Evaluation of Topical Anti-Inflammatory Effects of a Gel Formulation with Plantago Lanceolata, Achillea Millefolium, Aesculus Hippocastanum and Taxodium Distichum
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Plant Material and Extracts Preparation
2.3. Active Gel Preparation
2.4. Toxicity Assay on Daphnia magna
2.5. Animals
2.6. Anti-Inflammatory Effect
2.7. Tail-Flick Test
2.8. Statistical Analysis
3. Results
3.1. Gel Preparation
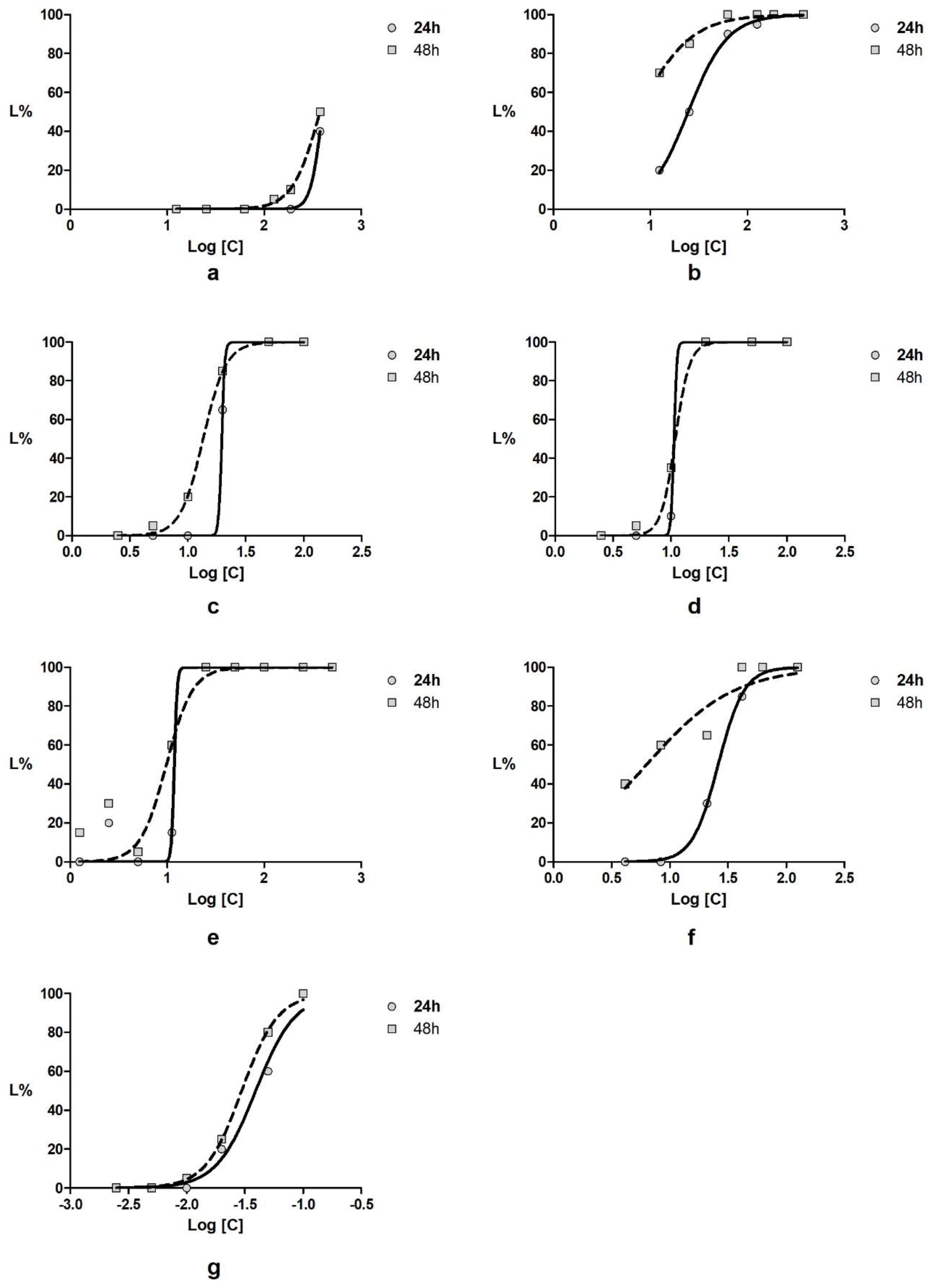
3.2. Toxicity Assay on Daphnia Magna
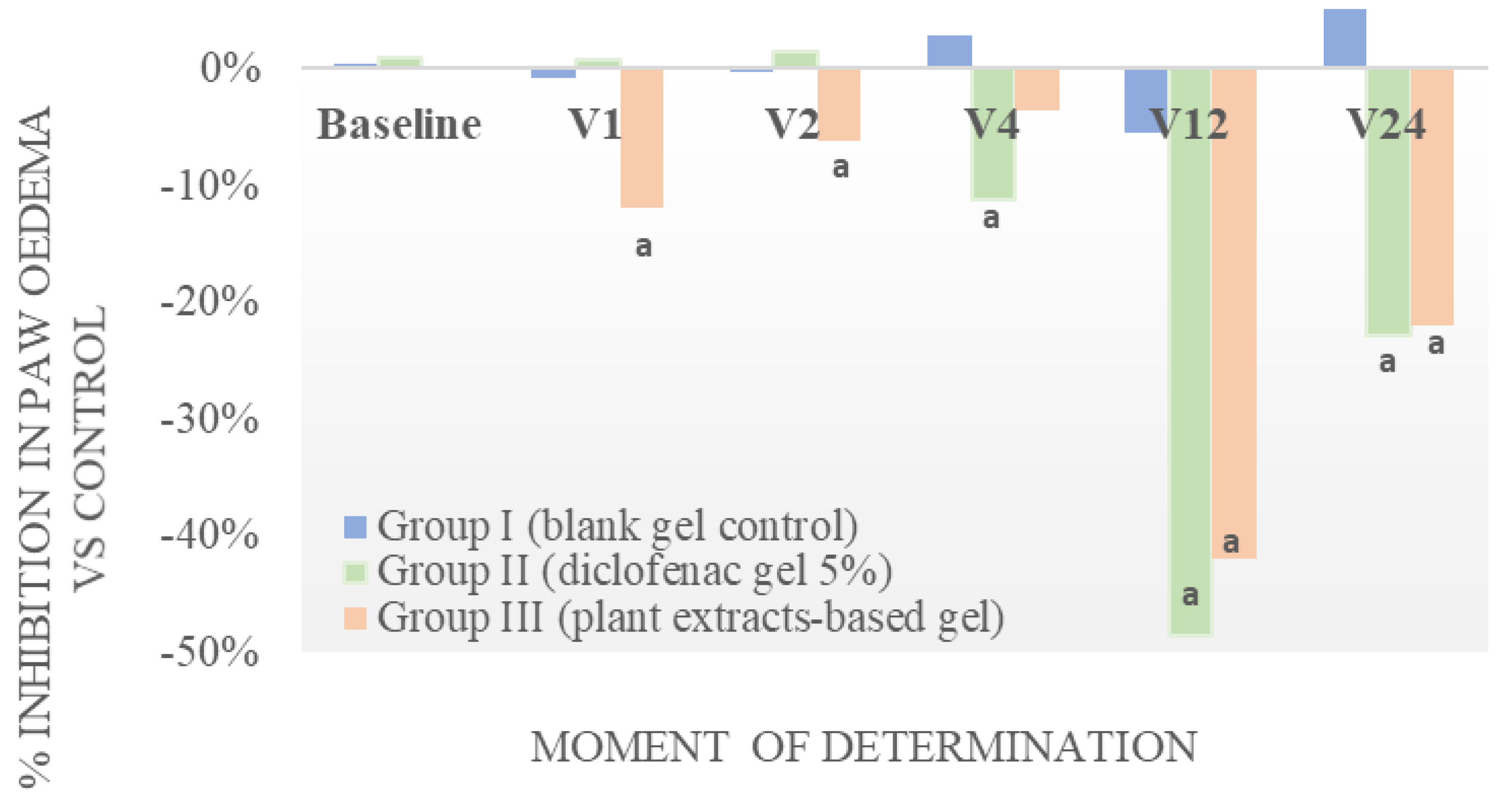
3.3. Anti-Inflammatory Activity
3.4. Anti-Nociceptive Efficacy
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ji, R.; Chamessian, A.; Zhang, Y. Pain regulation by non-neuronal cells and inflammation. Pain Res. 2016, 354, 572–577. [Google Scholar] [CrossRef]
- Ambriz-Perez, D.L.; Leyva-Lopez, N.; Gutierrez-Grijalva, E.P.; Heredia, J.B. Phenolic compounds: Natural alternative in inflammation treatment. A Review. Cogent Food Agric. 2016, 2, 1131412. [Google Scholar]
- Miyasaka, M.; Takatsu, K. Chronic Neuroinflammation Mechanism and Regulation; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Ashley, N.T.; Weil, Z.M.; Nelson, R.J. Inflammation: Mechanisms, Costs, and Natural Variation. Annu. Rev. Ecol. Evol. Syst. 2012, 43, 385–406. [Google Scholar] [CrossRef]
