The Effect of Cladode Drying Techniques on the Prebiotic Potential and Molecular Characteristics of the Mucilage Extracted from Opuntia ficus-indica and Opuntia joconostle
Abstract
1. Introduction
2. Materials and Methods
2.1. Vegetable Material and Drying Methods
2.2. Purification of the Powdered Cladodes
2.3. Purification of Methanolic Extracts
2.4. Mucilage Extraction
2.5. Trifluoroacetic Acid (TFA) Hydrolysis
2.6. Thin Layer Chromatography (TLC)
2.7. High Performance Anion Exchange Chromatography–Pulsed Amperometric Detection (HPAEC-PAD)
2.8. Size Exclusion Chromatography (SEC) Analysis
2.9. Total Carbohydrate Content
2.10. Protein Content
2.11. Mineral Content
2.12. Evaluation of Prebiotic Potential
2.13. Statistical Analysis
3. Results
3.1. Drying Methods and Composition of the Cladodes
3.2. Mucilage Composition
3.2.1. Main Components
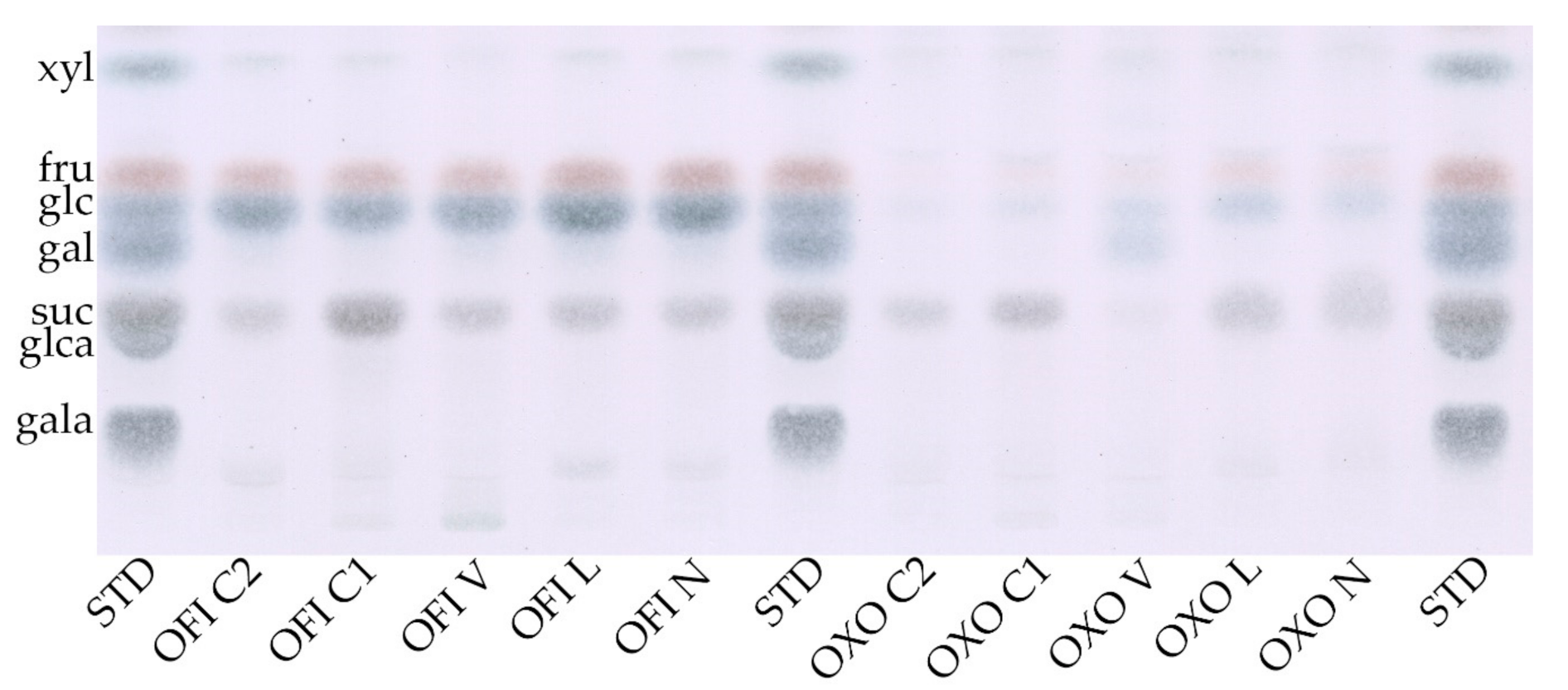
3.2.2. Monosaccharide Composition
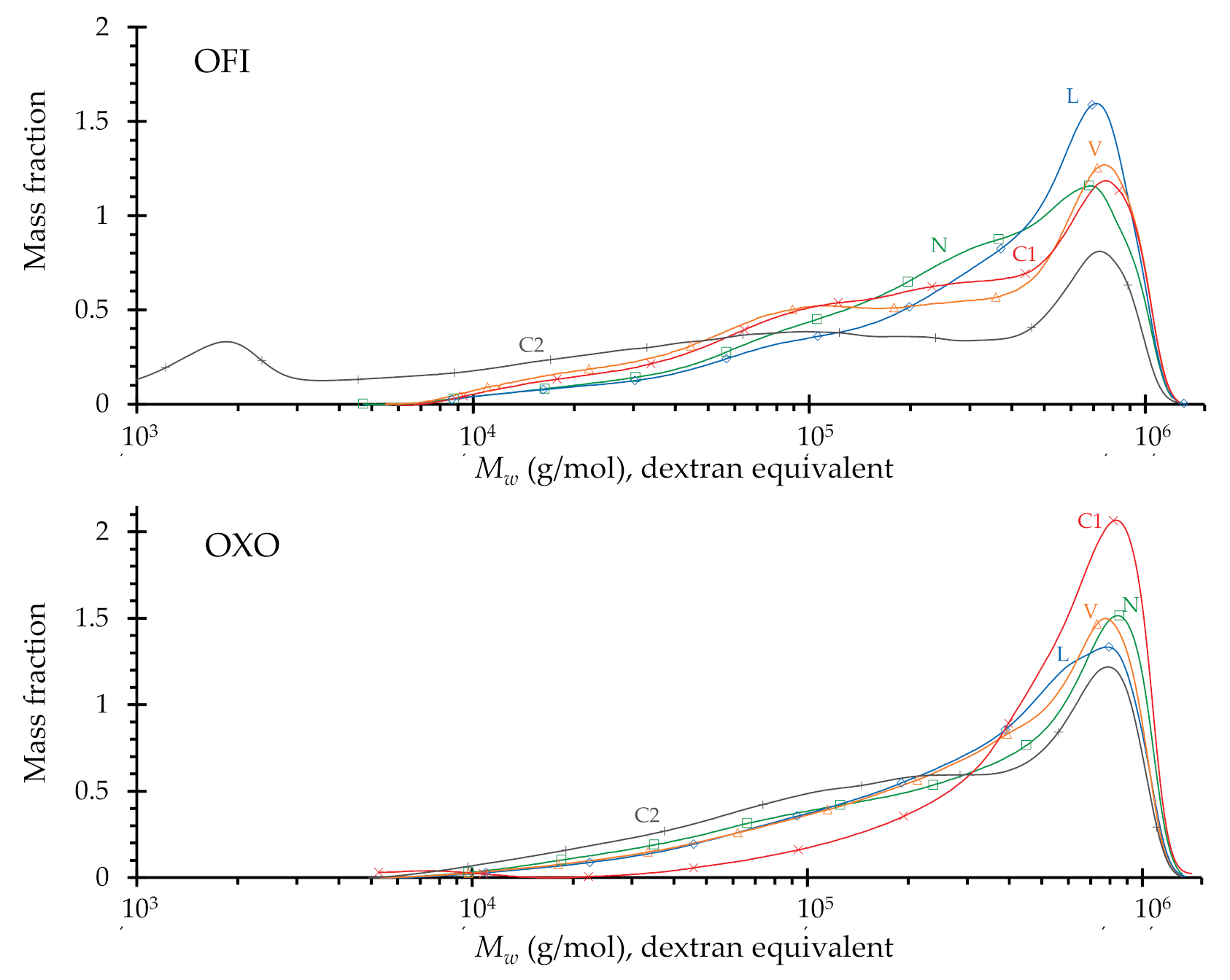
3.2.3. Molecular Dimensions
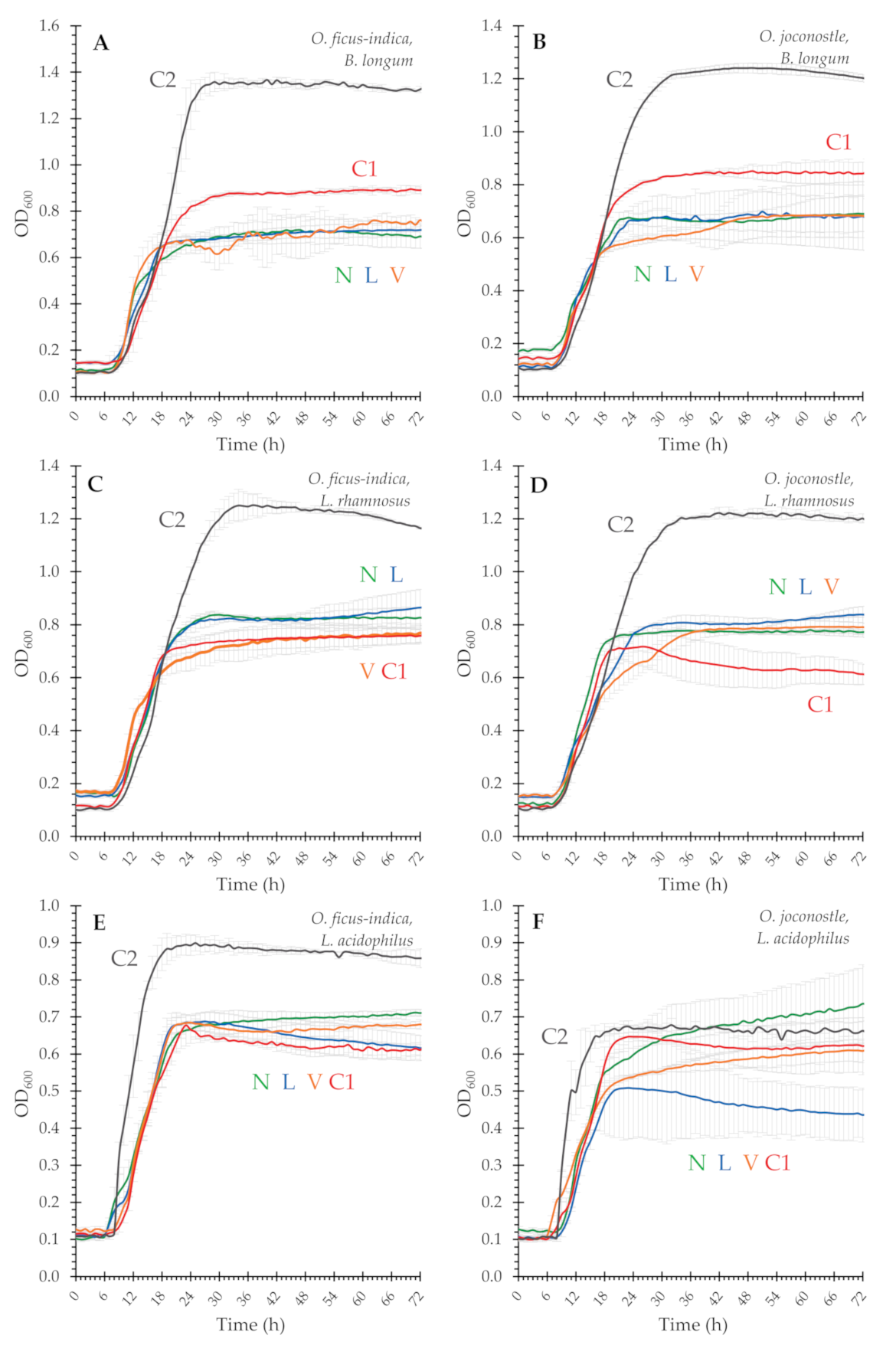
3.3. Prebiotic Potential of Mucilage
4. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nobel, P.S.; Bobich, E.G. Environmental Biology. In Cacti: Biology and Uses; Nobel, P.S., Ed.; University of California Press: Berkeley, CA, USA, 2002; p. 280. [Google Scholar]
- Durán, A.G.; López, M.E.R.; Nava, H.J.A. El Género Opuntia en Jalisco: Guía de Campo; Universidad de Guadalajara: Guadalajara, Mexico, 2001. [Google Scholar]
- Garcia de Cortázar, V.; Nobel, P.S. Biomass and Fruit Production for the Prickly Pear Cactus, Opuntia ficus-indica. J. Am. Soc. Hortic. Sci. 1992, 117, 558–562. [Google Scholar] [CrossRef]
- Majure, L.C.; Puente, R.; Griffith, M.P.; Judd, W.S.; Soltis, P.S.; Soltis, D.E. Phylogeny of Opuntia s.s. (Cactaceae): Clade delineation, geographic origins, and reticulate evolution. Am. J. Bot. 2012, 99, 847–864. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.P. The origins of an important cactus crop, Opuntia ficus-indica (Cactaceae): New molecular evidence. Am. J. Bot. 2004, 91, 1915–1921. [Google Scholar] [CrossRef] [PubMed]
- Caruso, M.; Currò, S.; Las Casas, G.; La Malfa, S.; Gentile, A. Microsatellite markers help to assess genetic diversity among Opuntia ficus indica cultivated genotypes and their relation with related species. Plant Syst. Evol. 2010, 290, 85–97. [Google Scholar] [CrossRef]
