Cinnamaldehyde Relieves Induced Hepatocellular Carcinoma in Rat Model via Targeting Wnt/β-Catenin Pathway
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
2.3. Experimental Design
2.4. Sample Collection
2.5. Assessed Parameters
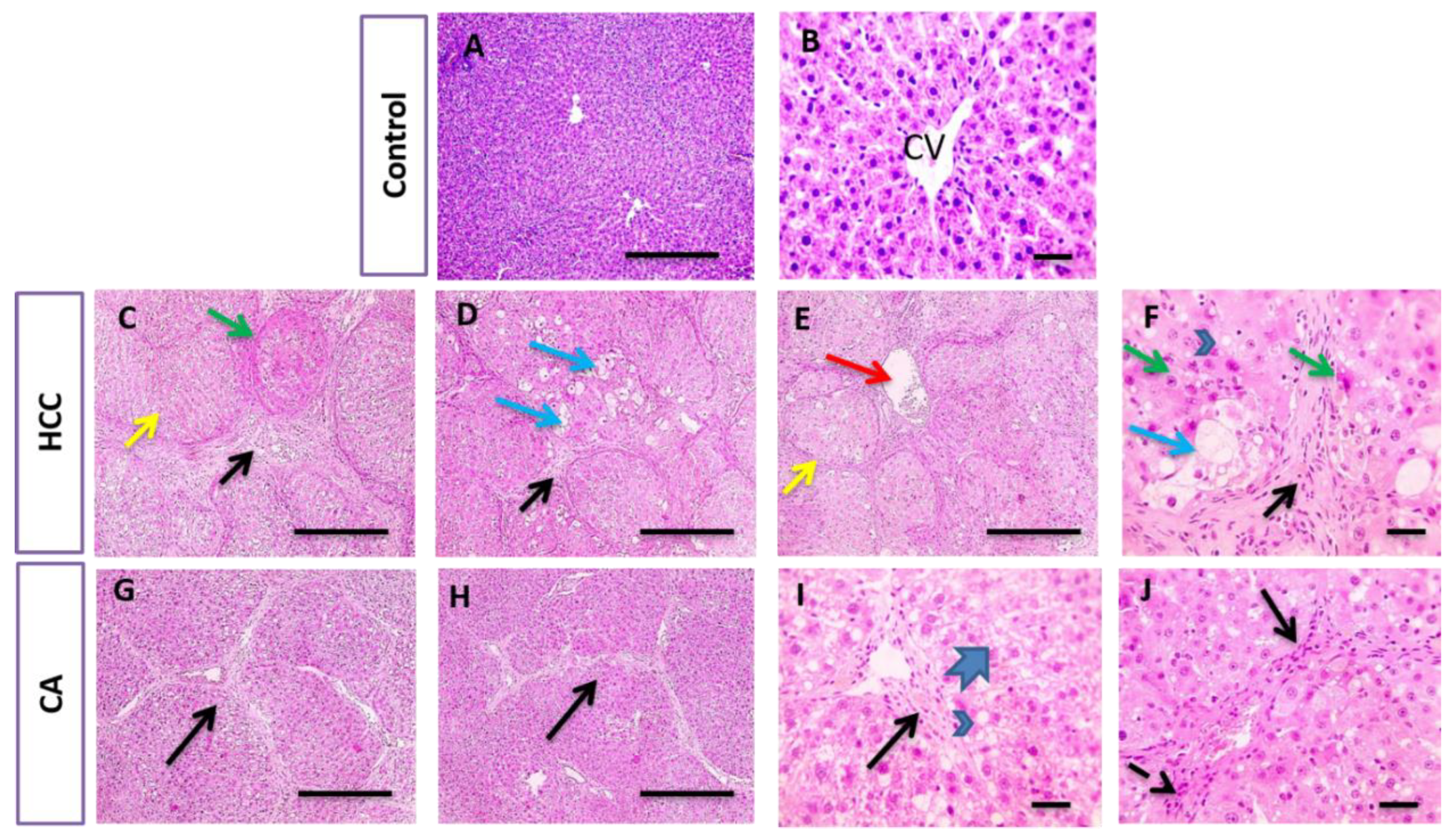
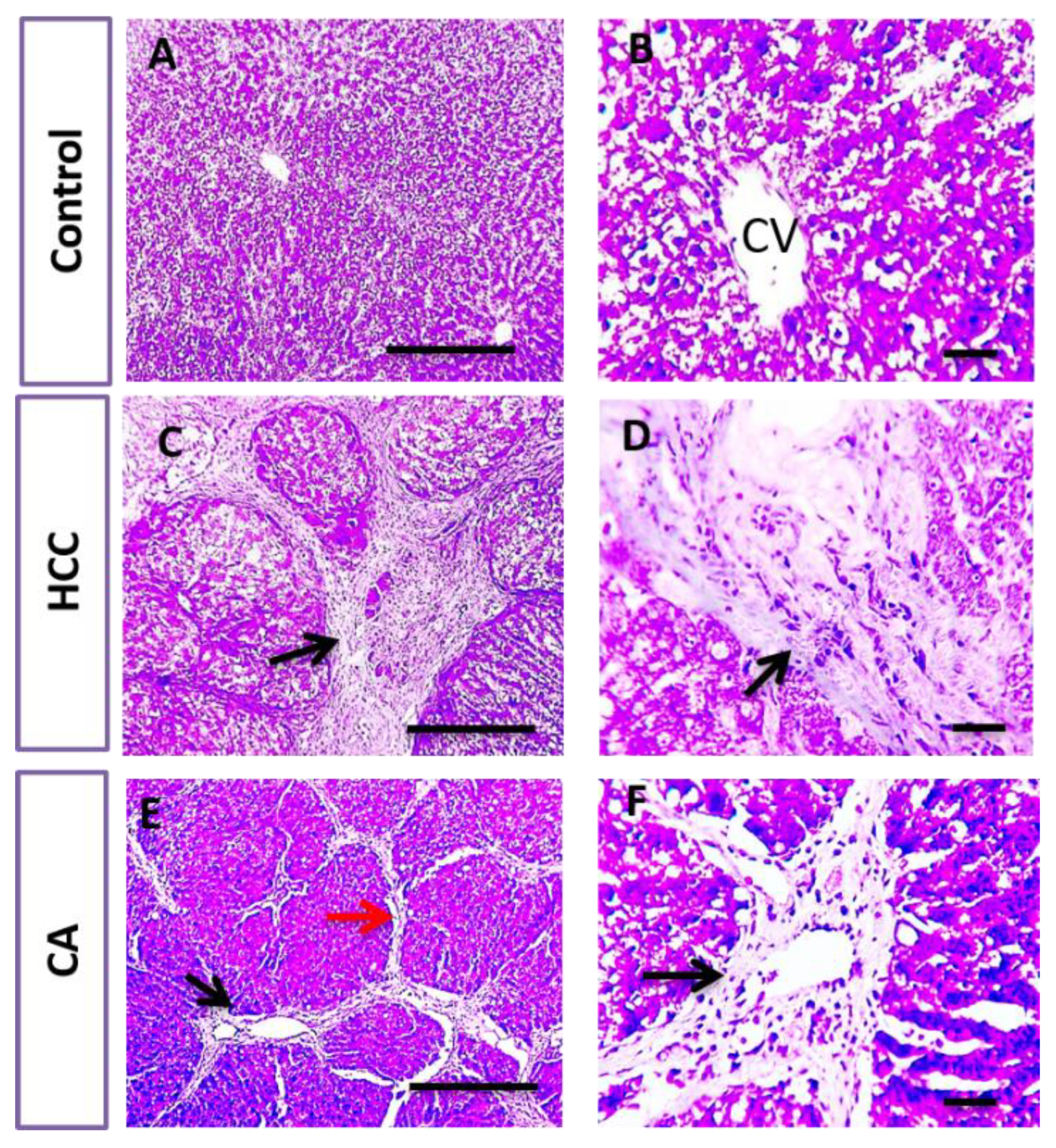
2.6. Histopathological Examination of Liver Tissue
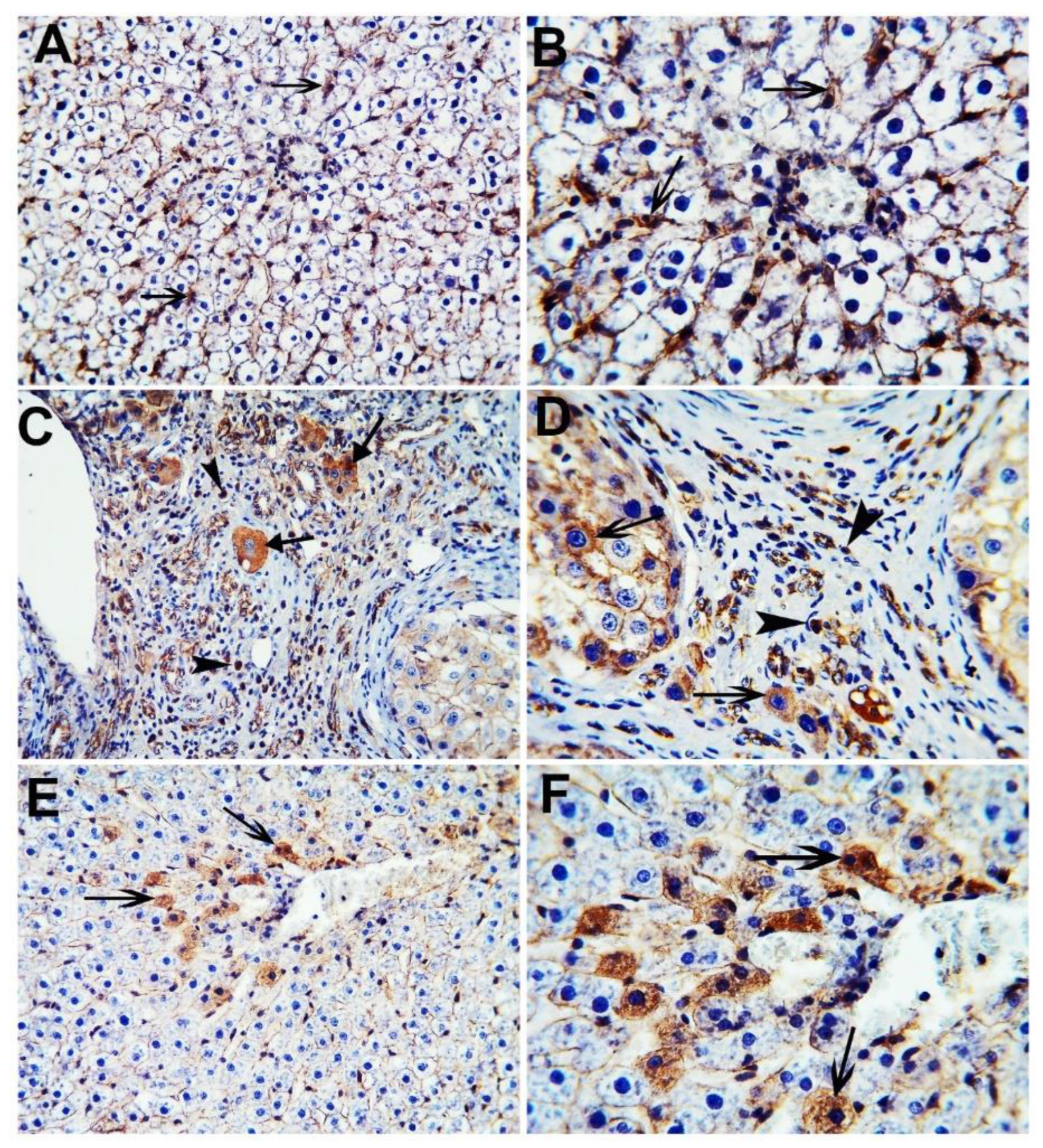
2.7. Immunohistochemistry (IHC)
2.8. Statistical Analysis
3. Results
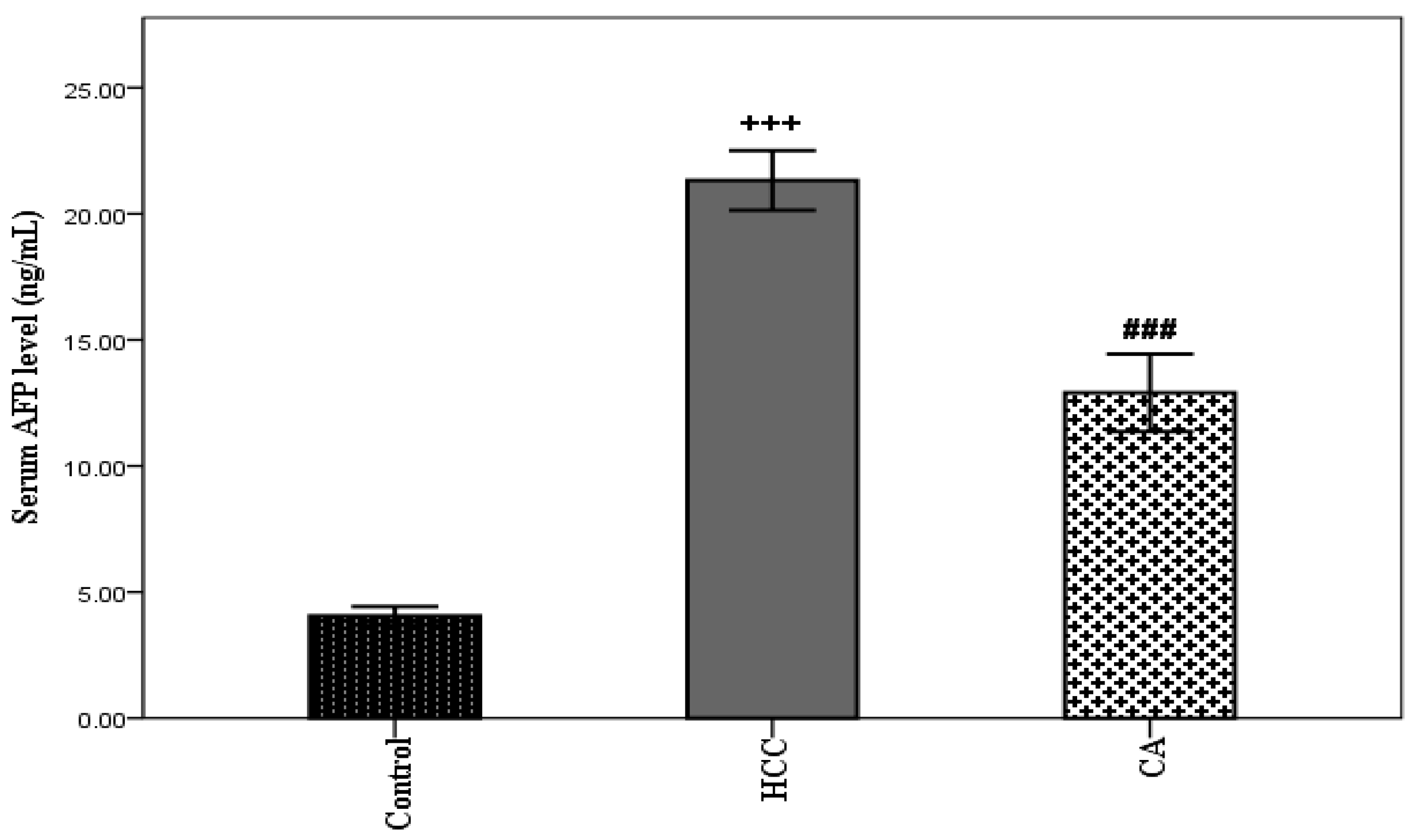
3.1. Effect of CA on Liver Function
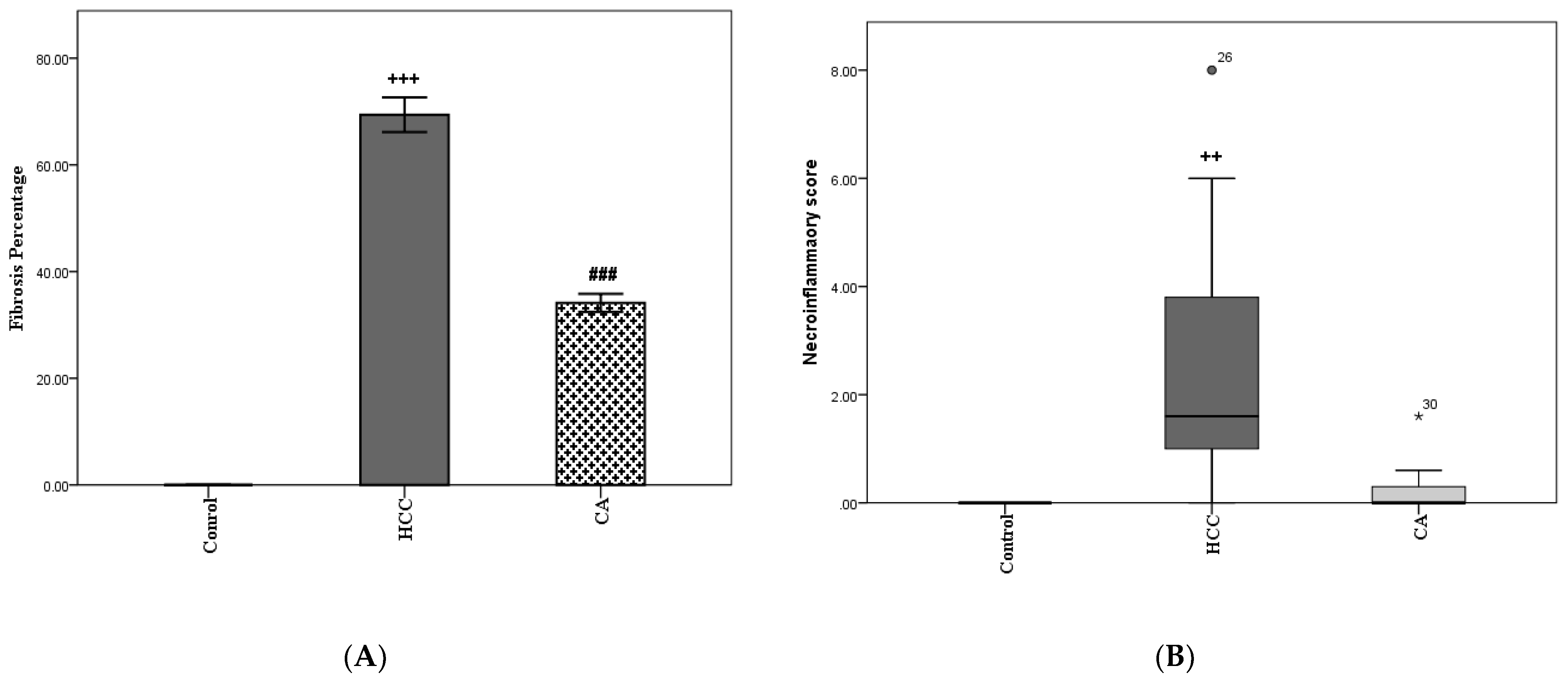
3.2. CA Decreases Fibrosis Percentage and Necroinflammatory Scores in Liver Homogenates
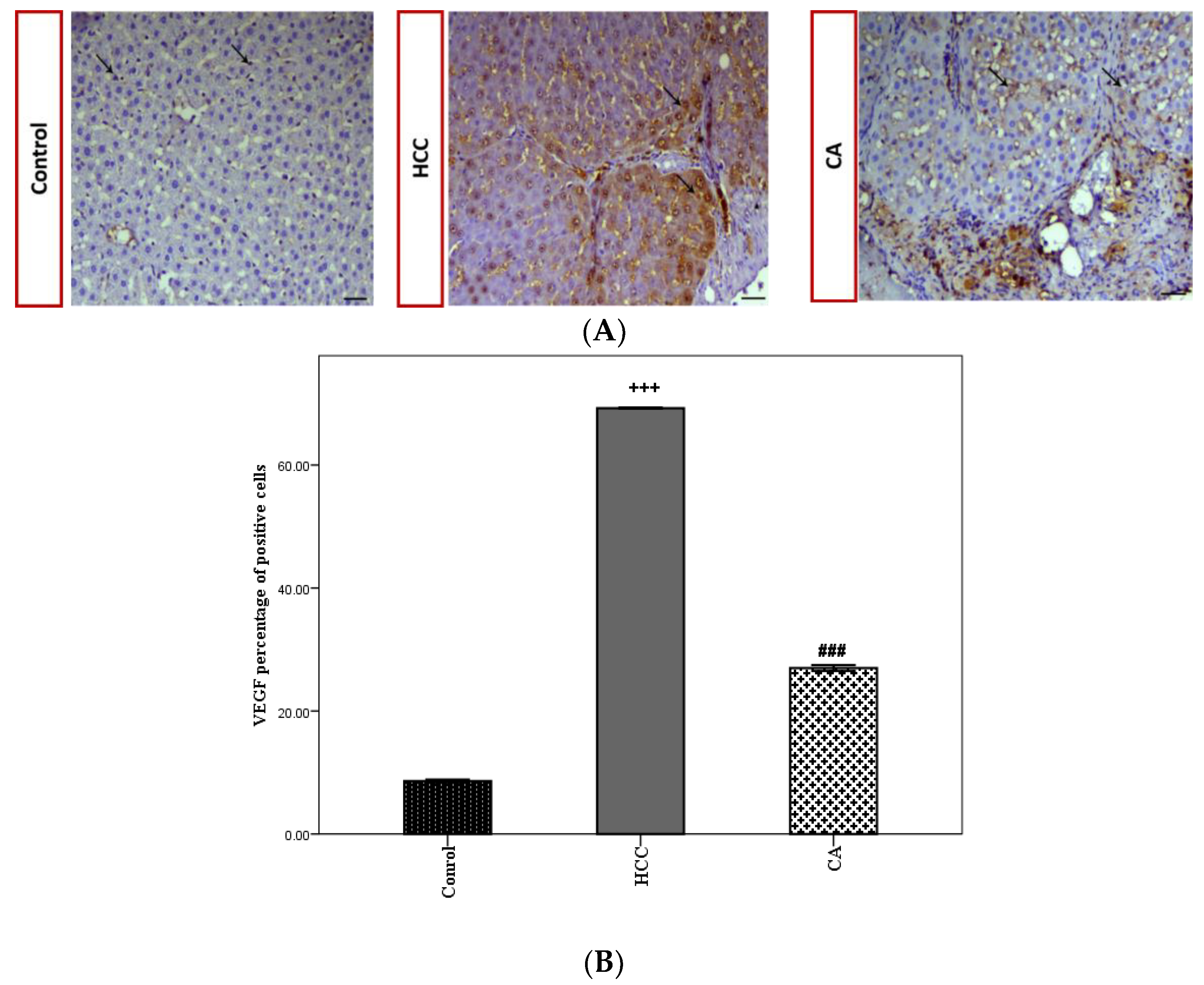
3.3. CA Attenuates TAA Induced HCC
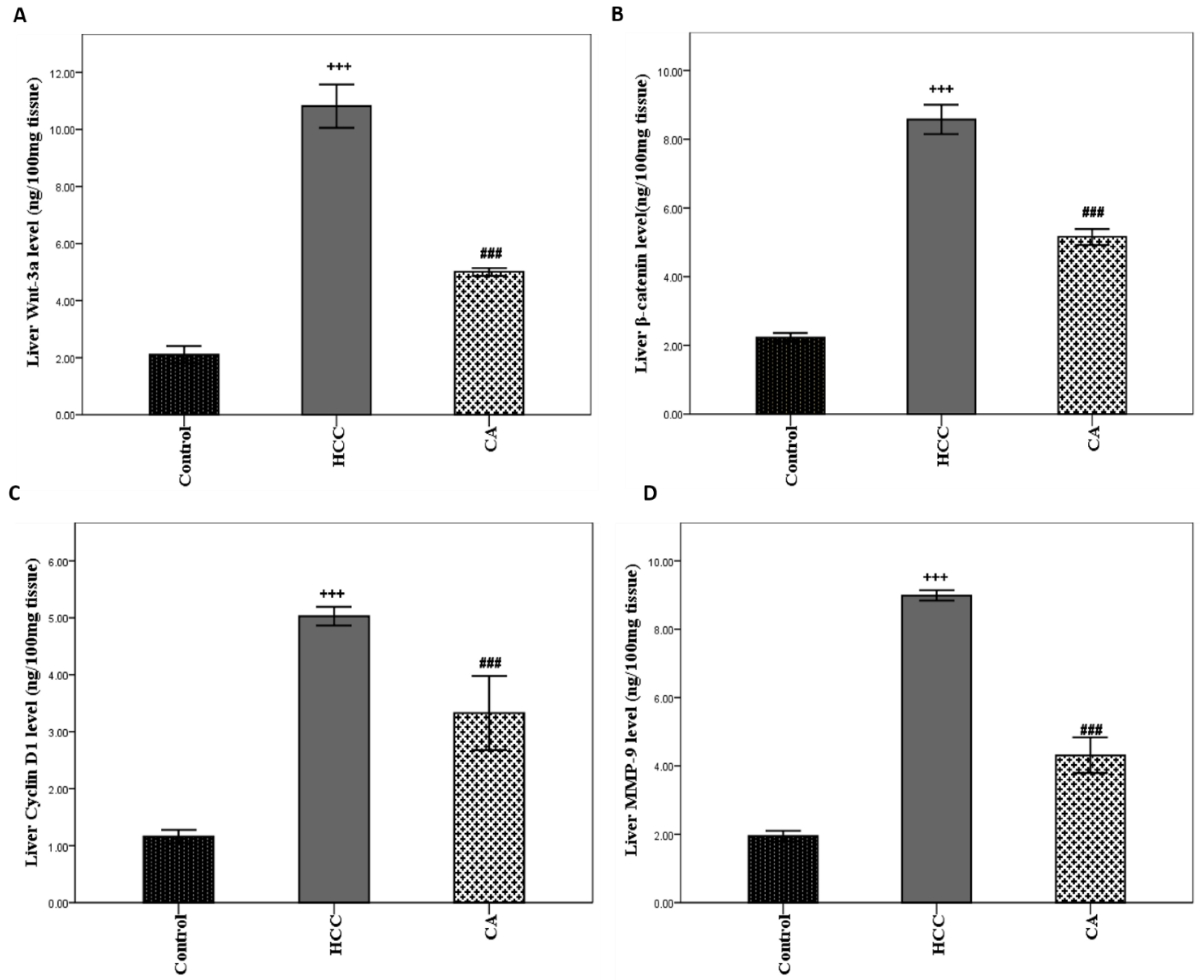
3.4. CA Inhibits Wnt/β-Catenin Signaling Pathway
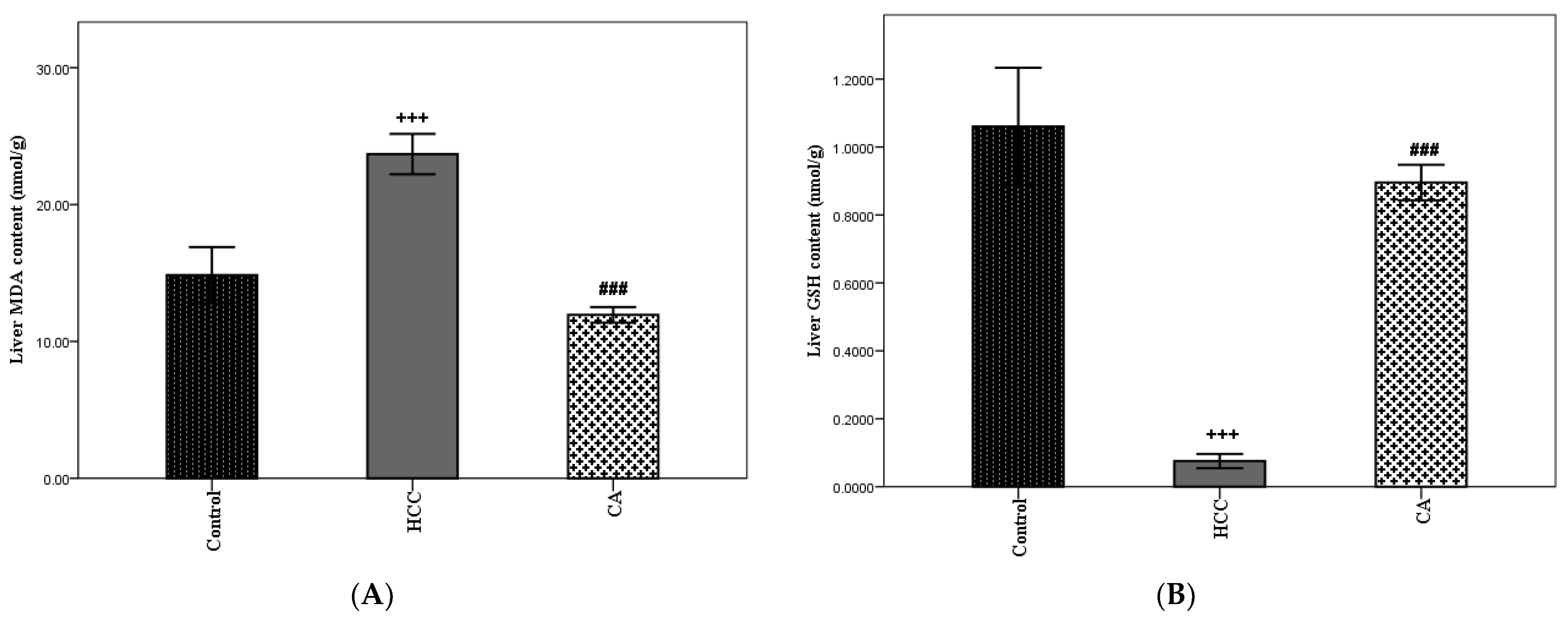
3.5. Antioxidant Effect of CA
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, J.D.; Hainaut, P.; Gores, G.J.; Amadou, A.; Plymoth, A.; Roberts, L.R. A global view of hepatocellular carcinoma: Trends, risk, prevention and management. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 589–604. [Google Scholar] [CrossRef] [PubMed]
- Khalaf, A.M.; Fuentes, D.; Morshid, A.I.; Burke, M.R.; Kaseb, A.O.; Hassan, M.; Hazle, J.D.; Elsayes, K.M. Role of Wnt/β-catenin signaling in hepatocellular carcinoma, pathogenesis, and clinical significance. J. Hepatocell. Carcinoma 2018, 5, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Wu, Y.; Awadasseid, A.; Tanaka, Y.; Zhang, W. New Advances in Canonical Wnt/β-Catenin Signaling in Cancer. Cancer Manag. Res. 2020, 12, 6987–6998. [Google Scholar] [CrossRef] [PubMed]