- Duthie, G.G.; Wood, A.D. Natural salicylates: Foods, functions and disease prevention. Food Funct. 2011, 2, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Desborough, M.J.R.; Keeling, D.M. The aspirin story–from willow to wonder drug. Br. J. Haematol. 2017, 177, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Pahwa, R.; Jialal, I. Chronic Inflammation-StatPearls-NCBI Bookshelf. In Stat Pearls. Available online: https://www.ncbi.nlm.nih.gov/books/NBK493173/ (accessed on 24 May 2020).
- Persson, M.S.M.; Stocks, J.; Walsh, D.A.; Doherty, M.; Zhang, W. The relative efficacy of topical non-steroidal anti-inflammatory drugs and capsaicin in osteoarthritis: A network meta-analysis of randomised controlled trials. Osteoarthr. Cartil. 2018, 26, 1575–1582. [Google Scholar] [CrossRef]
- Brune, K.; Patrignani, P. New insights into the use of currently available non-steroidal anti-inflammatory drugs. J. Pain Res. 2015, 8, 105–118. [Google Scholar] [CrossRef]
- Green, G.A. Understanding NSAIDs: From aspirin to COX-2. Clin. Cornerstone 2001, 3, 50–58. [Google Scholar] [CrossRef]
- Harirforoosh, S.; Asghar, W.; Jamali, F. Adverse effects of nonsteroidal antiinflammatory drugs: An update of gastrointestinal, cardiovascular and renal complications. J. Pharm. Pharm. Sci. 2013, 16, 821–847. [Google Scholar] [CrossRef]