- Ciriminna, R.; Chavarría-Hernández, N.; Rodríguez-Hernández, A.I.; Pagliaro, M. Toward unfolding the bioeconomy of nopal (Opuntia spp.). Biofuels Bioprod. Biorefining 2019, 13, 1417–1427. [Google Scholar] [CrossRef]
- Billiard, E. When tradition becomes trendy: Social distinction in Maltese food culture. Anthropol. Noteb. 2006, 12, 113–126. [Google Scholar]
- Sawaya, W.N.; Khatchadourian, H.A.; Safi, W.M.; Al-muhammad, H.M. Chemical characterization of prickly pear pulp, Opuntia ficus-indica, and the manufacturing of prickly pear jam. Int. J. Food Sci. Technol. 1983, 18, 183–193. [Google Scholar] [CrossRef]
- Ramírez-Moreno, E.; Córdoba-Díaz, D.; de Cortes Sánchez-Mata, M.; Díez-Marqués, C.; Goñi, I. Effect of boiling on nutritional, antioxidant and physicochemical characteristics in cladodes (Opuntia ficus indica). LWT Food Sci. Technol. 2013, 51, 296–302. [Google Scholar] [CrossRef]
- Morales, P.; Ramírez-Moreno, E.; Sanchez-Mata, M.d.C.; Carvalho, A.M.; Ferreira, I.C.F.R. Nutritional and antioxidant properties of pulp and seeds of two xoconostle cultivars (Opuntia joconostle F.A.C. Weber ex Diguet and Opuntia matudae Scheinvar) of high consumption in Mexico. Food Res. Int. 2012, 46, 279–285. [Google Scholar] [CrossRef]
- Poinar, H.N.; Kuch, M.; Sobolik, K.D.; Barnes, I.; Stankiewicz, A.B.; Kuder, T.; Spaulding, W.G.; Bryant, V.M.; Cooper, A.; Paabo, S. A molecular analysis of dietary diversity for three archaic Native Americans. Proc. Natl. Acad. Sci. USA 2001, 98, 4317–4322. [Google Scholar] [CrossRef]
- Alonso-Castro, A.J.; Dominguez, F.; Zapata-Morales, J.R.; Carranza-Alvarez, C. Plants used in the traditional medicine of Mesoamerica (Mexico and Central America) and the Caribbean for the treatment of obesity. J. Ethnopharmacol. 2015, 175, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Galati, E.M.; Monforte, M.T.; Tripodo, M.M.; d’Aquino, A.; Mondello, M.R. Antiulcer activity of Opuntia ficus indica (L.) Mill. (Cactaceae): Ultrastructural study. J. Ethnopharmacol. 2001, 76, 1–9. [Google Scholar] [CrossRef]
- McGarvie, D.; Parolis, H. The mucilage of Opuntia ficus-indica. Carbohydr. Res. 1979, 69, 171–179. [Google Scholar] [CrossRef]
- Astello-García, M.G.; Cervantes, I.; Nair, V.; Santos-Díaz, M.d.S.; Reyes-Agüero, A.; Guéraud, F.; Negre-Salvayre, A.; Rossignol, M.; Cisneros-Zevallos, L.; de la Barba Rosa, A.P. Chemical composition and phenolic compounds profile of cladodes from Opuntia spp. cultivars with different domestication gradient. J. Food Compos. Anal. 2015, 43, 119–130. [Google Scholar] [CrossRef]
