Emerging Approach for the Application of Hibiscus sabdariffa Extract Ointment in the Superficial Burn Care
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection of Plant and Preparation of the Extract
2.2. Qualitative Phytochemical Analysis
2.3. Chemicals and Reagents
2.4. Formulation of the Ointment
2.5. Evaluation of Ointment Stability
2.6. Testing for Acute Skin Irritation and Toxicity of the Ointment
2.7. Animals and Experimental Protocols
2.8. Induction of Burn
2.9. Collection of Tissues
2.10. Assessment of Burn Intensity
2.11. Histological and Immunohistochemical Analysis
2.12. Determination of Total Antioxidant Capacity
2.13. Determination of Inflammatory Markers
2.14. Western Blot Analysis for TLR4 Expression
2.15. Antibiotic Sensitivity and Evaluation of Antibacterial Activity
2.16. Statistical Analysis
3. Results
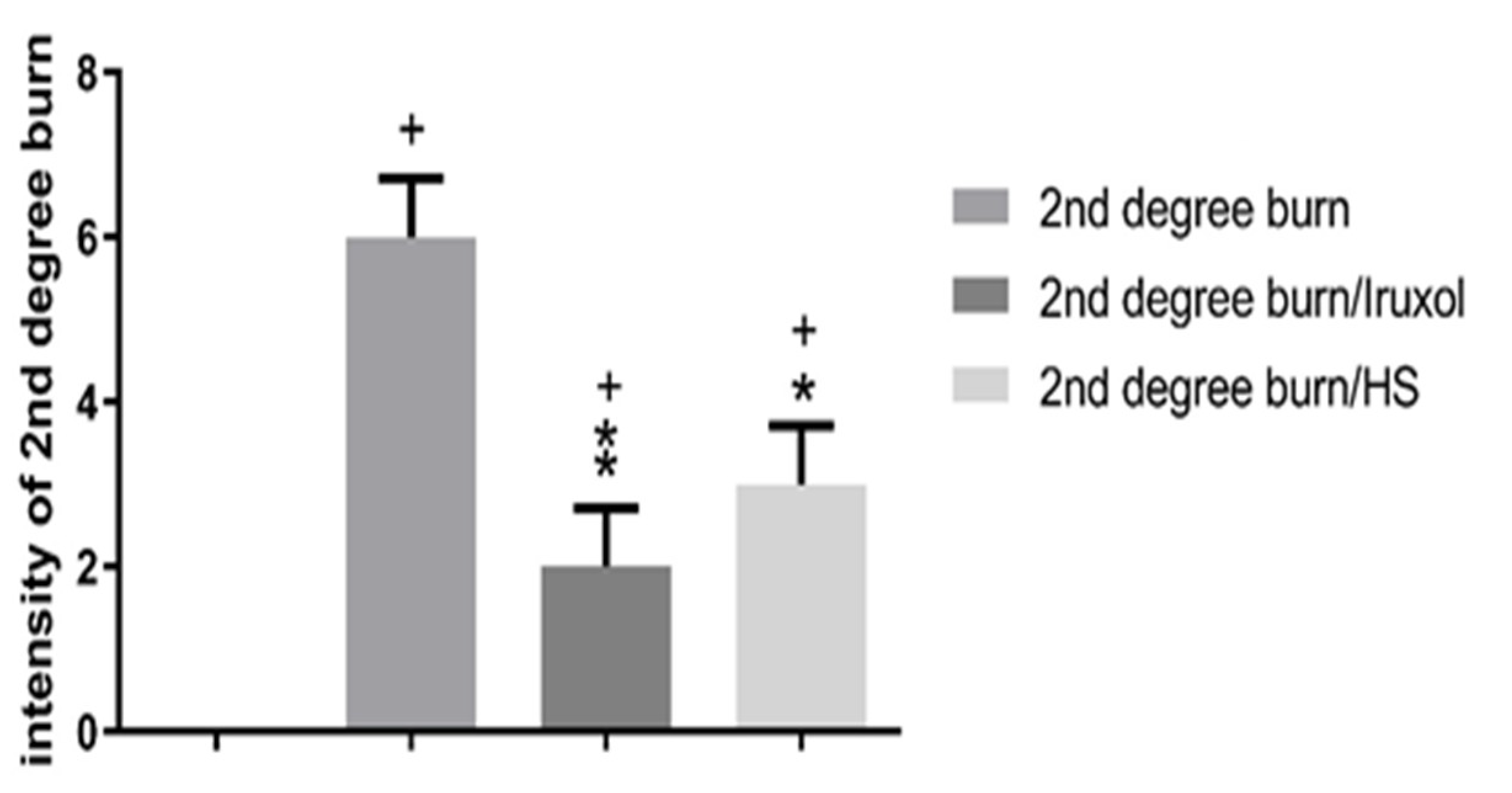
3.1. Effect of HS on the Intensity of 2nd Degree Burn Wound
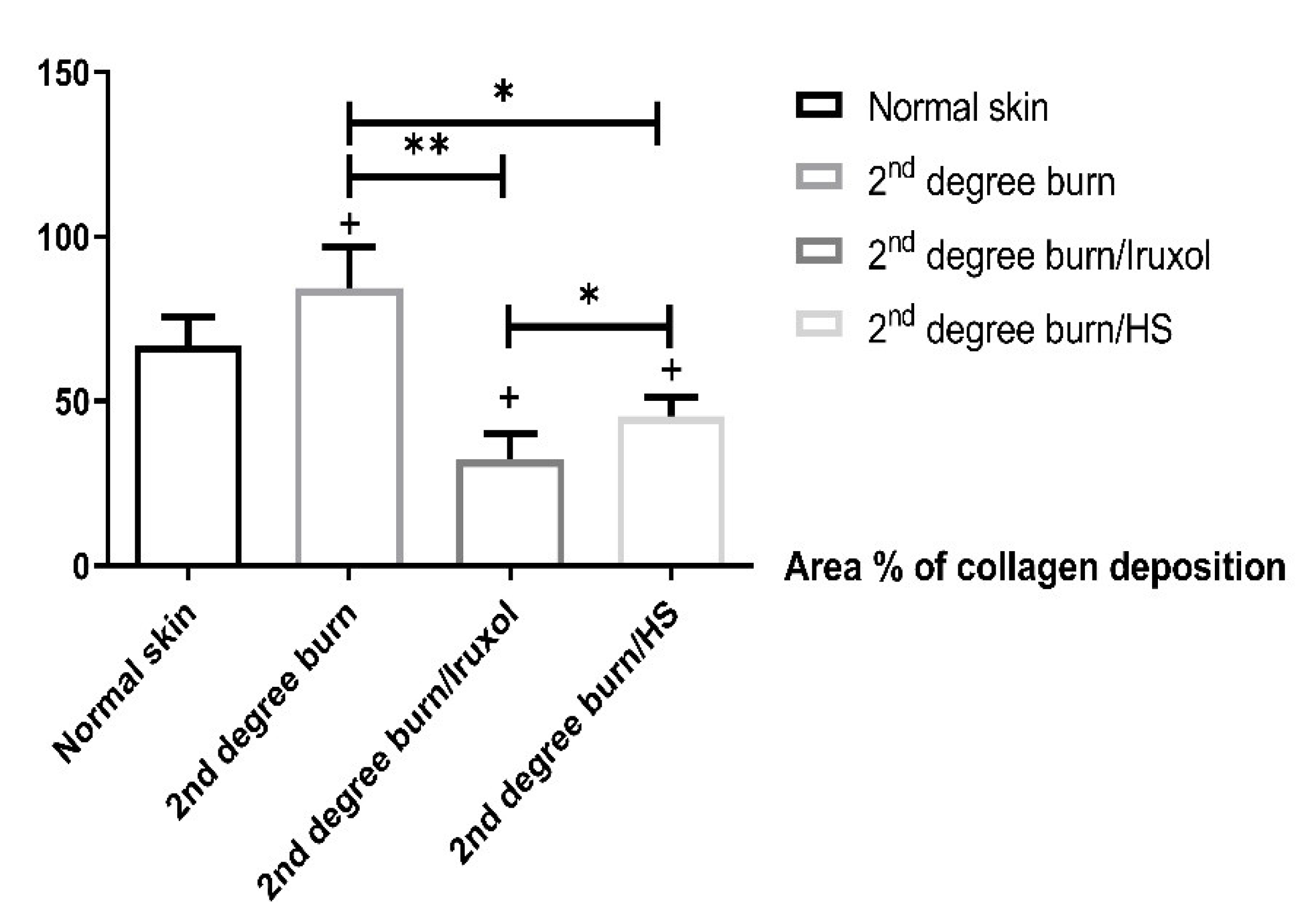
3.2. Effect of HS on Collagen Deposition
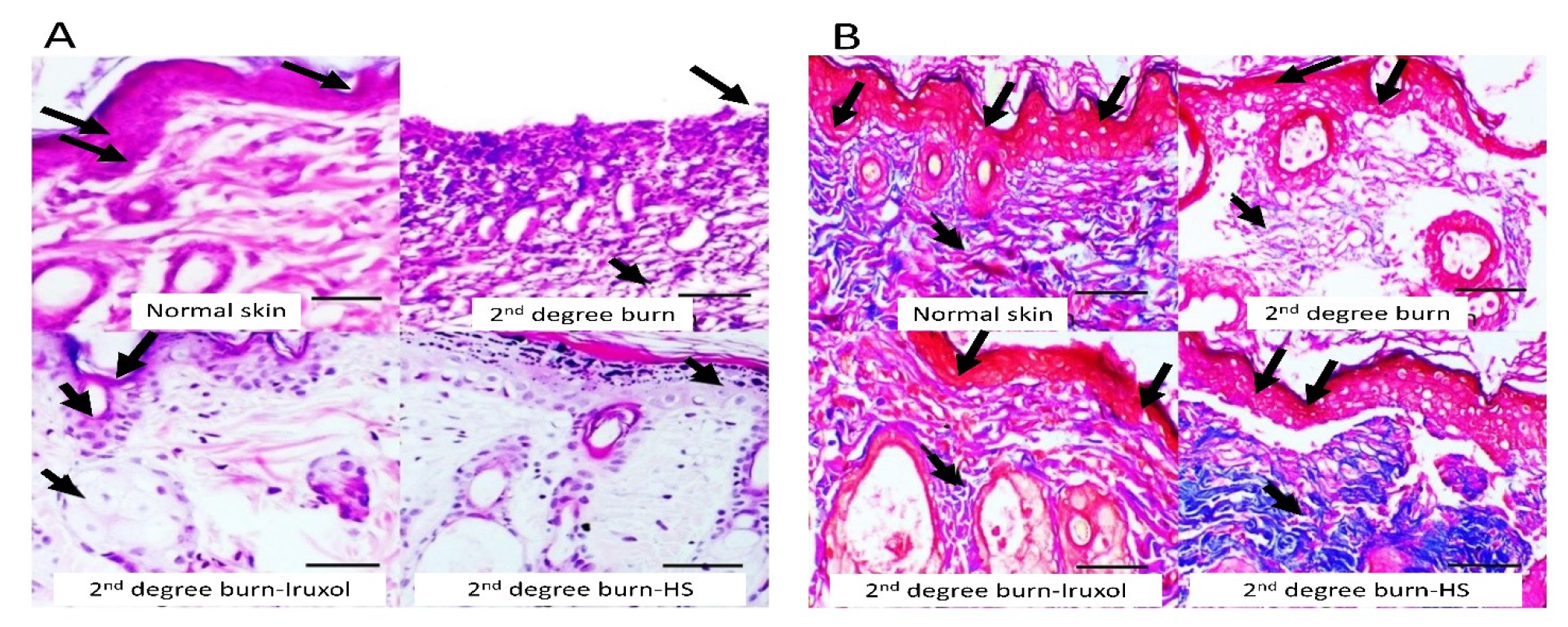
3.3. Histopathological Examination of Skin Tissues
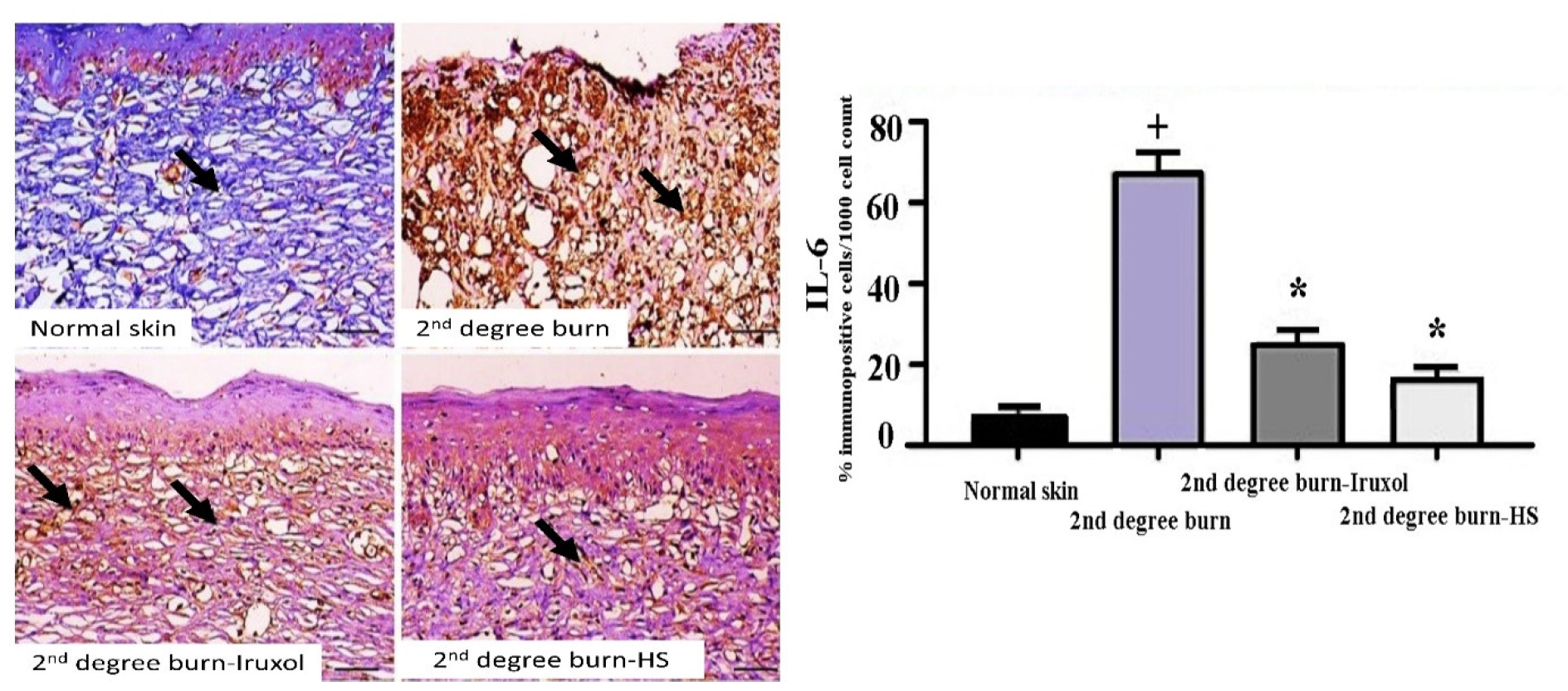
3.4. Immunostaining of IL-6
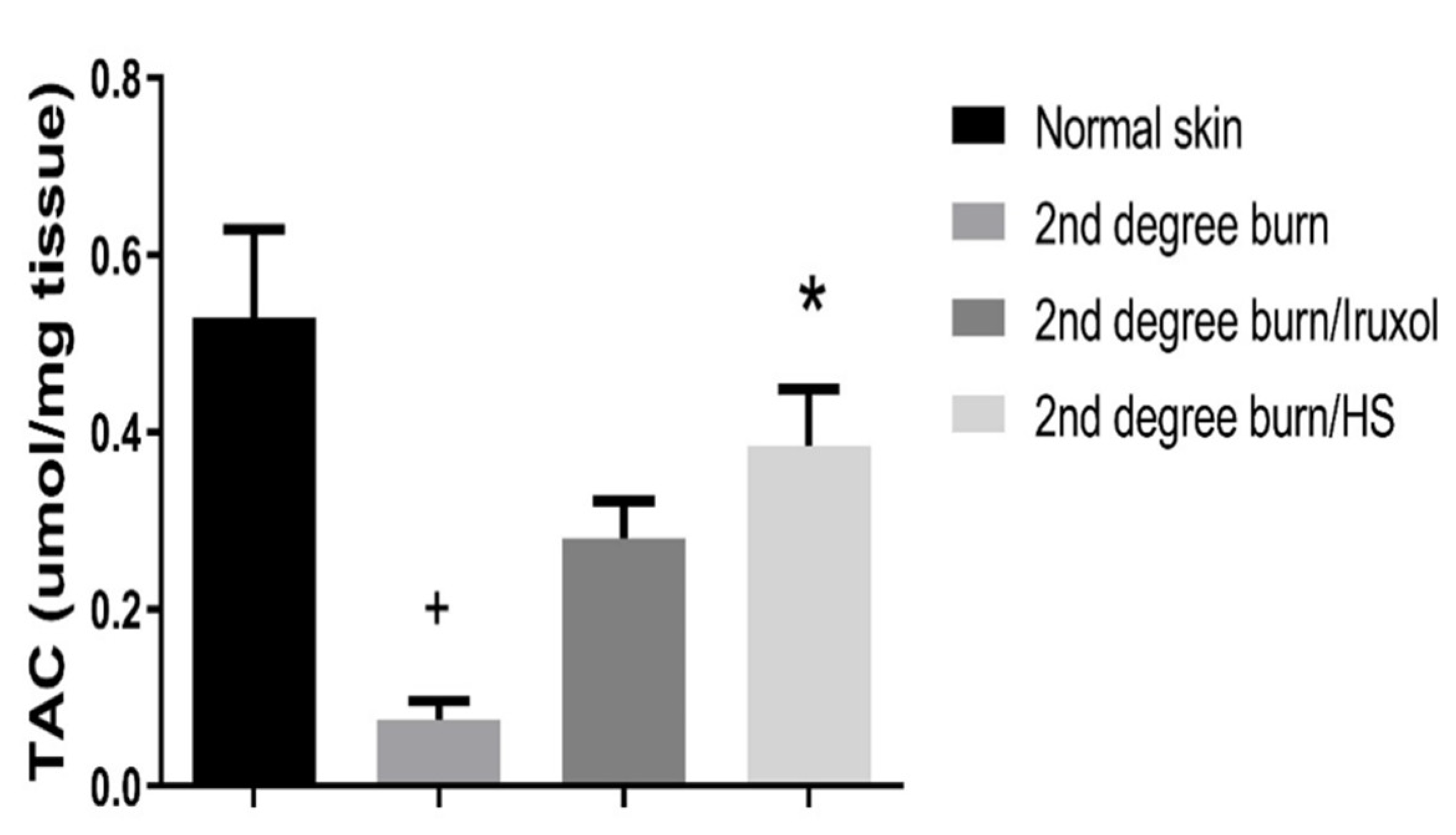
3.5. Effect of HS on Total Antioxidant Capacity (TAC)
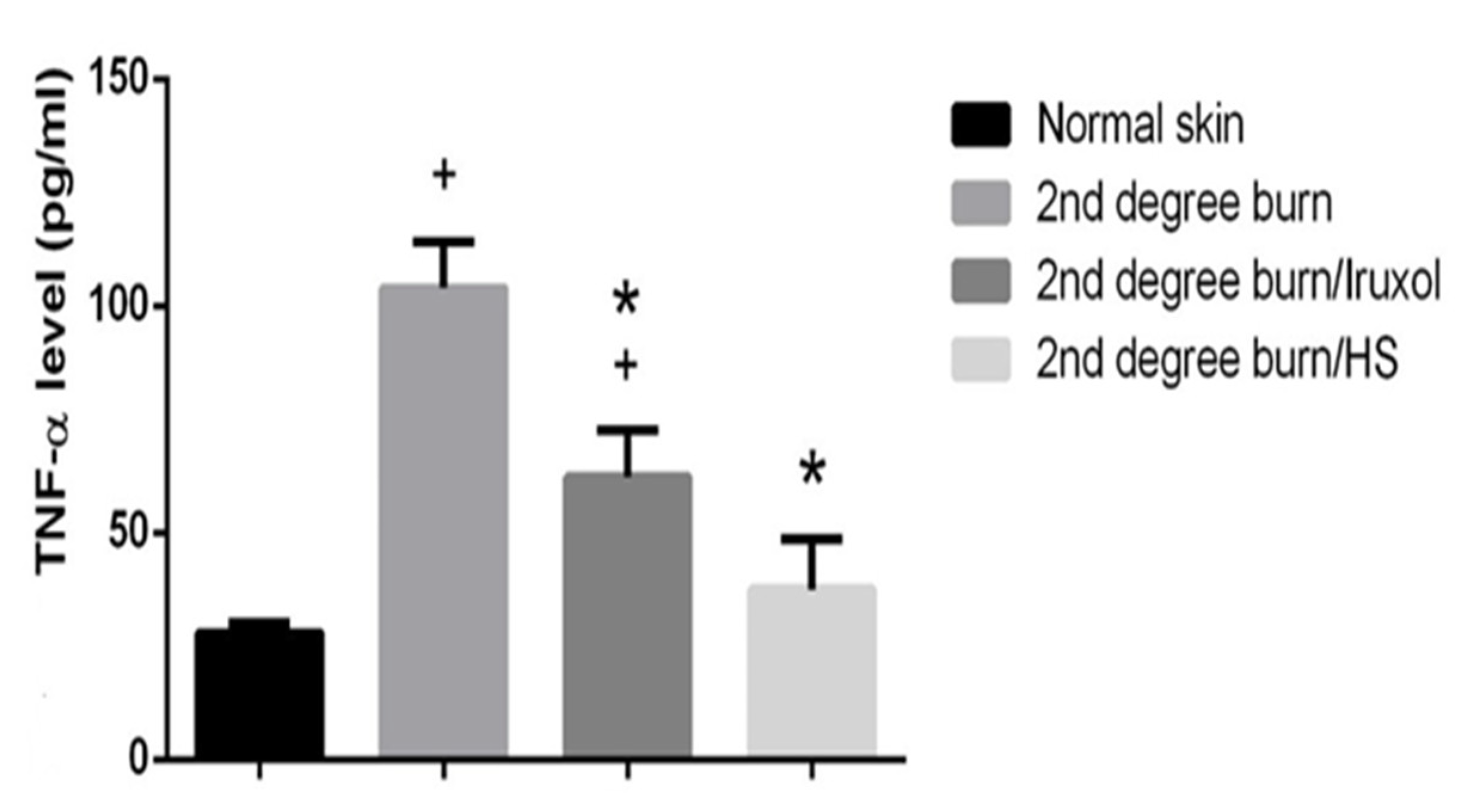
3.6. Effect of HS on TNF-α
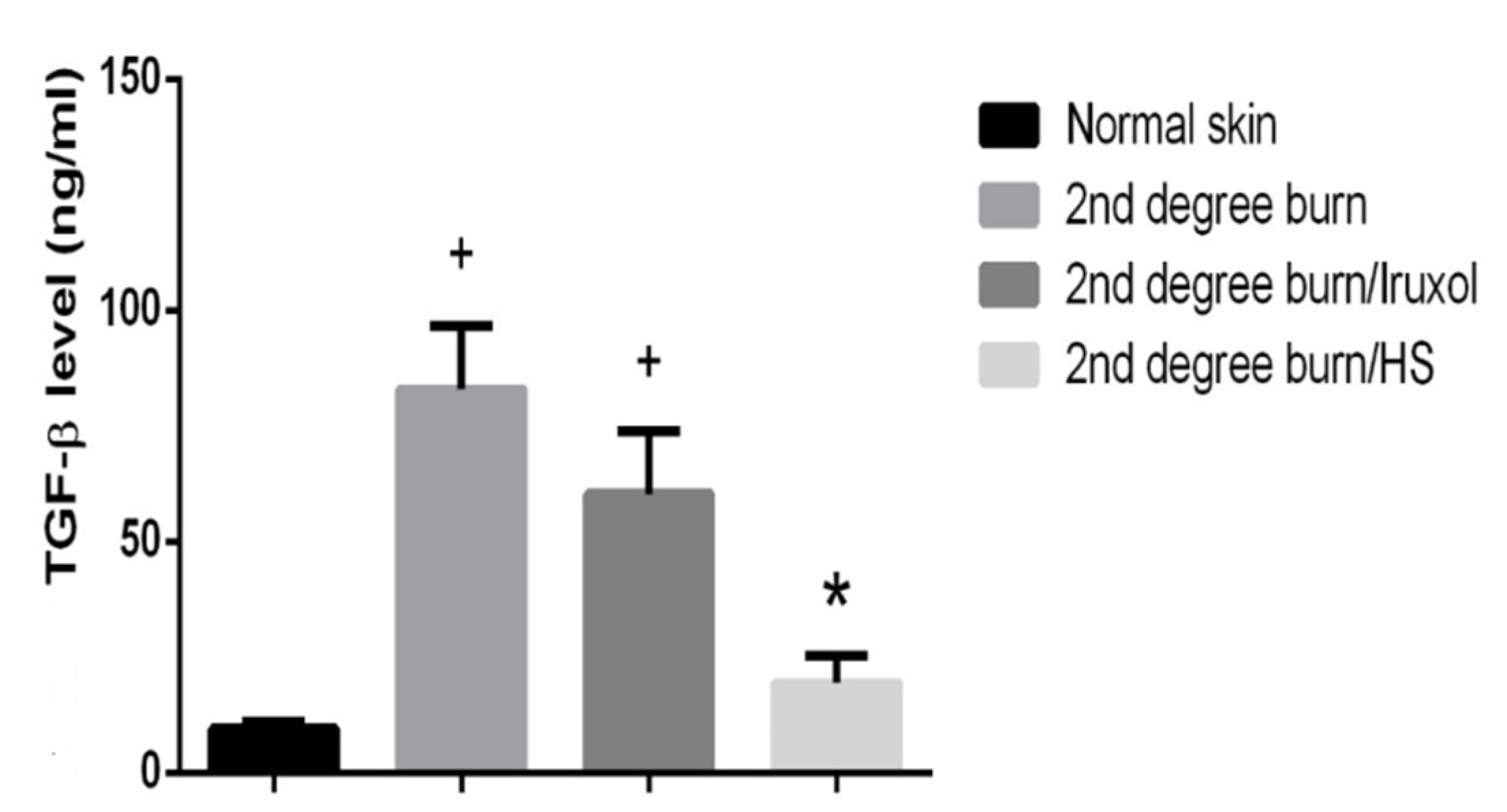
3.7. Effect of HS on TGF-β
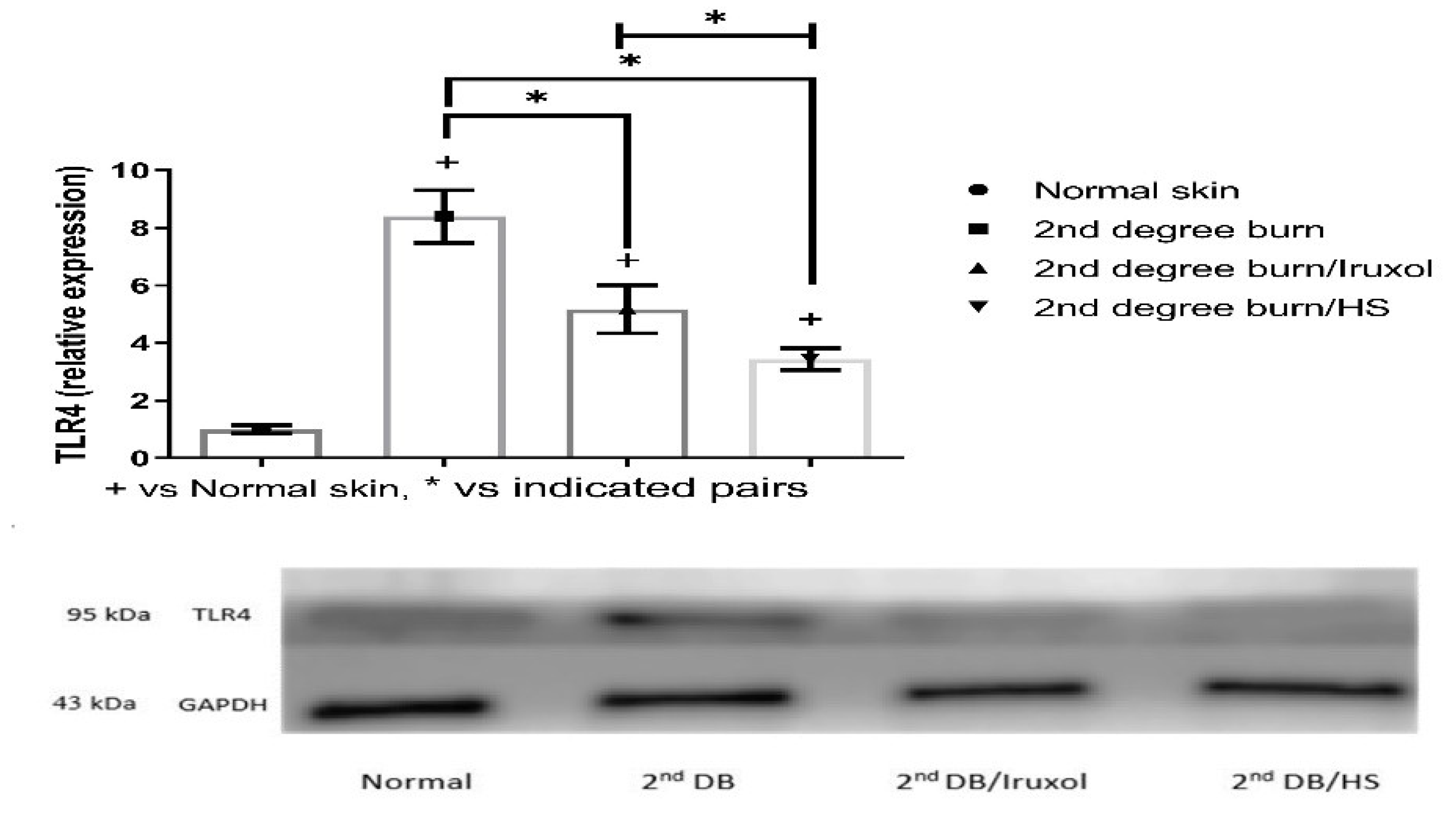
3.8. Effect of HS on TLR4 Expression in Different Groups
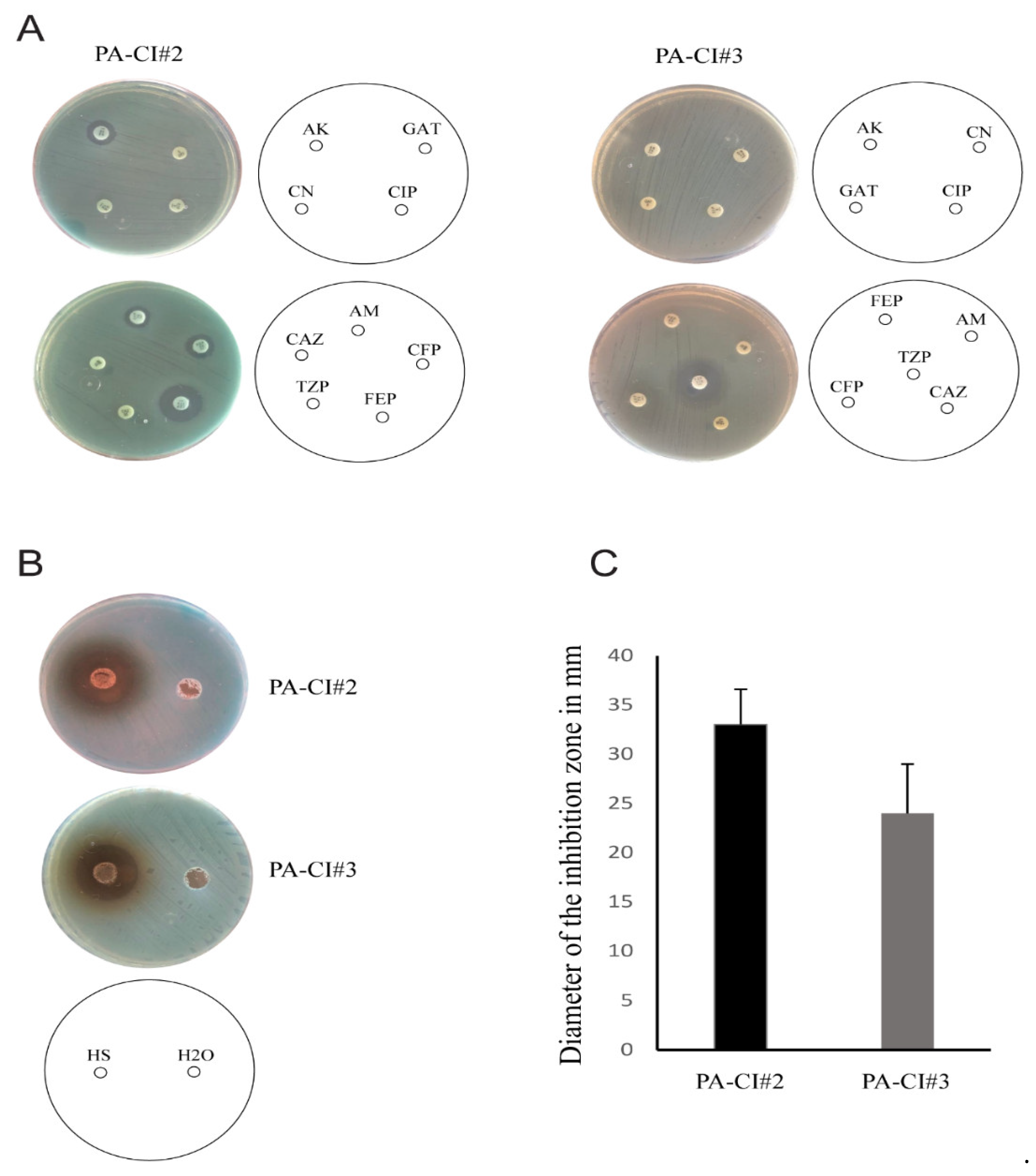
3.9. Antibacterial Activity of HS on Burn Wound Isolates of P. aeruginosa
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yadav, E.; Singh, D.; Yadav, P.; Verma, A. Antioxidant and anti-inflammatory properties of Prosopis cineraria based phenolic rich ointment in wound healing. Biomed. Pharmacother. 2018, 108, 1572–1583. [Google Scholar] [CrossRef] [PubMed]
- Antonescu, I.A.; Antonescu, A.; Miere, F.; Fritea, L.; Teușdea, A.C.; Vicaș, L.; Vicaș, S.I.; Brihan, I.; Domuța, M.; Zdrinca, M.; et al. Evaluation of Wound Healing Potential of Novel Hydrogel Based on Ocimum basilicum and Trifolium pratense Extracts. Processes 2021, 9, 2096. [Google Scholar] [CrossRef]