- Sevilla, L.M.; Pérez, P. Roles of the glucocorticoid and mineralocorticoid receptors in skin pathophysiology. Int. J. Mol. Sci. 2018, 19, 1906. [Google Scholar] [CrossRef]
- Landriscina, A.; Rosen, J.; Friedman, A.J. Nanotechnology, inflammation and the skin barrier: Innovative approaches for skin health and cosmesis. Cosmetics 2015, 2, 177–186. [Google Scholar] [CrossRef]
- Barnes, P.J.; Adcock, I.M. Glucocorticoid resistance in inflammatory diseases. Lancet 2009, 373, 1905–1917. [Google Scholar] [CrossRef]
- Derry, S.; Conaghan, P.; Jap, D.S.; Pj, W.; Ra, M. Topical NSAIDs for chronic musculoskeletal pain in adults. Cochrane Database Syst. Rev. 2016, 2016, CD007400. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Wei, J.; Persson, M.S.M.; Sarmanova, A.; Doherty, M.; Xie, D.; Wang, Y.; Li, X.; Li, J.; Long, H.; et al. Relative efficacy and safety of topical non-steroidal anti-inflammatory drugs for osteoarthritis: A systematic review and network meta-analysis of randomised controlled trials and observational studies. Br. J. Sports Med. 2018, 52, 642–650. [Google Scholar] [CrossRef]
- Kumar, A.H.S. Rediscovering the Drug Discovery with Natural Products as Therapeutic Tools. J. Nat. Sci. Biol. Med. 2018, 9, 1. [Google Scholar] [CrossRef]
- McClements, D.J. Future Foods; Springer Nature, 2019; Available online: https://www.springer.com/gp/book/9783030129941 (accessed on 24 May 2020).
