Curcumin-Loaded Nanoemulsion for Better Cellular Permeation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Curcumin-Loaded Nanoemulsion
2.3. Morphological Characterisation
2.4. Chemical Characterisation and Antioxidant Activity
2.5. In Vitro Skin Permeation of Nano-Encapsulated Curcumin
2.6. Fluorescent Imaging of Permeated Curcumin
2.7. Scratch Assay for Wound Healing
2.8. Cell Cultivation and Proliferation Assay
3. Results
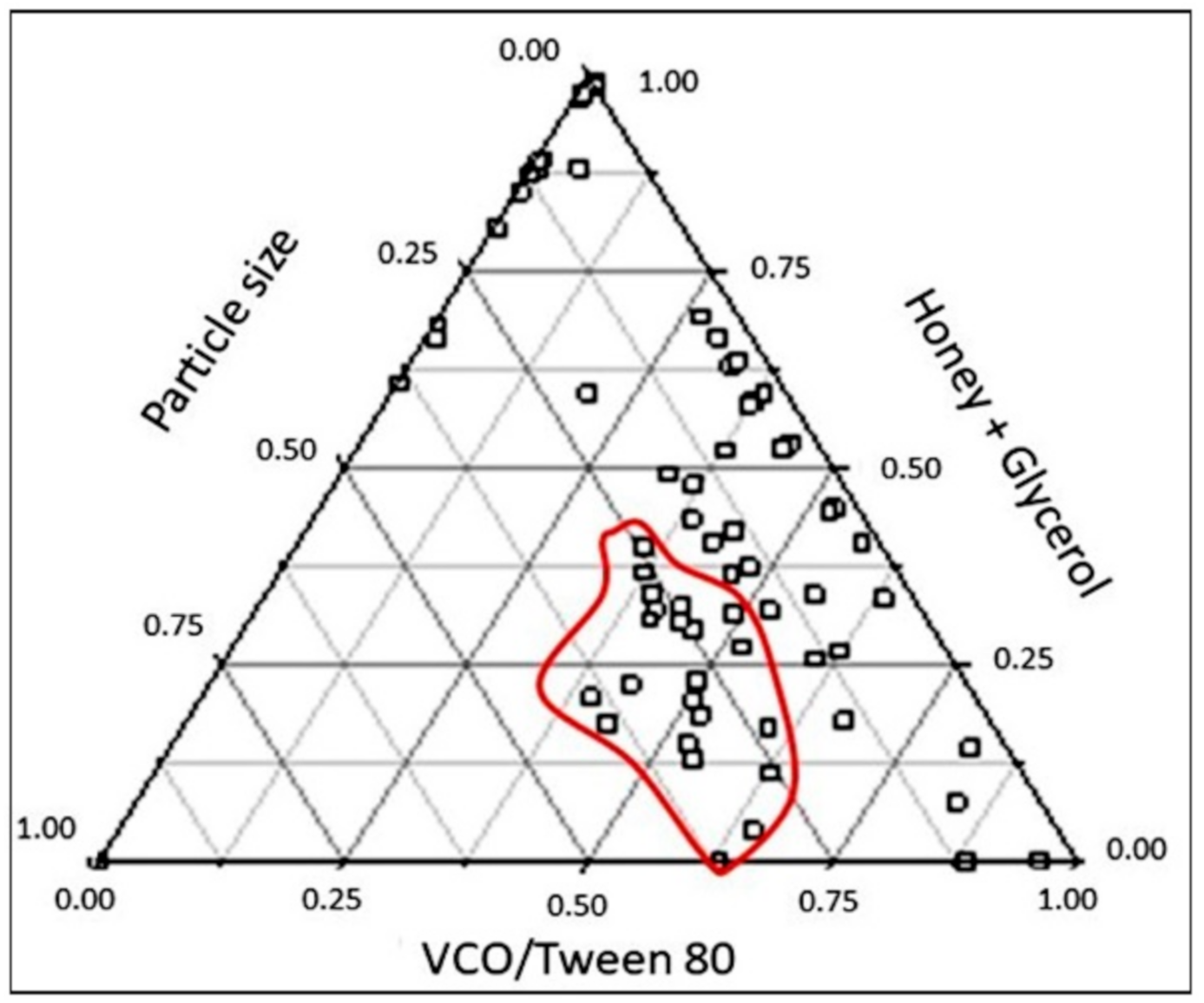
3.1. One-Factor-At-A-Time Optimization of Nanoemulsion Preparation
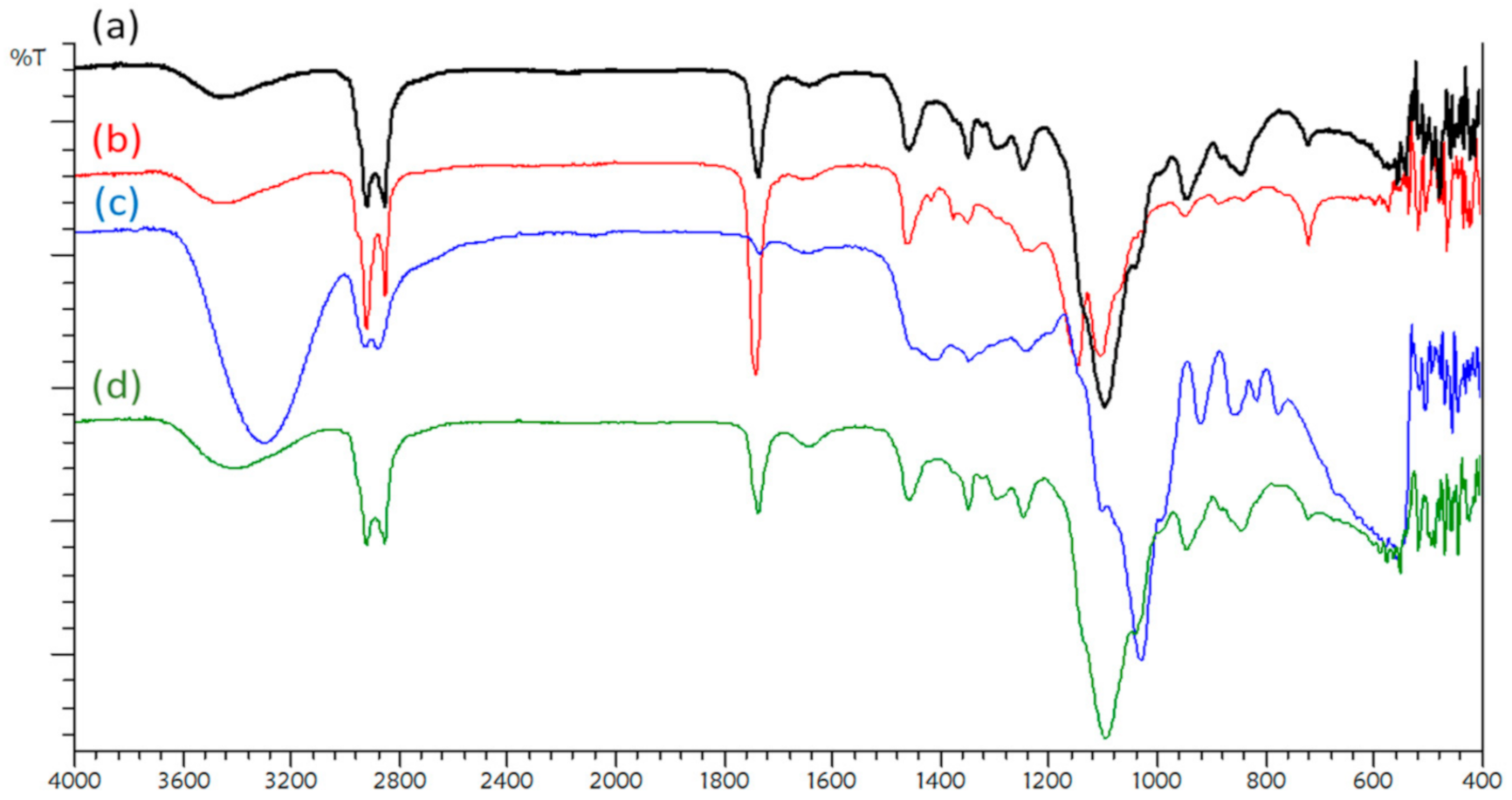
3.2. FTIR Spectrum Analysis
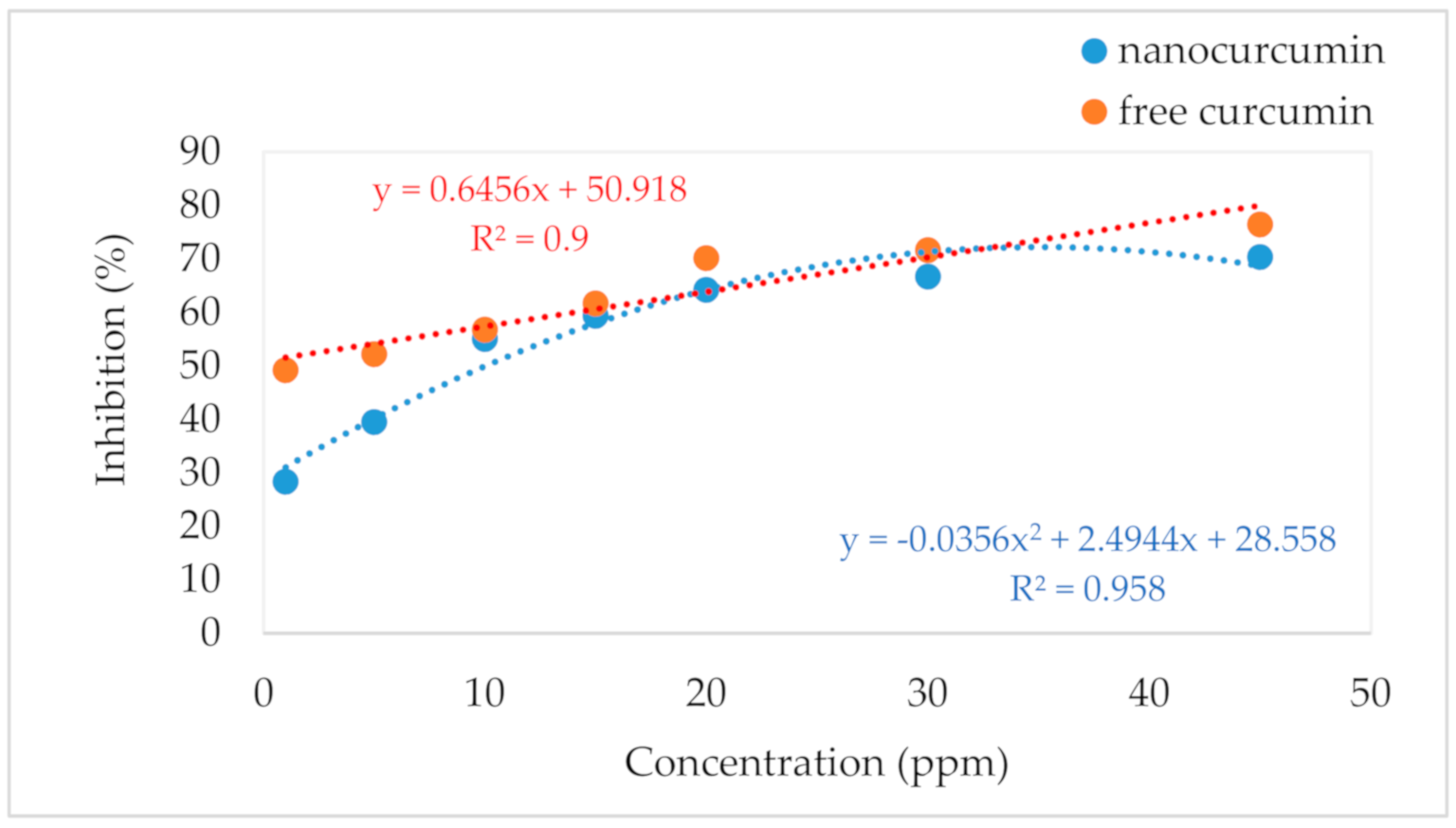
3.3. Antioxidant Activity
3.4. Skin Permeation and Deposition Study
3.5. Release Kinetics
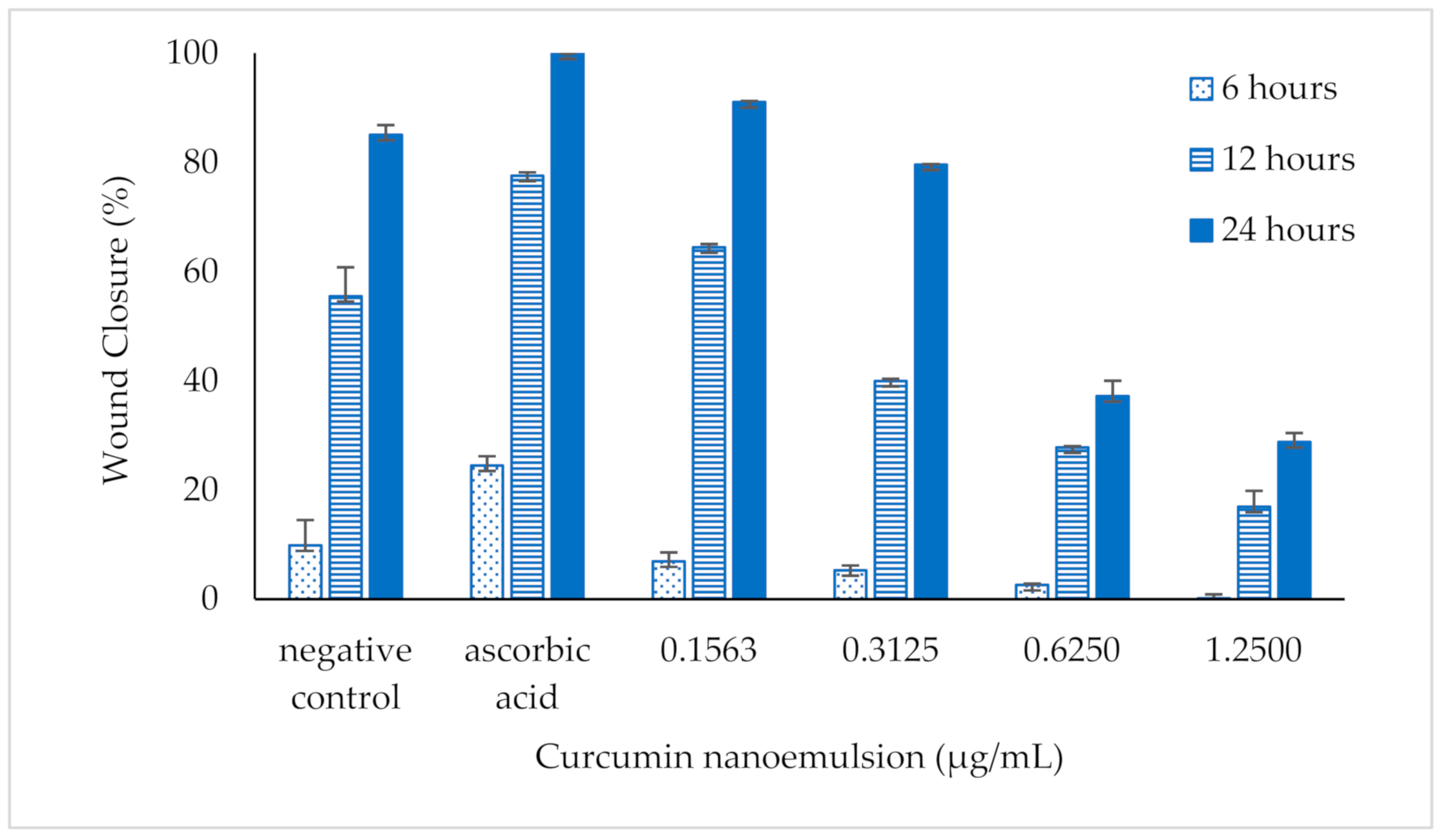
3.6. Performance on Wound Closure by Scratch Assay
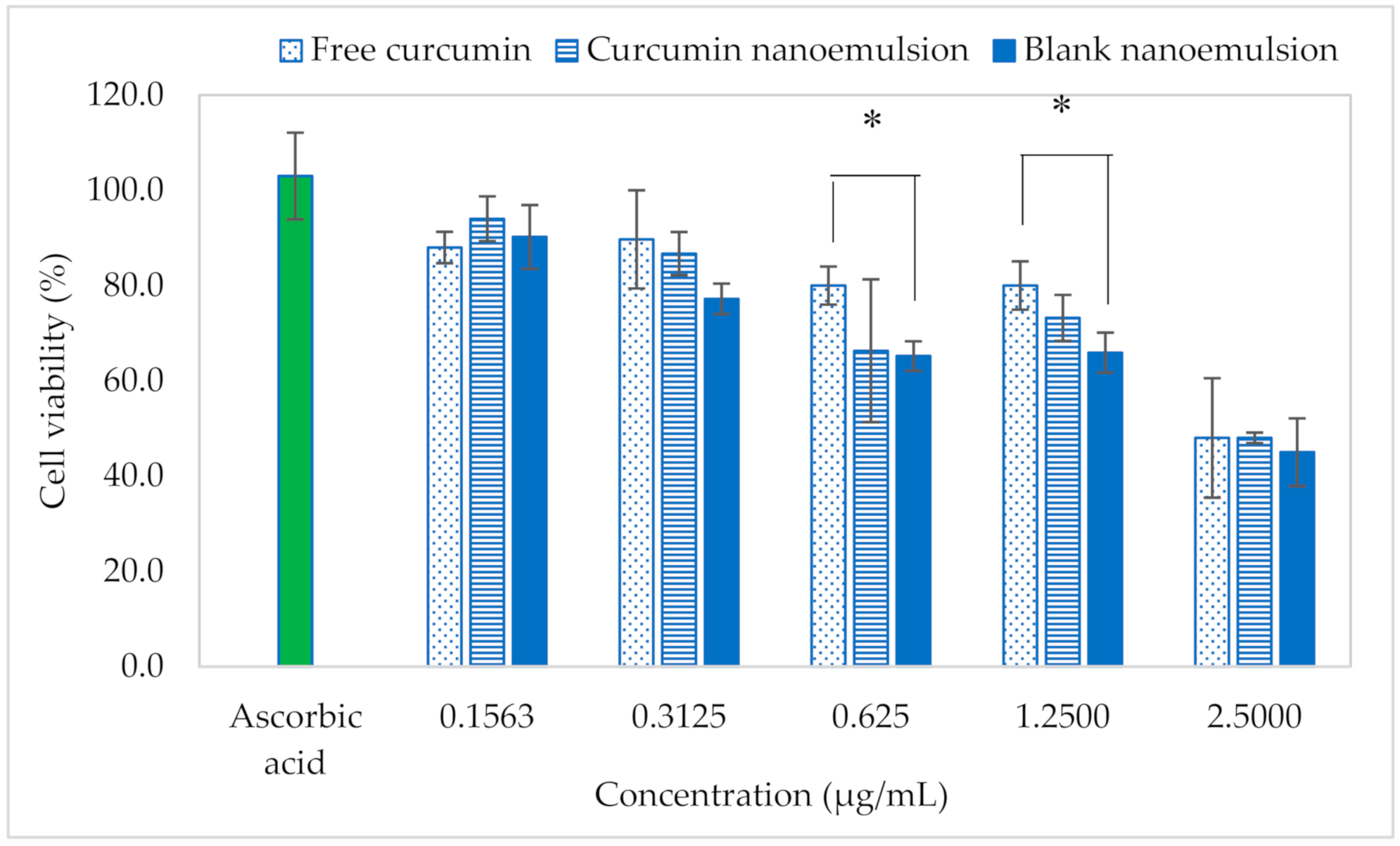
3.7. Cytotoxic Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hashish, E.A.; Elgaml, S.A. Hepatoprotective and nephroprotective effect of curcumin against copper toxicity in rats. Indian. J. Clin. Biochem. 