- Ezzat, S.M.; Choucry, M.A.; Kandil, Z.A. Antibacterial, antioxidant, and topical anti-inflammatory activities of Bergia ammannioides: A wound-healing plant. Pharm. Biol. 2016, 54, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Khalil, R.; Shata, A.; El-Kader, E.M.A.; Sharaf, H.; Abdo, W.S.; Amin, N.A.; Saber, S. Vildagliptin, a DPP-4 inhibitor, attenuates carbon tetrachloride-induced liver fibrosis by targeting ERK1/2, p38α, and NF-κB signaling. Toxicol. Appl. Pharmacol. 2020, 407, 115246. [Google Scholar] [CrossRef]
- Saber, S.; Youssef, M.E.; Sharaf, H.; Amin, N.A.; El-Shedody, R.; Aboutouk, F.H.; El-Galeel, Y.A.; El-Hefnawy, A.; Shabaka, D.; Khalifa, A.; et al. BBG enhances OLT1177-induced NLRP3 inflammasome inactivation by targeting P2X7R/NLRP3 and MyD88/NF-κB signaling in DSS-induced colitis in rats. Life Sci. 2021, 270, 119123. [Google Scholar] [CrossRef]
- El-Rous, M.A.; Saber, S.; Raafat, E.M.; Ahmed, A.A.E. Dapagliflozin, an SGLT2 inhibitor, ameliorates acetic acid-induced colitis in rats by targeting NFκB/AMPK/NLRP3 axis. Inflammopharmacology 2021, 29, 1169–1185. [Google Scholar] [CrossRef]
- Portou, M.J.; Baker, D.; Abraham, D.; Tsui, J. The innate immune system, toll-like receptors and dermal wound healing: A review. Vasc. Pharmacol. 2015, 71, 31–36. [Google Scholar] [CrossRef]
- Miere, F.; Teușdea, A.C.; Laslo, V.; Cavalu, S.; Fritea, L.; Dobjanschi, L.; Zdrinca, M.; Zdrinca, M.; Ganea, M.; Pașc, P.; et al. Evaluation of In Vitro Wound-Healing Potential, Antioxidant Capacity, and Antimicrobial Activity of Stellaria media (L.) Vill. Appl. Sci. 2021, 11, 11526. [Google Scholar] [CrossRef]
- Youssef, M.E.; El-Fattah, E.E.A.; Abdelhamid, A.M.; Eissa, H.; El-Ahwany, E.; Amin, N.A.; Hetta, H.F.; Mahmoud, M.H.; Batiha, G.E.-S.; Gobba, N.; et al. Interference with the AMPKα/mTOR/NLRP3 Signaling and the IL-23/IL-17 Axis Effectively Protects Against the Dextran Sulfate Sodium Intoxication in Rats: A New Paradigm in Empagliflozin and Metformin Reprofiling for the Management of Ulcerative Colitis. Front Pharmacol. 2021, 12, 719984. [Google Scholar] [CrossRef]
