Purple Corn Silk Extract Attenuates UVB-Induced Inflammation in Human Keratinocyte Cells
Abstract
:1. Introduction
2. Results
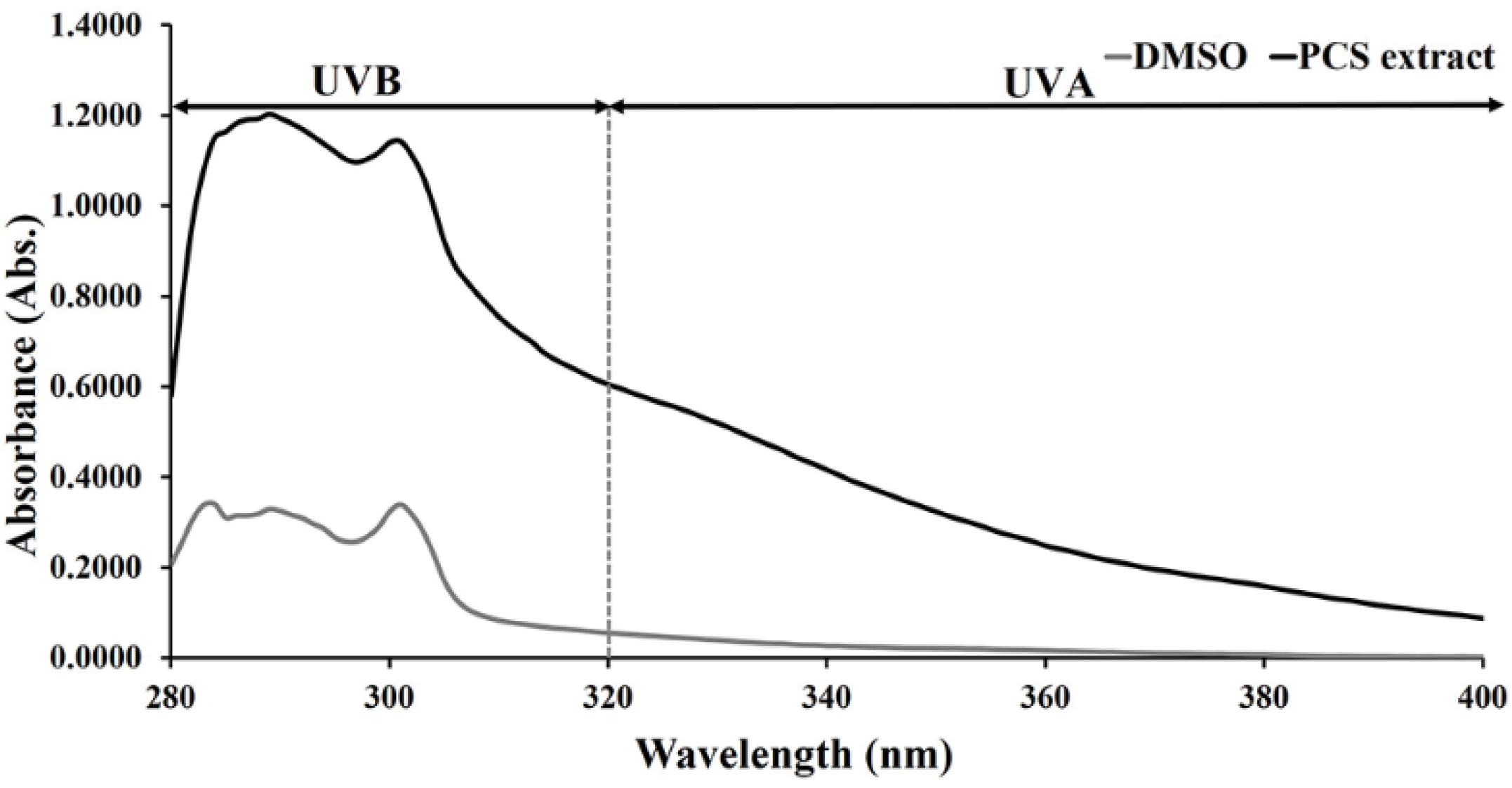
2.1. UV-Absorbing Properties of PCS Extract
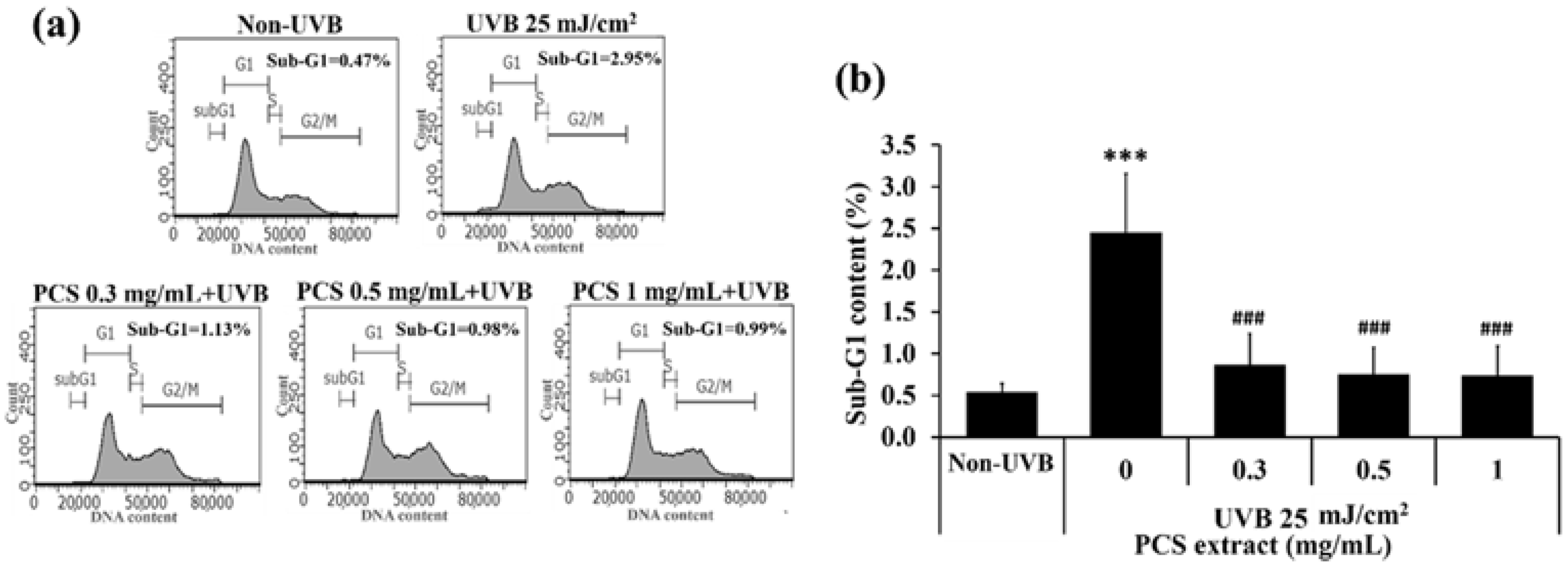
2.2. PCS Extract Attenuated UVB-Induced Cell Death in HaCaT Cells
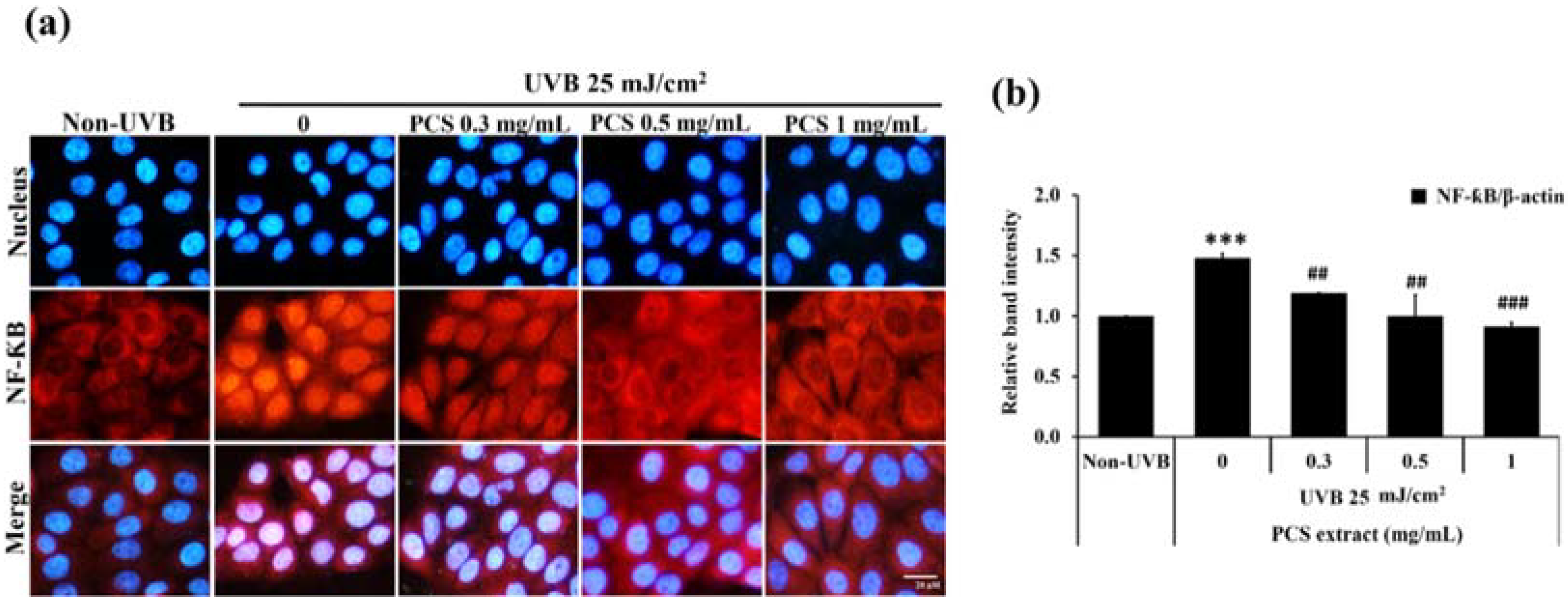
2.3. PCS Extract Decreases NF-κB Activity in UVB-Treated HaCaT Cells
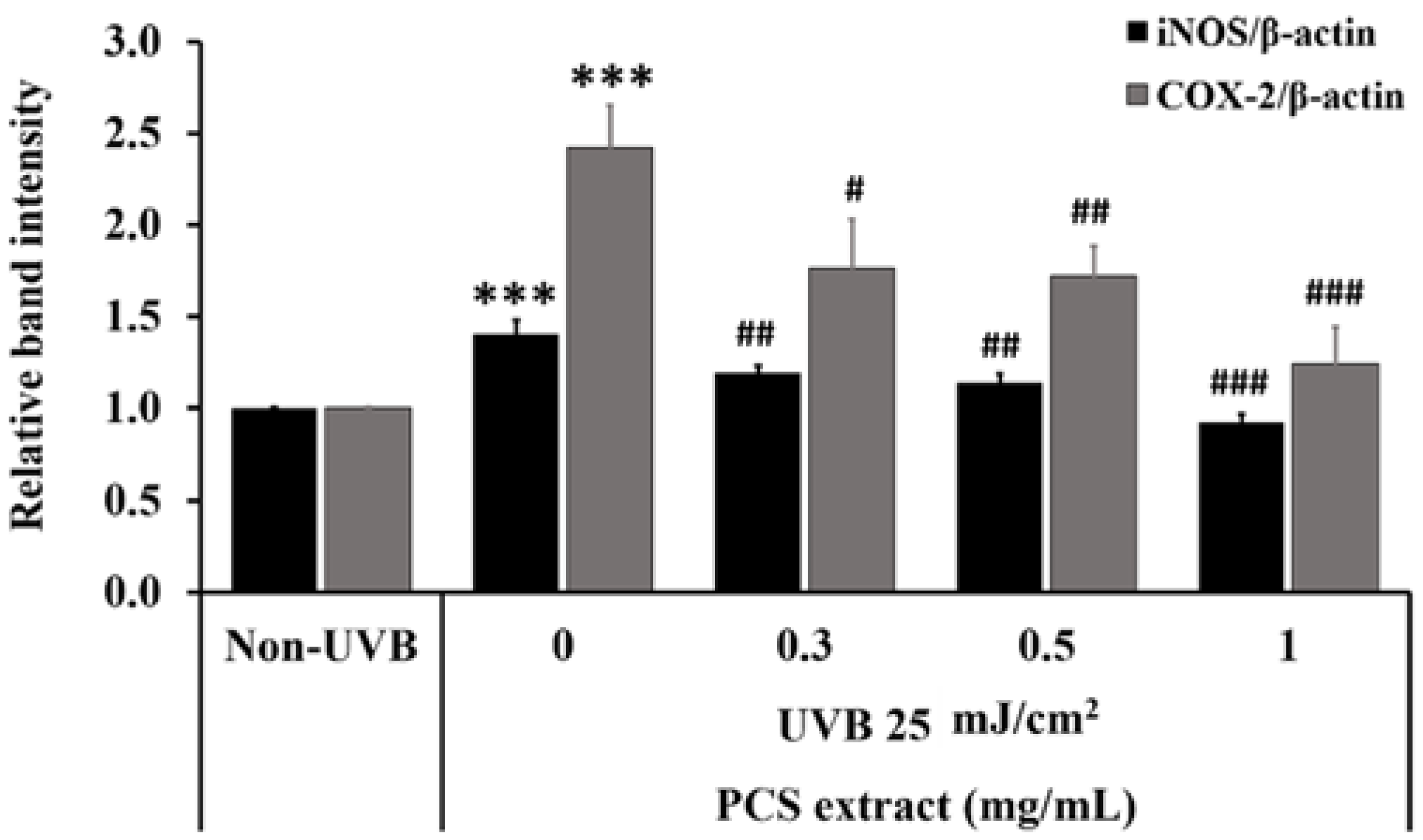
2.4. PCS Extract Reduced Proinflammatory Cytokine Expression in UVB-Treated HaCaT Cells
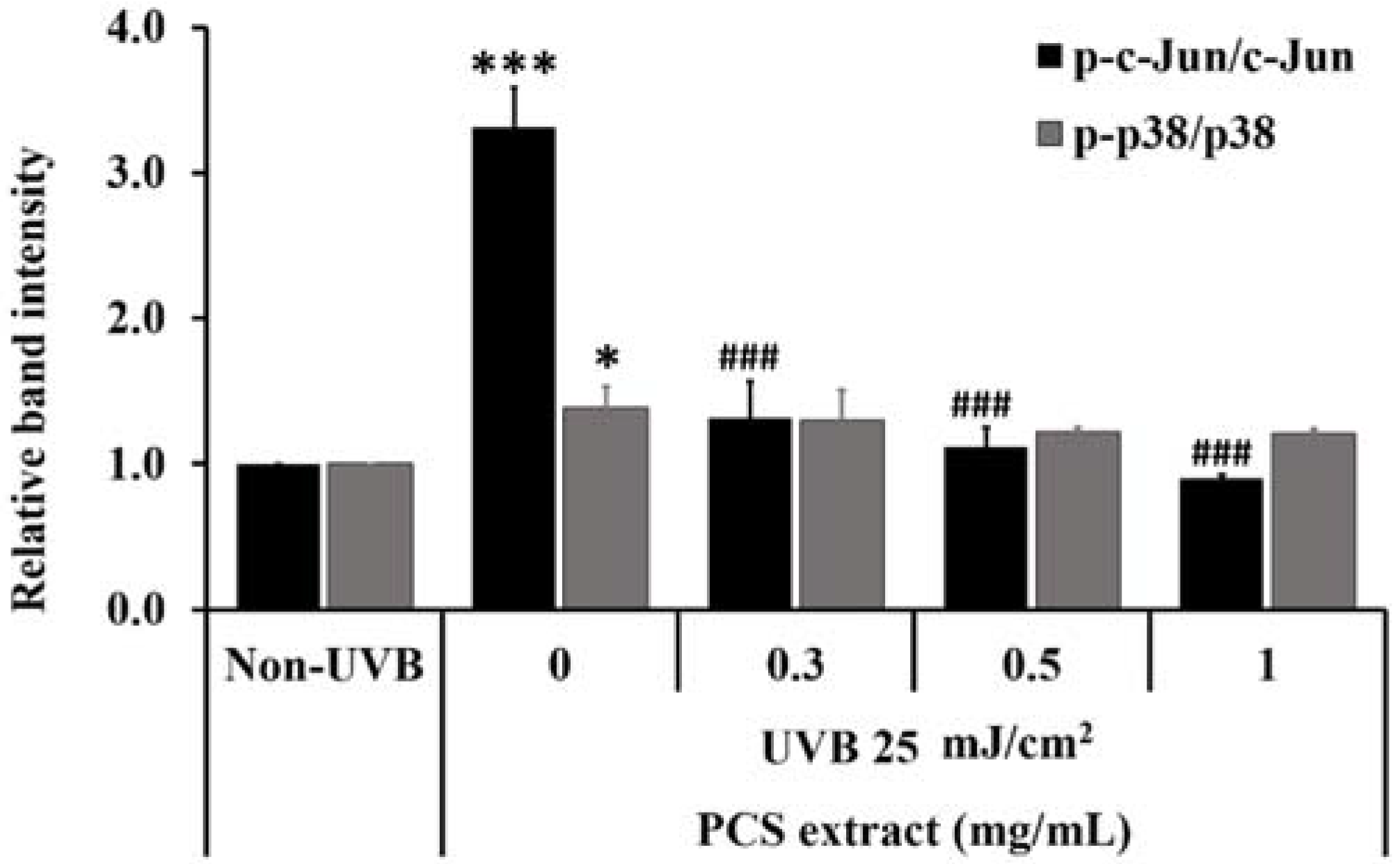
2.5. PCS Extract Decreased UVB-Induced MAPKs Phosphorylation in UVB-Treated HaCaT Cells
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Plant Extraction
4.3. UVB Irradiation System
4.4. UV-Absorbing Properties of PCS Extract
4.5. Cell Culture
4.6. Flow Cytometry
4.7. Immunofluorescence NF-κB Staining
4.8. Western Blot Analysis
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Afaq, F.; Mukhtar, H. Effects of solar radiation on cutaneous detoxification pathways. J. Photochem. Photobiol. B 2001, 63, 61–69. [Google Scholar] [CrossRef]
- Ivan, A.L.; Campanini, M.Z.; Martinez, R.M.; Ferreira, V.S.; Steffen, V.S.; Vicentini, F.T.; Vilela, F.M.; Martins, F.S.; Zarpelon, A.C.; Cunha, T.M.; et al. Pyrrolidine dithiocarbamate inhibits UVB-induced skin inflammation and oxidative stress in hairless mice and exhibits antioxidant activity in vitro. J. Photochem. Photobiol. B 2014, 138, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Park, G.; Kim, H.G.; Hong, S.P.; Kim, S.Y.; Oh, M.S. Walnuts (seeds of Juglandis sinensis L.) protect human epidermal keratinocytes against UVB-induced mitochondria-mediated apoptosis through upregulation of ROS elimination pathways. Skin Pharmacol. Physiol. 