- Chua, M.-S.; Ma, L.; Wei, W.; So, S. WNT/β-catenin pathway activation in hepatocellular carcinoma: A clinical perspective. Gastrointest. Cancer Targets Ther. 2014, 4, 49–63. [Google Scholar] [CrossRef]
- Mohammed, M.K.; Shao, C.; Wang, J.; Wei, Q.; Wang, X.; Collier, Z.; Tang, S.; Liu, H.; Zhang, F.; Huang, J.; et al. Wnt/β-catenin signaling plays an ever-expanding role in stem cell self-renewal, tumorigenesis and cancer chemoresistance. Genes Dis. 2016, 3, 11–40. [Google Scholar] [CrossRef]
- Komiya, Y.; Habas, R. Wnt signal transduction pathways. Organogenesis 2008, 4, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Wong, H.-C.; Bourdelas, A.; Krauss, A.; Lee, H.-J.; Shao, Y.; Wu, D.; Mlodzik, M.; Shi, D.-L.; Zheng, J. Direct binding of the PDZ domain of Dishevelled to a conserved internal sequence in the C-terminal region of Frizzled. Mol. Cell 2003, 12, 1251–1260. [Google Scholar] [CrossRef]
- Yang, K.; Wang, X.; Zhang, H.; Wang, Z.; Nan, G.; Li, Y.; Zhang, F.; Mohammed, M.K.; Haydon, R.C.; Luu, H.H.; et al. The evolving roles of canonical WNT signaling in stem cells and tumorigenesis: Implications in targeted cancer therapies. Lab Invest. 2016, 96, 116–136. [Google Scholar] [CrossRef] [PubMed]
- Nejak-Bowen, K.N.; Monga, S.P.S. Beta-catenin signaling, liver regeneration and hepatocellular cancer: Sorting the good from the bad. Semin. Cancer Biol. 2011, 21, 44–58. [Google Scholar] [CrossRef]