- Zohny, M.H.; Cavalu, S.; Youssef, M.E.; Kaddah, M.M.Y.; Mourad, A.A.E.; Gaafar, A.G.A.; El-Ahwany, E.; Amin, N.A.; Arakeep, H.M.; Shata, A.; et al. Coomassie brilliant blue G-250 dye attenuates bleomycin-induced lung fibrosis by regulating the NF-κB and NLRP3 crosstalk: A novel approach for filling an unmet medical need. Biomed. Pharmacother. 2022, 148, 112723. [Google Scholar] [CrossRef]
- Saber, S.; Abd El-Fattah, E.E.; Yahya, G.; Gobba, N.A.; Maghmomeh, A.O.; Khodir, A.E.; Mourad, A.A.; Saad, A.S.; Mohammed, H.G.; Nouh, N.A.; et al. A Novel Combination Therapy Using Rosuvastatin and Lactobacillus Combats Dextran Sodium Sulfate-Induced Colitis in High-Fat Diet-Fed Rats by Targeting the TXNIP/NLRP3 Interaction and Influencing Gut Microbiome Composition. Pharmaceuticals 2021, 14, 341. [Google Scholar] [CrossRef] [PubMed]
- Lafyatis, R.; Farina, A. New insights into the mechanisms of innate immune receptor signalling in fibrosis. Open Rheumatol. J. 2012, 6, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Midwood, K.S.; Yin, H.; Varga, J. Toll-Like Receptor-4 Signaling Drives Persistent Fibroblast Activation and Prevents Fibrosis Resolution in Scleroderma. Adv. Wound Care 2017, 6, 356–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saber, S.; Yahya, G.; Gobba, N.A.; Sharaf, H.; Alshaman, R.; Alattar, A.; Amin, N.A.; El-Shedody, R.; Aboutouk, F.H.; El-Galeel, Y.A.; et al. The Supportive Role of NSC328382, a P2X7R Antagonist, in Enhancing the Inhibitory Effect of CRID3 on NLRP3 Inflammasome Activation in Rats with Dextran Sodium Sulfate-Induced Colitis. J. Inflamm. Res. 2021, 14, 3443–3463. [Google Scholar] [CrossRef]
- Youssef, M.E.; El-Azab, M.F.; Abdel-Dayem, M.A.; Yahya, G.; Alanazi, I.S.; Saber, S. Electrocardiographic and histopathological characterizations of diabetic cardiomyopathy in rats. Environ. Sci. Pollut. Res. 2022, 29, 25723–25732. [Google Scholar] [CrossRef]
- Chen, L.; DiPietro, L.A. Toll-Like Receptor Function in Acute Wounds. Adv. Wound Care 2017, 6, 344–355. [Google Scholar] [CrossRef] [Green Version]