2014, 27, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Park, G.H.; Ahn, E.M.; Kim, B.A.; Park, C.I.; Jang, J.H. Protective effect of Codium fragile against UVB-induced pro-inflammatory and oxidative damages in HaCaT cells and BALB/c mice. Fitoterapia 2013, 86, 54–63. [Google Scholar] [CrossRef]
- Chatterjee, S. Oxidative Stress. Inflammation and Disease. In Oxidative Stress and Biomaterials; Dziubla, T., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 35–58. [Google Scholar]
- Kawashima, S.; Funakoshi, T.; Sato, Y.; Saito, N.; Ohsawa, H.; Kurita, K.; Nagata, K.; Yoshida, M.; Ishigami, A. Protective effect of pre- and post-vitamin C treatments on UVB-irradiation-induced skin damage. Sci. Rep. 2018, 8, 16199. [Google Scholar] [CrossRef]
- Liu, X.; Shi, S.; Ye, J.; Liu, L.; Sun, M.; Wang, C. Effect of polypeptide from Chlamys farreri on UVB-induced ROS/NF-kappaB/COX-2 activation and apoptosis in HaCaT cells. J. Photochem. Photobiol. B. 2009, 96, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Hur, S.; Lee, Y.S.; Yoo, H.; Yang, J.H.; Kim, T.Y. Homoisoflavanone inhibits UVB-induced skin inflammation through reduced cyclooxygenase-2 expression and NF-kappaB nuclear localization. J. Dermatol. Sci. 2010, 59, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Isoherranen, K.; Punnonen, K.; Jansen, C.; Uotila, P. Ultraviolet irradiation induces cyclooxygenase-2 expression in keratinocytes. Br. J. Dermatol. 1999, 140, 1017–1022. [Google Scholar] [CrossRef]
