Prevalence of Drug Interaction in Severely Obese Individuals and Associated Factors: Baseline Results from a Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting and Study Design
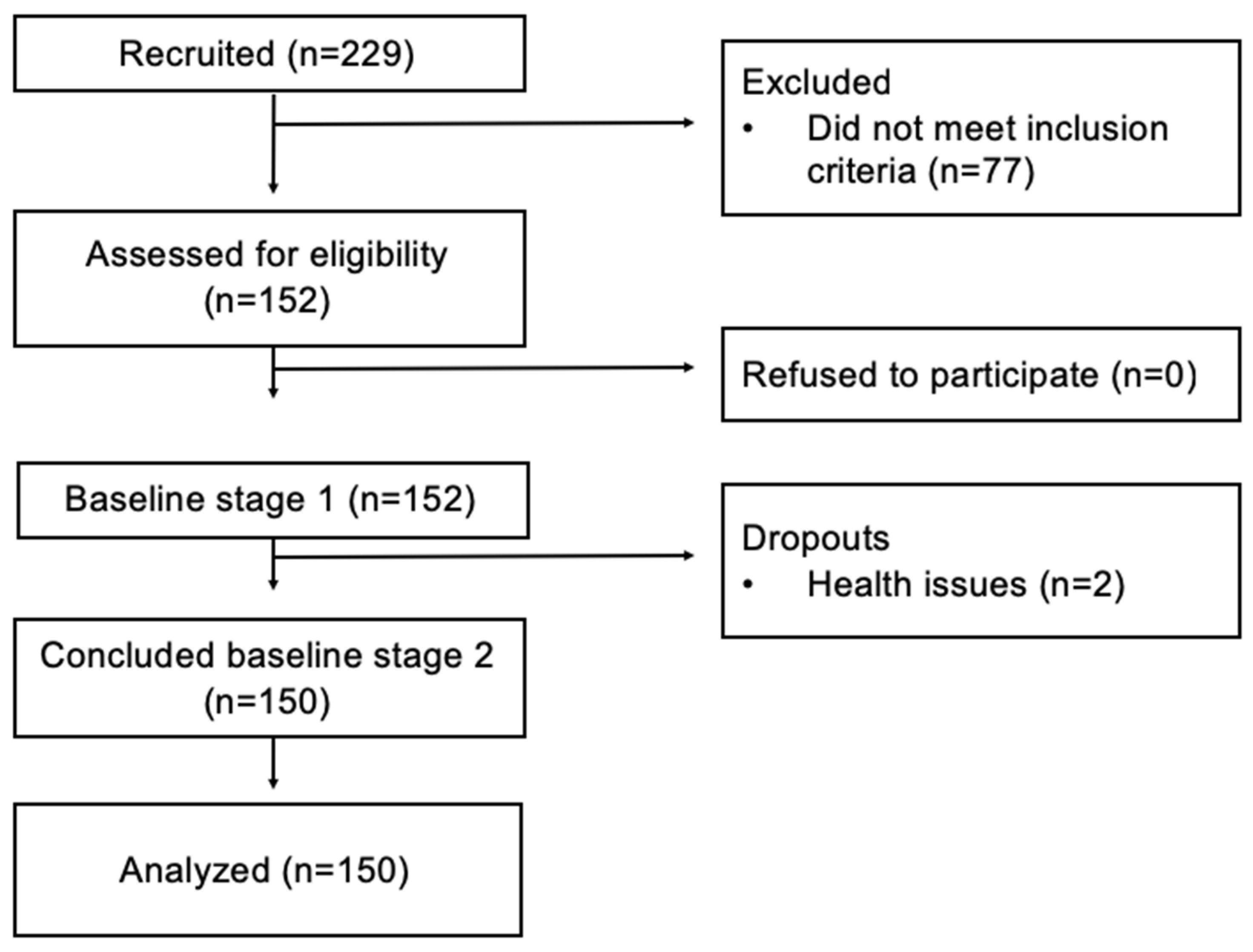
2.2. Subjects
2.3. Variables
2.4. Data Collection
2.5. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Di Cesare, M.; Bentham, J.; Stevens, G.A.; Rühli, F.J.; Staub, K.; Faeh, D.; Gutzwiller, F.; Al, E. NCD Risk Factor Collaboration (NCD-RisC) Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19·2 million participants. Lancet 2016, 387, 1377–1396. [Google Scholar] [CrossRef]
- Cheng, F.W.; Gao, X.; Mitchell, D.C.; Wood, C.; Still, C.D.; Rolston, D.; Jensen, G.L. Body mass index and all-cause mortality among older adults. Obesity 2016, 24, 2232–2239. [Google Scholar] [CrossRef] [PubMed]
- Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; Naghavi, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Malta, D.C.; Silva Júnior, J.B. O Plano de Ações Estratégicas para o Enfrentamento das Doenças Crônicas Não Transmissíveis no Brasil e a definição das metas globais para o enfrentamento dessas doenças até 2025: Uma revisão. Epidemiol. Serviços Saúde 2013, 22, 151–164. [Google Scholar] [CrossRef]
- Cohen, J.B. Hypertension in Obesity and the Impact of Weight Loss. Curr. Cardiol. Rep. 2017, 19, 1–8. [Google Scholar] [CrossRef]
- Bae, J.; Lage, M.; Mo, D.; Nelson, D.R.; Hoogwerf, B. Obesity and glycemic control in patients with diabetes mellitus: Analysis of physician electronic health records in the US from 2009–2011. J. Diabetes Complicat. 2016, 30, 212–220. [Google Scholar] [CrossRef]
- Allam-Ndoul, B.; Guénard, F.; Garneau, V.; Cormier, H.; Barbier, O.; Pérusse, L.; Vohl, M.-C. Association between Metabolite Profiles, Metabolic Syndrome and Obesity Status. Nutrients 2016, 8, 324. [Google Scholar] [CrossRef]
- Vitolo, E.; Santini, E.; Salvati, A.; Volterrani, D.; Duce, V.; Bruno, R.; Solini, A. Metabolic and Hormonal Determinants of Glomerular Filtration Rate and Renal Hemodynamics in Severely Obese Individuals. Obes. Facts 2016, 9, 310–320. [Google Scholar] [CrossRef]
- Liew, P.-L.; Lee, W.-J.; Lee, Y.-C.; Wang, H.-H.; Wang, W.; Lin, Y.-C. Hepatic Histopathology of Morbid Obesity: Concurrence of Other Forms of Chronic Liver Disease. Obes. Surg. 2006, 16, 1584–1593. [Google Scholar] [CrossRef]
- Calenzani, G.; Dos Santos, F.F.; Wittmer, V.L.; Freitas, G.K.F.; Paro, F.M. Prevalence of musculoskeletal symptoms in obese patients candidates for bariatric surgery and its impact on health related quality of life. Arch. Endocrinol. Metab. 2017, 61, 319–325. [Google Scholar] [CrossRef][Green Version]