- Vilchez, V.; Turcios, L.; Marti, F.; Gedaly, R. Targeting Wnt/β-catenin pathway in hepatocellular carcinoma treatment. World J. Gastroenterol. 2016, 22, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Logan, C.Y.; Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.O.; Monga, S.P. Wnt/β-Catenin Signaling in Liver Development, Homeostasis, and Pathobiology. Annu. Rev. Pathol. 2018, 13, 351–378. [Google Scholar] [CrossRef] [PubMed]
- Mandlik, D.S.; Mandlik, S.K. Herbal and Natural Dietary Products: Upcoming Therapeutic Approach for Prevention and Treatment of Hepatocellular Carcinoma. Nutr. Cancer 2020, 73, 2130–2154. [Google Scholar] [CrossRef] [PubMed]
- Cocchiara, J.; Letizia, C.S.; Lalko, J.; Lapczynski, A.; Api, A.M. Fragrance material review on cinnamaldehyde. Food Chem. Toxicol. 2005, 43, 867–923. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Chen, J.; Wang, K.; Geng, Y.; Lai, W.; Huang, X.; Chen, D.; Guo, H.; Fang, J.; Chen, Z.; et al. Study the antibacterial mechanism of cinnamaldehyde against drug-resistant Aeromonas hydrophila in vitro. Microb. Pathog. 2020, 145, 104208. [Google Scholar] [CrossRef] [PubMed]
- Doyle, A.A.; Stephens, J.C. A review of cinnamaldehyde and its derivatives as antibacterial agents. Fitoterapia 2019, 139, 104405. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.C.; Deng, J.S.; Chiu, C.S.; Hou, W.C.; Huang, S.S.; Shie, P.H.; Huang, G.J. Anti-Inflammatory Activities of Cinnamomum cassia Constituents In Vitro and In Vivo. Evid. Based Complement. Alternat. Med. 2012, 2012, 429320. [Google Scholar] [CrossRef]
- Lin, L.-T.; Wu, S.-J.; Lin, C.-C. The Anticancer Properties and Apoptosis-inducing Mechanisms of Cinnamaldehyde and the Herbal Prescription Huang-Lian-Jie-Du-Tang ( Huáng Lián Jiě Dú Tang) in Human Hepatoma Cells. J. Tradit. Complement. Med. 2013, 3, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Mateen, S.; Rehman, M.T.; Shahzad, S.; Naeem, S.S.; Faizy, A.F.; Khan, A.Q.; Khan, M.S.; Husain, F.M.; Moin, S. Anti-oxidant and anti-inflammatory effects of cinnamaldehyde and eugenol on mononuclear cells of rheumatoid arthritis patients. Eur. J. Pharm. 