Antiproliferative Activity of Triterpenoid and Steroid Compounds from Ethyl Acetate Extract of Calotropis gigantea Root Bark against P388 Murine Leukemia Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extraction of C. gigantea Root Bark
2.3. Selection of the Combined Fractions Based on Their Cytotoxicity
2.4. Isolation of Triterpenoids and Steroids
2.5. 3-(4,5-Dimethylthiazol-2-yl) -2-5 Diphenyl Tetrazolium Bromide (MTT) Assay
2.6. Structure Characterization
3. Results
3.1. Extraction Yield and Phytochemical Properties
3.2. Fractions and Their Cytotoxicity against Brine Shrimp Lethality Test
3.3. MTT Assay of the Extracts from C. gigantea Root Bark
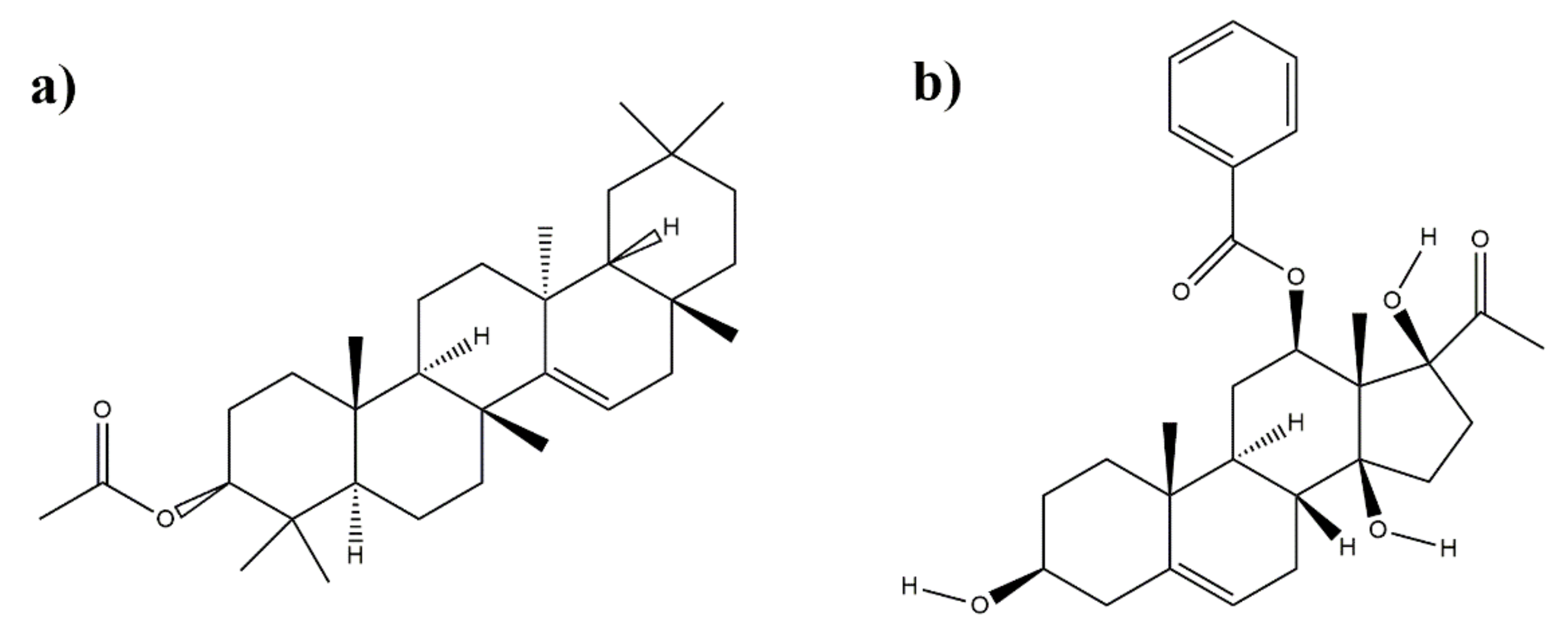
3.4. Structure Characterization of Subfraction C2 and D1
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clarke, J.N.; Everest, M.M. Cancer in the mass print media: Fear, uncertainty and the medical model. Soc. Sci. Med. 2006, 62, 2591–2600. [Google Scholar] [CrossRef] [PubMed]
- Baskar, R.; Lee, K.; Yeo, R.; Yeoh, K.-W. Cancer and radiation therapy: Current advances and future directions. Int. J. Med. Sci. 2012, 9, 193. [Google Scholar] [CrossRef]
- Chinembiri, T.; du Plessis, L.; Gerber, M.; Hamman, J.; du Plessis, J. Review of Natural Compounds for Potential Skin Cancer Treatment. Molecules 2014, 19, 11679–11721. [Google Scholar] [CrossRef]
- Molassiotis, A.; Fernadez-Ortega, P.; Pud, D.; Ozden, G.; Scott, J.A.; Panteli, V.; Margulies, A.; Browall, M.; Magri, M.; Selvekerova, S.; et al. Use of complementary and alternative medicine in cancer patients: A European survey. Ann. Oncol. 2005, 16, 655–663. [Google Scholar] [CrossRef]
- Kumar, G.; Karthik, L.; Rao, K. Antibacterial activity of aqueous extract of Calotropis gigantea leaves–an in vitro study. Int. J. Pharm. Sci. Rev. Res. 2010, 4, 141–144. [Google Scholar]
- Elakkiya, P.; Prasanna, G. A study on phytochemical screening and in vitro antioxidant activity of Calotropis gigantea L. Int. J. Chemtech. Res. 2012, 4, 1428–1431. [Google Scholar]