- Coban, Y.K. Infection control in severely burned patients. World J. Crit. Care Med. 2012, 1, 94–101. [Google Scholar] [CrossRef]
- Palmieri, T.L.; Greenhalgh, D.G. Topical Treatment of Pediatric Patients with Burns. Am. J. Clin. Dermatol. 2002, 3, 529–534. [Google Scholar] [CrossRef]
- Momtaz, S.; Dibaj, M.; Abdollahi, A.; Amin, G.; Bahramsoltani, R.; Abdollahi, M.; Mahdaviani, P.; Abdolghaffari, A.H. Wound healing activity of the flowers of Lilium candidum L. in burn wound model in rats. JMPIR 2020, 19, 109–118. [Google Scholar] [CrossRef]
- Manuel, R.G.; Fleuchot, B.; Lauciello, L.; Jafari, P.; Lee, A.A.; Raffoul, W.; Que, Y.-A.; Perron, K.; Blokesch, M. Effect of Human Burn Wound Exudate on Pseudomonas aeruginosa virulence. mSphere 2016, 1, e00111-15. [Google Scholar]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.-J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Adewuni, T.M.; Ademiluyi, A.O.; Olasehinde, T.A.; Ademosun, A.O. Phenolic Constituents and Inhibitory Effects of Hibiscus sabdariffa L. (Sorrel) Calyx on Cholinergic, Monoaminergic, and Purinergic Enzyme Activities. J. Diet. Suppl. 2018, 15, 910–922. [Google Scholar] [CrossRef] [PubMed]
- Jabeur, I.; Pereira, E.; Caleja, C.; Calhelha, R.C.; Soković, M.; Catarino, L.; Barros, L.; Ferreira, I. Exploring the chemical and bioactive properties of Hibiscus sabdariffa L. calyces from Guinea-Bissau (West Africa). Food Funct. 2019, 10, 2234–2243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdallah, E.M. Antibacterial efficiency of the Sudanese Roselle (Hibiscus sabdariffa L.), a famous beverage from Sudanese folk medicine. J. Intercult. Ethnopharmacol. 2016, 5, 186–190. [Google Scholar] [CrossRef]
- Abdallah, E.M. Antibacterial activity of Hibiscus sabdariffa L. calyces against hospital isolates of multidrug resistant Acinetobacter baumannii. J. Acute Dis. 2016, 5, 512–516. [Google Scholar] [CrossRef] [Green Version]
- Bhaskar, A.; Nithya, V. Evaluation of the wound-healing activity of Hibiscus rosa sinensis L. (Malvaceae) in Wistar albino rats. Indian J. Pharmacol. 2012, 44, 694–698. [Google Scholar] [CrossRef] [Green Version]