2016, 31, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Nagahama, K.; Utsumi, T.; Kumano, T.; Maekawa, S.; Oyama, N.; Kawakami, J. Discovery of a new function of curcumin which enhances its anticancer therapeutic potency. Sci. Rep. 2016, 6, 30962. [Google Scholar] [CrossRef] [PubMed]
- Azami, S.J.; Teimouri, A.; Keshavarz, H.; Amani, A.; Esmaeili, F.; Hasanpour, H.; Elikaee, S.; Salehiniya, H.; Shojaee, S. Curcumin nanoemulsion as a novel chemical for the treatment of acute and chronic toxoplasmosis in mice. Int. J. Nanomedicine 2018, 13, 7363–7374. [Google Scholar] [CrossRef] [PubMed]
- Sari, T.P.; Mann, B.; Kumar, R.; Singh, R.R.B.; Sharma, R.; Bhardwaj, M.; Athira, S. Preparation and characterization of nanoemulsion encapsulating curcumin. Food Hydrocoll. 2015, 43, 540–546. [Google Scholar] [CrossRef]
- Rachmawati, H.; Budiputra, D.K.; Mauludin, R. Curcumin nanoemulsion for transdermal application: Formulation and evaluation. Drug Dev. Ind. Pharm. 2015, 41, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Ahmad, R.; Al-Qudaihi, A.; Alaseel, S.E.; Fita, I.Z.; Khalid, M.S.; Pottoo, F.H. Preparation of a novel curcumin nanoemulsion by ultrasonication and its comparative effects in wound healing and the treatment of inflammation. RSC. Adv. 2019, 9, 20192–20206. [Google Scholar] [CrossRef]
- Guerrero, S.; Inostroza-Riquelme, M.; Contreras-Orellana, P.; Diaz-Garcia, V.; Lara, P.; Vivanco-Palma, A.; Cárdenas, A.; Miranda, V.; Robert, P.; Leyton, L.; et al. Curcumin-loaded nanoemulsion: A new safe and effective formulation to prevent tumor reincidence and metastasis. Nanoscale 2018, 10, 22612–22622. [Google Scholar] [CrossRef]
- De Matos, R.P.A.; Calmon, M.F.; Amantino, C.F.; Villa, L.L.; Primo, F.L.; Claudio Tedesco, A.; Rahal, P. Effect of curcumin-nanoemulsion associated with photodynamic therapy in cervical carcinoma cell lines. BioMed. Res. Int 2018, 2018, 4057959. [Google Scholar] [CrossRef] [PubMed]
- Mohan, P.K.; Sreelakshmi, G.; Muraleedharan, C.V.; Joseph, R. Water soluble complexes of curcumin with cyclodextrins: Characterization by FT-Raman spectroscopy. Vib. Spectrosc. 2012, 62, 77–84. [Google Scholar] [CrossRef]