- Choi, Y.J.; Moon, K.M.; Chung, K.W.; Jeong, J.W.; Park, D.; Kim, D.H.; Yu, B.P.; Chung, H.Y. The underlying mechanism of proinflammatory NF-kappaB activation by the mTORC2/Akt/IKKalpha pathway during skin aging. Oncotarget 2016, 7, 52685–52694. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Jung, E.; Kim, S.; Kim, J.H.; Kim, E.G.; Lee, J.; Park, D. Antagonizing effects and mechanisms of afzelin against UVB-induced cell damage. PLoS ONE 2013, 8, e61971. [Google Scholar] [CrossRef]
- Zhi, Q.; Lie, L.; Li, F.; Zhao, J.; Yin, R.; Ming, J. The anthocyanin extracts from purple-fleshed sweet potato exhibited anti-photoaging effects on ultraviolent B-irradiated BALB/c-nu mouse skin. J. Funct. Foods 2020, 64, 103640. [Google Scholar] [CrossRef]
- Kuanpradit, C.; Jaisin, Y.; Jungudomjaroen, S.; Akter, M.S.; Puttikamonkul, S.; Sobhon, P.; Cummins, S.F. Attenuation of UV-B exposure-induced inflammation by abalone hypobranchial gland and gill extracts. Int. J. Mol. Med. 2017, 39, 1083–1090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, F.; Guan, H.; Liu, D.; Wu, X.; Fan, M.; Han, J. Flavonoids from sea buckthorn inhibit the lipopolysaccharide-induced inflammatory response in RAW264.7 macrophages through the MAPK and NF-kappaB pathways. Food Funct. 2017, 8, 1313–1322. [Google Scholar] [CrossRef] [PubMed]
- Papavassiliou, A.G.; Musti, A.M. The Multifaceted output of c-Jun biological activity: Focus at the junction of CD8 T cell activation and exhaustion. Cells 2020, 9, 2470. [Google Scholar] [CrossRef]
- Khan, N.; Syed, D.N.; Pal, H.C.; Mukhtar, H.; Afaq, F. Pomegranate fruit extract inhibits UVB-induced inflammation and proliferation by modulating NF-kappaB and MAPK signaling pathways in mouse skin. Photochem. Photobiol. 2012, 88, 1126–1134. [Google Scholar] [CrossRef] [Green Version]
