Neuroprotective and Cognitive Enhancement Potentials of Angelica gigas Nakai Root: A Review
Abstract
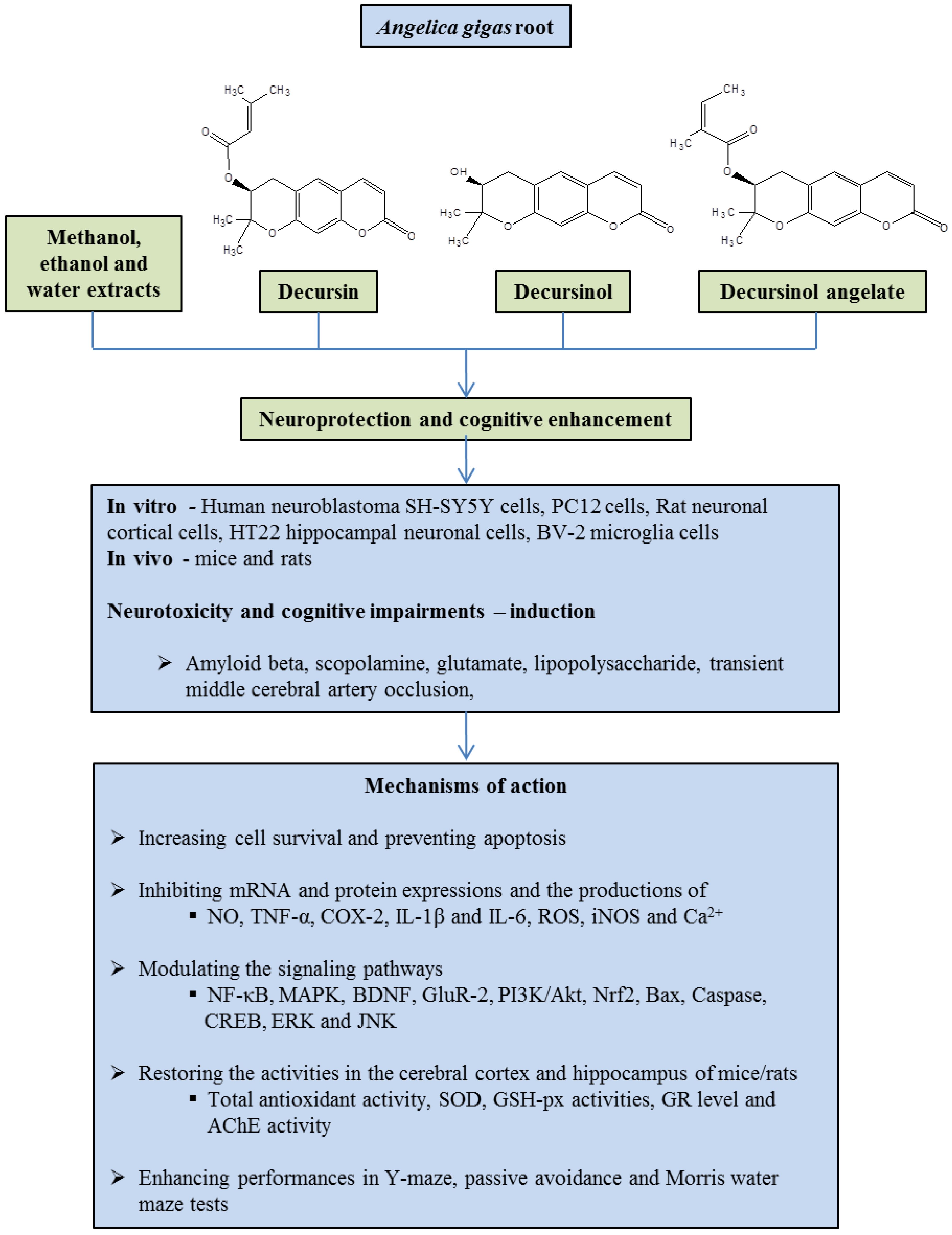
:1. Introduction
2. Neuroprotective Properties of A. gigas Root
2.1. Extracts
2.2. Decursin
2.3. Decursinol and Decursinol Angelate
2.4. Other Compounds
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tejada, S.; Setzer, W.; Daglia, M.; Nabavi, S.F.; Sureda, A.; Braidy, N.; Gortzi, O.; Nabavi, S.M. Neuroprotective effects of Ellagitannins: A brief review. Curr. Drug Targets 2016, 7, 26–33. [Google Scholar] [CrossRef]
- Hirsch, E.C.; Vyas, S.; Hunot, S. Neuroinflammation in Parkinson’s disease. Parkinsonism Relat. Disord. 2012, 18, S210–S212. [Google Scholar] [CrossRef]
- Chen, W.W.; Zhang, X.; Huang, W.J. Role of neuroinflammation in neurodegenerative diseases (Review). Mol. Med. Rep. 2016, 13, 3391–3396. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.; Khodagholi, F. Natural products as promising drug candidates for the treatment of Alzheimer’s disease: Molecular mechanism aspect. Curr. Neuropharmacol. 2013, 11, 414–429. [Google Scholar] [CrossRef] [PubMed]
- Sultana, R.; Butterfield, D.A. Role of oxidative stress in the progression of Alzheimer’s disease. J. Alzheimers Dis. 2010, 19, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Jalsrai, A.; Numakawa, T.; Kunugi, H.; Dieterich, D.C.; Becker, A. The neuroprotective effects and possible mechanism of action of a methanol extract from Asparagus cochinchinensis: In vitro and in vivo studies. Neuroscience 2016, 322, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.; Khodagholi, F.; Amini, M. 2-Ethoxy-4,5-diphenyl-1,3-oxazine-6-one activates the Nrf2/HO-1 axis and protects against oxidative stress-induced neuronal death. Eur. J. Pharmacol. 2011, 658, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Ashabi, G.; Ramin, M.; Azizi, P.; Taslimi, Z.; Alamdary, S.Z.; Haghparast, A.; Ansari, N.; Motamedi, F.; Khodagholi, F. ERK and p38 inhibitors attenuate memory deficits and increase CREB phosphorylation and PGC-1α levels in Aβ-injected rats. Behav. Brain Res. 2012, 232, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Son, B.G.; Park, Y.H.; Kim, C.M.; Park, G.; Choi, Y.W. The neuroprotective effects of α-iso-cubebene on dopaminergic cell death: Involvement of CREB/Nrf2 signaling. Neurochem. Res. 2014, 39, 1759–1766. [Google Scholar] [CrossRef] [PubMed]
