Comparative Investigation of Amino Acids Content in the Dry Extracts of Juno bucharica, Gladiolus Hybrid Zefir, Iris Hungarica, Iris Variegata and Crocus Sativus Raw Materials of Ukrainian Flora
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Methods
2.2. Plant Raw Materials
2.3. Preparation of Extracts
2.4. Sample Preparation and Derivatization
2.5. GC–MS Analysis
2.6. Statistical Analyses
3. Results
3.1. Amino Acids Analysis
3.2. Method Validation
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Capasso, F.; Gaginella, T.S.; Grandolini, G.; Izzo, A.A. (Eds.) Phytotherapy: A Quick Reference to Herbal Medicine; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2003; 500p. [Google Scholar] [CrossRef]
- Essafi, H.; Trambelsi, N.; Benincasa, C.; Tamaalli, A.; Perri, E.; Zarrouk, M. Phytochemical profile, antioxidant and antiproliferative activities of olive leaf extracts from autochthonous Tunisian cultivars. Acta Aliment. 2019, 48, 384–390. [Google Scholar] [CrossRef]
- Essafi, H.; Trambelsi, N.; Chimento, A.; Benincasa, C.; Tamaalli, A.; Perri, E.; Zarrouk, M.; Pezzi, V. Olea europaea L. Flowers as a new promising anticancer natural product: Phenolic composition, antiproliferative activity and apoptosis induction. Nat. Prod. Res. 2019, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Anjum, S.; Ahmed Bazai, Z.; Rizwan, S.; Benincasa, C.; Mehmood, K.; Siddique, N.; Shaheen, G.; Mehmood, Z.; Azam, M.; Sajjad, A. Elemental characterization of medicinal plants and soils from Hazarganji Chiltan National Park and nearby unprotected areas of Balochistan, Pakistan. J. Oleo Sci. 2019, 68, 443–461. [Google Scholar] [CrossRef] [PubMed]
- Niu, B.; Zheng, F.; Xu, J. Protective effect of gui zhi (Ramulus Cinnamomi) on abnormal levels of four amino acid neurotransmitters by chronically ma huang (Herb Ephedra) intoxicated prefrontal cortex in rats treated with a ma huang-gui zhi herb pair. J. Ethnopharmacol. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Huang, S.; Wu, Y.; Zhang, W.; Wang, F.; Cao, J.; Sheng, Q.; Liang, Z.; Liu, L.; Ou, W.B. Comparative study on the composition of free amino acids and derivatives in the two botanical origins of an edible Chinese herb “Xiebai”, i.e., Allium chinense G. Don and Allium macrostemon Bunge species. Food Res. Int. 2018, 106, 446–457. [Google Scholar] [CrossRef]
- Firenzuoli, F.; Gori, L. Herbal medicine today: Clinical and research issues. Evid. Based Complement. Altern. Med. 2007, 4, 37–40. [Google Scholar] [CrossRef]
- Papes, F.; Surpili, M.J.; Langone, F.; Trigo, J.R.; Arruda, P. The essential amino acid lysine acts as precursor of glutamate in the mammalian central nervous system. FEBS Lett. 2001, 488, 34–38. [Google Scholar] [CrossRef]