- Cranwell, P.B.; Harwood, L.M.; Moody, C.J. Experimental Organic Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Kodym, A.; Bujak, T. Physicochemical and microbiological properties as well as stability of ointments containing aloe extract (Aloe arborescens Mill.) or aloe extract associated to neomycin sulphate. Die Pharm. 2002, 57, 834–837. [Google Scholar]
- Fikru, A.; Makonnen, E.; Eguale, T.; Debella, A.; Mekonnen, G.A. Evaluation of in vivo wound healing activity of methanol extract of Achyranthes aspera L. J. Ethnopharmacol. 2012, 143, 469–474. [Google Scholar] [CrossRef]
- Cai, E.Z.; Ang, C.H.; Raju, A.; Tan, K.B.; Hing, E.C.; Loo, Y.; Wong, Y.C.; Lee, H.; Lim, J.; Moochhala, S.M.; et al. Creation of consistent burn wounds: A rat model. Arch. Plast. Surg. 2014, 41, 317–324. [Google Scholar] [CrossRef] [Green Version]
- Nasr, M.; Teiama, M.; Ismail, A.; Ebada, A.; Saber, S. In vitro and in vivo evaluation of cubosomal nanoparticles as an ocular delivery system for fluconazole in treatment of keratomycosis. Drug Deliv. Transl. Res. 2020, 10, 1841–1852. [Google Scholar] [CrossRef]
- El-Baz, A.M.; Yahya, G.; Mansour, B.; El-Sokkary, M.M.A.; Alshaman, R.; Alattar, A.; El-Ganiny, A.M. The Link between Occurrence of Class I Integron and Acquired Aminoglycoside Resistance in Clinical MRSA Isolates. Antibiotics 2021, 10, 488. [Google Scholar] [CrossRef] [PubMed]
- Yahya, G.; Ebada, A.; Khalaf, E.M.; Mansour, B.; Nouh, N.A.; Mosbah, R.A.; Saber, S.; Moustafa, M.; Negm, S.; El-Sokkary, M.M.A.; et al. Soil-Associated Bacillus Species: A Reservoir of Bioactive Compounds with Potential Therapeutic Activity against Human Pathogens. Microorganisms 2021, 9, 1131. [Google Scholar] [CrossRef] [PubMed]
- Riaz, G.; Chopra, R. A review on phytochemistry and therapeutic uses of Hibiscus sabdariffa L. Biomed. Pharmacother. 2018, 102, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Patry, J.; Blanchette, V. Enzymatic debridement with collagenase in wounds and ulcers: A systematic review and meta-analysis. Int. Wound J. 2017, 14, 1055–1065. [Google Scholar] [CrossRef]
