Dührkop, K.; Fleischauer, M.; Ludwig, M.; Aksenov, A.A.; Melnik, A.V.; Meusel, M.; Dorrestein, P.C.; Rousu, J.; Böcker, S. SIRIUS 4: A rapid tool for turning tandem mass spectra into metabolite structure information. Nat. Methods 2019, 16, 299–302. [Google Scholar] [CrossRef] [Green Version]
- Gaugg, M.T.; Bruderer, T.; Nowak, N.; Eiffert, L.; Martinez-Lozano Sinues, P.; Kohler, M.; Zenobi, R. Mass-Spectrometric Detection of Omega-Oxidation Products of Aliphatic Fatty Acids in Exhaled Breath. Anal. Chem. 2017, 89, 10329–10334. [Google Scholar] [CrossRef] [PubMed]
- Ratjen, F.; Bell, S.C.; Rowe, S.M.; Goss, C.H.; Quittner, A.L.; Bush, A. Cystic fibrosis. Nat. Rev. Dis. Prim. 2015, 1, 15010. [Google Scholar] [CrossRef] [PubMed]
- Gaisl, T.; Bregy, L.; Stebler, N.; Gaugg, M.T.; Bruderer, T.; García-Gómez, D.; Moeller, A.; Singer, F.; Schwarz, E.I.; Benden, C.; et al. Real-time exhaled breath analysis in patients with cystic fibrosis and controls. J. Breath Res. 2018, 12, 036013. [Google Scholar] [CrossRef]
- Rees, C.A.; Burklund, A.; Stefanuto, P.H.; Schwartzman, J.D.; Hill, J.E. Comprehensive volatile metabolic fingerprinting of bacterial and fungal pathogen groups. J. Breath Res. 2018, 12, 026001. [Google Scholar] [CrossRef] [PubMed]
- Schulz-Bohm, K.; Martín-Sánchez, L.; Garbeva, P. Microbial Volatiles: Small Molecules with an Important Role in Intra- and Inter-Kingdom Interactions. Front. Microbiol. 2017, 8, 2484. [Google Scholar] [CrossRef]
- O’Hara, M.; Mayhew, C.A. A preliminary comparison of volatile organic compounds in the headspace of cultures of Staphylococcus aureus grown in nutrient, dextrose and brain heart bovine broths measured using a proton transfer reaction mass spectrometer. J. Breath Res. 2009, 3, 027001. [Google Scholar] [CrossRef]
- Lazazzara, V.; Perazzolli, M.; Pertot, I.; Biasioli, F.; Puopolo, G.; Cappellin, L. Growth media affect the volatilome and antimicrobial activity against Phytophthora infestans in four Lysobacter type strains. Microbiol. Res. 2017, 201, 52–62. [Google Scholar] [CrossRef]
- Küntzel, A.; Fischer, S.; Bergmann, A.; Oertel, P.; Steffens, M.; Trefz, P.; Miekisch, W.; Schubert, J.K.; Reinhold, P.; Köhler, H. Effects of biological and methodological factors on volatile organic compound patterns during cultural growth of Mycobacterium avium ssp. paratuberculosis. J. Breath Res. 2016, 10, 037103. [Google Scholar] [CrossRef]
- Rees, C.A.; Nordick, K.V.; Franchina, F.A.; Lewis, A.E.; Hirsch, E.B.; Hill, J.E. Volatile metabolic diversity of Klebsiella pneumoniae in nutrient-replete conditions. Metabolomics 2017, 13, 18. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Liang, J.; Chingin, K.; Hang, Y.; Wu, X.; Chen, H. Early release of 1-pyrroline by Pseudomonas aeruginosa cultures discovered using ambient corona discharge ionization mass spectrometry. RSC Adv. 2016, 6, 8449–8455. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef] [Green Version]
- Kreßel, U.H.G. Pairwise Classification and Support Vector Machines. In Advances in Kernel Methods: Support Vector Learning; MIT Press: Cambridge, MA, USA, 1999; pp. 255–268. [Google Scholar]
- Guyon, I.; Weston, J.; Barnhill, S.; Vapnik, V. Gene Selection for Cancer Classification using Support Vector Machines. Mach. Learn. 2002, 46, 389–422. [Google Scholar] [CrossRef]
- Hochberg, Y. A sharper Bonferroni procedure for multiple tests of significance. Biometrika 1988, 75, 800–802. [Google Scholar] [CrossRef]