- Freese, J.L.; Pino, D.; Pleasure, S.J. Wnt signaling in development and disease. Neurobiol. Dis. 2010, 38, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hu, D.; Zhang, L.; Lian, G.; Zhao, S.; Wang, C.; Yin, J.; Wu, C.; Yang, J. Gomisin A inhibits lipopolysaccharide-induced inflammatory responses in N9 microglia via blocking the NF-κB/MAPKs pathway. Food. Chem. Toxicol. 2014, 63, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.T.; Cohan, C.H.; Dave, K.R.; Wright, C.B.; Perez-Pinzon, M.A. Global cerebral ischemia: Synaptic and cognitive dysfunction. Curr. Drug Targets 2013, 14, 20–35. [Google Scholar] [CrossRef] [PubMed]
- Pak, M.E.; Kim, Y.R.; Kim, H.N.; Ahn, S.M.; Shin, H.K.; Baek, J.U.; Choi, B.T. Studies on medicinal herbs for cognitive enhancement based on the text mining of Dongeuibogam and preliminary evaluation of its effects. J. Ethnopharmacol. 2016, 179, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, Y. Recent research progress in and future perspective on treatment of Parkinson’s disease. Integr. Med. Int. 2014, 1, 67–79. [Google Scholar] [CrossRef]
- Crane, P.K.; Doody, R.S. Donepezil treatment of patients with MCI: A 48-week randomized, placebo-controlled trial. Neurology 2009, 73, 1514–1516. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.K.; Grossberg, G.T. Diagnosis and treatment of Alzheimer’s disease. Neurology 2005, 64 (Suppl. 3), S34–S39. [Google Scholar] [CrossRef] [PubMed]
- Okun, M.S. Deep-brain stimulation—Entering the era of human neural-network modulation. N. Engl. J. Med. 2014, 371, 1369–1373. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Pan, W. The treatment strategies for neurodegenerative diseases by integrative medicine. Integr. Med. Int. 2014, 1, 223–225. [Google Scholar] [CrossRef]
- Daulatzai, M.A. Neurotoxic saboteurs: Straws that break the hippo’s (hippocampus) back drive cognitive impairment and Alzheimer’s disease. Neurotox. Res. 2013, 24, 407–459. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.K.; Kumar, R.; Mishra, A.; Gupta, R. Problems associated with clinical trials of Ayurvedic medicines. Braz. J. Pharmacogn. 2010, 20, 276–281. [Google Scholar] [CrossRef]