- Peña-Valdivia, C.B.; Trejo, C.; Arroyo-Pena, V.B.; Sanchez Urdaneta, A.B.; Balois Morales, R. Diversity of unavailable polysaccharides and dietary fiber in domesticated nopalito and cactus pear fruit (Opuntia spp.). Chem. Biodivers 2012, 9, 1599–1610. [Google Scholar] [CrossRef]
- du Toit, A.; de Wit, M.; Hugo, A. Cultivar and Harvest Month Influence the Nutrient Content of Opuntia spp. Cactus Pear Cladode Mucilage Extracts. Molecules 2018, 23, 916. [Google Scholar] [CrossRef]
- Hernandez-Urbiola, M.I.; Perez-Torrero, E.; Rodriguez-Garcia, M.E. Chemical analysis of nutritional content of prickly pads (Opuntia ficus indica) at varied ages in an organic harvest. Int. J. Environ. Res. Public Health 2011, 8, 1287–1295. [Google Scholar] [CrossRef]
- Butterweck, V.; Semlin, L.; Feistel, B.; Pischel, I.; Bauer, K.; Verspohl, E.J. Comparative evaluation of two different Opuntia ficus-indica extracts for blood sugar lowering effects in rats. Phytother. Res. 2011, 25, 370–375. [Google Scholar] [CrossRef]
- Zhao, L.Y.; Lan, Q.J.; Huang, Z.C.; Ouyang, L.J.; Zeng, F.H. Antidiabetic effect of a newly identified component of Opuntia dillenii polysaccharides. Phytomedicine 2011, 18, 661–668. [Google Scholar] [CrossRef]
- Alarcon-Aguilar, F.J.; Valdes-Arzate, A.; Xolalpa-Molina, S.; Banderas-Dorantes, T.; Jimenez-Estrada, M.; Hernandez-Galicia, E.; Roman-Ramos, R. Hypoglycemic activity of two polysaccharides isolated from Opuntia ficus-indica and O. streptacantha. Proc. West. Pharm. Soc. 2003, 46, 139–142. [Google Scholar]
- Godard, M.P.; Ewing, B.A.; Pischel, I.; Ziegler, A.; Benedek, B.; Feistel, B. Acute blood glucose lowering effects and long-term safety of OpunDia supplementation in pre-diabetic males and females. J. Ethnopharmacol. 2010, 130, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Nuñez-Lopez, M.A.; Paredes-Lopez, O.; Reynoso-Camacho, R. Functional and hypoglycemic properties of nopal cladodes (O. ficus-indica) at different maturity stages using in vitro and in vivo tests. J. Agric. Food Chem. 2013, 61, 10981–10986. [Google Scholar] [CrossRef] [PubMed]
- Gouws, C.A.; Georgousopoulou, E.N.; Mellor, D.D.; McKune, A.; Naumovski, N. Effects of the Consumption of Prickly Pear Cacti (Opuntia spp.) and its Products on Blood Glucose Levels and Insulin: A Systematic Review. Medicina 2019, 55, 138. [Google Scholar] [CrossRef] [PubMed]
