Antileishmanial Activity and Influence on Mitochondria of the Essential Oil from Tagetes lucida Cav. and Its Main Component
Abstract
1. Introduction
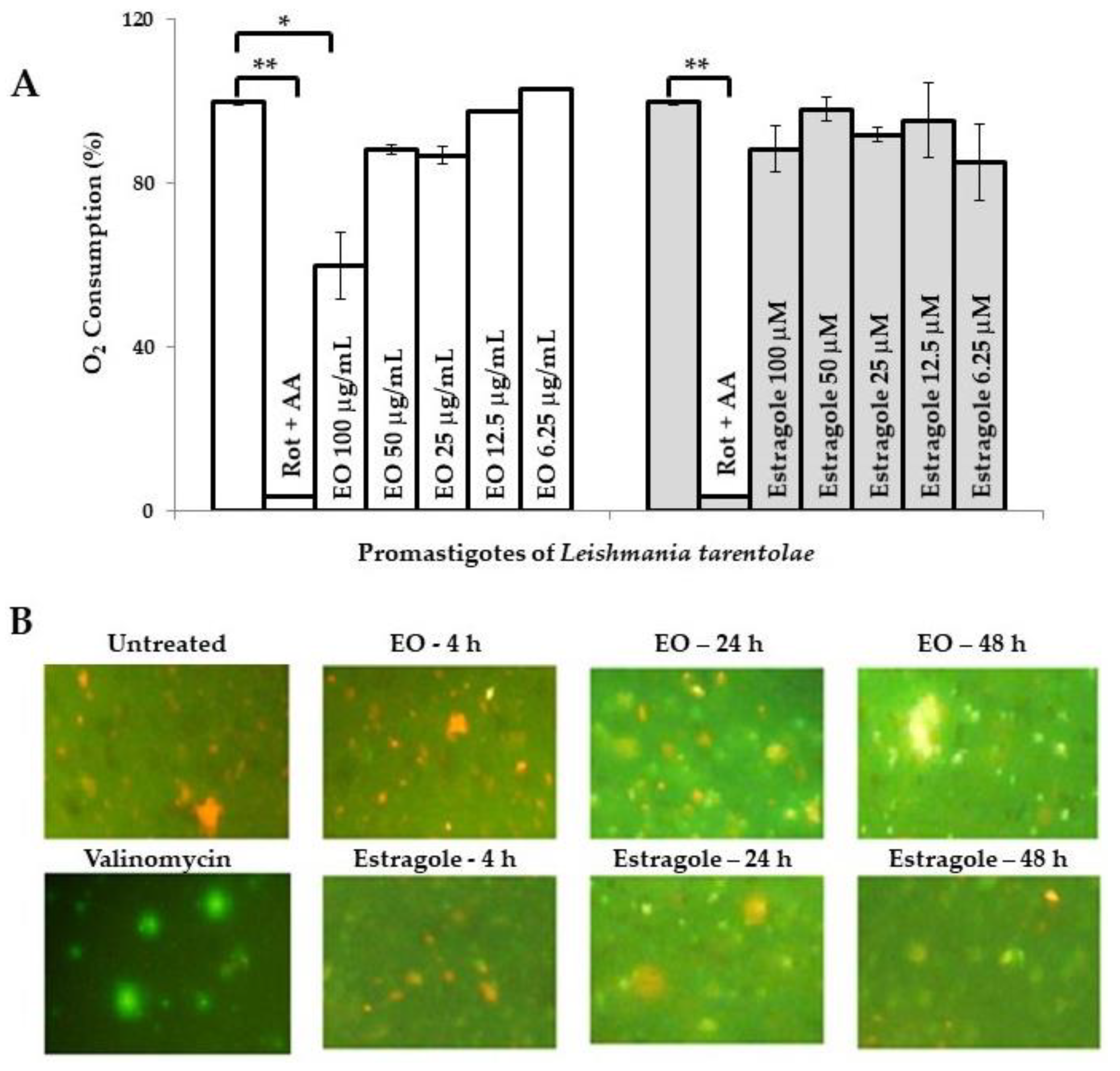
2. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Δψm | Variation of mitochondrial membrane potential |
| aBHI | BHI medium saturated with air |
| BHI | brain heart infusion |
| CC50 | Median cytotoxic concentration |
| CEI-IPK | Institutional Ethical Committee from the Institute of Tropical Medicine Pedro Kouri |
| dBHI | BHI medium reduced with dithionite |
| DMEM | Dulbecco’s modified eagle medium |
| DMSO | Dimethyl sulfoxide |
| EO | Essential oil |
| GC-MS | Gas chromatography-mass spectrometry |
| HFBS | Heat-inactivated fetal bovine serum |
| IC50 | Median inhibitory concentration |
| JC-1 | 5,5’,6,6´-Tetrachloro-1,1´,3,3´-tetraethyl-benzimidazolylcarbocyanine iodide |
| LaA | Leishmania amazonensis amastigotes |
| LaP | Leishmania amazonensis promastigotes |
| LtP | Leishmania tarentolae promastigotes |
| MMP | Mitochondrial membrane potential |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| PBS | Phosphate buffered saline |
| PMM | Peritoneal macrophage from mice |
| ROS | Reactive oxygen species |
| SD | Standard deviation |
| SI | Selectivity index |
| YEM | Yeast extract medium |
References
- Burza, S.; Croft, S.L.; Boelaert, M. Leishmaniasis. Lancet 2018, 392, 951–970. [Google Scholar] [CrossRef]
- Meira, C.M.; Gedamu, L. Protective or detrimental? Understanding the role of host Immunity in leishmaniasis. Microorganisms 2019, 7, 695. [Google Scholar] [CrossRef] [PubMed]