- Mahar, R.; Dixit, S.; Joshi, T.; Kanojiya, S.; Mishra, D.; Konwar, R.; Al, E. Bioactivity guided isolation of oxypregnane-oligoglycosides (calotroposides) from the root bark of Calotropis gigantea as potent anticancer agents. RSC Adv. 2016, 6, 104215–104226. [Google Scholar] [CrossRef]
- Seniya, C.; Trivedia, S.; Verma, S. Antibacterial efficacy and phytochemical analysis of organic solvent extracts of Calotropis gigantea. J. Chem. Pharm. Res. 2011, 3, 330–336. [Google Scholar]
- Ansari, S.; Ali, M. Norditerpenic ester and pentacyclic triterpenoids from root bark of Calotropis procera (Ait) R. Br. Die Pharm. 2001, 56, 175–177. [Google Scholar]
- Saratha, V.; Pillai, S.; Subramanian, S. Isolation and characterization of lupeol, a triterpenoid from Calotropis gigantea latex. Int. J. Pharm. Sci. Rev. Res. 2011, 10, 54–57. [Google Scholar]
- Hasballah, K. Cytotoxic Bioactive Compounds from Calotropis Gigantea Stem Bark. Int. J. Pharm. Pharm. Sci. 2016, 8, 111–115. [Google Scholar] [CrossRef][Green Version]
- Mohaimenul, I.M.; Hossain, M.; Osman, A.; Aziz, M.; Habib, M.; Karim, R. A terpenoid and a steroid from Calotropis gigantea (L.). Nov. Sci. Int. J. Pharm. Sci. 2012, 1, 580–584. [Google Scholar]
- Habib, M.R.; Karim, M.R. Evaluation of antitumour activity of Calotropis gigantea L. root bark against Ehrlich ascites carcinoma in Swiss albino mice. Asian Pac. J. Trop. Med. 2011, 4, 786–790. [Google Scholar] [CrossRef]
- Wang, Z.-N.; Wang, M.-Y.; Mei, W.-L.; Han, Z.; Dai, H.-F. A new cytotoxic pregnanone from Calotropis gigantea. Molecules 2008, 13, 3033–3039. [Google Scholar] [CrossRef]
- Etti, I.C.; Abdullah, R.; Kadir, A.; Hashim, N.M.; Yeap, S.K.; Imam, M.U.; Ramli, F.; Malami, I.; Lam, K.L.; Etti, U.; et al. The molecular mechanism of the anticancer effect of Artonin E in MDA-MB 231 triple negative breast cancer cells. PLoS ONE 2017, 12, e0182357. [Google Scholar] [CrossRef]
