Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Standard Solution Preparation
2.3. Chromatographic Conditions
2.4. Preparation of Physical Blend and HGCs
2.5. Sample Preparation
2.6. Validation of the Method Parameters
3. Results and Discussion
3.1. Optimization of the Mobile Phase
3.2. Method Validation
3.3. Chromatographic Conditions
3.4. Assay of Commercial Formulation (HGC Samples)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Guo, Q.; Li, W.; Wang, C.; Mao, X.; Wang, X.; Chen, W.; Xu, H.; Wang, Q.; Zhang, Y.; Lin, N. Biomolecular network-based synergistic drug combination discovery: A combination of paeoniflorin and liquiritin alleviates neuropathic pain by inhibiting neuroin-flammation via suppressing the chemokine signaling pathway. Signal Transduct. Target. Ther. 2020, 5, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Aldewachi, H.S.; Gurram, A. A comparative study of two chromatographic techniques for the determination of group of im-idazoles simultaneously. Int. J. Pharm. Sci. Rev. Res. 2013, 20, 153–158. [Google Scholar]
- Al-Khadhra, R.S. The determination of common anabolic steroid and stimulants in nutritional supplements by HPLC-DAD and LC-MS. J. Chromatogr. Sci. 2020, 58, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Kavathia, A.; Misra, M. Development and validation of RP-HPLC and UV-spectrophotometric methods for rapid simultaneous estimation of amlodipine and benazepril in pure and fixed dose combination. Arab. J. Chem. 2017, 10, S3021–S3028. [Google Scholar] [CrossRef]
- Mangla, B.; Beg, S.; Alam, O.; Ahsan, W.; Haque, A.; Patel, K.S.; Almalki, W.H.; Alrobaian, M.; Kohli, K. Systematic development and validation of RP-HPLC method for simultaneous estimation of tamoxifen and sulphoraphane with specific application for nanolipidic formulations. Arab. J. Chem. 2020, 13, 7909–7920. [Google Scholar] [CrossRef]
- Chiarugi, A.; Camaioni, A. Update on the pathophysiology and treatment of rhinogenic headache: Focus on the ibu-profen/pseudoephedrine combination. Acta Otorhinolaryngol. Ital. 2019, 39, 22. [Google Scholar] [CrossRef]
- El-Maraghy, C.M.; Lamie, N.T. Three smart spectrophotometric methods for resolution of severely overlapped binary mixture of Ibuprofen and Paracetamol in pharmaceutical dosage form. BMC Chem. 2019, 13, 1–8. [Google Scholar] [CrossRef]
- Magdy, G.; Belal, F.; Abdel-Megied, A.M.; Hakiem, A.F.A. Two different synchronous spectrofluorimetric approaches for simultaneous determination of febuxostat and ibuprofen. R. Soc. Open Sci. 2021, 8, 210354. [Google Scholar] [CrossRef]
- Farrar, H.; Letzig, L.; Gill, M. Validation of a liquid chromatographic method for the determination of ibuprofen in human plasma. J. Chromatogr. B 2002, 780, 341–348. [Google Scholar] [CrossRef]
- Şenol, Y.; DOĞAN, A.; Çelebier, M. Ultrafiltration-Based extraction of Ibuprofen from human plasma samples and HPLC analysis: Developing an innovative bioanalytical analysis method. Cumhur. Sci. J. 2021, 42, 276–284. [Google Scholar] [CrossRef]
- Paik, M.J.; Kim, K.R. Optical purity determination of ibuprofen in tablets by achiral gas chromatography. Arch. Pharm. Res. 2004, 27, 820–824. [Google Scholar]
- A A Ragab, M.; Abdel-Hay, M.H.; Ahmed, H.M.; Mohyeldin, S.M. Determination of Ibuprofen and Phenylephrine in Tablets by High-Performance Thin Layer Chromatography and in Plasma by High-Performance Liquid Chromatography with Diode Array Detection. J. Chromatogr. Sci. 2019, 57, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Geetha, L.E.; Rama, S.M. A novel RP-HPLC method for simultaneous estimation of codeine phosphate, chlorpheniramine maleate and its preservative in syrup formulation. Int. J. Pharm. Pharm. Sci. 2012, 4 (Suppl. S3), 585–590. [Google Scholar]
- Ge, Q.-H.; Zhou, Z.; Zhi, X.-J.; Wang, H. Simultaneous determination of pseudoephedrine and chlorpheniramine in human plasma by HPLC-UV detection method. Yao Xue Xue Bao Acta Pharm. Sin. 2001, 19, 236–238. [Google Scholar]