- McMillan, A.; Renaud, J.B.; Gloor, G.B.; Reid, G.; Sumarah, M.W. Post-acquisition filtering of salt cluster artefacts for LC-MS based human metabolomic studies. J. Cheminform. 2016, 8, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Filipiak, W.; Sponring, A.; Baur, M.M.; Filipiak, A.; Ager, C.; Wiesenhofer, H.; Nagl, M.; Troppmair, J.; Amann, A. Molecular analysis of volatile metabolites released specifically by staphylococcus aureus and pseudomonas aeruginosa. BMC Microbiol. 2012, 12, 113. [Google Scholar] [CrossRef] [Green Version]
- Bean, H.; Rees, C.; Hill, J. Comparative analysis of the volatile metabolomes of Pseudomonas aeruginosa clinical isolates. J. Breath Res. 2016, 10, 047102. [Google Scholar] [CrossRef] [PubMed]
- He, C.N.; Ye, W.Q.; Zhu, Y.Y.; Zhou, W.W. Antifungal Activity of Volatile Organic Compounds Produced by Bacillus methylotrophicus and Bacillus thuringiensis against Five Common Spoilage Fungi on Loquats. Molecules 2020, 25, 3360. [Google Scholar] [CrossRef] [PubMed]
- Routray, W.; Rayaguru, K. 2-Acetyl-1-pyrroline: A key aroma component of aromatic rice and other food products. Food Rev. Int. 2018, 34, 539–565. [Google Scholar] [CrossRef]
- Yap, I.K.S.; Li, J.V.; Saric, J.; Martin, F.P.; Davies, H.; Wang, Y.; Wilson, I.D.; Nicholson, J.K.; Utzinger, J.; Marchesi, J.R.; et al. Metabonomic and Microbiological Analysis of the Dynamic Effect of Vancomycin-Induced Gut Microbiota Modification in the Mouse. J. Proteome Res. 2008, 7, 3718–3728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.Y.; Mao, Z.C.; Wu, Y.X.; Ho, H.H.; He, Y.Q. Comprehensive volatile organic compounds profiling of Bacillus species with biocontrol properties by head space solid phase microextraction with gas chromatography-mass spectrometry. Biocontrol Sci. Technol. 2015, 25, 132–143. [Google Scholar] [CrossRef]
- Ji, D.; Yi, Y.; Kang, G.H.; Choi, Y.H.; Kim, P.; Baek, N.I.; Kim, Y. Identification of an antibacterial compound, benzylideneacetone, from Xenorhabdus nematophila against major plant-pathogenic bacteria. FEMS Microbiol. Lett. 2004, 239, 241–248. [Google Scholar] [CrossRef] [Green Version]
- Ding, L.; Pfoh, R.; Rühl, S.; Qin, S.; Laatsch, H. T-Muurolol Sesquiterpenes from the Marine Streptomyces sp. M491 and Revision of the Configuration of Previously Reported Amorphanes. J. Nat. Prod. 2009, 72, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Sokal, R.; Michener, C. A Statistical Method of Evaluating Systematic Relationships. Univ. Kans. Sci. Bull. 1958, 38, 1409–1438. [Google Scholar]
- Nizio, K.; Perrault, K.; Troobnikoff, A.; Ueland, M.; Mohsin, S.; Iredell, J.; Middleton, P.; Forbes, S. In vitro volatile organic compound profiling using GC×GC-TOFMS to differentiate bacteria associated with lung infections: A proof-of-concept study. J. Breath Res. 2016, 10, 026008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitzgerald, S.; Duffy, E.; Holland, L.; Morrin, A. Multi-strain volatile profiling of pathogenic and commensal cutaneous bacteria. Sci. Rep. 2020, 10, 17971. [Google Scholar] [CrossRef] [PubMed]
- Kessner, D.; Chambers, M.; Burke, R.; Agus, D.; Mallick, P. ProteoWizard: Open source software for rapid proteomics tools development. Bioinformatics 2008, 24, 2534–2536. [Google Scholar] [CrossRef]
- Fritsch, F.N.; Carlson, R.E. Monotone Piecewise Cubic Interpolation. SIAM J. Numer. Anal. 1980, 17, 238–246. [Google Scholar] [CrossRef]