- Etti, I.; Abdullah, R.; Hashim, N.; Kadir, A.; Abdul, A.; Etti, C.; Malami, I.; Waziri, P.; How, C. Artonin E and Structural Analogs from Artocarpus Species Abrogates Estrogen Receptor Signaling in Breast Cancer. Molecules 2016, 21, 839. [Google Scholar] [CrossRef]
- Palleros, D. Experimental Organic Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2000. [Google Scholar]
- De Hoffmann, E. Mass Spectrometry; John Wiley & Sons: Hoboken, NJ, USA, 2000. [Google Scholar]
- Harborne, J. Metode Fitokimia: Penuntun Cara Modern Menganalisis Tumbuhan; Institut Teknologi Bandung Press: Bandung, Indonesia, 1987. [Google Scholar]
- Fessenden, R.J.; Fessenden, J.S. Organic Chemistry; Brooks/Cole Publishing Company: Pacific Grove, CA, USA, 1986; ISBN 9780534050887. [Google Scholar]
- Shiojima, K.; Arai, Y.; Masuda, K.; Takase, Y.; Ageta, T.; Ageta, H. Mass spectra of pentacyclic triterpenoids. Chem. Pharm. Bull. 1992, 40, 1683–1690. [Google Scholar] [CrossRef]
- Caragay, A. Cancer-preventive foods and ingredients. Food Technol. 1992, 46, 65–68. [Google Scholar]
- Kumar, P.; Suresh, E.; Kalavathy, S. Review on a potential herb Calotropis gigantea (L.) R. Br. Sch. Acad. J. Biosci. 2013, 2, 135–143. [Google Scholar]
- Verma, N.; Shukla, S. Impact of various factors responsible for fluctuation in plant secondary metabolites. J. Appl. Res. Med. Aromat. Plants 2015, 2, 105–113. [Google Scholar] [CrossRef]
- Yoshiki, Y.; Kudou, S.; Okubo, K. Relationship between chemical structures and biological activities of triterpenoid saponins from soybean. Biosci. Biotechnol. Biochem. 1998, 62, 2291–2299. [Google Scholar] [CrossRef]
- GuangLie, C.; WeiShi, G.; GaiLing, H.; JianPing, C. Effect of Paris saponin on antitumor and immune function in U14 tumor-bearing mice. Afr. J. Tradit. Complement Altern. Med. 2013, 10, 503–507. [Google Scholar] [CrossRef]
- Galanty, A.; Michalik, M.; Sędek, Ł.; Podolak, I. The influence of LTS-4, a saponoside from Lysimachia thyrsiflora L., on human skin fibroblasts and human melanoma cells. Cell Mol. Biol. Lett. 2008, 13, 585. [Google Scholar] [CrossRef]