- Galati, E.M.; Pergolizzi, S.; Miceli, N.; Monforte, M.T.; Tripodo, M.M. Study on the increment of the production of gastric mucus in rats treated with Opuntia ficus indica (L.) Mill. cladodes. J. Ethnopharmacol. 2002, 83, 229–233. [Google Scholar] [CrossRef]
- Lee, E.-B.; Hyun, J.-E.; Li, D.-W.; Moon, Y.-I. The Effect of Opuntia ficus-indica var. saboten Fruit on Gastric Lesion and Ulcer in Rats. Nat. Prod. Sci. 2001, 7, 90–93. [Google Scholar]
- Vazquez-Ramirez, R.; Olguin-Martinez, M.; Kubli-Garfias, C.; Hernandez-Munoz, R. Reversing gastric mucosal alterations during ethanol-induced chronic gastritis in rats by oral administration of Opuntia ficus-indica mucilage. World J. Gastroenterol. 2006, 12, 4318–4324. [Google Scholar] [CrossRef]
- Galati, E.M.; Tripodo, M.M.; Trovato, A.; d’Aquino, A.; Monforte, M.T. Biological Activity of Opuntia ficus indica Cladodes II: Effect on Experimental Hypercholesterolemia in Rats. Pharm. Biol. 2008, 41, 175–179. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Xie, G.; Kirpotina, L.N.; Klein, R.A.; Jutila, M.A.; Quinn, M.T. Macrophage immunomodulatory activity of polysaccharides isolated from Opuntia polyacantha. Int. Immunopharmacol. 2008, 8, 1455–1466. [Google Scholar] [CrossRef]
- Aguilera-Barreiro, M.d.L.; Rivera-Marquez, J.A.; Trujillo-Arriaga, H.M.; Tamayo, Y.O.J.A.; Barreira-Mercado, E.; Rodriguez-Garcia, M.E. Intake of dehydrated nopal (Opuntia ficus indica) improves bone mineral density and calciuria in adult Mexican women. Food Nutr. Res. 2013, 57. [Google Scholar] [CrossRef]
- Mueller, M.; Cavarkapa, A.; Unger, F.M.; Viernstein, H.; Praznik, W. Prebiotic potential of neutral oligo- and polysaccharides from seed mucilage of Hyptis suaveolens. Food Chem. 2017, 221, 508–514. [Google Scholar] [CrossRef]
- Koenen, M.E.; Cruz Rubio, J.M.; Mueller, M.; Venema, K. The effect of agave fructan products on the activity and composition of the microbiota determined in a dynamic in vitro model of the human proximal large intestine. J. Funct. Foods 2016, 22, 201–210. [Google Scholar] [CrossRef]
- Mueller, M.; Reiner, J.; Fleischhacker, L.; Viernstein, H.; Loeppert, R.; Praznik, W. Growth of selected probiotic strains with fructans from different sources relating to degree of polymerization and structure. J. Funct. Foods 2016, 24, 264–275. [Google Scholar] [CrossRef]
- Yahia, E.M.; Ornelas, J.D.J.; Anaya, A. Extraction and Chemical Characteristics of Mucilage from Mesquite, Aloe Vera, Maguey and Prickly Pear Cactus Cladodes (Nopal) and Evaluation of Its Prebiotic Effect on the Growth of 2 Probiotic Bacteria. Acta Hortic. 2009, 625–628. [Google Scholar] [CrossRef]