- Musgrave, T.; Tenorio, G.; Rauw, G.; Baker, G.B.; Kerr, B.J. Tissue concentration changes of amino acids and biogenic amines in the central nervous system of mice with experimental autoimmune encephalomyelitis (EAE). Neurochem. Int. 2011, 59, 28–38. [Google Scholar] [CrossRef]
- Singab, A.N.B.; Ayoub, I.M.; El-Shazly, M.; Korinek, M.; Wu, T.Y.; Cheng, Y.B.; Wu, Y.C. Shedding the light on Iridaceae: Ethnobotany, phytochemistry and biological activity. Ind. Crop. Prod. 2016, 92, 308–335. [Google Scholar] [CrossRef]
- Mykhailenko, O.; Kovalyov, V.; Goryacha, O.; Ivanauskas, L.; Georgiyants, V. Biologically active compounds and pharmacological activities of species of the genus Crocus: A review. Phytochemistry 2019, 162, 56–89. [Google Scholar] [CrossRef]
- Gohari, A.R.; Saeidnia, S.; Mahmoodabadi, M.K. An overview on saffron, phytochemicals, and medicinal properties. Pharmacogn. Rev. 2013, 7, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.Y.; Zhu, Y.; Shu, P.; Qin, X.Y.; Wu, G.; Wang, Q.; Qin, M.J. Phenolic metabolite profiles and antioxidants assay of three Iridaceae medicinal plants for traditional Chinese medicine “She-gan” by on-line HPLC–DAD coupled with chemiluminescence (CL) and ESI-Q-TOF-MS/MS. J. Pharm. Biomed. Anal. 2014, 98, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Kostić, A.Ž.; Gašić, U.M.; Pešić, M.B.; Stanojević, S.P.; Barać, M.B.; Mačukanović-Jocić, M.P.; Avramov, S.N.; Tešić, Ž.L. Phytochemical analysis and total antioxidant capacity of rhizome, above-ground vegetative parts and flower of three Iris species. Chem. Biodivers. 2019, 16, e1800565. [Google Scholar] [CrossRef] [PubMed]
- Mykhailenko, O.; Kovalyov, V.; Kovalyov, S.; Toryanik, E.; Osolodchenko, T.; Buidin, Y. Fatty acid composition of lipids of Iris sibirica. Cesk. Sloven. Farm. 2017, 66, 220–227. [Google Scholar]
- Munyemana, F.; Mondego, A.P.; Cumbane, P. Qualitative Phytochemical Screening and Antimicrobial Activity Evaluation of the Bulb Extracts of Gladiolus Psittacinus Hook (Iridaceae); International Network Environmental Management Conflicts: Santa Catarina, Brazil, 2013; Volume 2, pp. 14–31. [Google Scholar]
- Jadouali, S.M.; Rachid, H.A.; Majourhat, M.K.; Abdelatif, Z.B.; Faouzi, L.A. Chemical characterization and antioxidant compounds of flower parts of Moroccan Crocus sativus L. J. Saudi Soc. Agric. Sci. 2019, 18, 476–480. [Google Scholar] [CrossRef]
- Basgedik, B.; Ugurb, A.; Sarac, N. Antimicrobial, antioxidant, and antimutagenic activities of Gladiolus illyricus. J. Pharm. Pharmacogn. Res. 2014, 2, 93–99. [Google Scholar]
- Wollenweber, E.; Stevens, J.F.; Klimo, K.; Knauft, J.; Frank, N.; Gerhauser, C. Cancer chemopreventive in vitro activities of isoflavones isolated from Iris germanica. Planta Med. 2003, 69, 15–20. [Google Scholar] [CrossRef]