2019, 852, 14–24. [Google Scholar] [CrossRef]
- Elewa, M.A.; Al-Gayyar, M.M.; Schaalan, M.F.; Abd El Galil, K.H.; Ebrahim, M.A.; El-Shishtawy, M.M. Hepatoprotective and anti-tumor effects of targeting MMP-9 in hepatocellular carcinoma and its relation to vascular invasion markers. Clin. Exp. Metastasis 2015, 32, 479–493. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhuang, Y.; Jiang, S.; Tian, F.; Teng, Y.; Chen, X.; Zheng, P.; Liu, S.; Zhou, J.; Wu, J.; et al. Cinnamaldehyde induces apoptosis and reverses epithelial-mesenchymal transition through inhibition of Wnt/β-catenin pathway in non-small cell lung cancer. Int. J. Biochem. Cell Biol. 2017, 84, 58–74. [Google Scholar] [CrossRef] [PubMed]
- Ishak, K.; Baptista, A.; Bianchi, L.; Callea, F.; De Groote, J.; Gudat, F.; Denk, H.; Desmet, V.; Korb, G.; MacSween, R.N.M.; et al. Histological grading and staging of chronic hepatitis. J. Hepatol. 1995, 22, 696–699. [Google Scholar] [CrossRef]
- Saber, S.; Khalil, R.M.; Abdo, W.S.; Nassif, D.; El-Ahwany, E. Olmesartan ameliorates chemically-induced ulcerative colitis in rats via modulating NFκB and Nrf-2/HO-1 signaling crosstalk. Toxicol. Appl. Pharmacol. 2019, 364, 120–132. [Google Scholar] [CrossRef]
- Liu, Y.; An, T.; Wan, D.; Yu, B.; Fan, Y.; Pei, X. Targets and Mechanism Used by Cinnamaldehyde, the Main Active Ingredient in Cinnamon, in the Treatment of Breast Cancer. Front. Pharmacol. 2020, 11, 582719. [Google Scholar] [CrossRef]