- Mann, H.B.; Whitney, D.R. On a Test of Whether one of Two Random Variables is Stochastically Larger than the Other. Ann. Math. Stat. 1947, 18, 50–60. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling The False Discovery Rate - A Practical And Powerful Approach To Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Cortes, C.; Vapnik, V. Support-vector networks. Mach. Learn. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Bottou, L.; Cortes, C.; Denker, J.S.; Drucker, H.; Guyon, I.; Jackel, L.D.; LeCun, Y.; Muller, U.A.; Sackinger, E.; Simard, P.; et al. Comparison of classifier methods: A case study in handwritten digit recognition. In Proceedings of the 12th IAPR International Conference on Pattern Recognition, Jerusalem, Israel, 9–13 October 1994; Volume 2, pp. 77–82. [Google Scholar]
- Varma, S.; Simon, R. Bias in error estimation when using cross-validation for model selection. BMC Bioinform. 2006, 7, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bermingham, M.L.; Pong-Wong, R.; Spiliopoulou, A.; Hayward, C.; Rudan, I.; Campbell, H.; Wright, A.F.; Wilson, J.F.; Agakov, F.; Navarro, P.; et al. Application of high-dimensional feature selection: Evaluation for genomic prediction in man. Sci. Rep. 2015, 5, 10312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dührkop, K.; Shen, H.; Meusel, M.; Rousu, J.; Böcker, S. Searching molecular structure databases with tandem mass spectra using CSI:FingerID. Proc. Natl. Acad. Sci. USA 2015, 112, 12580–12585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ludwig, M.; Nothias, L.F.; Dührkop, K.; Koester, I.; Fleischauer, M.; Hoffmann, M.A.; Petras, D.; Vargas, F.; Morsy, M.; Aluwihare, L.; et al. ZODIAC: Database-independent molecular formula annotation using Gibbs sampling reveals unknown small molecules. bioRxiv 2019. [Google Scholar] [CrossRef] [Green Version]
- Gaugg, M.T.; Engler, A.; Bregy, L.; Nussbaumer-Ochsner, Y.; Eiffert, L.; Bruderer, T.; Zenobi, R.; Sinues, P.; Kohler, M. Molecular breath analysis supports altered amino acid metabolism in idiopathic pulmonary fibrosis. Respirology 2019, 24, 437–444. [Google Scholar] [CrossRef] [Green Version]
- Gaugg, M.; Engler, A.; Nussbaumer-Ochsner, Y.; Bregy, L.; Stöberl, A.; Gaisl, T.; Bruderer, T.; Zenobi, R.; Kohler, M.; Sinues, P. Metabolic effects of inhaled salbutamol determined by exhaled breath analysis. J. Breath Res. 2017, 11, 046004. [Google Scholar] [CrossRef] [PubMed]
- Pence, H.E.; Williams, A. ChemSpider: An Online Chemical Information Resource. J. Chem. Educ. 2010, 87, 1123–1124. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaeslin, J.; Micic, S.; Weber, R.; Müller, S.; Perkins, N.; Berger, C.; Zenobi, R.; Bruderer, T.; Moeller, A. Differentiation of Cystic Fibrosis-Related Pathogens by Volatile Organic Compound Analysis with Secondary Electrospray Ionization Mass Spectrometry. Metabolites 2021, 11, 773. https://doi.org/10.3390/metabo11110773
Kaeslin J, Micic S, Weber R, Müller S, Perkins N, Berger C, Zenobi R, Bruderer T, Moeller A. Differentiation of Cystic Fibrosis-Related Pathogens by Volatile Organic Compound Analysis with Secondary Electrospray Ionization Mass Spectrometry. Metabolites. 2021; 11(11):773. https://doi.org/10.3390/metabo11110773
Chicago/Turabian StyleKaeslin, Jérôme, Srdjan Micic, Ronja Weber, Simona Müller, Nathan Perkins, Christoph Berger, Renato Zenobi, Tobias Bruderer, and Alexander Moeller. 2021. "Differentiation of Cystic Fibrosis-Related Pathogens by Volatile Organic Compound Analysis with Secondary Electrospray Ionization Mass Spectrometry" Metabolites 11, no. 11: 773. https://doi.org/10.3390/metabo11110773
APA StyleKaeslin, J., Micic, S., Weber, R., Müller, S., Perkins, N., Berger, C., Zenobi, R., Bruderer, T., & Moeller, A. (2021). Differentiation of Cystic Fibrosis-Related Pathogens by Volatile Organic Compound Analysis with Secondary Electrospray Ionization Mass Spectrometry. Metabolites, 11(11), 773. https://doi.org/10.3390/metabo11110773