- Stenholm, S.; Head, J.; Aalto, V.; Kivimäki, M.; Kawachi, I.; Zins, M.; Goldberg, M.; Platts, L.G.; Zaninotto, P.; Hanson, L.L.M.; et al. Body mass index as a predictor of healthy and disease-free life expectancy between ages 50 and 75: A multicohort study. Int. J. Obes. 2017, 41, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Sonntag, D.; Jarczok, M.N.; Ali, S. DC-Obesity: A New Model for Estimating Differential Lifetime Costs of Overweight and Obesity by Socioeconomic Status. Obesity 2017, 25, 1603–1609. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Laxy, M.; Stark, R.G.; Peters, A.; Hauner, H.; Holle, R.; Teuner, C.M. The Non-Linear Relationship between BMI and Health Care Costs and the Resulting Cost Fraction Attributable to Obesity. Int. J. Environ. Res. Public Health 2017, 14, 984. [Google Scholar] [CrossRef] [PubMed]
- Gazmararian, J.A.; Frisvold, D.; Zhang, K.; Koplan, J.P. Obesity is associated with an increase in pharmaceutical expenses among university employees. J. Obes. 2015, 2015, 7. [Google Scholar] [CrossRef]
- Narbro, K.; Agren, G.; Jonsson, E.; Näslund, I.; Sjöström, L.; Peltonen, M. Swedish Obese Subjects Intervention Study Pharmaceutical Costs in Obese Individuals. Arch. Intern. Med. 2002, 162, 2061–2069. [Google Scholar] [CrossRef] [PubMed]
- Ferner, R.E.; Easton, C.; Cox, A.R. Deaths from Medicines: A Systematic Analysis of Coroners’ Reports to Prevent Future Deaths. Drug Saf. 2018, 42, 445–451. [Google Scholar] [CrossRef]
- Ferner, R.E. The epidemiology of medication errors: The methodological difficulties. Br. J. Clin. Pharmacol. 2009, 67, 614–620. [Google Scholar] [CrossRef]
- Gosho, M.; Maruo, K.; Tada, K.; Hirakawa, A. Utilization of chi-square statistics for screening adverse drug-drug interactions in spontaneous reporting systems. Eur. J. Clin. Pharmacol. 2017, 3, 383–786. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Medication without Harm-Global Patient Safety Challenge on Medication Safety; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Souza, T.T.; de Godoy, R.R.; Rotta, I.; Pontarolo, R.; Fernandez-llimos, F.; Correr, C.J. Morbidade e mortalidade relacionadas a medicamentos no Brasil: Revisão sistemática de estudos observacionais. Rev. Ciênc. Farm Básica Appl. 2014, 35, 519–532. [Google Scholar]