- Avin, B.; Thirusangu, P.; Ranganatha, V.; Firdouse, A.; Prabhakar, B.; Khanum, S. Synthesis and tumor inhibitory activity of novel coumarin analogs targeting angiogenesis and apoptosis. Eur. J. Med. Chem. 2014, 75, 211–221. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, G.; Yao, F.; He, Y.; Liang, G.; Zhang, Y.; Et, A. Growth inhibition and apoptosis induced by osthole, a natural coumarin, in hepatocellular carcinoma. PLoS ONE 2012, 7, e37865. [Google Scholar] [CrossRef]
- Budzisz, E.; Brzezinska, E.; Krajewska, U.; Rozalski, M. Cytotoxic effects, alkylating properties and molecular modelling of coumarin derivatives and their phosphonic analogues. Eur. J. Med. Chem. 2003, 38, 597–603. [Google Scholar] [CrossRef]
- Darmawan, A.; Kosela, S.; Kardono, L.; Syah, Y. Scopoletin, a coumarin derivative compound isolated from Macaranga gigantifolia Merr. J. Appl. Pharm. Sci. 2012, 2, 175. [Google Scholar]
- Taraphdar, A.; Roy, M.; Bhattacharya, R. Natural products as inducers of apoptosis: Implication for cancer therapy and prevention. Curr. Sci. 2001, 80, 1387–1396. [Google Scholar]
- Fabiani, R.; de Bartolomeo, A.; Rosignoli, P.; Servili, M.; Selvaggini, R.; Montedoro, G.; Et, A. Virgin olive oil phenols inhibit proliferation of human promyelocytic leukemia cells (HL60) by inducing apoptosis and differentiation. J. Nutr. 2006, 136, 614–619. [Google Scholar] [CrossRef]
- Fabiani, R.; de Bartolomeo, A.; Rosignoli, P.; Servili, M.; Montedoro, G.; Morozzi, G. Cancer chemoprevention by hydroxytyrosol isolated from virgin olive oil through G1 cell cycle arrest and apoptosis. Eur. J. Cancer Prev. 2002, 11, 351–358. [Google Scholar] [CrossRef]
- Makino, T.; Ono, T.; Muso, E.; Yoshida, H.; Honda, G.; Sasayama, S. Inhibitory effects of rosmarinic acid on the proliferation of cultured murine mesangial cells. Nephrol. Dial. Transpl. 2000, 15, 1140–1145. [Google Scholar] [CrossRef]