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.; Lightfoot, D. Phytochemicals: Extraction, Isolation, and Identification of Bioactive Compounds from Plant Extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef]
- Xiao, J.; Bai, W. Bioactive phytochemicals. Crit. Rev. Food Sci. Nutr. 2019, 59, 827–829. [Google Scholar] [CrossRef]
- Daliu, P.; Santini, A.; Novellino, E. From pharmaceuticals to nutraceuticals: Bridging disease prevention and management. Expert Rev. Clin. Pharmacol. 2019, 12, 1–7. [Google Scholar] [CrossRef]
- Howes, M.J.R.; Perry, N.S.L.; Vásquez-Londoño, C.; Perry, E.K. Role of phytochemicals as nutraceuticals for cognitive functions affected in ageing. Br. J. Pharmacol. 2019, 177, 1294–1315. [Google Scholar] [CrossRef]
- Applequist, W.L.; Moerman, D.E. Yarrow (Achillea millefolium L.): A neglected panacea? A review of ethnobotany. Econ. Bot. 2011, 65, 209–225. [Google Scholar] [CrossRef]
- Ali, S.I.; Gopalakrishnan, B.; Venkatesalu, V. Pharmacognosy, Phytochemistry and Pharmacological Properties of Achillea millefolium L.: A Review. Phyther. Res. 2017, 31, 1140–1161. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.; Peethambaran, B. Anti-Inflammatory and Anti-Microbial Properties of Achillea Millefolium in Acne Treatment; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Wilkinson, J.A.; Brown, A.M.G. Horse Chestnut-Aesculus Hippocastanum: Potential Applications in Cosmetic Skin-care Products. Int. J. Cosmet. Sci. 1999, 21, 437–447. [Google Scholar] [CrossRef]
- Küçükkurt, I.; Ince, S.; Keleş, H.; Küpeli Akkol, E.; Avcı, G.; Yeşilada, E.; Bacak, E. Beneficial effects of Aesculus hippocastanum L. seed extract on the body’s own antioxidant defense system on subacute administration. J. Ethnopharmacol. 2010, 129, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Varinská, L.; Fáber, L.; Kello, M.; Petrovová, E.; Balážová, L.; Solár, P.; Coma, M.; Urdzík, P.; Mojžiš, J.; Vajdlenka, E.; et al. β-escin effectively modulates HUVECs proliferation and tube formation. Molecules 2018, 23, 197. [Google Scholar] [CrossRef] [PubMed]
- Foca, G.; Ulrici, A.; Cocchi, M.; Durante, C.; Vigni, M.L.; Marchetti, A.; Sighinolfi, S.; Tassi, L. Seeds of Horse Chestnut (Aesculus hippocastanum L.) and Their Possible Utilization for Human Consumption. Nuts Seeds Heal. Dis. Prev. 2011. [Google Scholar] [CrossRef]
- Margină, D.; Olaru, O.T.; Ilie, M.; Grădinaru, D.; Guțu, C.; Voicu, S.; Dinischiotu, A.; Spandidos, D.A.; Tsatsakis, A.M. Assessment of the potential health benefits of certain total extracts from Vitis vinifera, Aesculus hyppocastanum and Curcuma longa. Exp. Ther. Med. 2015, 10, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Mihai, P.D.; Seremet, C.O.; Nitulescu, G.; Ivopol, M.; Sevastre, A.-S.; Negres, S.; Ivopol, G.; Nitulescu, M.G.; Olaru, T.O. Evaluation of Natural Extracts in Animal Models of Pain and Inflammation for a Potential Therapy of Hemorrhoidal Disease. Sci. Pharm. 2019, 87, 14. [Google Scholar] [CrossRef]