- Fouad, M.M.; El-Maraghy, C.M. Rapid validated thin-layer chromatography–densitometry for the simultaneous determination of three co-formulated drugs used for common cold treatment. JPC J. Planar Chromatogr. Mod. TLC 2019, 32, 127–131. [Google Scholar] [CrossRef]
- Dong, Y.; Chen, X.; Chen, Y.; Chen, X.; Hu, Z. Separation and determination of pseudoephedrine, dextromethorphan, diphenhydramine and chlorpheniramine in cold medicines by nonaqueous capillary electrophoresis. J. Pharm. Biomed. Anal. 2005, 39, 285–289. [Google Scholar] [CrossRef]
- Chawe, L.C.; Tittikpina, N.K.; Ndiaye, S.M.; Diop, A.; Ndiaye, B.; Fall, D.; Diop, Y.M.; Sarr, S.O. Validation of an UV-Visible spectro-photometry assay method for the determination of chlorpheniramine maleate tablets without prior extraction. Int. J. Biol. Chem. Sci. 2021, 15, 273–281. [Google Scholar] [CrossRef]
- Al-Shaalan, N.H. Determination of phenylephrine hydrochloride and chlorpheniramine maleate in binary mixture using chemometric-assisted spectrophotometric and high-performance liquid chromatographic-UV methods. J. Saudi Chem. Soc. 2010, 14, 15–21. [Google Scholar] [CrossRef]
- Sanchaniya, P.M.; Mehta, F.A.; Uchadadiya, N.B. Development and validation of an RP-HPLC method for estimation of chlor- pheniramine maleate, ibuprofen, and phenylephrine hydrochloride in combined pharmaceutical dosage form. Chromatogr. Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Aşçi, B.; Dönmmez, Ö.A.; Bozdoğan, A.; Sungur, S. Experimental design of reversed-phase high performance liquid chromato-graphic conditions for simultaneous determination of ibuprofen, pseudoephedrine hydrochloride, chlorpheniramine male-ate, and nipagen. J. Anal. Chem. 2010, 65, 743–748. [Google Scholar] [CrossRef]
- Sarılmışer, H.K.; Özçelik, N.O.; Arslan, B.; Gökalp, M.; Ustaoğlu, A.; Dude, U.K.; Knezevic, Z. Development and validation of a simple HPLC assay method for ibuprofen, pseudoephedrine hydrochloride and chlorpheniramine maleate in syrup form. J. Chem. Metrol. 2017, 11, 61–67. [Google Scholar] [CrossRef]
- Mazzoccanti, G.; Gasparrini, F.; Calcaterra, A.; Villani, C.; Ciogli, A. Static vs. Dynamic Electrostatic Repulsion Reversed Phase Liquid Chromatography: Solutions for Pharmaceutical and Biopharmaceutical Basic Compounds. Separations 2021, 8, 59. [Google Scholar] [CrossRef]
- Langlois, M.-H.; Dallet, P.; Kauss, T.; Dubost, J.-P. Simultaneous Determination of Ibuprofen and Pseudoephedrine Hydrochloride in Pharmaceutical Tablets by Reversed-Phase HPLC. Anal. Lett. 2009, 42, 2951–2961. [Google Scholar] [CrossRef]
- Yang, M.; Fazio, S.; Munch, D.; Drumm, P. Impact of methanol and acetonitrile on separations based on π–π interactions with a reversed-phase phenyl column. J. Chromatogr. A 2005, 1097, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Moyano, M.A.; Rosasco, M.A.; Pizzorno, M.T.; Segall, A.I. Simultaneous determination of chlorpheniramine maleate and dexame-thasone in a tablet dosage form by liquid chromatography. J. AOAC Int. 2005, 88, 1677–1683. [Google Scholar] [CrossRef]
- Singh, J. International conference on harmonization of technical requirements for registration of pharmaceuticals for human use. J. Pharmacol. Pharmacother. 2015, 6, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.M.; Bai, L. Sampling Distribution of the Coefficient of Variation when the Population Takes Normal Distribution N(0,σ2). Adv. Mater. Res. 2011, 291–294, 3300–3304. [Google Scholar] [CrossRef]
- Ahmed, M.; Ali, A.; Mahmud, T.; Qadir, M.; Nadeem, K.; Saleem, A. Stability-indicating High-performance liquid chromatography method for simultaneous determination of aminophylline and chlorpheniramine maleate in pharmaceutical formulations. Indian J. Pharm. Sci. 2015, 77, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Darmawati, A.; Annuryanti, F.; Primaharinastiti, R.; Isnaeni, I. Determination and stability testing method of chlorpheniramine maleate in the presence of tartrazine using HPLC. Pharmaciana 2020, 10, 269–280. [Google Scholar] [CrossRef]
- British Pharmacopoeia Commission. British Pharmacopoeia; British Pharmacopoeia Commission: London, UK, 2020.