- Gilligan, A.M.; Waycaster, C.R.; Bizier, R.; Chu, B.C.; Carter, M.J.; Fife, C.E. Comparative Effectiveness of Clostridial Collagenase Ointment to Medicinal Honey for Treatment of Pressure Ulcers. Adv. Wound Care 2017, 6, 125–134. [Google Scholar] [CrossRef] [Green Version]
- Zielins, E.R.; Brett, E.A.; Luan, A.; Hu, M.S.; Walmsley, G.G.; Paik, K.; Senarath-Yapa, K.; Atashroo, D.A.; Wearda, T.; Lorenz, H.P.; et al. Emerging drugs for the treatment of wound healing. Expert Opin. Emerg. Drugs 2015, 20, 235–246. [Google Scholar] [CrossRef]
- El-Fattah, E.E.A.; Saber, S.; Mourad, A.A.E.; El-Ahwany, E.; Amin, N.A.; Cavalu, S.; Yahya, G.; Saad, A.S.; Alsharidah, M.; Shata, A.; et al. The dynamic interplay between AMPK/NFκB signaling and NLRP3 is a new therapeutic target in inflammation: Emerging role of dapagliflozin in overcoming lipopolysaccharide-mediated lung injury. Biomed. Pharmacother. 2022, 147, 112628. [Google Scholar] [CrossRef]
- Saber, S.; Nasr, M.; Saad, A.S.; Mourad, A.A.E.; Gobba, N.A.; Shata, A.; Hafez, A.-M.; Elsergany, R.N.; Elagamy, H.I.; El-Ahwany, E.; et al. Albendazole-loaded cubosomes interrupt the ERK1/2-HIF-1α-p300/CREB axis in mice intoxicated with diethylnitrosamine: A new paradigm in drug repurposing for the inhibition of hepatocellular carcinoma progression. Biomed. Pharmacother. 2021, 142, 112029. [Google Scholar] [CrossRef]
- Wang, J.; Li, X.; Gao, L.; Wang, X. In vitro anti-inflammatory mechanism of Folium Hibisci Mutabilis leaves ethanol extracts, African journal of traditional, complementary, and alternative medicines. AJTCAM 2014, 11, 127–130. [Google Scholar]
- Shah, A.; Amini-Nik, S. The Role of Phytochemicals in the Inflammatory Phase of Wound Healing. Int. J. Mol. Sci. 2017, 18, 1068. [Google Scholar] [CrossRef] [Green Version]
- Finnerty, C.C.; Jeschke, M.G.; Branski, L.K.; Barret, J.P.; Dziewulski, P.; Herndon, D.N. Hypertrophic scarring: The greatest unmet challenge after burn injury. Lancet 2016, 388, 1427–1436. [Google Scholar] [CrossRef] [Green Version]