- Shinwari, K.J.; Rao, P.S. Thermal-assisted high hydrostatic pressure extraction of nutraceuticals from saffron (Crocus sativus): Process optimization and cytotoxicity evaluation against cancer cells. Innov. Food Sci. Emerg. Technol. 2018, 48, 296–303. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, H.; Tian, X.; Zhao, C.; Cai, L.; Liu, Y.; Jia, L.; Yin, H.X.; Chen, H.X. Antioxidant potential of crocins and ethanol exracts of Gardenia jasminoides ELLIS and Crocus sativus L.: A relationahip investigation between antioxidant activity and crocin content. Food Chem. 2008, 109, 484–492. [Google Scholar] [CrossRef]
- Montoro, P.; Maldini, M.; Luciani, L.; Tuberoso, C.I.G.; Congiu, F.; Pizza, C. Radical scavenging activity and LC-MS metabolic profiling of petals, stamens, and flowers of Crocus sativus L. J. Food Sci. 2012, 77, 893–900. [Google Scholar] [CrossRef]
- Huwaitat, S.; Al-Khateeb, E.; Finjan, S.; Maraqa, A. Antioxidant and antimicrobial activities of Iris nigricans methanolic extracts containing phenolic compounds. Eur. Sci. J. 2013, 9, 83–91. [Google Scholar]
- Korinek, M.; Mykhailenko, O.; Hsieh, C.F.; Georgiyants, V.; El-Shazly, M.; Handoussa, H.; Horng, J.T.; Hwang, T.L. Antiviral effects of herbs from Ukraine against influenza and enterovirus. In Proceedings of the 34th Symposium of Natural Products, Taoyuan, Taiwan, 17–19 October 2019. [Google Scholar]
- Alam, A.; Jaiswal, V.; Akhtar, S.; Jayashree, B.S.; Dhar, K.L. Isolation of isoflavones from Iris kashmiriana Baker as potential anti proliferative agents targeting NF-kappaB. Phytochemistry 2017, 136, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Lamshöft, M.; Marner, F.J. Analysis of the iridals in rhizome extracts of Iris variegata Linn. Nat. Prod. Res. 2005, 19, 57–60. [Google Scholar] [CrossRef]
- Peder, O.; Larsena, M.; Sundahla, F.; Torben, S.; Wieczorkowskaa, E.; Goldblat, P. Relationship between subfamilies, tribes and genera in Iridaceae inferred from chemical characters. Biochem. Syst. Ecol. 1987, 15, 575–579. [Google Scholar] [CrossRef]
- Peder, O.; Larsena, M.; Sundahla, F.; Torben, S.; Wieczorkowskaa, E.; Goldblat, P. Meta-carboxy-substituted aromatic amino acids and γ-glutamyl peptides: Chemical characters for classification in the Iridaceae. Biochem. Syst. Ecol. 1981, 9, 313–323. [Google Scholar] [CrossRef]
- Chen, Y.; Fu, X.; Mei, X.; Zhou, Y.; Cheng, S.; Zeng, L.; Dong, F.; Yang, Z. Proteolysis of chloroplast proteins is responsible for accumulation of free amino acidsin dark-treated tea (Camellia sinensis) leaves. J. Proteom. 2017, 157, 10–17. [Google Scholar] [CrossRef]
- Hung, Y.T.; Chen, P.C.; Chen, R.L.C.; Cheng, T.J. Sequential determination oftannin and total amino acid contents in tea for taste assessment by a fluorescent flow-injection analytical system. Food Chem. 2010, 118, 876–881. [Google Scholar] [CrossRef]