- Poorahong, W.; Innalak, S.; Ungsurungsie, M.; Watanapokasin, R. Protective effect of purple corn silk extract against ultraviolet-B-induced cell damage in human keratinocyte cells. J. Adv. Pharm. Technol. Res. 2021, 12, 140–146. [Google Scholar]
- Ramos-Escudero, F.; Muñoz, A.M.; Alvarado-Ortíz, C.; Alvarado, Á.; Yáñez, J.A. Purple corn (Zea mays L.) phenolic compounds profile and its assessment as an agent against oxidative stress in isolated mouse organs. J. Med. Food. 2012, 15, 206–215. [Google Scholar] [CrossRef] [Green Version]
- Pedreschi, R.; Cisneros-Zevallos, L. Antimutagenic and antioxidant properties of phenolic fractions from Andean purple corn (Zea mays L.). J. Agric. Food Chem. 2006, 54, 4557–4567. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.A.; Pourmorad, P.; Hafezi, S. Antioxidant activities of Iranian corn silk. Turk. J. Biol. 2008, 32, 43–49. [Google Scholar]
- Zilic, S.; Jankovic, M.; Basic, Z.; Vancetovic, J.; Maksimovic, V. Antioxidant activity, phenolic profile, chlorophyll and mineral matter content of corn silk (Zea mays L): Comparison with medicinal herbs. J. Cereal Sci. 2016, 69, 363–370. [Google Scholar] [CrossRef]
- Chaiittianan, R.; Sutthanut, K.; Rattanathongkom, A. Purple corn silk: A potential anti-obesity agent with inhibition on adipogenesis and induction on lipolysis and apoptosis in adipocytes. J. Ethnopharmacol. 2017, 6, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Sarepoua, E.; Tangwongchai, R.; Suriharn, B.; Lertrat, K. Relationships between phytochemicals and antioxidant activity in corn silk. Int. Food Res. J. 2013, 20, 2073–2079. [Google Scholar]