- Wong, S.; Lim, Y.; Abdullah, N.; Nordin, F. Assessment of antiproliferative and antiplasmodial activities of five selected Apocynaceae species. BMC Complement. Altern. Med. 2011, 11, 3. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Liu, L.; Zhen, X.; Wu, X.; Zhang, Y. Anti-tumor activity of triterpenoid-rich extract from bamboo shavings (Caulis bamfusae in Taeniam). Afr. J. Biotechnol. 2010, 9, 6430–6436. [Google Scholar]
- Wu, Y.; Lou, Y. A steroid fraction of chloroform extract from bee pollen of Brassica campestris induces apoptosis in human prostate cancer PC-3 cells. Phytother. Res. 2007, 21, 1087–1091. [Google Scholar] [CrossRef] [PubMed]
- Habib, M.; Nikkon, F.; Rahman, M.; Haque, Z.; Karim, M. Isolation of stigmasterol and ß-sitosterol from methanolic extract of root. Pak. J. Biol. Sci. 2007, 10, 4174–4176. [Google Scholar] [PubMed]
- Ghosh, T.; Maity, T.; Singh, J. Evaluation of antitumor activity of stigmasterol, a constituent isolated from Bacopa monnieri Linn aerial parts against Ehrlich Ascites Carcinoma in mice. Orient. Pharm. Exp. Med. 2011, 11, 41–49. [Google Scholar] [CrossRef]
- Bin Sayeed, M.; Ameen, S. Beta-sitosterol: A promising but orphan nutraceutical to fight against cancer. Nutr. Cancer 2015, 67, 1216–1222. [Google Scholar] [CrossRef]
- Suffness, M.; Pezzuto, J.M. Assays related to cancer drug discovery. Methods Plant Biochem. Assays Bioact. 1990, 6, 71–133. [Google Scholar]
- Caesar, L.K.; Cech, N.B. Synergy and antagonism in natural product extracts: When 1 + 1 does not equal 2. Nat. Prod. Rep. 2019, 36, 869–888. [Google Scholar] [CrossRef]
- Hong, J.; Song, Y.; Liu, Z.; Zheng, Z.; Chen, H.; Wang, S. Anticancer activity of taraxerol acetate in human glioblastoma cells and a mouse xenograft model via induction of autophagy and apoptotic cell death, cell cycle arrest and inhibition of cell migration. Mol. Med. Rep. 2016, 13, 4541–4548. [Google Scholar] [CrossRef]