- Reyes-Reyes, M.; Salazar-Montoya, J.A.; Rodriguez-Paez, L.I.; Ramos-Ramirez, E.G. In vitro fermentation of oligosaccharides obtained from enzymatic hydrolysis of Opuntia streptacantha mucilage. J. Sci. Food Agric. 2019, 99, 2883–2891. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Tapia, M.; Aguilar-Lopez, M.; Perez-Cruz, C.; Pichardo-Ontiveros, E.; Wang, M.; Donovan, S.M.; Tovar, A.R.; Torres, N. Nopal (Opuntia ficus indica) protects from metabolic endotoxemia by modifying gut microbiota in obese rats fed high fat/sucrose diet. Sci. Rep. 2017, 7, 4716. [Google Scholar] [CrossRef]
- Moran-Ramos, S.; He, X.; Chin, E.L.; Tovar, A.R.; Torres, N.; Slupsky, C.M.; Raybould, H.E. Nopal feeding reduces adiposity, intestinal inflammation and shifts the cecal microbiota and metabolism in high-fat fed rats. PLoS ONE 2017, 12, 171672. [Google Scholar] [CrossRef]
- Guevara-Arauza, J.C.; de Jesús Ornelas-Paz, J.; Pimentel-González, D.J.; Rosales Mendoza, S.; Soria Guerra, R.E.; Paz Maldonado, L.M.T. Prebiotic effect of mucilage and pectic-derived oligosaccharides from nopal (Opuntia ficus-indica). Food Sci. Biotechnol. 2012, 21, 997–1003. [Google Scholar] [CrossRef]
- Contreras-Padilla, M.; Gutierrez-Cortez, E.; Valderrama-Bravo, M.d.C.; Rojas-Molina, I.; Espinosa-Arbelaez, D.G.; Suarez-Vargas, R.; Rodriguez-Garcia, M.E. Effects of drying process on the physicochemical properties of nopal cladodes at different maturity stages. Plant Foods Hum. Nutr. 2012, 67, 44–49. [Google Scholar] [CrossRef]
- León-Martínez, F.M.; Rodríguez-Ramírez, J.; Medina-Torres, L.L.; Méndez Lagunas, L.L.; Bernad-Bernad, M.J. Effects of drying conditions on the rheological properties of reconstituted mucilage solutions (Opuntia ficus-indica). Carbohydr. Polym. 2011, 84, 439–445. [Google Scholar] [CrossRef]
- Martínez-Soto, G.; Celis-Fabián, F.; Hernández-Pérez, T.; López, O.P. Effect of Drying Methods on the Nutraceutical Potential of Cactus Cladodes (Opuntia spp.). Int. J. Food Nutr. Sci. 2016, 2, 1–6. [Google Scholar] [CrossRef]
- Kocourek, J.; Tichá, M.; Koštíř, J. The use of diphenylamine-aniline-phosphoric acid reagent in the detection and differentiation of monosaccharides and their derivatives on paper chromatograms. J. Chromatogr. A 1966, 24, 117–124. [Google Scholar] [CrossRef]
- Rao, P.; Pattabiraman, T.N. Reevaluation of the phenol-sulfuric acid reaction for the estimation of hexoses and pentoses. Anal. Biochem. 1989, 181, 18–22. [Google Scholar] [CrossRef]