- Gradoni, L. Canine Leishmania vaccines: Still a long way to go. Vet. Parasitol. 2015, 208, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Bekhit, A.A.; El-Agroudy, E.; Helmy, A.; Ibrahim, T.M.; Shavandi, A.; Bekhit, A.E.A. Leishmania treatment and prevention: Natural and synthesized drugs. Eur. J. Med. Chem. 2018, 5, 229–244. [Google Scholar] [CrossRef] [PubMed]
- Simoben, C.V.; Ntie-Kang, F.; Akone, S.H.; Sippl, W. Compounds from African medicinal plants with activities against selected parasitic diseases: Schistosomiasis, trypanosomiasis and leishmaniasis. Nat. Prod. Bioprospect. 2018, 8, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Moraes Neto, R.N.; Setúbal, R.F.B.; Higino, T.M.M.; Brelaz-de-Castro, M.C.A.; Da Silva, L.C.N.; Aliança, A.S.S. Asteraceae plants as sources of compounds against leishmaniasis and Chagas disease. Front. Pharm. 2019, 10, 477. [Google Scholar] [CrossRef]
- Pérez-Ortega, G.; González-Trujano, M.E.; Ángeles-López, G.E.; Brindis, F.; Vibrans, H.; Reyes-Chilpa, R. Tagetes lucida Cav.: Ethnobotany, phytochemistry and pharmacology of its tranquilizing properties. J. Ethnopharmacol. 2016, 181, 221–228. [Google Scholar] [CrossRef]
- Bonilla-Jaime, H.; Guadarrama-Cruz, G.; Alarcon-Aguilar, F.J.; Limón-Morales, O.; Vazquez-Palacios, G. Antidepressant-like activity of Tagetes lucida Cav. is mediated by 5-HT(1A) and 5-HT(2A) receptors. J. Nat. Med. 2015, 69, 463–470. [Google Scholar] [CrossRef]
- Gutiérrez, G.Y.; Scull, L.R.; Montes, A.A.; García, S.G. Pharmacognostic, phytochemical and biological evaluation of a hydroalcoholic extract of Tagetes lucida Cavanilles. Rev. Cub. Plantas Med. 2018. Available online: http://revplantasmedicinales.sld.cu/index.php/pla/article/view/669 (accessed on 28 July 2020).