- Zhu, Y.; Luo, Y.; Wang, P.; Zhao, M.; Li, L.; Hu, X.; Chen, F. Simultaneous determination of free amino acids in Pu-erh tea and their changes during fermentation. Food Chem. 2015, 194, 643–649. [Google Scholar] [CrossRef]
- Horanni, R.; Engelhardt, U.H. Determination of amino acids in white, green, black, oolong, puerh teas and tea products. J. Food Compos. Anal. 2013, 31, 94–100. [Google Scholar] [CrossRef]
- Zhou, P.; Zhao, F. Determination of 21 free amino acids in 5 types of tea by ultra-high performance liquid chromatography coupled with tandem mass spectrometry (UHPLC–MS/MS) using a modified 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate (AQC) method. J. Food Compos. Anal. 2019, 81, 46–54. [Google Scholar] [CrossRef]
- Fu, G.; He, Y.; Wang, X.; Wang, L. Determination of amino acids in tea samples bycapillary electrophoresis with partition cell and indirect ultraviolet detection. Chin. J. Chromatogr. 2007, 25, 193–196. [Google Scholar]
- Hener, C.; Hummel, S.; Suarez, J.; Stahl, M.; Kolukisaoglu, Ü. d-Amino acids are exuded by arabidopsis thaliana roots to the rhizosphere. Int. J. Mol. Sci. 2018, 19, 1109. [Google Scholar] [CrossRef]
- Prokopenko, Y.; Jakštas, V.; Žvikas, V.; Georgiyants, V.; Ivanauskas, L. Hilic MS/MS determination of amino acids in herbs of Fumaria schleicheri L., Ocimum basilicum L., and leaves of Corylus avellana L. Nat.Prod. Res. 2018, 33, 1961–1963. [Google Scholar] [CrossRef]
- Koek, M.M.; Jellema, R.H.; Van der Greef, J.; Tas, A.C.; Hankemeier, T. Quantitative metabolomics based on gaschromatography mass spectrometry: Status and perspectives. Metabolomics 2011, 7, 307–328. [Google Scholar] [CrossRef]
- Bruheim, P.; Kvitvang, H.F.N.; Villas-Boas, S.G. Stable isotope coded derivatizing reagents as internal standardsin metabolite profiling. J. Chromatogr. A 2013, 1296, 196–203. [Google Scholar] [CrossRef]
- Vielhauer, O.; Zakhartsev, M.; Horn, T.; Takors, R.; Reuss, M. Simplified absolute metabolite quantification by gaschromatography-isotope dilution mass spectrometry on the basisof commercially available source material. J. Chromatogr. B 2011, 879, 3859–3870. [Google Scholar] [CrossRef]
- Villas-Boas, S.G.; Smart, K.F.; Sivakumaran, S.; Lane, G.A. Alkylation or silylation for analysis of amino and non-amino organic acids by GC-MS? Metabolites 2011, 1, 3–20. [Google Scholar] [CrossRef]
- Tumanov, S.; Zubenko, Y.; Obolonkin, V.; Greenwood, D.R.; Shmanai, V.; Villas-Bôas, S.G. Calibration curve-free GC-MS method for quantification of amino and non-amino organic acids in biological samples. Metabolomics 2016, 12, 64. [Google Scholar] [CrossRef]
- Hryzodub, O.I.; Leontyev, D.A.; Dmitriyeva, M.V.; Rudyk, Z.S.; Kotov, A.G.; Kotova, E.E.; Kishenets, N.V.; Klestova, Z.S.; Zhemerova, K.G.; Chekalova, S.O.; et al. State Pharmacopoeia of Ukraine: At 3 vol, 2nd ed.; State Enterprise “Ukrainian Scientific Pharmacopoeial Center of Medicines Quality”: Kharkiv, Ukraine, 2015; Volume 1, p. 1128. [Google Scholar]