- Ramos, G.V.; Guaraldo, L.; Japiassu, A.M.; Bozza, F.A. Comparison of two databases to detect potential drug-drug interactions between prescriptions of HIV/AIDS patients in critical care. J. Clin. Pharm. Ther. 2015, 40, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Sönnerstam, E.; Sjölander, M.; Lövheim, H.; Gustafsson, M. Clinically relevant drug-drug interactions among elderly people with dementia. Eur. J. Clin. Pharmacol. 2018, 74, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- Leger, D.Y.; Moreau, S.; Signol, N.; Fargeas, J.-B.; Picat, M.-A.; Penot, A.; Abraham, J.; Laroche, M.-L.; Bordessoule, D. Polypharmacy, potentially inappropriate medications and drug-drug interactions in geriatric patients with hematologic malignancy: Observational single-center study of 122 patients. J. Geriatr. Oncol. 2017, 9, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Apovian, C.M.; Aronne, L.J.; Bessesen, D.H.; McDonnell, M.E.; Murad, M.H.; Pagotto, U.; Ryan, D.H.; Still, C.D. Pharmacological Management of Obesity: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2014, 100, 342–362. [Google Scholar] [CrossRef] [PubMed]
- Narayanaswami, V.; Dwoskin, L.P. Obesity: Current and potential pharmacotherapeutics and targets. Pharmacol. Ther. 2017, 170, 116–147. [Google Scholar] [CrossRef] [PubMed]
- Solmi, F.; Morris, S. Association between childhood obesity and use of regular medications in the UK: Longitudinal cohort study of children aged 5–11 years. BMJ Open 2015, 5, e007373. [Google Scholar] [CrossRef] [PubMed]
- Silveira, E.A.; Rosa, L.P.S.; Santos, A.S.A.C.; Cardoso, C.K.S. Type 2 Diabetes Mellitus in Class II and III Obesity: Prevalence, Associated Factors, and Correlation between Glycemic Parameters and Body Mass Index. Int. J. Environ. Res. Public Health 2020, 17, 3930. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.S.; Rodrigues, A.P.S.; Rosa, L.P.; Sarrafzadegan, N.; Silveira, E.A. Cardiometabolic risk factors and Framingham Risk Score in severely obese patients: Baseline data from DieTBra trial. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Truven Health Analytics. Micromedex; Truven Health Analytics: Greenwood Village, CO, USA, 2017; Available online: http://www.thomsonhc.com (accessed on 27 January 2017).
- Abep. Critério Padrão de Classificação Econômica Brasil. Available online: http://www.abep.org/criterio-brasil (accessed on 27 January 2017).
- Bloomfield, K.; Allamani, A.; Beck, F.; Bergmark, K.H.; Csemy, L.; Eisenbach-Stangl, I.; Elekes, Z.; Gmel, G.; Ker-Correa, F.; Knibbe, R.; et al. Gender, Culture and Alcohol Problems: A Multi-National Study; Project Final Report; Institute for Medical Informatics: Berlin, Germany, 2005. [Google Scholar]
- Organizacíón Panamericana de la Salud. Guias Para el Control y Monitoreo de la Epidemia Tabaquica; Organizacíón Panamericana de la Salud: Caracas, Venezuela, 1995. [Google Scholar]
- Obesity: Preventing and Managing the Global Epidemic; Report of a WHO Consultation; TRS 931; World Health Organization: Geneva, Switzerland, 2000.