- Regalado, E.L.; Fernández, M.D.; Pino, J.A.; Mendiola, J.; Echemendia, O.A. Chemical composition and biological properties of the leaf essential oil of Tagetes lucida Cav. from Cuba. J. Essent. Oil Res. 2011, 23, 63–67. [Google Scholar] [CrossRef]
- Omer, E.A.; Hendawy, S.F.; Ismail, R.F.; Petretto, G.L.; Rourke, J.P.; Pintore, G. Acclimatization study of Tagetes lucida L. in Egypt and the chemical characterization of its essential oils. Nat. Prod. Res. 2017, 31, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Bandeira Reidel, R.V.; Nardoni, S.; Mancianti, F.; Anedda, C.; El Gendy, A.E.G.; Omer, E.A.; Pistelli, L. Chemical composition and antifungal activity of essential oils from four Asteraceae plants grown in Egypt. Z. Nat. C J. Biosci. 2018, 73, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Caballero-Gallardo, K.; Olivero-Verbel, J.; Stashenko, E.E. Repellent activity of essential oils and some of their individual constituents against Tribolium castaneum Herbst. J. Agric. Food Chem. 2011, 59, 1690–1696. [Google Scholar] [CrossRef] [PubMed]
- Cicció, J.F. A source of almost pure methyl chavicol: Volatile oil from the aerial parts of Tagetes lucida (Asteraceae) cultivated in Costa Rica. Rev. Biol. Trop. 2004, 52, 853–857. [Google Scholar] [PubMed]
- Zarate-Escobedo, J.; Castañeda-González, E.L.; Cuevas-Sánchez, J.A.; Carrillo-Fonseca, C.L.; Ortiz-Torres, C.; Ibarra-Estrada, E.; Serrato-Cruz, M.A. Essential oil of some populations of Tagetes lucida Cav. from the northern and southern regions of the state of México. Rev. Fitotec. Mex. 2018, 41, 199–209. [Google Scholar]
- López López, E.; Peña Ortega, M.G.; Colinas León, M.T.B.; Díaz Cedillo, F.; Serrato Cruz, M.A. Fungistasis of essential oil extracted from a Tagetes lucida population of Hidalgo, México. Rev. Mex. Cienc. Agric. 2018, 9, 329–341. [Google Scholar]
- Zarate-Escobedo, J.; Castañeda-González, E.L.; Cuevas-Sánchez, J.A.; Carrillo-Fonseca, C.L.; Mendoza-Garcia, E.E.; Serrato-Cruz, M.A. Concentrations and application intervals of the essential oil of Tagetes lucida Cav. against Nacobbus aberrans. Rev. Mexicana Cienc. Agric. 2018, 9, 589–600. [Google Scholar]
- Hernandez-Leon, A.; González-Trujano, M.E.; Narváez-González, F.; Pérez-Ortega, G.; Rivero-Cruz, F.; Aguilar, M.I. Role of β-caryophyllene in the antinociceptive and anti-inflammatory effects of Tagetes lucida Cav. essential oil. Molecules 2020, 25, 675. [Google Scholar] [CrossRef]
- Khan, A.; Ahmad, A.; Khan, L.A.; Manzoor, N. Ocimum sanctum (L.) essential oil and its lead molecules induce apoptosis in Candida albicans. Res. Microbiol. 2014, 165, 411–419. [Google Scholar] [CrossRef]
- Mota, M.L.; Lobo, L.T.; Costa, J.M.; Costa, L.S.; Rocha, H.A.; Rocha e Silva, L.F.; Pohlit, A.M.; Neto, V.F. In vitro and in vivo antimalarial activity of essential oils and chemical components from three medicinal plants found in northeastern Brazil. Planta Med. 2012, 78, 658–664. [Google Scholar] [CrossRef]
- Jitviriyanon, S.; Phanthong, P.; Lomarat, P.; Bunyapraphatsara, N.; Porntrakulpipat, S.; Paraksa, N. In vitro study of anti-coccidial activity of essential oils from indigenous plants against Eimeria tenella. Vet. Parasitol. 2016, 228, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.B.; Oliveira Brito Pereira Bezerra Martins, A.; Cesário, F.R.; Ferreira E Castro, F.; de Albuquerque, T.R.; Martins Fernandes, M.N.; Fernandes da Silva, B.A.; Quintans Júnior, L.J.; da Costa, J.G.; Melo Coutinho, H.D.; et al. Anti-inflammatory and antiedematogenic activity of the Ocimum basilicum essential oil and its main compound estragole: In vivo mouse models. Chem. Biol. Interact. 2016, 257, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Al-Musayeib, N.M.; Mothana, R.A.; Matheeussen, A.; Cos, P.; Maes, L. In vitro antiplasmodial, antileishmanial and antitrypanosomal activities of selected medicinal plants used in the traditional Arabian Peninsular region. BMC Complement. Altern. Med. 2012, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Britta, E.A.; Scariot, D.B.; Falzirolli, H.; Ueda-Nakamura, T.; Silva, C.C.; Dias Filho, B.P.; Borsali, R.; Nakamura, C.V. Cell death and ultrastructural alterations in Leishmania amazonensis caused by new compound 4-nitrobenzaldehyde thiosemicarbazone derived from S-limonene. BMC Microbiol. 2014, 14, 236. [Google Scholar] [CrossRef] [PubMed]