- Council of Europe. European Pharmacopoeia, 9.2th ed.; Council of Europe: Strasbourg, France, 2017. [Google Scholar]
- International Conference on Harmonization (ICH). Validation of Analytical Procedures: Text and Methodology Q2 (R1); ICH Secretariat: Geneva, Switzerland, 2005. [Google Scholar]
- Jarukas, L.; Kamarauskaitė, J.; Marksa, M.; Trumbeckaitė, S.; Banienė, R.; Ivanauskas, L. Bio-based succinic acid sample preparation and derivatization procedure optimisation for gas chromatography-mass spectrometry analysis. Sci. J. Sci. Rise Pharm. Sci. 2018, 4, 9–13. [Google Scholar] [CrossRef][Green Version]
- Chen, Z.; Landman, P.; Colmer, T.D.; Adams, M.A. Simultaneous analysis of amino and organic acids in extracts of plant leaves astert-butyldimethylsilyl derivatives by capillary gas chromatography. Anal. Biochem. 1998, 259, 203–211. [Google Scholar] [CrossRef]
- Ayalew, Y.; Retta, N.; Desse, G.; Mohammed, A.; Mellesse, A. Amino acid profile and protein quality in tuber and leaf of Coccnia abyssinica (Lam.) (Cogn.) accessions of Ethiopia. Food Sci. Nutr. 2016, 5, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Dash, P.; Ghosh, G. Amino acid composition, antioxidant and functional properties of protein hydrolysates from Cucurbitaceae seeds. J. Food Sci. Technol. 2017, 54, 4162–4172. [Google Scholar] [CrossRef] [PubMed]
- Sonklin, C.; Laohakunjit, N.; Kerdchoechuen, O. Assessment of antioxidant properties of membrane ultrafiltration peptides from mungbean meal protein hydrolysates. Peer J. 2018, 6, e5337. [Google Scholar] [CrossRef] [PubMed]
- Pompella, A.; Visvikis, A.; Paolicchi, A.; De Tata, V.; Casini, A.F. The changing faces of glutathione, a cellular protagonist. Biochem. Pharmacol. 2003, 66, 1499–1503. [Google Scholar] [CrossRef]
- Del Campo, C.P.; Garde-Cerdán, T.; Sánchez, A.M.; Maggi, L.; Carmona, M.; Alonso, G.L. Determination of free amino acids and ammonium ion in saffron (Crocus sativus L.) from different geographical origins. Food Chem. 2009, 114, 1542–1548. [Google Scholar] [CrossRef]
- Covarrubias-Cárdenas, A.G.; Martínez-Castillo, J.I.; Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; García-Cruz, N.U.; Pacheco, N. Antioxidant capacity and UPLC-PDA ESI-MS phenolic profile of Stevia rebaudiana dry powder extracts obtained by ultrasound assisted extraction. Agronomy 2018, 8, 170. [Google Scholar] [CrossRef]
- Joshi, A.J.; Kumar, A.S. Seasonal variations of proteins and amino acids in three salt marsh species. Plant Sci. 1989, 99, 287–292. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, G. Research advance in nitrogen metabolism of plant and its environmental regulation. Ying Yong Sheng Tai Xue Bao 2004, 15, 511–516. (In Chinese) [Google Scholar]