- Samuelsen, A.B. The traditional uses, chemical constituents and biological activities of Plantago major L. A review. J. Ethnopharmacol. 2000, 71, 1–21. [Google Scholar] [CrossRef]
- Yang, L.; Wen, K.S.; Ruan, X.; Zhao, Y.X.; Wei, F.; Wang, Q. Response of plant secondary metabolites to environmental factors. Molecules 2018, 23, 762. [Google Scholar] [CrossRef]
- Beara, I.N.; Lesjak, M.M.; Orčić, D.Z.; Simin, N.D.; Četojević-Simin, D.D.; Božin, B.N.; Mimica-Dukić, N.M. Comparative analysis of phenolic profile, antioxidant, anti-inflammatory and cytotoxic activity of two closely-related Plantain species: Plantago altissima L. and Plantago lanceolata L. LWT Food Sci. Technol. 2012, 47, 64–70. [Google Scholar] [CrossRef]
- Najafian, Y.; Hamedi, S.S.; Kaboli Farshchi, M.; Feyzabadi, Z. Plantago major in Traditional Persian Medicine and modern phytotherapy: A narrative review. Electron. Physician 2018, 10, 6390–6399. [Google Scholar] [CrossRef] [PubMed]
- Genc, Y.; Harput, U.S.; Saracoglu, I. Cytotoxic and Antiinflammatory Activity Guided Studies on Plantago holosteum Scop. Proceedings 2017, 1, 997. [Google Scholar] [CrossRef]
- Zubair, M.; Widén, C.; Renvert, S.; Rumpunen, K. Water and ethanol extracts of Plantago major leaves show anti-inflammatory activity on oral epithelial cells. J. Tradit. Complement. Med. 2019, 9, 169–171. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Thummasuwan, S.; Sehgal, S.K.; Chouvarine, P.; Peterson, D.G. Characterization of the genome of bald cypress. BMC Genom. 2011, 12, 553. [Google Scholar] [CrossRef]
- Cortés-Arroyo, A.R.; Domínguez-Ramírez, A.M.; Gómez-Hernández, M.; López, J.R.M.; de la Peña, M.H.; López-Muñoz, F.J. Antispasmodic and bronchodilator activities of Taxodium mucronatum ten leaf extract. Afr. J. Biotechnol. 2011, 10, 54–64. [Google Scholar]
- Li, S. Ethnobotany, Phytochemistry, and Biological Activities of Taxodium Rich. Pharm. Crop. 2013, 4, 1–14. [Google Scholar] [CrossRef][Green Version]
- El Tantawy, M.E.; El Sakhawy, F.S.; El Sohly, M.A.; Ross, S.A. Chemical composition and biological activity of the essential oil of the fruit of taxodium distichum L. Rich growing in egypt. J. Essent. Oil Res. 1999, 11, 386–392. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Savidov, N.; Poroikov, V.V.; Gloriozova, T.A.; Imbs, A.B. Naturally occurring aromatic steroids and their biological activities. Appl. Microbiol. Biotechnol. 2018, 102, 4663–4674. [Google Scholar] [CrossRef]
- Nitulescu, G.; Mihai, D.P.; Nicorescu, I.M.; Olaru, O.T.; Ungurianu, A.; Zanfirescu, A.; Nitulescu, G.M.; Margina, D. Discovery of natural naphthoquinones as sortase A inhibitors and potential anti-infective solutions against Staphylococcus aureus. Drug Dev. Res. 2019, 80, 1136–1145. [Google Scholar] [CrossRef]
- Seremet, O.C.; Olaru, O.T.; Gutu, C.M.; Nitulescu, G.M.; Ilie, M.; Negres, S.; Zbarcea, C.E.; Purdel, C.N.; Spandidos, D.A.; Tsatsakis, A.M.; et al. Toxicity of plant extracts containing pyrrolizidine alkaloids using alternative invertebrate models. Mol. Med. Rep. 2018, 17, 7757–7763. [Google Scholar] [CrossRef]