- Drozda, J.; Messer, J.V.; Spertus, J.; Abramowitz, B.; Alexander, K.; Beam, C.T.; Bonow, R.O.; Burkiewicz, J.S.; Crouch, M.; Goff, D.C.; et al. Performance Measures for Adults with Coronary Artery Disease and Hypertension: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Performance Measures and the American Medical Association. Circulation 2011, 124, 248–270. [Google Scholar]
- American Diabetes Association. Standard of medical care in diabetes—2017. Diabetes Care 2017, 40 (Suppl. S1), s4–s128. [Google Scholar] [CrossRef]
- National Cholesterol Education Program Expert Panel on Detection E and T of HBC in A. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143. [Google Scholar] [CrossRef]
- Keith, S.W.; Redden, D.T.; Katzmarzyk, P.T.; Boggiano, M.M.; Hanlon, E.C.; Benca, R.M.; Ruden, D.; Pietrobelli, A.; Barger, J.L.; Fontaine, K.R.; et al. Putative contributors to the secular increase in obesity: Exploring the roads less traveled. Int. J. Obes. 2006, 30, 1585–1594. [Google Scholar] [CrossRef]
- Apovian, C.M. Obesity: Definition, comorbidities, causes, and burden. Am. J. Manag. Care 2016, 22, s176–s185. [Google Scholar] [PubMed]
- Domecq, J.P.; Prutsky, G.; Leppin, A.; Sonbol, M.B.; Altayar, O.; Undavalli, C.; Wang, Z.; Elraiyah, T.; Brito, J.P.; Mauck, K.F.; et al. Drugs Commonly Associated with Weight Change: A Systematic Review and Meta-analysis. J. Clin. Endocrinol. Metab. 2015, 100, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Guharoy, R. Polypharmacy: America’s other drug problem. Am. J. Health Pharm. 2017, 74, 1305–1306. [Google Scholar] [CrossRef]
- World Health Organization. The Role of the Pharmacist in Self-Care and Self-Medication Contents; World Health Organization: Geneva, Switzerland, 1998. [Google Scholar]
- World Health Organization. WHO Collaborating Centre for Drug Statistics Methodology, Guidelines for ATC Classification and DDD Assignment; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Kennedy-Dixon, T.-G.; Gossell-Williams, M.; Hall, J.; Anglin-Brown, B. The prevalence of major potential drug-drug interactions at a University health centre pharmacy in Jamaica. Pharm. Pr. 2015, 13, 601. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes—2020. Diabetes Care 2020, 43, S98–S110. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ciaraldi, T.P.; Samad, A.M. Tissue Factor Expression in Obese Type 2 Diabetic Subjects and Its Regulation by Antidiabetic Agents. J. Obes. 2015, 2015, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, M.L.; Stengel, B.; Legrand, K.; Briançon, S.; Jacquelinet, C.; Combe, C.; Fouque, D.; Massy, Z.A.; Laville, M.; Frimat, L.; et al. Obesity phenotype and patient-reported outcomes in moderate and severe chronic kidney disease: A cross-sectional study from the CKD-REIN cohort study. Qual. Life Res. 2019, 28, 1873–1883. [Google Scholar] [CrossRef] [PubMed]
- Al Hamid, A.; Ghaleb, M.; Aljadhey, H.; Aslanpour, Z. A systematic review of qualitative research on the contributory factors leading to medicine-related problems from the perspectives of adult patients with cardiovascular diseases and diabetes mellitus. BMJ Open 2014, 4, e005992. [Google Scholar] [CrossRef] [PubMed]
- Ikäheimo, I.; Karjalainen, M.; Tiihonen, M.; Haanpää, M.; Kautiainen, H.; Saltevo, J.; Mäntyselkä, P. Clinically relevant drug-drug interactions and the risk for drug adverse effects among home-dwelling older persons with and without type 2 diabetes. J. Clin. Pharm. Ther. 2019, 44, 735–741. [Google Scholar] [CrossRef]