- Mykhailenko, O.; Gudžinskas, Z.; Kovalyov, V.; Desenko, V.; Ivanauskas, L.; Bezruk, I.; Georgiyants, V. Effect of ecological factors on the accumulation of phenolic compounds in Iris species from Latvia, Lithuania and Ukraine. Phytochem. Anal. 2020, 1, 1–19. [Google Scholar] [CrossRef]
- Cao, M.; Gao, M.; Suástegui, M.; Mei, Y.; Shao, Z. Building microbial factories for the production of aromatic amino acid pathway derivatives: From commodity chemicals to plant-sourced natural products. Metab. Eng. 2019, in press. [Google Scholar] [CrossRef]
- Filiz, E.; Cetin, D.; Akbudak, M.A. Aromatic amino acids biosynthesis genes identification and expression analysis under salt and drought stresses in Solanum lycopersicum L. Sci. Hortic. 2019, 250, 127–137. [Google Scholar] [CrossRef]
- Panday, S.K.; Prasad, J.; Dikshit, D.K. Pyroglutamic acid: A unique chiral synthon. Tetrahedron Asymmetry 2009, 20, 1581–1632. [Google Scholar] [CrossRef]
- Stefanucci, A.; Novellino, E.; Costante, R.; Mollica, A. Pyroglutamic acid derivatives: Building blocks for drug discovery. Heterocycles 2014, 89, 1801–1825. [Google Scholar] [CrossRef]
- Wang, G.; Xu, M.; Wang, W.; Galili, G. Fortifying horticultural crops with essential amino acids: A review. Int. J. Mol. Sci. 2017, 18, 1306. [Google Scholar] [CrossRef]
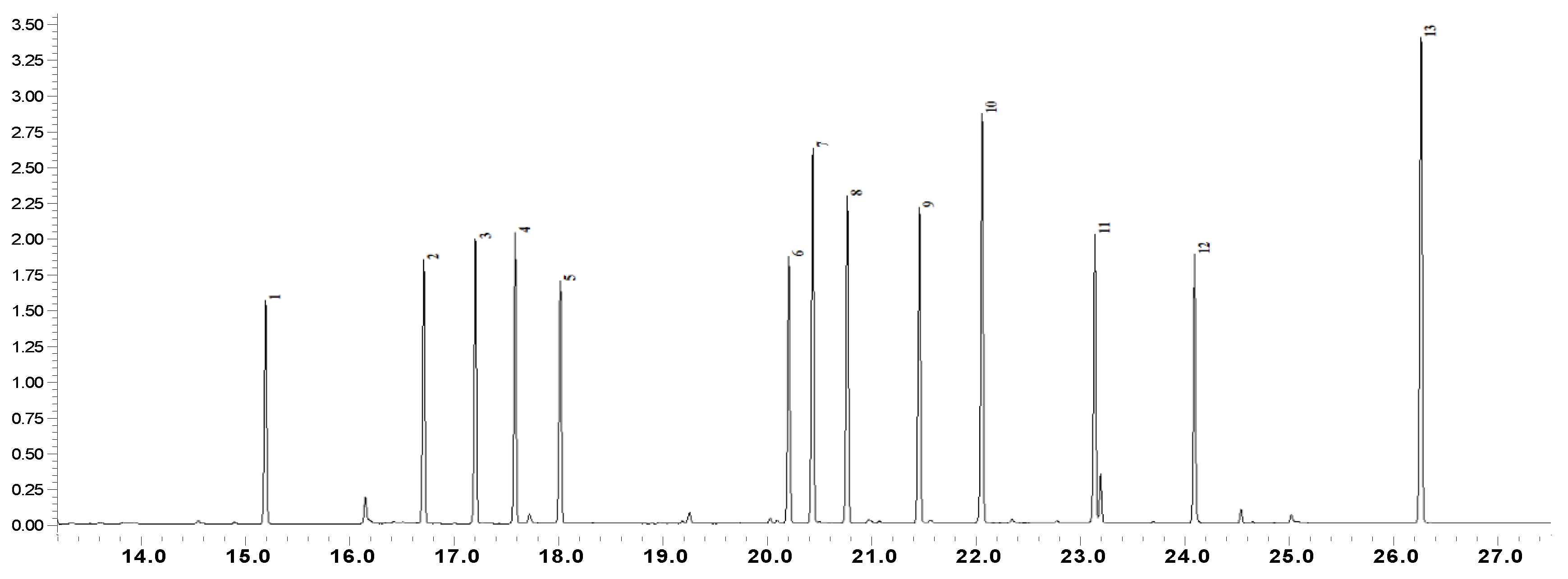
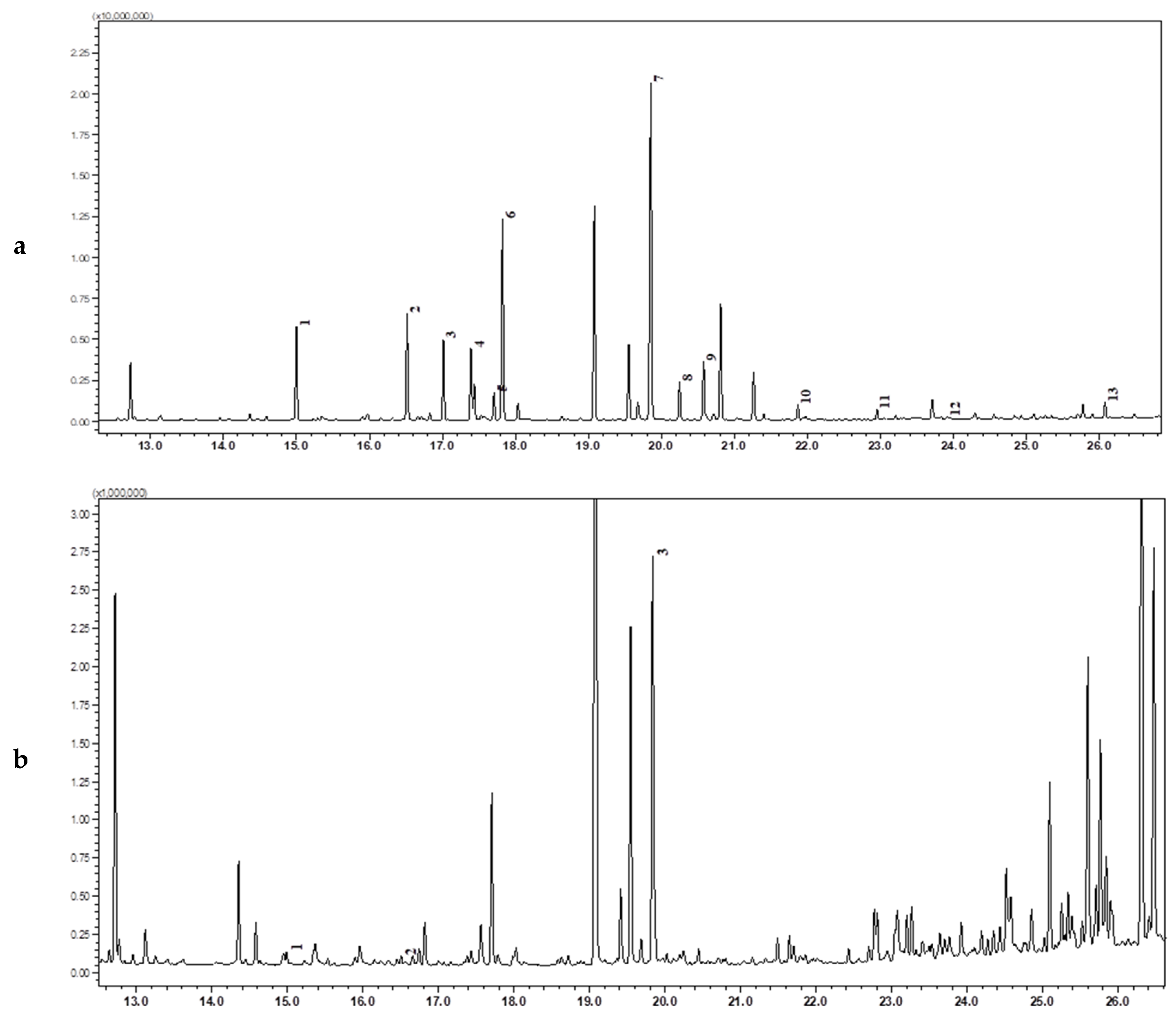
| N | R.Time | Name of Amino Acid | Gladiolus Leaves | Juno Leaves | Crocus Leaves | Crocus Flowers | Crocus Stigma | Crocus Corms | Juno Corms | Iris Hungarica Rhizome | Iris Variegata Rhizome |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 14,98 | l-Alanine | 34.9 ± 0.7 | 1032.2 ± 15.1 | 2709.5 ± 75.1 | 156.3 ± 1.7 | 60.0 ± 2.1 | 204.1 ± 2.1 | 52.1 ± 1.5 | n/d | 114.9 ± 1.4 |
| 2 | 16,51 | l-Valine | 24.3 ± 0.5 | 1411.1 ± 20.7 | 3023.0 ± 51.1 | 105.8 ± 2.1 | n/d | 458.7 ± 19.3 | 24.5 ± 0.5 | n/d | 81.2 ± 2.6 |
| 3 | 17,00 | l-Leucine | n/d | 1016.2 ± 18.5 | 2401.5 ± 75.2 | 72.2 ± 1.6 | n/d | 80.7 ± 1.3 | n/d | n/d | n/d |
| 4 | 17,34 | Isoleucine | n/d | 1049.2 ± 25.4 | 2322.5 ± 67.8 | 101.7 ± 4.8 | n/d | 174.9 ± 4.8 | 31.2 ± 0.6 | n/d | 50.2 ± 1.2 |
| 5 | 17,70 | 4-Aminobutanoic acid (recalculated to l-Glutamic acid) | n/d | 255.6± 7.2 | 728.8 ± 29.6 | n/d | n/d | n/d | n/d | n/d | n/d |
| 6 | 17,81 | l-Proline | n/d | 73.8 ± 1.9 | 4683.5 ± 171.0 | n/d | n/d | 2251.6 ± 82.5 | 49.6 ± 1.8 | n/d | 68.8 ± 2.4 |
| 7 | 19,84 | l-Pyroglutamic acid (recalculated to l-Glutamic acid) | 1695.2 ± 46.1 | 8538.8 ± 196.4 | 12347.5 ± 517.3 | 1158.4 ± 29.7 | n/d | 6318.5 ± 295.1 | 330.9 ± 6.4 | 1340.1 ± 56.7 | 783.9 ± 19.3 |
| 8 | 19,80 | l-Methionine | n/d | n/d | n/d | n/d | 84.1 ± 2.4 | n/d | n/d | n/d | n/d |
| 9 | 20,24 | l-Serine | n/d | 348.6 ± 3.7 | 790.6 ± 25.3 | 77.5 ± 1.7 | 14.2 ± 0.2 | 213.6 ± 8.6 | n/d | n/d | 24.2 ± 0.8 |