- Han, L.; Mei, J.; Ma, J.; Wang, F.; Gu, Z.; Li, J.; Zhang, Z.; Zeng, Y.; Lou, X.; Yao, X.; et al. Cinnamaldehyde induces endogenous apoptosis of the prostate cancer-associated fibroblasts via interfering the Glutathione-associated mitochondria function. Med. Oncol. 2020, 37, 91. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-e.; Zhuang, Y.-w.; Zhou, J.-y.; Liu, S.-l.; Wang, R.-p.; Shu, P. Cinnamaldehyde enhances apoptotic effect of oxaliplatin and reverses epithelial-mesenchymal transition and stemnness in hypoxic colorectal cancer cells. Exp. Cell Res. 2019, 383, 111500. [Google Scholar] [CrossRef] [PubMed]
- Ka, H.; Park, H.-J.; Jung, H.-J.; Choi, J.-W.; Cho, K.-S.; Ha, J.; Lee, K.-T. Cinnamaldehyde induces apoptosis by ROS-mediated mitochondrial permeability transition in human promyelocytic leukemia HL-60 cells. Cancer Lett. 2003, 196, 143–152. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, J.; Yang, S.; Tan, T.; Wang, N.; Wang, Y.; Zhang, L.; Yang, C.; Huang, H.; Luo, J.; et al. Cinnamaldehyde Inhibits the Function of Osteosarcoma by Suppressing the Wnt/β-Catenin and PI3K/Akt Signaling Pathways. Drug Des. Dev. Ther. 2020, 14, 4625–4637. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.-J.; Xie, S.-X.; Chen, Y.-T.; Xue, J.-L.; Zhang, C.-J.; Zhu, F. Aberrant regulation of Wnt signaling in hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 7486–7499. [Google Scholar] [CrossRef]
- Lu, C.; He, Y.; Duan, J.; Yang, Y.; Zhong, C.; Zhang, J.; Liao, W.; Huang, X.; Zhu, R.; Li, M. Expression of Wnt3a in hepatocellular carcinoma and its effects on cell cycle and metastasis. Int. J. Oncol. 2017, 51, 1135–1145. [Google Scholar] [CrossRef]