- Hall, B.G.; Acar, H.; Nandipati, A.; Barlow, M. Growth rates made easy. Mol. Biol. Evol. 2014, 31, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Morreale, V.; Pecoraino, M.; Pagliaro, M. Solar air drying for innovative Opuntia ficus-indica cladode dehydration. 4open 2019, 2. [Google Scholar] [CrossRef]
- Crispín-Isidro, G.; Hernández-Rodríguez, L.; Ramírez-Santiago, C.; Sandoval-Castilla, O.; Lobato-Calleros, C.; Vernon-Carter, E.J. Influence of purification on physicochemical and emulsifying properties of tamarind (Tamarindus indica L.) seed gum. Food Hydrocoll. 2019, 93, 402–412. [Google Scholar] [CrossRef]
- Sepúlveda, E.; Sáenz, C.; Aliaga, E.; Aceituno, C. Extraction and characterization of mucilage in Opuntia spp. J. Arid Environ. 2007, 68, 534–545. [Google Scholar] [CrossRef]
- Trachtenberg, S.; Mayer, A.M. Composition and properties of Opuntia ficus-indica mucilage. Phytochemistry 1981, 20, 2665–2668. [Google Scholar] [CrossRef]
- Manhivi, V.E.; Venter, S.; Amonsou, E.O.; Kudanga, T. Composition, thermal and rheological properties of polysaccharides from amadumbe (Colocasia esculenta) and cactus (Opuntia spp.). Carbohydr. Polym. 2018, 195, 163–169. [Google Scholar] [CrossRef]
- Medina-Torres, L.; Brito-De La Fuente, E.; Torrestiana-Sanchez, B.; Katthain, R. Rheological properties of the mucilage gum (Opuntia ficus indica). Food Hydrocoll. 2000, 14, 417–424. [Google Scholar] [CrossRef]
- Reyes-Ocampo, I.; Córdova-Aguilar, M.S.; Guzmán, G.; Blancas-Cabrera, A.; Ascanio, G. Solvent-free mechanical extraction of Opuntia ficus-indica mucilage. J. Food Process Eng. 2019, 42. [Google Scholar] [CrossRef]
- Gunness, P.; Gidley, M.J. Mechanisms underlying the cholesterol-lowering properties of soluble dietary fibre polysaccharides. Food Funct. 2010, 1, 149–155. [Google Scholar] [CrossRef] [PubMed]
| Treatment a | Cladode b | Mucilage c | |||||
|---|---|---|---|---|---|---|---|
| Moisture | Insoluble Material | Crude Mucilage | MeOH Extract | Protein 1 | Mineral Content 2 | Carbohydrate Content 1 | |
| (%w/w) | (%w/w) | (%w/w) | (%w/w) | (%dm) | (%dm) | (%dm) | |
| OFI N | 92.4 | 4.4 | 1.62 | 1.44 | 1.37 ± 0.24 | 3.86 | 81.2 ± 0.12 |
| OFI L | 92.7 | 4.2 | 1.56 | 1.44 | 1.39 ± 0.18 | n.a. | 82.3 ± 0.17 |
| OFI V | 93.7 | 4.0 | 1.39 | 1.08 | 1.47 ± 0.27 | n.a. | 81.8 ± 0.09 |
| OFI C1 | 93.5 | 4.1 | 1.29 | 1.03 | 1.67 ± 0.29 | n.a. | 78.8 ± 0.11 |