- Guillén, M.D.; Cabo, N. Characterization of edible oils and lard by fourier transform infrared spectroscopy. Relationships between composition and frequency of concrete bands in the fingerprint region. J. Am. Oil. Chem. Soc. 1997, 74, 1281–1286. [Google Scholar] [CrossRef]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for nutrition and health: A review. J. Am. Coll. Nutr. 2008, 27, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Machado De-Melo, A.A.; Almeida-Muradian, L.B.D.; Sancho, M.T.; Pascual-Maté, A. Composition and properties of Apis mellifera honey: A review. J. Apicul. Res. 2018, 57, 5–37. [Google Scholar] [CrossRef]
- Kędzierska-Matysek, M.; Matwijczuk, A.; Florek, M.; Barłowska, J.; Wolanciuk, A.; Matwijczuk, A.; Chruściel, E.; Walkowiak, R.; Karcz, D.; Gładyszewska, B. Application of FTIR spectroscopy for analysis of the quality of honey. BIO Web of Conferences 2018, 10, 02008. [Google Scholar]
- Wiebe, J.P.; Dinsdale, C.J. Inhibition of cell proliferation by glycerol. Life Sci. 1991, 48, 1511–1517. [Google Scholar] [CrossRef]
- ISO10993-5. Biological evaluation of medical devices—Part. 5: Tests for in vitro cytotoxicity, third edition; International Standard: Geneva, Switzerland, 2009; p. 34. [Google Scholar]
- Zhang, W.; Zhu, D.; Qin, Y.; Ou, W.; Bao, Y.; Song, L.; Zhang, Q. Effects of compositions on the stability of polyols-in-oil-in-water (P/O/W) multiple emulsions. J. Disper. Sci. Technol. 2017, 39, 1344–1351. [Google Scholar] [CrossRef]
- Aniesrani Delfiyaa, D.S.; Thangavel, K. Evaluation of in vitro release pattern of curcumin loaded egg albumin nanoparticles prepared using acetone as desolvation agent. Curr. Tr. Biotechnol. Pharm. 2016, 10, 125–135. [Google Scholar]
- Wojcik-Pastuszka, D.; Krzak, J.; Macikowski, B.; Berkowski, R.; Osiński, B.; Musiał, W. Evaluation of the release kinetics of a pharmacologically active substance from model intra-articular implants replacing the cruciate ligaments of the knee. Materials 2019, 12, 1202. [Google Scholar] [CrossRef]