- Larsson, J.; Goumans, M.J.; Sjöstrand, L.J.; van Rooijen, M.A.; Ward, D.; Levéen, P.; Xu, X.; Dijke, P.t.; Mummery, C.L.; Karlsson, S. Abnormal angiogenesis but intact hematopoietic potential in TGF-beta type I receptor-deficient mice. EMBO J. 2001, 20, 1663–1673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.I.; Kwak, J.H.; Na, H.J.; Kim, J.K.; Ding, Y.; Choi, M.E. Transforming growth factor-beta (TGF-beta1) activates TAK1 via TAB1-mediated autophosphorylation, independent of TGF-beta receptor kinase activity in mesangial cells. J. Biol. Chem. 2009, 284, 22285–22296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Da-Costa-Rocha, I.; Bonnlaender, B.; Sievers, H.; Pischel, I.; Heinrich, M. Hibiscus sabdariffa L.—A phytochemical and pharmacological review. Food Chem. 2014, 165, 424–443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Süntar, I.; Akkol, E.K.; Nahar, L.; Sarker, S.D. Wound healing and antioxidant properties: Do they coexist in plants? Free. Radic. Antioxid. 2012, 2, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Frank, T.; Netzel, G.; Kammerer, D.R.; Carle, R.; Kler, A.; Kriesl, E.; Bitsch, I.; Bitsch, R.; Netzel, M. Consumption of Hibiscus sabdariffa L. aqueous extract and its impact on systemic antioxidant potential in healthy subjects. J. Sci. Food Agric. 2012, 92, 2207–2218. [Google Scholar] [CrossRef] [PubMed]
- Florina, M.; Luminița, F.; Simona, C.; Ioana, V.S. Formulation, Characterization, and Advantages of Using Liposomes in Multiple Therapies. Pharmacophore 2020, 11, 1–12. [Google Scholar]
- Peng, C.H.; Chyau, C.C.; Chan, K.C.; Chan, T.H.; Wang, C.J.; Huang, C.N. Hibiscus sabdariffa polyphenolic extract inhibits hyperglycemia, hyperlipidemia, and glycation-oxidative stress while improving insulin resistance. J. Agric. Food Chem. 2011, 59, 9901–9909. [Google Scholar] [CrossRef]
- Bakhtiari, E.; Hosseini, A.; Mousavi, S.H. Protective effect of Hibiscus sabdariffa against serum/glucose deprivation-induced PC12 cells injury. Avicenna J. Phytomedicine 2015, 5, 231–237. [Google Scholar]