- Senphan, T.; Yakong, N.; Aurtae, K.; Songchanthuek, S.; Choommongkol, V.; Fuangpaiboon, N.; Phing, P.L.; Yarnpakdee, S. Comparative studies on chemical composition and antioxidant activity of corn silk from two varieties of sweet corn and purple waxy corn as influenced by drying methods. Food Appl. Biosci. J. 2019, 7, 64–80. [Google Scholar]
- Perez-Sanchez, A.; Barrajon-Catalan, E.; Herranz-Lopez, M.; Castillo, J.; Micol, V. Lemon balm extract (Melissa officinalis L.) promotes melanogenesis and prevents UVB-induced oxidative stress and DNA damage in a skin cell model. J. Dermatol. Sci. 2016, 84, 169–177. [Google Scholar] [CrossRef]
- Tsoyi, K.; Park, H.B.; Kim, Y.M.; Chung, J.I.; Shin, S.C.; Lee, W.S.; Seo, H.G.; Lee, J.H.; Chang, K.C.; Kim, H.J. Anthocyanins from black soybean seed coats inhibit UVB-induced inflammatory cylooxygenase-2 gene expression and PGE2 production through regulation of the nuclear factor-kappaB and phosphatidylinositol 3-kinase/Akt pathway. J. Agric. Food Chem. 2008, 56, 8969–8974. [Google Scholar] [CrossRef]
- Buckman, S.Y.; Gresham, A.; Hale, P.; Hruza, G.; Anast, J.; Masferrer, J.; Pentland, A.P. COX-2 expression is induced by UVB exposure in human skin: Implications for the development of skin cancer. Carcinogenesis 1998, 19, 723–729. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Zhai, W. Identification and antioxidant activity of anthocyanins extracted from the seed and cob of purple corn (Zea mays L.). Innov. Food Sci. Emerg. Technol. 2010, 11, 169–176. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, B.; Wang, H.; Wang, F.; Song, Y.; Liu, S.; Xi, S. Maize purple plant pigment protects against fluoride-induced oxidative damage of liver and kidney in rats. Int. J. Environ. Res. Public Health 2014, 11, 1020–1033. [Google Scholar] [CrossRef] [Green Version]