- Nitulescu, G.; Nicorescu, I.; Olaru, O.; Ungurianu, A.; Mihai, D.; Zanfirescu, A.; Nitulescu, G.; Margina, D. Molecular Docking and Screening Studies of New Natural Sortase A Inhibitors. Int. J. Mol. Sci. 2017, 18, 2217. [Google Scholar] [CrossRef] [PubMed]
- Candan, F.; Unlu, M.; Tepe, B.; Daferera, D.; Polissiou, M.; Sökmen, A.; Akpulat, H.A. Antioxidant and antimicrobial activity of the essential oil and methanol extracts of Achillea millefolium subsp. millefolium Afan. (Asteraceae). J. Ethnopharmacol. 2003, 87, 215–220. [Google Scholar] [CrossRef]
- Ogunwande, I.A.; Olawore, N.O.; Ogunmola, O.O.; Walker, T.M.; Schmidt, J.M.; Setzer, W.N. Cytotoxic effects of Taxodium distichum oils. Pharm. Biol. 2007, 45, 106–110. [Google Scholar] [CrossRef]
- Charan, J.; Kantharia, N. How to calculate sample size in animal studies? J. Pharmacol. Pharmacother. 2013, 4, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Koblin, D.D. Urethane: Help or hindrance? Anesth. Analg. 2002, 94, 241–242. [Google Scholar]
- Ukrainets, V.I.; Petrushova, A.L.; Fedosov, I.A.; Voloshchuk, I.N.; Bondarenko, S.P.; Shishkina, V.S.; Sidorenko, V.L.; Sim, G. Crystal Habits and Biological Properties of N-(4-Trifluoromethylphenyl)-4-Hydroxy-2,2-Dioxo-1H-2λ6,1-Benzothiazine-3-Carboxamide. Sci. Pharm. 2020, 88, 1. [Google Scholar] [CrossRef]
- Benbow, T.; Campbell, J. Comparison of the Topical Analgesic Effects of a Novel Diclofenac Microemulsion to a Marketed Diclofenac Macroemulsion Formulation in Rats Using the Tail Flick Test. J. Dev. Drugs 2018, 7, 1–6. [Google Scholar]
- Seo, S.M.; Park, H.M.; Park, I.K. Larvicidal activity of ajowan (Trachyspermum ammi) and Peru balsam (Myroxylon pereira) oils and blends of their constituents against mosquito, Aedes aegypti, acute toxicity on water flea, Daphnia magna, and aqueous residue. J. Agric. Food Chem. 2012, 60, 5909–5914. [Google Scholar] [CrossRef]
- Park, H.-M.; Kim, J.; Chang, K.-S.; Kim, B.-S.; Yang, Y.-J.; Kim, G.-H.; Shin, S.-C.; Park, I.-K. Larvicidal Activity of Myrtaceae Essential Oils and Their Components Against Aedes aegypti, Acute Toxicity on Daphnia magna, and Aqueous Residue. J. Med. Entomol. 2011, 48, 405–410. [Google Scholar] [CrossRef]
- Smelcerovic, A.; Lamshoeft, M.; Radulovic, N.; Ilic, D.; Palic, R. LC-MS analysis of the essential oils of Achillea millefolium and Achillea crithmifolia. Chromatographia 2010, 71, 113–116. [Google Scholar] [CrossRef]
- Rufino, A.T.; Ribeiro, M.; Judas, F.; Salgueiro, L.; Lopes, M.C.; Cavaleiro, C.; Mendes, A.F. Anti-inflammatory and Chondroprotective Activity of ( + )-α-Pinene: Structural and Enantiomeric Selectivity. J. Nat. Prod. 2014, 77, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Paun, G.; Neagu, E.; Moroeanu, V.; Albu, C.; Ursu, T.M.; Zanfirescu, A.; Negres, S.; Chirita, C.; Radu, G.L. Anti-inflammatory and antioxidant activities of the Impatiens noli-tangere and Stachys officinalis polyphenolic-rich extracts. Braz. J. Pharmacogn. 2018, 28, 57–64. [Google Scholar] [CrossRef]