- Dolai, S.; Pal, S.; Yadav, R.K.; Adak, S. Endoplasmic reticulum stress induced apoptosis in Leishmania through Ca2+-dependent and caspase independent mechanism. J. Biol. Chem. 2011, 286, 13638–13646. [Google Scholar] [CrossRef]
- Serrano-Martín, X.; García-Marchan, Y.; Fernandez, A.; Rodriguez, N.; Rojas, H.; Visbal, G.; Benaim, G. Amiodarone destabilizes intracellular Ca2+ homeostasis and biosynthesis of sterols in Leishmania mexicana. Antimicrob. Agents Chemother. 2009, 53, 1403–1410. [Google Scholar] [CrossRef]
- Macedo-Silva, S.T.; Silva, T.L.A.O.; Urbina, J.Á.; De Souza, W.; Rodrigues, J.C.F. Antiproliferative, ultrastructural, and physiological effects of amiodarone on promastigote and amastigote forms of Leishmania amazonensis. Mol. Biol. Int. 2011, 876021–876112. [Google Scholar] [CrossRef]
- Ly, J.D.; Grubb, D.R.; Lawen, A. The mitochondrial membrane potential (deltapsi(m)) in apoptosis; an update. Apoptosis 2003, 8, 115–128. [Google Scholar] [CrossRef]
- Green, D.R.; Kroemer, G. The pathophysiology of mitochondrial cell death. Science 2004, 305, 626–629. [Google Scholar] [CrossRef]
- Dos Santos, A.O.; Costa, M.A.; Ueda-Nakamura, T.; Dias-Filho, B.P.; da Veiga-Júnior, V.F.; de Souza Lima, M.M.; Nakamura, C.V. Leishmania amazonensis: Effects of oral treatment with copaiba oil in mice. Exp. Parasitol. 2011, 129, 145–151. [Google Scholar] [CrossRef]
- De Lima, J.P.S.; Pinheiro, M.L.B.; Santos, A.M.G.; Pereira, J.L.S.; Santos, D.M.F.; Barison, A.; Silva-Jardim, I.; Costa, E.V. In vitro antileishmanial and cytotoxic activities of Annona mucosa (Annonaceae). Rev. Virtual Quim. 2012, 4, 692–702. [Google Scholar] [CrossRef]
- Katsuno, K.; Burrows, J.N.; Duncan, K.; van Huijsduijnen, R.H.; Kaneko, T.; Kita, K.; Mowbray, C.E.; Schmatz, D.; Warner, P.; Slingsby, B.T. Hit and lead criteria in drug discovery for infectious diseases of the developing world. Nat. Rev. Drug. 2015, 14, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
- Edris, A.E. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: A review. Phytother. Res. 2007, 21, 308–323. [Google Scholar] [CrossRef] [PubMed]
| Products | IC50 ± SD LaA | CC50 ± SD | SI | ||
|---|---|---|---|---|---|
| PMM | J774A | PMM | J774A | ||
| EO from T. lucida | 14.2 ± 1.6 a µg/mL | 80.8 ± 3.6 a µg/mL | 156.0 ± 4.5 a µg/mL | 6 | 11 |
| Estragole | 1.4 ± 0.1 b µg/mL 9.6 ± 0.9 µM | 20.6 ± 0.8 b µg/mL 139.1 ± 5.7 µM | 14.5 ± 0.4 b µg/mL 97.8 ± 2.4 µM | 15 | 6 |
| Pentamidine | 1.3 ± 0.1 b µg/mL 3.8 ± 0.3 µM | 13.6 ± 0.4 c µg/mL 39.9 ± 1.1 µM | 8.1± 2.8 c µg/mL 23.8 ± 4.1 µM | 11 | 6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monzote, L.; Gutiérrez, Y.; Machin, L.; Staniek, K.; Scull, R.; Satyal, P.; Gille, L.; Setzer, W.N. Antileishmanial Activity and Influence on Mitochondria of the Essential Oil from Tagetes lucida Cav. and Its Main Component. Sci. Pharm. 2020, 88, 31. https://doi.org/10.3390/scipharm88030031
Monzote L, Gutiérrez Y, Machin L, Staniek K, Scull R, Satyal P, Gille L, Setzer WN. Antileishmanial Activity and Influence on Mitochondria of the Essential Oil from Tagetes lucida Cav. and Its Main Component. Scientia Pharmaceutica. 2020; 88(3):31. https://doi.org/10.3390/scipharm88030031
Chicago/Turabian StyleMonzote, Lianet, Yamilet Gutiérrez, Laura Machin, Katrin Staniek, Ramón Scull, Prabodh Satyal, Lars Gille, and William N. Setzer. 2020. "Antileishmanial Activity and Influence on Mitochondria of the Essential Oil from Tagetes lucida Cav. and Its Main Component" Scientia Pharmaceutica 88, no. 3: 31. https://doi.org/10.3390/scipharm88030031
APA StyleMonzote, L., Gutiérrez, Y., Machin, L., Staniek, K., Scull, R., Satyal, P., Gille, L., & Setzer, W. N. (2020). Antileishmanial Activity and Influence on Mitochondria of the Essential Oil from Tagetes lucida Cav. and Its Main Component. Scientia Pharmaceutica, 88(3), 31. https://doi.org/10.3390/scipharm88030031