- Pashirzad, M.; Fiuji, H.; Khazei, M.; Moradi-Binabaj, M.; Ryzhikov, M.; Shabani, M.; Avan, A.; Hassanian, S.M. Role of Wnt3a in the pathogenesis of cancer, current status and prospective. Mol. Biol. Rep. 2019, 46, 5609–5616. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Yao, M.; Zheng, W.; Gu, J.; Yang, X.; Qiu, L.; Cai, Y.; Wu, W.; Yao, D. Abnormality of Wnt3a expression as novel specific biomarker for diagnosis and differentiation of hepatocellular carcinoma. Tumor Biol. 2016, 37, 5561–5568. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.-H.; Yao, M.; Cai, Y.; Gu, J.-J.; Yang, X.-L.; Wang, L.; Yao, D.-F. Oncogenic Wnt3a expression as an estimable prognostic marker for hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 3829–3836. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Yao, M.; Fang, M.; Pan, L.; Wang, L.; Yang, J.; Dong, Z.; Yao, D. Oncogenic Wnt3a: A Candidate Specific Marker and Novel Molecular Target for Hepatocellular Carcinoma. J. Cancer 2019, 10, 5862–5873. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Fang, M.; Zheng, W.-J.; Yao, D.-F. Oncogenic Wnt3a: A promising specific biomarker in hepatocellular carcinoma. Hepatoma Res. 2018, 4, 30. [Google Scholar] [CrossRef][Green Version]
- Deane, N.G.; Parker, M.A.; Aramandla, R.; Diehl, L.; Lee, W.J.; Washington, M.K.; Nanney, L.B.; Shyr, Y.; Beauchamp, R.D. Hepatocellular carcinoma results from chronic cyclin D1 overexpression in transgenic mice. Cancer Res. 2001, 61, 5389–5395. [Google Scholar] [PubMed]