- Park, C.; Park, J.; Kim, W.J.; Kim, W.; Cheong, H.; Kim, S.J. Malonic acid isolated from Pinus densiflora inhibits UVB-induced oxidative stress and inflammation in HaCaT keratinocytes. Polymers 2021, 13, 816. [Google Scholar] [CrossRef]
- Li, X.Q.; Cai, L.M.; Liu, J.; Ma, Y.L.; Kong, Y.H.; Li, H.; Jiang, M. Liquiritin suppresses UVBinduced skin injury through prevention of inflammation, oxidative stress and apoptosis through the TLR4/MyD88/NFkappaB and MAPK/caspase signaling pathways. Int. J. Mol. Med. 2018, 42, 1445–1459. [Google Scholar]
- Hinata, K.; Gervin, A.M.; Jennifer-Zhang, Y.; Khavari, P.A. Divergent gene regulation and growth effects by NF-kappa B in epithelial and mesenchymal cells of human skin. Oncogene 2003, 22, 1955–1964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, Y.; Jiang, Q.; Gao, H.; Fan, J.; Wang, Z.; Zhong, F.; Zheng, Y.; Gong, Z.; Wang, C. The anti-apoptotic effect of polypeptide from Chlamys farreri (PCF) in UVB-exposed HaCaT cells involves inhibition of iNOS and TGF-beta1. Cell Biochem. Biophys. 2015, 71, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal. Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.W.; Cheng, Y.C.; Hung, Y.C.; Lee, C.H.; Fang, J.Y.; Li, W.T.; Wu, Y.R.; Pan, T.L. Red raspberry extract protects the skin against UVB-induced damage with antioxidative and anti-inflammatory properties. Oxid. Med. Cell Longev. 2019, 9529676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasparrini, M.; Forbes-Hernandez, T.Y.; Afrin, S.; Reboredo-Rodriguez, P.; Cianciosi, D.; Mezzetti, B.; Quiles, J.L.; Bompadre, S.; Battino, M.; Giampieri, F. Strawberry-based cosmetic formulations protect human dermal fibroblasts against UVA-induced damage. Nutrients 2017, 14, 