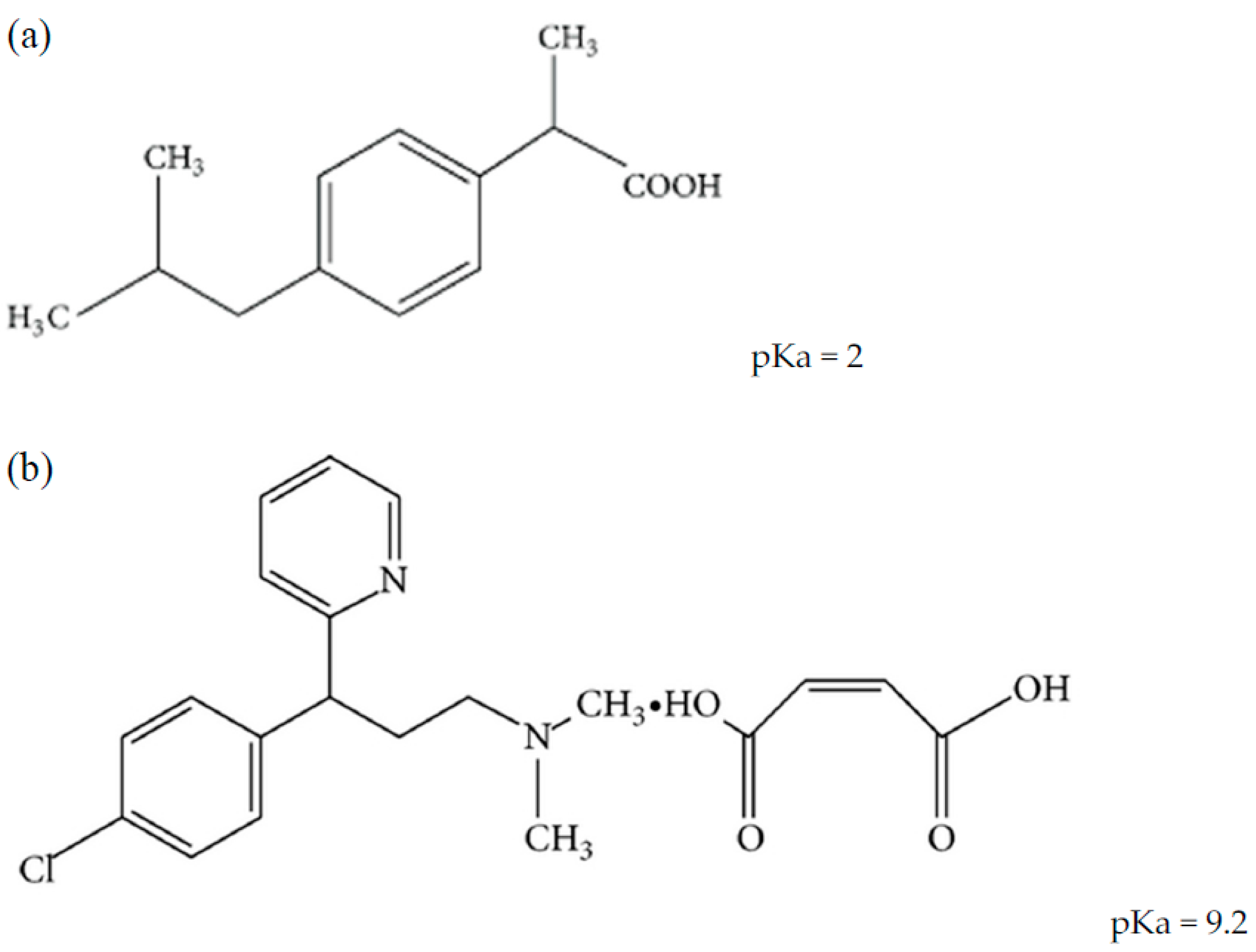
| Method (Year) | Run Time | Mobile Phase | Column & Gradient |
|---|---|---|---|
| Sanchaniya et al. [19] | 14 min | Acetonitrile : methanol : phoshphate buffer (50:20:30, v/v/v) | C 18 column (5 μm × 250 mm × 4.6 mm) isocratic |
| Asçi et al. [20] | 15 min | Acetonitrile buffer (15:85) for 5.5 min, (45:55) for 5.5–12 min, (60:40) for 12–17 min. | C18 (300 mm × 3.9 mm) gradient |
| Sarılmışer et al. [21] | 28 min | Buffer: methanol (80:20) | ODS4 column (250 mm × 4.6 mm; 5 μm) isocratic |
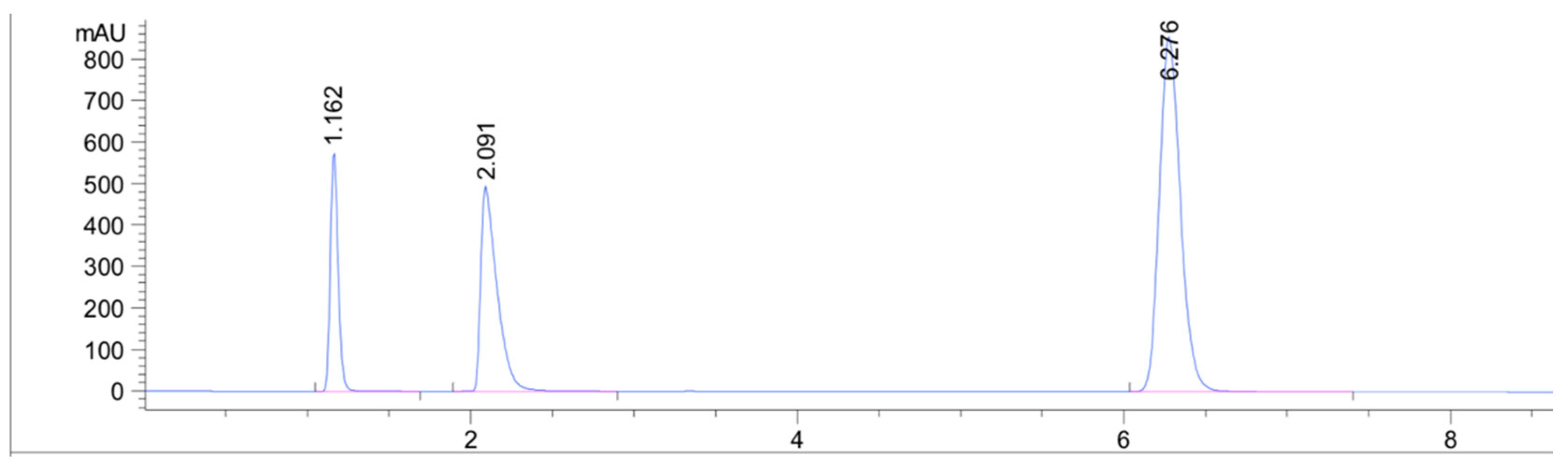
| This method | 7 min | Acetonitrile and acetate buffer at pH 3.8 (55:45; v/v) | Eclipse Plus C18 isocratic |
| Parameters | CPM | IBF |
|---|---|---|
| Linearity (range) (mg/mL) | 0.3–3.5 | 0.4–4.0 |
| Retention time (min) | 2.09 | 6.27 |
| Detection limit (μg/mL) | 10 | 27 |
| Quantitation limit (μg/mL) | 33 | 90 |
| Accuracy (%) (n = 5) | 97.7–98.9 | 101.0–104.5 |
| Intra day precision. (n = 5) | 1.50 | 1.35 |
| Day to day precision (RSD%) * (n = 5) | 2.14 | 3.48 |
| Drug | Concentration Range, mg/mL | Correlation Coefficient |
|---|---|---|
| Ibuprofen | 0.4–4.0 | 0.9998 |
| Chlorpheniramine | 0.3–3.5 | 0.9992 |
| Malic acid | 0.3–3.2 | 0.9970 |
| Drug | Theoretical % | Measured % | Recovery % |
|---|---|---|---|
| Chlorpheniramine maleate | 10 | 10.85 | 108.50% |
| Ibuprofen | 20 | 18.95 | 94.75% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aldewachi, H.; Omar, T.A. Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate. Sci. Pharm. 2022, 90, 53. https://doi.org/10.3390/scipharm90030053
Aldewachi H, Omar TA. Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate. Scientia Pharmaceutica. 2022; 90(3):53. https://doi.org/10.3390/scipharm90030053
Chicago/Turabian StyleAldewachi, Hasan, and Thamer A. Omar. 2022. "Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate" Scientia Pharmaceutica 90, no. 3: 53. https://doi.org/10.3390/scipharm90030053
APA StyleAldewachi, H., & Omar, T. A. (2022). Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate. Scientia Pharmaceutica, 90(3), 53. https://doi.org/10.3390/scipharm90030053