- Joo, M.; Kang, Y.K.; Kim, M.R.; Lee, H.K.; Jang, J.J. Cyclin D1 overexpression in hepatocellular carcinoma. Liver 2001, 21, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Anna, C.H.; Iida, M.; Sills, R.C.; Devereux, T.R. Expression of potential beta-catenin targets, cyclin D1, c-Jun, c-Myc, E-cadherin, and EGFR in chemically induced hepatocellular neoplasms from B6C3F1 mice. Toxicol. Appl. Pharmacol. 2003, 190, 135–145. [Google Scholar] [CrossRef]
- Qu, B.; Liu, B.-R.; Du, Y.-J.; Chen, J.; Cheng, Y.-Q.; Xu, W.; Wang, X.-H. Wnt/β-catenin signaling pathway may regulate the expression of angiogenic growth factors in hepatocellular carcinoma. Oncol. Lett. 2014, 7, 1175–1178. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.J.; Wang, N.; Sun, Z.W.; Chen, J.; Cui, H.W. MiR-5692a promotes the invasion and metastasis of hepatocellular carcinoma via MMP9. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 4869–4878. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Guo, R.P.; Shi, M.; Wei, W.; Yu, W.S.; Li, J.Q. Expression and clinical significance of VEGF and MMP-9 in hepatocellular carcinoma. Ai Zheng Aizheng Chin. J. Cancer 2006, 25, 599–603. [Google Scholar]
- Atta, M.M.; Atta, H.M.; Gad, M.A.; Rashed, L.A.; Said, E.M.; Hassanien Sel, S.; Kaseb, A.O. Clinical significance of vascular endothelial growth factor in hepatitis C related hepatocellular carcinoma in Egyptian patients. J. Hepatocell. Carcinoma 2016, 3, 19–24. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cao, M.Q.; You, A.B.; Cui, W.; Zhang, S.; Guo, Z.G.; Chen, L.; Zhu, X.D.; Zhang, W.; Zhu, X.L.; Guo, H.; et al. Cross talk between oxidative stress and hypoxia via thioredoxin and HIF-2α drives metastasis of hepatocellular carcinoma. FASEB J. 2020, 34, 5892–5905. [Google Scholar] [CrossRef] [PubMed]