- Arullappan, S.; Muhamad, S.; Zakaria, Z. Cytotoxic activity of the leaf and stem extracts of Hibiscus rosa-sinensis (Malvaceae) against Leukaemic Cell Line (K-562). Trop. J. Pharm. Res. 2013, 12, 743–746. [Google Scholar] [CrossRef]
| Sample | IC50 (μg/mL) |
|---|---|
| N-hexane extract | 49.02 ± 0.06 |
| Ethyl acetate extract | 21.79 ± 0.03 |
| Methanol extract | 68.45 ± 0.02 |
| Subfraction C2 (ethyl acetate) | 50.64 ± 0.03 |
| Subfraction D1 (ethyl acetate) | 49.33 ± 0.01 |
| Artonin E (positive control) | 0.74 ± 0.27 |
| Functional Group | Wavenumber (cm−1) | ||
|---|---|---|---|
| Subfraction C2 | Subfraction D1 | Ref. [20] | |
| O-H | Not detected | 3007 | 3000–3700 |
| C-H (alkane) | 2920 | 2934 | 2800–3000 |
| C=O | 1701 | 1715 | 1700–1725 |
| C=C | 1462 | Not detected | 1400–1600 |
| C-O (ester) | 1190 | 1057 | 1050–1260 |
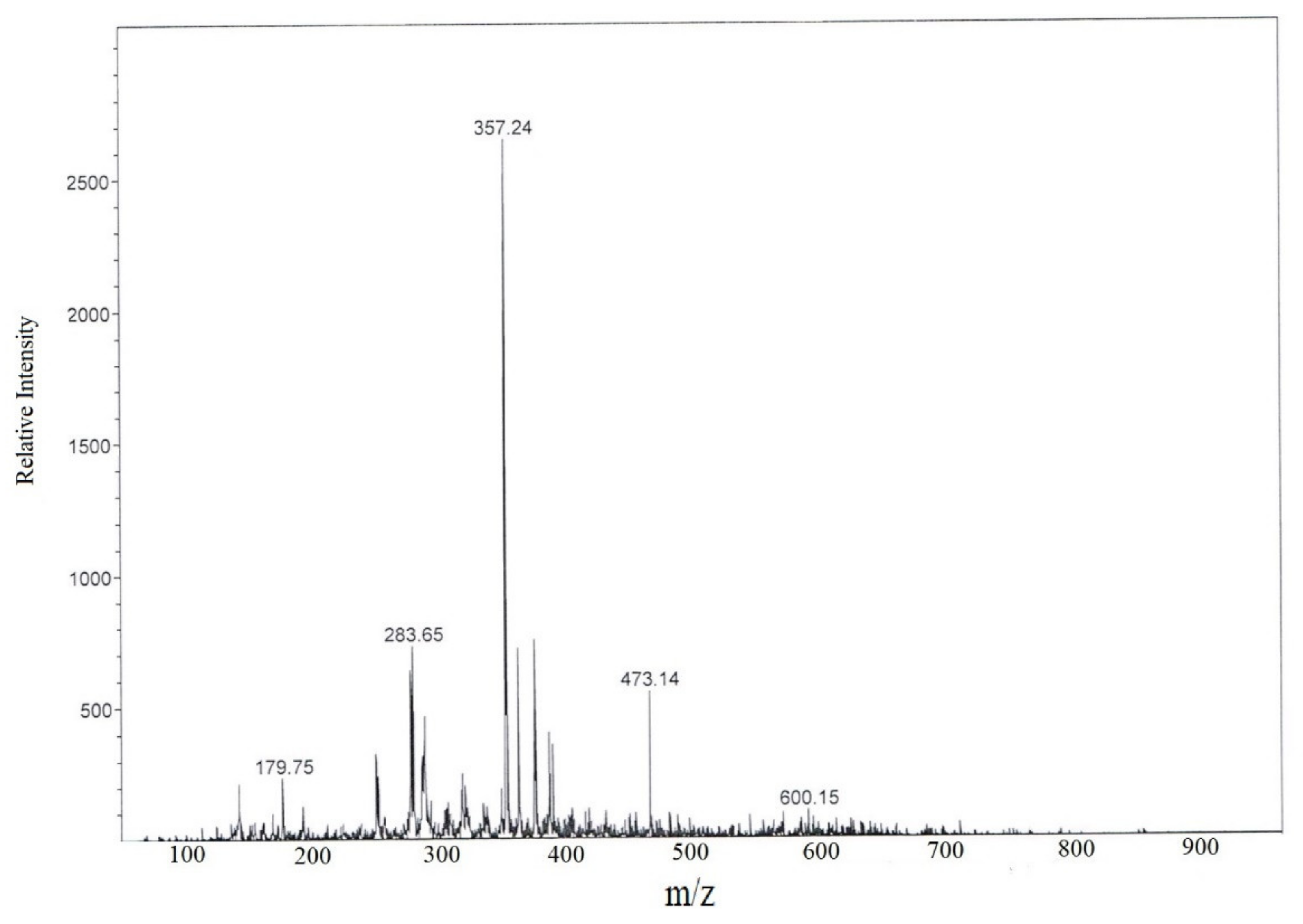
| Fragment ion (m/z) | |
|---|---|
| Subfraction D1 | Ref. [21] |
| 467 | 468 |
| 457 | 453 |
| 410 | 409 |
| 397 | - |
| 383 | - |
| 370 | - |
| 357 | 359 |
| 344 | 344 |
| 325 | 329 |
| - | 269 |
| 284 | 284 |
| 257 | 257 |
| 200 | 204 |
| - | 189 |
| 179 | - |
| 127 | - |
| - | 121 |
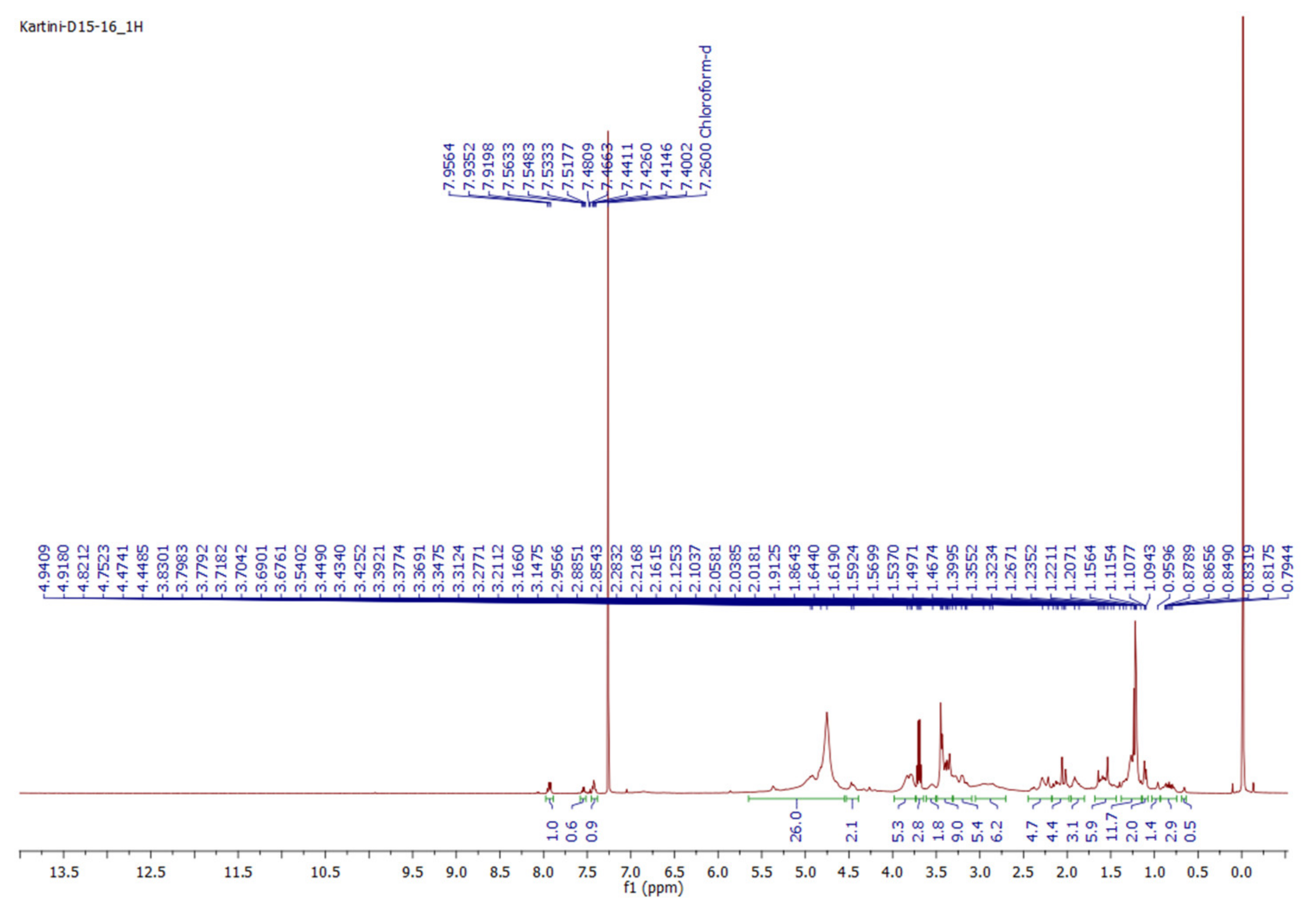
| Position | δH (μg/mL) | |
|---|---|---|
| This Study (CDCl3, 500 MHz) | Ref. [14] (CDCl3, 400 MHz) | |
| 1 | 1.74 (1H, m, H-1a), | 1.75 (1H, m, H-1a), |
| 2 | 1.12 (1H, m, H-1b) | 1.13 (1H, m, H-1b) |
| 3 | 1.82, 1.49 (each 1H, m) | 1.81, 1.46 (each 1H, m) |
| 4 | 3.54 (1H, m) | 3.53 (1H, m) |
| 5 | 2.31 (1H, m), | 2.33 (1H, dd, 12.8, 3.6 Hz) |
| 6 | 2.22 (1H, m, overlapped) | 2.25 (1H, m, overlapped) |
| 7 | NA | NA |
| 8 | 5.40 (1H, m) | 5.41 (1H, m) |
| 9 | 2.16, 1.92 (each 1H, m) | 2.20, 1.91 (each 1H, m) |
| 10 | 1.81 (1H, m) | 1.80 (1H, m) |
| 11 | 1.32 (1H, m) | 1.32 (1H, m) |
| 12 | NA | NA |
| 13 | 2.08, 1.47 (each 1H, m, overlapped) | 2.06, 1.45 (each 1H, m, overlapped) |
| 14 | 4.82 (1H, m) | 4.80 (1H, dd, 11.3, 4.5 Hz) |
| 15 | NA | NA |
| 16 | NA | NA |
| 17 | 2.13 (1H, m, H-15a), | 2.12 (1H, m, H-15a), |