| OFI C2 | 92.1 | 4.8 | 1.56 | 1.37 | 2.37 ± 0.26 | 2.71 | 79.1 ± 0.17 |
| mean | 92.9 | 4.3 | 1.48 | 1.27 | 1.65 | 3.29 | 80.6 |
| OXO N | 91.8 | 4.4 | 2.75 | 0.93 | 0.97 ± 0.26 | 3.32 | 80.9 ± 0.15 |
| OXO L | 91.9 | 4.5 | 2.42 | 1.01 | 0.74 ± 0.29 | n.a. | 80.1 ± 0.11 |
| OXO V | 92.1 | 4.3 | 2.56 | 0.91 | 0.69 ± 0.3 | n.a. | 78.6 ± 0.11 |
| OXO C1 | 92.2 | 4.6 | 2.37 | 0.69 | 0.98 ± 0.24 | n.a. | 77.9 ± 0.08 |
| OXO C2 | 91.2 | 5.3 | 2.55 | 0.81 | 1.1 ± 0.24 | 2.71 | 77.1 ± 0.16 |
| mean | 91.8 | 4.6 | 2.53 | 0.87 | 0.9 | 3.02 | 78.9 |
| Sample a | Ca 1 | Mg 1 | Na 1 | K 1 | Total |
|---|---|---|---|---|---|
| (%w/w) | (%w/w) | (%w/w) | (%w/w) | (%w/w) | |
| OFI N | 0.23 ± 0.02 | 0.36 ± 0.01 | 0.12 ± 0.01 | 3.08 ± 0.09 | 3.79 |
| OFI C2 | 0.18 ± 0.01 | 0.30 ± 0.01 | 0.04 ± 0.01 | 2.15 ± 0.09 | 2.67 |
| OXO N | 0.30 ± 0.02 | 0.46 ± 0.05 | 0.10 ± 0.01 | 2.41 ± 0.09 | 3.26 |
| OXO C2 | 0.27 ± 0.01 | 0.41 ± 0.02 | 0.06 ± 0.01 | 2.08 ± 0.11 | 2.83 |
| Sample a | Rhamnose | Arabinose | Xylose | Galactose | Glucose | Gal. Acid |
|---|---|---|---|---|---|---|
| (mol %) 1 | (mol %) 1 | (mol %) 1 | (mol %) 1 | (mol %) 1 | (mol %) 1 | |
| OFI N | 3.3 ± 0.0 | 18.0 ± 1.2 | 13.4 ± 0.4 | 31.6 ± 1.2 | 14.8 ± 0.1 | 18.9 ± 0.2 |
| OFI L | 3.6 ± 0.4 | 18.5 ± 0.7 | 13.1 ± 0.8 | 31.3 ± 2.2 | 12.7 ± 1.7 | 20.8 ± 0.0 |
| OFI V | 4.8 ± 0.1 | 20.9 ± 0.8 | 16.4 ± 1.7 | 29.9 ± 1.4 | 10.9 ± 0.9 | 17.2 ± 0.3 |
| OFI C1 | 4.1 ± 0.0 | 18.2 ± 1.0 | 13.9 ± 0.6 | 29.2 ± 0.4 | 13.6 ± 0.9 | 21.0 ± 1.0 |
| OFI C2 | 4.3 ± 0.1 | 18.9 ± 0.4 | 14.7 ± 0.8 | 28.4 ± 0.7 | 12.6 ± 1.1 | 21.1 ± 0.7 |
| mean | 4 | 18.9 | 14.3 | 30.1 | 12.9 | 19.8 |
| OXO N | 3.1 ± 0.3 | 30.0 ± 5.5 | 16.4 ± 1.1 | 35.1 ± 4.6 | 2.3 ± 0.0 | 13.2 ± 0.5 |
| OXO L | 3.4 ± 0.2 | 28.4 ± 0.5 | 16.7 ± 1.6 | 33.6 ± 0.1 | 2.3 ± 0.5 | 15.5 ± 1.7 |
| OXO V | 3.9 ± 0.0 | 28.1 ± 0.7 | 15.8 ± 1.4 | 34.7 ± 1.2 | 3.2 ± 0.2 | 14.3 ± 0.7 |
| OXO C1 | 3.9 ± 0.1 | 28.9 ± 1.4 | 15.9 ± 0.3 | 33.8 ± 1.8 | 4.1 ± 0.4 | 13.5 ± 0.2 |
| OXO C2 | 3.7 ± 0.3 | 27.4 ± 0.7 | 16.1 ± 1.0 | 33.8 ± 1.0 | 4.9 ± 0.6 | 14.2 ± 1.1 |
| mean | 3.6 | 28.5 | 16.2 | 34.2 | 3.3 | 14.1 |
| O. Ficus-Indica | O. Joconostle | |||||
|---|---|---|---|---|---|---|
| Treatment a | Mw1 | Ð2 | dpw 3 | Mw1 | Ð2 | dpw 3 |
| (g·mol−1) | (g·mol−1) | |||||
| N | 4.3 × 105 | 2.9 | 2850 | 4.3 × 105 | 2.8 | 2950 |
| L | 3.8 × 105 | 2.9 | 2480 | 4.5 × 105 | 3.4 | 3000 |
| V | 3.7 × 105 | 3.9 | 2460 | 4.4 × 105 | 3.0 | 3000 |
| C1 | 3.7 × 105 | 3.4 | 2460 | 4.7 × 105 | 2.6 | 3900 |