- Saastamoinen, L.K.; Verho, J. Register-based indicators for potentially inappropriate medication in high-cost patients with excessive polypharmacy. Pharmacoepidemiol. Drug Saf. 2015, 24, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Maideen, N.M.P.; Jumale, A.; Balasubramaniam, R. Drug Interactions of Metformin Involving Drug Transporter Proteins. Adv. Pharm. Bull. 2017, 7, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Kalafutova, S.; Juraskova, B.; Vlcek, J. The impact of combinations of non-steroidal anti-inflamatory and anti-hypertensive agents on blood-pressure. Adv. Clin. Exp. Med. 2014, 23, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Fournier, J.P.; Lapeyre-Mestre, M.; Sommet, A.; Dupouy, J.; Poutrain, J.C.; Montastruc, J.L. Laboratory monitoring of patients treated with antihypertensive drugs and newly exposed to nonsteroidal anti-inflammatory drugs: A cohort study. PLoS ONE 2012, 7, 1–6. [Google Scholar] [CrossRef]
- Srinivasan, A.; De Cruz, P. Review article: A practical approach to the clinical management of NSAID enteropathy. Scand. J. Gastroenterol. 2017, 52, 941–947. [Google Scholar] [CrossRef]
| Severity | n | % | More Prevalent Pairs of Drugs with Potential Drug Interactions | Management * |
|---|---|---|---|---|
| Contra-indicated | 1 | 0.42% | Trometamol cetrolac + Naproxen | Concurrent use may cause in enhanced gastrointestinal adverse effects (peptic ulcers, gastrointestinal bleeding and/or perforation. |
| Major | 95 | 40.25% | Diclofenac + Hydrochlorothiazide | Concurrent use may result in impaired renal function. When co-administration is required, monitor renal function at the beginning and during treatment, as well as blood pressure control. |
| Acetylsalicylic acid + Metformin | Concomitant use may cause hypoglycemia. A more frequent monitoring of blood glucose is suggested. Advise patients to recognize early signs and symptoms of hypoglycemia. | |||
| Diclofenac + Acetylsalicylic acid | Concurrent use may result in impaired renal function. When co-administration is required, monitor renal function at the beginning and during treatment, as well as blood pressure control. | |||
| Diclofenac + Fluoxetine | Concurrent use results in an increased risk of bleeding. Monitoring the patient for signs and symptoms of bleeding may be necessary. | |||
| Hydrochlorothiazide + Acetylsalicylic acid | Concurrent use may result in impaired renal function. When co-administration is required, monitor renal function at the beginning and during treatment, as well as blood pressure control. | |||
| Moderate | 137 | 58.05% | Ethinylestradiol + Caffeine | Concurrent use may cause central nervous stimulation and insomnia. Advise patients to decrease caffeine intake while using contraceptives. |
| Diclofenac + Losartan | Concurrent use may result in impaired renal function. When co-administration is required, monitor renal function at the beginning and during treatment, as well as blood pressure control. | |||
| Hydrochlorothiazide + Enalapril | Concurrent use may result in an excessive increase in blood pressure reduction. At the beginning of therapy, it may be necessary to decrease diuretic dose or increase salt intake. If such measures are not effective, reduce the initial dose of Enalapril. | |||