- Schäfer, M.; Werner, S. Transcriptional control of wound repair. Annu. Rev. Cell Dev. Biol. 2007, 23, 69–92. [Google Scholar] [CrossRef]
- Liu, M.L.; Zang, T.; Zou, Y.; Chang, J.C.; Gibson, J.R.; Huber, K.M.; Zhang, C.L. Small molecules enable neurogenin 2 to efficiently convert human fibroblasts into cholinergic neurons. Nat. Comm. 2013, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- De Campos, P.S.; Matte, B.F.; Diel, L.F.; Jesus, L.H.; Bernardi, L.; Alves, A.M.; Rados, P.V.; Lamers, M.L. Low doses of curcuma longa modulates cell migration and cell–cell adhesion. Phytother. Res. 2017, 31, 1433–1440. [Google Scholar] [CrossRef] [PubMed]



| Order | Kinetic Model | Linearized Kinetic Model | Constant (k) | Correlation Coefficient (r2) | ||
|---|---|---|---|---|---|---|
| Free Curcumin | Curcumin Nanoemulsion | Free Curcumin | Curcumin Nanoemulsion | |||
| Zero | Ct − Co = k.t | Ct = Co − k.t | 0.5701 mg/h | 0.806 mg/h | 0.7867 | 0.8112 |
| First | ln Ct = ln Co − k.t | 0.0674 mg/h | 0.0706 mg/h | 0.5291 | 0.4908 | |
| Second | 0.0062 mg−1.h−1 | 0.0048 mg−1.h−1 | 0.3244 | 0.2893 | ||