| C2 | 2.6 × 105 | 3.7 | 1685 | 3.6 × 105 | 2.9 | 2400 |
| Sample a | B. Longum ssp. infantis | L. Acidophilus | L. Rhamnosus GG | |||
|---|---|---|---|---|---|---|
| Max. OD600 1 | RGR 2 | Max. OD600 1 | RGR 2 | Max. OD600 1 | RGR 2 | |
| () | (%) | () | (%) | () | (%) | |
| glucose | 1.63 ± 0.17 | 100 | 1.55 ± 0.03 | 100 | 1.49 ± 0.24 | 100 |
| OFI N | 0.29 ± 0.51 | 18 | 0.29 ± 0.17 | 19 | 0.35 ± 0.13 | 23 |
| OFI L | 0.31 ± 0.25 | 19 b | 0.32 ± 0.15 | 20 | 0.41 ± 0.11 | 27 |
| OFI V | 0.35 ± 0.33 | 21 b | 0.26 ± 0.01 | 17 c | 0.31 ± 0.14 | 21 |
| OFI C1 | 0.47 ± 0.12 | 29 b | 0.23 ± 0.07 | 15 c | 0.26 ± 0.21 | 18 |
| OFI C2 | 0.95 ± 0.14 | 58 | 0.45 ± 0.20 | 29 | 0.84 ± 0.17 | 56 |
| OXO N | 0.28 ± 0.37 | 17 d | 0.31 ± 0.45 | 20 e | 0.28 ± 0.01 | 19 |
| OXO L | 0.51 ± 0.25 | 31 d | 0.15 ± 0.09 | 10 | 0.36 ± 0.37 | 24 f |
| OXO V | 0.21 ± 0.37 | 13 d | 0.21 ± 0.15 | 13 e | 0.30 ± 0.31 | 20 |
| OXO C1 | 0.44 ± 0.27 | 27 d | 0.27 ± 0.15 | 18 | 0.29 ± 0.38 | 19 |
| OXO C2 | 0.82 ± 0.17 | 50 | 0.25 ± 0.04 | 16 | 0.72 ± 0.17 | 48 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz-Rubio, J.M.; Mueller, M.; Loeppert, R.; Viernstein, H.; Praznik, W. The Effect of Cladode Drying Techniques on the Prebiotic Potential and Molecular Characteristics of the Mucilage Extracted from Opuntia ficus-indica and Opuntia joconostle. Sci. Pharm. 2020, 88, 43. https://doi.org/10.3390/scipharm88040043
Cruz-Rubio JM, Mueller M, Loeppert R, Viernstein H, Praznik W. The Effect of Cladode Drying Techniques on the Prebiotic Potential and Molecular Characteristics of the Mucilage Extracted from Opuntia ficus-indica and Opuntia joconostle. Scientia Pharmaceutica. 2020; 88(4):43. https://doi.org/10.3390/scipharm88040043
Chicago/Turabian StyleCruz-Rubio, José Manuel, Monika Mueller, Renate Loeppert, Helmut Viernstein, and Werner Praznik. 2020. "The Effect of Cladode Drying Techniques on the Prebiotic Potential and Molecular Characteristics of the Mucilage Extracted from Opuntia ficus-indica and Opuntia joconostle" Scientia Pharmaceutica 88, no. 4: 43. https://doi.org/10.3390/scipharm88040043
APA StyleCruz-Rubio, J. M., Mueller, M., Loeppert, R., Viernstein, H., & Praznik, W. (2020). The Effect of Cladode Drying Techniques on the Prebiotic Potential and Molecular Characteristics of the Mucilage Extracted from Opuntia ficus-indica and Opuntia joconostle. Scientia Pharmaceutica, 88(4), 43. https://doi.org/10.3390/scipharm88040043