| 10 | 20,57 | l-Threonine | n/d | 473.8 ± 9.6 | 1645.5 ± 27.5 | 63.9 ± 1.3 | n/d | 190.1 ± 5.4 | n/d | n/d | n/d |
| 11 | 21,25 | l-Phenylalanine | n/d | 391.0 ± 9.1 | n/d | n/d | n/d | 68.4 ± 2.3 | n/d | n/d | n/d |
| 12 | 21,86 | l-Aspartic acid | n/d | 63.1 ± 1.5 | 355.9 ± 9.7 | n/d | n/d | n/d | n/d | n/d | 31.4 ± 0.7 |
| 13 | 22,95 | l-Glutamic acid | n/d | n/d | 381.8 ± 9.9 | n/d | n/d | n/d | n/d | n/d | n/d |
| 14 | 23,91 | l-Lysine | n/d | n/d | 150.5 ± 40.9 | n/d | n/d | n/d | n/d | n/d | n/d |
| 15 | 26,08 | l-Tyrosine | n/d | n/d | 452.7 ± 16.2 | n/d | 326.6 ± 6.9 | n/d | n/d | n/d | n/d |
| Total amount | 1757.1 | 14653.2 | 31993.3 | 1735.8 | 484.9 | 9960.5 | 488.3 | 1340.1 | 1154.6 |
| Amino Acid | Calibration Curve | Correlation coefficient r2 | Linear Range (μm/mL) | LOD (ng/mL) | LOQ (ng/mL) |
|---|---|---|---|---|---|
| L-Alanine | f(x) = 276580.863935 × x − 353207.105647 | 0.983122 | 11.15–44.6 | 0.72 | 2.4 |
| L-Valine | f(x) = 243032.088633 × x + 49012.859468 | 0.975345 | 14.7–58.8 | 0.585 | 1.930 |
| L-Leucine | f(x) = 277306.790190 × x − 217513.405090 | 0.963603 | 16.5–58.8 | 0.1257 | 0.459 |
| Isoleucine | f(x) = 228260.308590 × x + 246041.176091 | 0.969531 | 16.45–65.8 | 0.125 | 0.491 |
| L-Proline | f(x) = 280385.380976 × x + 1325395.667639 | 0.972168 | 14.4–57.6 | 0.247 | 0.970 |
| L-Methionine | f(x) = 187897.462370 × x − 557659.056929 | 0.981929 | 18.65–74.6 | 0.184 | 0.213 |
| L-Serine | f(x) = 317111.017808 × x + 1640589.305389 | 0.957872 | 13.15–52.6 | 0.14 | 0.470 |
| L-Threonine | f(x) = 249467.821934 × x + 2284199.418438 | 0.953095 | 14.9–59.6 | 0.132 | 0.44 |
| L-Phenylalanine | f(x) = 206843.387916 × x + 74215.145383 | 0.963723 | 20.65–82.6 | 0.375 | 0.482 |
| L-Aspartic acid | f(x) = 267570.147433 × x + 2368216.421104 | 0.942835 | 16.65–66.6 | 0.308 | 0.362 |
| L-Glutamic acid | f(x) = 214747.719667 × x − 596641.230769 | 0.980993 | 18.4–73.6 | 0.195 | 0.651 |
| L-Lysine | f(x) = 232710.756329 × x − 3270542.068621 | 0.988024 | 18.25–73.00 | 0.34 | 0.144 |
| L-Tyrosine | f(x) = 213345.354626 × x + 3840401.269116 | 0.885138 | 22.65–90.6 | 0.53 | 1.76 |
| Amino Acid | RSD of Retention Time (%) | Precision | Repeatability |
|---|---|---|---|
| RSD of Area (%) | RSD of Area (%) | ||
| l-Alanine | 0.01 | 1.52 | 0.75 |
| l-Valine | 0.009 | 1.34 | 1.64 |
| l-Leucine | 0.01 | 1.78 | 1.70 |
| Isoleucine | 0.008 | 1.80 | 0.83 |
| 4-Aminobutanoic acid | 0.01 | 1.92 | 1.73 |
| l-Proline | 0.01 | 0.78 | 0.95 |
| l-Pyroglutamic acid | 0.1 | 1.71 | 0.94 |
| l-Methionine | 0.008 | 1.53 | 1.72 |
| l-Serine | 0.008 | 1.94 | 0.62 |
| l-Threonine | 0.01 | 0.83 | 1.48 |
| l-Phenylalanine | 0.009 | 1.71 | 0.72 |
| l-Aspartic acid | 0.01 | 2.39 | 1.37 |
| l-Glutamic acid | 0.009 | 1.56 | 1.41 |
| l-Lysine | 0.01 | 0.70 | 0.67 |
| l-Tyrosine | 0.04 | 1.75 | 0.88 |