- Gades, N.M.; Danneman, P.J.; Wixson, S.K.; Tolley, E.A. The Magnitude and Duration of the Analgesic Effect of Morphine, Butorphanol, and Buprenorphine in Rats and Mice. Contemp. Top. Lab. Anim. Sci. 2000, 39, 8–13. [Google Scholar] [PubMed]
- Arrau, S.; Delporte, C.; Cartagena, C.; Rodríguez-Díaz, M.; González, P.; Silva, X.; Cassels, B.K.; Miranda, H.F. Antinociceptive activity of Quillaja saponaria Mol. saponin extract, quillaic acid and derivatives in mice. J. Ethnopharmacol. 2011, 133, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Núñez Guillén, M.E.; Da Silva Emim, J.A.; Souccar, C.; Lapa, A.J. Analgesic and antiinflammatory activities of the aqueous extract of Plantago major L. Pharm. Biol. 1997, 35, 99–104. [Google Scholar]
- Türel, I.; Özbek, H.; Erten, R.; Öner, A.C.; Cengiz, N.; Yilmaz, O. Hepatoprotective and anti-inflammatory activities of Plantago major L. Indian J. Pharmacol. 2009, 41, 120–124. [Google Scholar]
- Palmeiro, N.S.; Esteves Almeida, C.; Ghedini, P.C.; Goulart, L.S.; Baldisserotto, B. Analgesic and anti-inflammatory properties of Plantago australis hydroalcoholic extract. Acta Farm. Bonaer. 2002, 21, 89–92. [Google Scholar]
- Oloumi, M.M.; Vosough, D.; Derakhshanfar, A.; Nematollahi, M.H. The Healing Potential of Plantago lanceolata Ointment on Collagenase-Induced Tendinitis in Burros (Equus asinus). J. Equine Vet. Sci. 2011, 31, 470–474. [Google Scholar] [CrossRef]
- Hussan, F.; Mansor, A.S.; Hassan, S.N.; Kamaruddin, T.N.E.; Budin, S.B.; Othman, F. Anti-Inflammatory Property of Plantago major Leaf Extract Reduces the Inflammatory Reaction in Experimental Acetaminophen-Induced Liver Injury. Evid. Based Complement. Altern. Med. 2015, 2015, 1–7. [Google Scholar] [CrossRef]
- Kazemi, M. Chemical composition and antimicrobial, antioxidant activities and anti-inflammatory potential of Achillea millefolium L., Anethum graveolens L., and Carum copticum L. essential oils. J. Herb. Med. 2015, 5, 217–222. [Google Scholar] [CrossRef]
- Abdossi, V.; Kazemi, M. Bioactivities of Achillea millefolium essential oil and its main terpenes from Iran. Int. J. Food Prop. 2015, 19, 1798–1808. [Google Scholar] [CrossRef]
- Villalva, M.; Jaime, L.; Villanueva-Bermejo, D.; Lara, B.; Fornari, T.; Reglero, G.; Santoyo, S. Supercritical anti-solvent fractionation for improving antioxidant and anti-inflammatory activities of an Achillea millefolium L. extract. Food Res. Int. 2019, 115, 128–134. [Google Scholar] [CrossRef] [PubMed]
| Ingredients | Active Gel | Blank Gel |
|---|---|---|
| Plantago lanceolata extract | 1.5 g | 0 |
| Aesculus hippocastanum extract | 0.5 g | 0 |
| Achillea millefolium oil | 1 mL | 0 |
| Taxodium distichum oil | 1 mL | 0 |
| Tween 20 | 0.15 g | 0.15 g |
| Ethanol | 0.3 g | 0.3 g |
| Carbopol base 1% | q.s. ad 30 g | q.s. ad 30 g |
| Sample | LC50 (24 h) | CI95% | LC50 (48 h) | CI95% |
| nL/mL | ||||
| Achillea millefolium volatile oil | 19.6 | ND ** | 13.6 | 11.7–15.7 |
| Taxodium distichum volatile oil | 10.6 | ND ** | 10.9 | 10.0–11.9 |