- Guardiola, S.; Mach, N. (Therapeutic potential of Hibiscus sabdariffa: A review of the scientific evidence), Endocrinologia y nutricion. Organo Soc. Esp. Endocrinol. Nutr. 2014, 61, 274–295. [Google Scholar] [CrossRef]
- Bielecki, P.; Glik, J.; Kawecki, M.; Santos, V.A.M.d. Towards understanding Pseudomonas aeruginosa burn wound infections by profiling gene expression. Biotechnol. Lett. 2008, 30, 777–790. [Google Scholar] [CrossRef] [PubMed]
- Márquez-Rodríguez, A.S.; Nevárez-Baca, S.; Lerma-Hernández, J.C.; Hernández-Ochoa, L.R.; Nevárez-Moorillon, G.V.; Gutiérrez-Méndez, N.; Muñoz-Castellanos, L.N.; Salas, E. In Vitro Antibacterial Activity of Hibiscus sabdariffa L. Phenolic Extract and Its in Situ Application on Shelf-Life of Beef Meat. Foods 2020, 9, 1080. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalil, R.; Yahya, G.; Abdo, W.S.; El-Tanbouly, G.S.; Johar, D.; Abdel-Halim, M.S.; Eissa, H.; Magheru, C.; Saber, S.; Cavalu, S. Emerging Approach for the Application of Hibiscus sabdariffa Extract Ointment in the Superficial Burn Care. Sci. Pharm. 2022, 90, 41. https://doi.org/10.3390/scipharm90030041
Khalil R, Yahya G, Abdo WS, El-Tanbouly GS, Johar D, Abdel-Halim MS, Eissa H, Magheru C, Saber S, Cavalu S. Emerging Approach for the Application of Hibiscus sabdariffa Extract Ointment in the Superficial Burn Care. Scientia Pharmaceutica. 2022; 90(3):41. https://doi.org/10.3390/scipharm90030041
Chicago/Turabian StyleKhalil, Rania, Galal Yahya, Walied S. Abdo, Ghada S. El-Tanbouly, Dina Johar, Mahmoud Saad Abdel-Halim, Hanan Eissa, Calin Magheru, Sameh Saber, and Simona Cavalu. 2022. "Emerging Approach for the Application of Hibiscus sabdariffa Extract Ointment in the Superficial Burn Care" Scientia Pharmaceutica 90, no. 3: 41. https://doi.org/10.3390/scipharm90030041
APA StyleKhalil, R., Yahya, G., Abdo, W. S., El-Tanbouly, G. S., Johar, D., Abdel-Halim, M. S., Eissa, H., Magheru, C., Saber, S., & Cavalu, S. (2022). Emerging Approach for the Application of Hibiscus sabdariffa Extract Ointment in the Superficial Burn Care. Scientia Pharmaceutica, 90(3), 41. https://doi.org/10.3390/scipharm90030041