| Amino Acid | Found (µg/mL) | Added (µg/mL) | Recovery (%) | Amino Acid | Found (µg/mL) | Added (µg/mL) | Recovery (%) |
|---|---|---|---|---|---|---|---|
| l-Alanine | 10.69 | 11.15 | 95.86 | l-Threonine | 14.88 | 14.90 | 99.87 |
| 19.40 | 22.30 | 86.99 | 30.51 | 37.25 | 81.91 | ||
| 34.16 | 33.45 | 102.11 | 50.51 | 44.70 | 113.00 | ||
| l-Valine | 13.98 | 14.70 | 95.11 | l-Phenylalanine | 18.16 | 20.65 | 87.92 |
| 25.66 | 29.40 | 87.27 | 35.63 | 41.30 | 86.27 | ||
| 46.95 | 44.10 | 106.46 | 68.05 | 61.95 | 109.85 | ||
| l-Leucine | 15.96 | 16.50 | 96.74 | l-Aspartic acid | 14.58 | 16.65 | 87.56 |
| 27.15 | 33.10 | 82.01 | 34.77 | 33.30 | 104.42 | ||
| 48.91 | 44.10 | 110.91 | 56.61 | 49.95 | 113.34 | ||
| Isoleucine | 16.05 | 16.45 | 97.57 | l-Glutamic acid | 15.66 | 18.40 | 85.11 |
| 28.17 | 32.90 | 85.63 | 31.65 | 36.80 | 86.02 | ||
| 54.87 | 49.35 | 111.18 | 57.23 | 55.20 | 103.67 | ||
| l-Proline | 13.79 | 14.40 | 95.73 | l-Lysine | 16.39 | 18.25 | 89.82 |
| 28.20 | 28.80 | 97.92 | 25.93 | 27.38 | 94.72 | ||
| 48.28 | 43.20 | 111.75 | 51.51 | 54.75 | 94.08 | ||
| l-Methionine | 16.58 | 18.65 | 88.89 | l-Tyrosine | 18.67 | 22.65 | 82.43 |
| 31.86 | 37.30 | 85.41 | 49.89 | 45.30 | 110.13 | ||
| 57.05 | 55.95 | 101.97 | 79.25 | 67.95 | 116.62 | ||
| l-Serine | 11.93 | 13.15 | 90.73 | ||||
| 26.30 | 26.30 | 100.00 | |||||
| 44.53 | 39.45 | 112.89 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mykhailenko, O.; Ivanauskas, L.; Bezruk, I.; Lesyk, R.; Georgiyants, V. Comparative Investigation of Amino Acids Content in the Dry Extracts of Juno bucharica, Gladiolus Hybrid Zefir, Iris Hungarica, Iris Variegata and Crocus Sativus Raw Materials of Ukrainian Flora. Sci. Pharm. 2020, 88, 8. https://doi.org/10.3390/scipharm88010008
Mykhailenko O, Ivanauskas L, Bezruk I, Lesyk R, Georgiyants V. Comparative Investigation of Amino Acids Content in the Dry Extracts of Juno bucharica, Gladiolus Hybrid Zefir, Iris Hungarica, Iris Variegata and Crocus Sativus Raw Materials of Ukrainian Flora. Scientia Pharmaceutica. 2020; 88(1):8. https://doi.org/10.3390/scipharm88010008
Chicago/Turabian StyleMykhailenko, Olha, Liudas Ivanauskas, Ivan Bezruk, Roman Lesyk, and Victoriya Georgiyants. 2020. "Comparative Investigation of Amino Acids Content in the Dry Extracts of Juno bucharica, Gladiolus Hybrid Zefir, Iris Hungarica, Iris Variegata and Crocus Sativus Raw Materials of Ukrainian Flora" Scientia Pharmaceutica 88, no. 1: 8. https://doi.org/10.3390/scipharm88010008
APA StyleMykhailenko, O., Ivanauskas, L., Bezruk, I., Lesyk, R., & Georgiyants, V. (2020). Comparative Investigation of Amino Acids Content in the Dry Extracts of Juno bucharica, Gladiolus Hybrid Zefir, Iris Hungarica, Iris Variegata and Crocus Sativus Raw Materials of Ukrainian Flora. Scientia Pharmaceutica, 88(1), 8. https://doi.org/10.3390/scipharm88010008