| Hydrochlorothiazide + Propranolol | Concurrent use may cause changes in patient’s glycemic and lipid profile. Monitoring of such parameters is suggested. | |||
| Enalapril + Metformin | Concomitant use may cause hypoglycemia. A more frequent monitoring of blood glucose is suggested. Advise patients to recognize early signs and symptoms of hypoglycemia. | |||
| Captopril + Metformin | Concomitant use may cause hypoglycemia. A more frequent monitoring of blood glucose is suggested. Advise patients to recognize early signs and symptoms of hypoglycemia. | |||
| Hydrochlorothiazide + Salbutamol | Concurrent use may increase the risk of hypokalemia as well as changes in electrocardiogram. Monitoring of potassium levels as well as cardiac function is recommended. | |||
| Diclofenac + Captopril | Concurrent use may result in impaired renal function. When co-administration is required, monitor renal function at the beginning and during treatment, as well as blood pressure control. | |||
| Hydrochlorothiazide + Captopril | Concurrent use may result in an excessive increase in blood pressure reduction. At the beginning of therapy, it may be necessary to decrease diuretic dose or increase salt intake. If such measures are not effective, reduce the initial dose of Captopril. | |||
| Levothyroxine + Omeprazole | Concurrent use may compromise the efficacy of Levothyroxine. Advise patient to use Levothyroxine four hours before or after omeprazole. Monitoring of TSH levels is suggested. | |||
| Total | 233 1 | 98.72 2 | ||
| Variables | n (%) | PDI Presence | PR (CI 95%) | p Value |
|---|---|---|---|---|
| Sex | ||||
| Female | 128 (85.33) | 63 (49.22) | 1.00 | 0.644 * |
| Male | 22 (14.67) | 12 (54.55) | 0.90 (0.59–1.37) | |
| Age | ||||
| 18–39 | 76 (50.67) | 34 (44.74) | 1.00 | 0.139 ** |
| 40–49 | 53 (35.33) | 28 (52.83) | 1.18 (0.82–1.68) | |
| ≥50 | 21 (14.00) | 13 (61.90) | 1.38 (0.90–2.10) | |
| Years of study | ||||
| <10 years | 75 (50.00) | 36 (48.00) | 1.00 | 0.624 * |
| >10 years | 75 (50.00) | 39 (52.00) | 1.08 (0.78–1.49) | |
| Skin color | ||||
| White | 46 (30.67) | 21 (45.65) | 1.00 | 0.530 ** |
| Brown | 83 (55.33) | 43 (51.81) | 1.13 (0.77–1.65) | |
| Black | 21 (14.00) | 11 (52.38) | 1.14 (0.68–1.92) | |
| Economic class | ||||
| Class A/B | 34 (22.67) | 18 (52.94) | 1.13 (0.76–1.66) | 0.556 * |
| Class C | 92 (61.33) | 43 (46.74) | 1.00 | |
| Class D/E | 24 (16.00) | 14 (58.33) | 1.24 (0.83–1.86) | |
| Lives with partner | ||||
| Yes | 55 (36.67) | 28 (50.91) | 1.02(0.73–143) | 0.865 * |
| No | 95 (63.33) | 47 (49.47) | 1.00 | |
| Smoking | ||||
| Non-smoker | 101 (67.33) | 48 (47.52) | 1.00 | 0.384 * |
| Smoker/Ex-smoker | 49 (32.67) | 27 (55.10) | 1.15 (0.83–1.60) | |
| Alcohol intake 1 | ||||
| No | 13 (15.48) | 5 (38.46) | 1.00 | 0.318 * |
| Yes | 71 (84.52) | 38 (53.52) | 1.39 (0.67–2.87) | |
| Variables | n (%) | PDI Presence | PR (CI 95%) | p Value * |
|---|---|---|---|---|
| Body Mass Index | ||||
| 35–39 | 27 (18.00) | 13 (48.15) | 1.00 | 0.832 |
| ≥40 | 123 (82.00) | 62 (50.41) | 1.04 (0.68–1.60) | |
| Arterial Hypertension | ||||
| No | 65 (43.33) | 15 (23.08) | 1.00 | <0.001 |
| Yes | 85 (56.67) | 60 (70.59) | 3.05 (1.91–4.87) | |
| Diabetes | ||||
| No | 90 (60.00) | 38 (42.22) | 1.00 | 0.020 |
| Yes | 60 (40.00) | 37 (61.67) | 1.46 (1.06–2.00) | |
| Hypercholesterolemia | ||||
| No | 94 (62.67) | 45 (47.87) | 1.00 | 0.500 |
| Yes | 56 (37.33) | 30 (53.57) | 1.11 (0.80–1.54) | |