| Higuchi | Q = k.t1/2 | Q = k.t1/2 | 0.3101 mg.h−1/2 | 0.2225 mg.h−1/2 | 0.9533 | 0.9512 |
| Korsmeyer–Peppas | n = 0.2965, k = 0.6531 h−n | n = 0.3057, k = 0.8816 h−n | 0.9867 | 0.9695 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Md Saari, N.H.; Chua, L.S.; Hasham, R.; Yuliati, L. Curcumin-Loaded Nanoemulsion for Better Cellular Permeation. Sci. Pharm. 2020, 88, 44. https://doi.org/10.3390/scipharm88040044
Md Saari NH, Chua LS, Hasham R, Yuliati L. Curcumin-Loaded Nanoemulsion for Better Cellular Permeation. Scientia Pharmaceutica. 2020; 88(4):44. https://doi.org/10.3390/scipharm88040044
Chicago/Turabian StyleMd Saari, Nur Hulwani, Lee Suan Chua, Rosnani Hasham, and Leny Yuliati. 2020. "Curcumin-Loaded Nanoemulsion for Better Cellular Permeation" Scientia Pharmaceutica 88, no. 4: 44. https://doi.org/10.3390/scipharm88040044
APA StyleMd Saari, N. H., Chua, L. S., Hasham, R., & Yuliati, L. (2020). Curcumin-Loaded Nanoemulsion for Better Cellular Permeation. Scientia Pharmaceutica, 88(4), 44. https://doi.org/10.3390/scipharm88040044