605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piao, M.J.; Kang, K.A.; Zhen, A.X.; Kang, H.K.; Koh, Y.S.; Kim, B.S.; Hyun, J.W. Horse oil mitigates oxidative damage to human HaCaT keratinocytes caused by ultraviolet B irradiation. Int. J. Mol. Sci. 2019, 25, 1490. [Google Scholar] [CrossRef] [Green Version]
- Spiegelman, V.S.; Slaga, T.J.; Pagano, M.; Minamoto, T.; Ronai, Z.; Fuchs, S.Y. Wnt/beta-catenin signaling induces the expression and activity of betaTrCP ubiquitin ligase receptor. Mol. Cell. 2000, 5, 877–882. [Google Scholar] [CrossRef]
- Chen, F.; Bower, J.; Demers, L.M.; Shi, X. Upstream Signal Transduction of NF-kB Activation. Atlas Genet. Cytogenet. Oncol. Haematol. 2002, 6, 156–170. [Google Scholar] [CrossRef] [Green Version]
- Yue, J.; López, J.M. Understanding MAPK Signaling Pathways in Apoptosis. Int. J. Mol. Sci. 2020, 21, 2346. [Google Scholar] [CrossRef] [Green Version]
- Gao, W.; Wang, Y.S.; Hwang, E.; Lin, P.; Bae, J.; Seo, S.A.; Yan, Z.; Yi, T.H. Rubus idaeus L. (red raspberry) blocks UVB-induced MMP production and promotes type I procollagen synthesis via inhibition of MAPK/AP-1, NF-kappabeta and stimulation of TGF-beta/Smad, Nrf2 in normal human dermal fibroblasts. J. Photochem. Photobiol. B 2018, 185, 241–253. [Google Scholar] [CrossRef]
- Hu, Y.; Ma, Y.; Wu, S.; Chen, T.; He, Y.; Sun, J.; Jiao, R.; Jiang, X.; Huang, Y.; Deng, L.; et al. Protective effect of cyanidin-3-o-glucoside against ultraviolet B radiation-induced cell damage in human HaCaT keratinocytes. Front. Pharmacol. 2016, 7, 301. [Google Scholar] [CrossRef] [Green Version]
- Cimino, F.; Ambra, R.; Canali, R.; Saija, A.; Virgili, F. Effect of cyanidin-3-o-glucoside on UVB-induced response in human keratinocytes. J. Agric. Food Chem. 2006, 54, 4041–4047. [Google Scholar] [CrossRef]