| α-pinene (control) | 39.2 | 30.4–50.6 | 29.7 | 23.9–37.0 |
| µg/mL | ||||
| Plantago lanceolata extract | 396.5 | 315.7–498.0 | 375.5 | 357.5–394.5 |
| Aesculus hippocastanum extract | 24.5 | 21.6–27.7 | 7.5 | 5.8–9.7 |
| Mixture of active components * | 11.8 | ND ** | 9.8 | 7.2–13.3 |
| Diclofenac (control) | 26.1 | 22.7–29.9 | 6.3 | 4.0–9.9 |
| Treatment | V0 (0 h) | V1 (1 h) | V2 (2 h) | V4 (4 h) | V5 (12 h) | V6 (24 h) |
|---|---|---|---|---|---|---|
| Active gel | 288.4 ± 29.0 | 308.7 ± 24.3 | 354.8 ± 41.7 | 371.0 ± 84.4 | 401.1 ± 62.8 | 680.9 ± 24.6 |
| Control | 289.3 ± 24.6 | 344.1 ± 26.8 | 373.9 ± 31.4 | 382.9 ± 47.5 | 523.9 ± 51.5 | 746.7 ± 96.2 |
| Blank gel | 305.4 ± 39.5 | 359.9 ± 39.6 | 386.9 ± 45.6 | 421.8 ± 77.2 | 444.9 ± 97.4 | 744.0 ± 27.9 |
| Control | 314.0 ± 27.9 | 372.7 ± 46.2 | 398.7 ± 39.9 | 424.5 ± 33.2 | 475.0 ± 35.6 | 745.1 ± 85.2 |
| Diclofenac 5% gel | 230.9 ± 25.5 | 312.8 ± 49.5 | 330.9 ± 61.1 | 310.9 ± 94.8 | 325.8 ± 69.9 | 557.8 ± 26.5 |
| Control | 249.2 ± 26.5 | 350.6 ± 31.6 | 369.2 ± 35.1 | 379.9 ± 37.4 | 493.9 ± 32.6 | 688.3 ± 61.8 |
| Treatment | Baseline | 60 Minutes after Administration | AI% | 120 Minutes after Administration | AI% |
|---|---|---|---|---|---|
| No treatment | 1.9 ± 0.5 | 2.3 ± 0.6 | 2.1 ± 0.8 | ||
| Active gel | 1.7 ± 0.6 | 3.8 a ± 1.0 | 19.7 | 3.0 a,b ± 0.6 | 11.8 |
| Blank gel | 2.0 ± 0.9 | 3.4 a ± 0.8 | 17.9 | 2.4 ± 0.8 | 4.4 |
| Diclofenac 5% gel | 2.2 ± 0.7 | 4.4 a ± 1.2 | 27.3 | 3.4 a,b ± 0.9 | 16.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zanfirescu, A.; Nitulescu, G.; Stancov, G.; Radulescu, D.; Trif, C.; Nitulescu, G.M.; Negres, S.; Olaru, O.T. Evaluation of Topical Anti-Inflammatory Effects of a Gel Formulation with Plantago Lanceolata, Achillea Millefolium, Aesculus Hippocastanum and Taxodium Distichum. Sci. Pharm. 2020, 88, 26. https://doi.org/10.3390/scipharm88020026
Zanfirescu A, Nitulescu G, Stancov G, Radulescu D, Trif C, Nitulescu GM, Negres S, Olaru OT. Evaluation of Topical Anti-Inflammatory Effects of a Gel Formulation with Plantago Lanceolata, Achillea Millefolium, Aesculus Hippocastanum and Taxodium Distichum. Scientia Pharmaceutica. 2020; 88(2):26. https://doi.org/10.3390/scipharm88020026
Chicago/Turabian StyleZanfirescu, Anca, Georgiana Nitulescu, Gheorghe Stancov, Denise Radulescu, Cosmin Trif, George Mihai Nitulescu, Simona Negres, and Octavian Tudorel Olaru. 2020. "Evaluation of Topical Anti-Inflammatory Effects of a Gel Formulation with Plantago Lanceolata, Achillea Millefolium, Aesculus Hippocastanum and Taxodium Distichum" Scientia Pharmaceutica 88, no. 2: 26. https://doi.org/10.3390/scipharm88020026
APA StyleZanfirescu, A., Nitulescu, G., Stancov, G., Radulescu, D., Trif, C., Nitulescu, G. M., Negres, S., & Olaru, O. T. (2020). Evaluation of Topical Anti-Inflammatory Effects of a Gel Formulation with Plantago Lanceolata, Achillea Millefolium, Aesculus Hippocastanum and Taxodium Distichum. Scientia Pharmaceutica, 88(2), 26. https://doi.org/10.3390/scipharm88020026