| 18 | 1.94 (1H, m, H-15b) | 1.92 (1H, m, H-15b) |
| 19 | 2.89 (1H, m, H-16a), 1.89 (1H, m, H-16b) | 2.90 (1H, m, H-16a), 1.88 (1H, m, H-16b) |
| 20 | NA | NA |
| 21 | 1.39 (3H, s) | 1.41 (3H, s) |
| 1′ | 0,96 (3H, s) | 0,98 (3H, s) |
| 2′ | NA | NA |
| 3′ | 2.08 (3H, overlapped) | 2.06 (3H, overlapped) |
| 4′ | NA | NA |
| 5′ | 7.94 (1H, d, 7.3 Hz) | 7.93 (1H, d, 7.5 Hz) |
| 6′ | 7.44 (1H, t, 7.3 Hz) | 7.43 (1H, t, 7.5 Hz) |
| 7′ | 7.55 (1H, t, 7.5 Hz) | 7.56 (1H, t, 7.5 Hz) |
| 1 | 7.44 (1H, t, 7.3 Hz) | 7.43 (1H, t, 7.5 Hz) |
| 2 | 7.94 (1H, d, 7.7 Hz) | 7.93 (1H, d, 7.5 Hz) |
| 3 | NA | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasballah, K.; Sarong, M.; Rusly, R.; Fitria, H.; Maida, D.R.; Iqhrammullah, M. Antiproliferative Activity of Triterpenoid and Steroid Compounds from Ethyl Acetate Extract of Calotropis gigantea Root Bark against P388 Murine Leukemia Cell Lines. Sci. Pharm. 2021, 89, 21. https://doi.org/10.3390/scipharm89020021
Hasballah K, Sarong M, Rusly R, Fitria H, Maida DR, Iqhrammullah M. Antiproliferative Activity of Triterpenoid and Steroid Compounds from Ethyl Acetate Extract of Calotropis gigantea Root Bark against P388 Murine Leukemia Cell Lines. Scientia Pharmaceutica. 2021; 89(2):21. https://doi.org/10.3390/scipharm89020021
Chicago/Turabian StyleHasballah, Kartini, Murniana Sarong, Renzavaldy Rusly, Herdina Fitria, Dewi Rara Maida, and Muhammad Iqhrammullah. 2021. "Antiproliferative Activity of Triterpenoid and Steroid Compounds from Ethyl Acetate Extract of Calotropis gigantea Root Bark against P388 Murine Leukemia Cell Lines" Scientia Pharmaceutica 89, no. 2: 21. https://doi.org/10.3390/scipharm89020021
APA StyleHasballah, K., Sarong, M., Rusly, R., Fitria, H., Maida, D. R., & Iqhrammullah, M. (2021). Antiproliferative Activity of Triterpenoid and Steroid Compounds from Ethyl Acetate Extract of Calotropis gigantea Root Bark against P388 Murine Leukemia Cell Lines. Scientia Pharmaceutica, 89(2), 21. https://doi.org/10.3390/scipharm89020021