- Pratheeshkumar, P.; Son, Y.O.; Wang, X.; Divya, S.P.; Joseph, B.; Hitron, J.A.; Wang, L.; Kim, D.; Yin, Y.; Roy, R.V.; et al. Cyanidin-3-glucoside inhibits UVB-induced oxidative damage and inflammation by regulating MAP kinase and NF-kappaB signaling pathways in SKH-1 hairless mice skin. Toxicol. Appl. Pharmacol. 2014, 280, 127–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kunchana, K.; Jarisarapurin, W.; Chularojmontri, L.; Wattanapitayakul, S.K. Potential Use of Amla (Phyllanthus emblica L.) Fruit Extract to Protect Skin Keratinocytes from Inflammation and Apoptosis after UVB Irradiation. Antioxidants 2021, 10, 703. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poorahong, W.; Innajak, S.; Ungsurungsie, M.; Watanapokasin, R. Purple Corn Silk Extract Attenuates UVB-Induced Inflammation in Human Keratinocyte Cells. Sci. Pharm. 2022, 90, 18. https://doi.org/10.3390/scipharm90010018
Poorahong W, Innajak S, Ungsurungsie M, Watanapokasin R. Purple Corn Silk Extract Attenuates UVB-Induced Inflammation in Human Keratinocyte Cells. Scientia Pharmaceutica. 2022; 90(1):18. https://doi.org/10.3390/scipharm90010018
Chicago/Turabian StylePoorahong, Watcharaporn, Sukanda Innajak, Malyn Ungsurungsie, and Ramida Watanapokasin. 2022. "Purple Corn Silk Extract Attenuates UVB-Induced Inflammation in Human Keratinocyte Cells" Scientia Pharmaceutica 90, no. 1: 18. https://doi.org/10.3390/scipharm90010018
APA StylePoorahong, W., Innajak, S., Ungsurungsie, M., & Watanapokasin, R. (2022). Purple Corn Silk Extract Attenuates UVB-Induced Inflammation in Human Keratinocyte Cells. Scientia Pharmaceutica, 90(1), 18. https://doi.org/10.3390/scipharm90010018