| Group | ALT Activity (IU/L) | AST Activity (IU/L) | ALP Activity (IU/L) | GGT Activity (IU/L) | Total Bilirubin (mg/dL) | Albumin (g/dL) | Total Protein (g/dL) |
|---|---|---|---|---|---|---|---|
| Control | 46.60 ± 2.50 | 123.80 ± 4.60 | 311.60 ± 23.653 | 6.60 ± 0.81 | 0.24 ± 0.03 | 3.98 ± 0.086 | 7.40 ± 0.27 |
| HCC | 396.60 ± 7.83 +++ | 503.60 ± 3.80 +++ | 743.60 ± 12.002 +++ | 29.80 ± 0.735 +++ | 1.20 ± 0.084 +++ | 2.42 ± 0.037 +++ | 3.94 ± 0.093 +++ |
| CA | 282.17 ± 27.83 ## | 377.00 ± 16.93 ### | 593.67 ± 19.178 ### | 21.83 ± 0.477 ### | 0.72 ± 0.05 ### | 2.98 ± 0.048 # | 5.67 ± 0.17 ### |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd El Salam, A.S.G.; Samra, Y.A.; El-Shishtawy, M.M. Cinnamaldehyde Relieves Induced Hepatocellular Carcinoma in Rat Model via Targeting Wnt/β-Catenin Pathway. Sci. Pharm. 2022, 90, 22. https://doi.org/10.3390/scipharm90020022
Abd El Salam ASG, Samra YA, El-Shishtawy MM. Cinnamaldehyde Relieves Induced Hepatocellular Carcinoma in Rat Model via Targeting Wnt/β-Catenin Pathway. Scientia Pharmaceutica. 2022; 90(2):22. https://doi.org/10.3390/scipharm90020022
Chicago/Turabian StyleAbd El Salam, Al Shaima G., Yara A. Samra, and Mamdouh M. El-Shishtawy. 2022. "Cinnamaldehyde Relieves Induced Hepatocellular Carcinoma in Rat Model via Targeting Wnt/β-Catenin Pathway" Scientia Pharmaceutica 90, no. 2: 22. https://doi.org/10.3390/scipharm90020022
APA StyleAbd El Salam, A. S. G., Samra, Y. A., & El-Shishtawy, M. M. (2022). Cinnamaldehyde Relieves Induced Hepatocellular Carcinoma in Rat Model via Targeting Wnt/β-Catenin Pathway. Scientia Pharmaceutica, 90(2), 22. https://doi.org/10.3390/scipharm90020022