| Self-reported morbidities | ||||
| ≤3 | 89 (59.33) | 30 (33.71) | 1.00 | <0.001 |
| ≥4 | 61 (40.67) | 45 (73.77) | 2.18 (1.57–3.03) | |
| Drugs that cause weight gain | ||||
| No | 106 (70.67) | 42 (39.62) | 1.00 | <0.001 |
| Yes | 44 (29.33) | 33 (75.00) | 1.89 (1.41–2.53) | |
| Self-medication | ||||
| 0 | 23 (15.33) | 12 (52.17) | 1.13 (0.72–1.78) | 0.463 |
| 1–2 | 89 (59.33) | 41 (46.07) | 1.00 | |
| ≥3 | 38 (25.33) | 22 (57.89) | 1.25 (0.88–1.78) | |
| Polypharmacy | ||||
| No | 101 (67.33) | 29 (28.71) | 1.00 | <0.001 |
| Yes | 49 (32.67) | 46 (93.88) | 3.26 (2.38–4.48) | |
| Variables | Adjusted Prevalence Ratio | Adjusted CI 95% | p Value * |
|---|---|---|---|
| Arterial Hypertension | |||
| No | 1 | ||
| Yes | 1.82 | 1.10–3.04 | 0.020 |
| Diabetes | |||
| No | 1 | ||
| Yes | 0.60 | 0.45–0.81 | 0.001 |
| Drugs that cause weight gain | |||
| No | 1 | ||
| Yes | 1.35 | 0.99–1.84 | 0.059 |
| Polypharmacy | |||
| No | 1 | ||
| Yes | 3.12 | 2.17–4.50 | <0.001 |
| ATC Classification | n (%) | PDI Presence | p Value | |
|---|---|---|---|---|
| A | Alimentary Tract and metabolism | 57 (38.00%) | 39 (68.42%) | <0.001 * |
| B | Blood and forming blood organs | 11 (7.33%) | 10 (90.91%) | 0.005 ** |
| C | Cardiovascular system | 65 (43.33%) | 48 (73.85%) | <0.001 * |
| D | Dermatologicals | 0 | 0 | - |
| G | Genito urinary system and sex hormones | 12 (8.00%) | 7 (58.33%) | 0.547 * |
| H | Systemic hormonal preparations, excluding sex hormones and insulins | 10 (6.67%) | 10 (100%) | 0.001 ** |
| J | Antiinfectives for systemic use | 6 (4.00%) | 4 (66.67%) | 0.341 ** |
| L | Antineoplasic and immunomodulating agents | 0 | 0 | - |
| M | Musculo-skeletal system | 97 (64.67%) | 60 (61.86%) | <0.001 * |
| N | Nervous system | 86 (57.33%) | 44 (51.16%) | 0.741 * |
| P | Antiparasitic products, insecticides and repellents | 1 (0.67%) | 0 (0.00%) | - |
| R | Respiratory system | 17 (11.33%) | 10 (58.82%) | 0.440 * |
| S | Sensory organs | 1 (0.67%) | 0 (0.00%) | - |
| V | Various | 0 | 0 | - |
| NC | Non-Classifiable | 16 (10.67%) | 10 (62.50%) | 0.290 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Modesto, A.C.F.; Silveira, E.A.; dos Santos Rodrigues, A.P.; Lima, D.M.; Provin, M.P.; Amaral, R.G. Prevalence of Drug Interaction in Severely Obese Individuals and Associated Factors: Baseline Results from a Clinical Trial. Sci. Pharm. 2020, 88, 48. https://doi.org/10.3390/scipharm88040048
Modesto ACF, Silveira EA, dos Santos Rodrigues AP, Lima DM, Provin MP, Amaral RG. Prevalence of Drug Interaction in Severely Obese Individuals and Associated Factors: Baseline Results from a Clinical Trial. Scientia Pharmaceutica. 2020; 88(4):48. https://doi.org/10.3390/scipharm88040048
Chicago/Turabian StyleModesto, Ana Carolina Figueiredo, Erika Aparecida Silveira, Ana Paula dos Santos Rodrigues, Dione Marçal Lima, Mércia Pandolfo Provin, and Rita Goreti Amaral. 2020. "Prevalence of Drug Interaction in Severely Obese Individuals and Associated Factors: Baseline Results from a Clinical Trial" Scientia Pharmaceutica 88, no. 4: 48. https://doi.org/10.3390/scipharm88040048
APA StyleModesto, A. C. F., Silveira, E. A., dos Santos Rodrigues, A. P., Lima, D. M., Provin, M. P., & Amaral, R. G. (2020). Prevalence of Drug Interaction in Severely Obese Individuals and Associated Factors: Baseline Results from a Clinical Trial. Scientia Pharmaceutica, 88(4), 48. https://doi.org/10.3390/scipharm88040048