- Sen, S.; Chakraborty, R. Revival, modernization and integration of Indian traditional herbal medicine in clinical practice: Importance, challenges and future. J. Tradit. Complement. Med. 2016, 7, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Sarker, S.D.; Nahar, L. Natural medicine: The genus Angelica. Curr. Med. Chem. 2004, 11, 1479–1500. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.W.; Che, J.H.; Kwon, E.; Kim, Y.S.; Kim, S.H.; You, J.R.; Kim, W.H.; Kim, H.H.; Kang, B.C. Safety evaluation of Angelica gigas: Genotoxicity and 13-weeks oral subchronic toxicity in rats. Regul. Toxicol. Pharmacol. 2015, 72, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.J.; Kim, D.H.; Moon, Y.S.; Jung, J.S.; Ahn, E.M.; Baek, N.I.; Song, D.K. Protection against beta-amyloid peptide-induced memory impairment with long-term administration of extract of Angelica gigas or decursinol in mice. Prog. Neuro-Psychopharmacol. Biol. Psychiatr. 2004, 28, 25–30. [Google Scholar] [CrossRef]
- Piao, J.; Lee, J.Y.; Weon, J.B.; Ma, C.J.; Ko, H.J.; Kim, D.D.; Kang, W.S.; Cho, H.J. Angelica gigas Nakai and soluplus-based solid formulations prepared by hot-melting extrusion: oral absorption enhancing and memory ameliorating effects. PLoS ONE 2015, 10, e0124447. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Du, J.K.; Zou, L.Y.; Wu, T.; Lee, Y.W.; Kim, Y.H. Decursin isolated from Angelica gigas Nakai rescues PC12 cells from amyloid β-protein-induced neurotoxicity through Nrf2-mediated upregulation of heme oxygenase-1: Potential roles of MAPK. Evid. Based Complement. Altern. Med. 2013, 2013, 467245. [Google Scholar]
- Li, L.; Zhang, J.; Xing, C.; Kim, S.H.; Jiang, C.; Lu, J. In vitro metabolism of pyranocoumarin isomers decursin and decursinol angelate by liver microsomes from man and rodents. Planta Med. 2013, 79, 1536–1544. [Google Scholar] [CrossRef] [PubMed]
- Oh, T.W.; Park, K.H.; Jung, H.W.; Park, Y.K. Neuroprotective effect of the hairy root extract of Angelica gigas NAKAI on transient focal cerebral ischemia in rats through the regulation of angiogenesis. BMC Complement. Altern. Med. 2015, 15, 101. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.J.; Park, Y.K. Effects of different parts of Angelica gigas Nakai on brain damages and neuronal death in transient middle artery occlusion/reperfusion-induced ischemic rats. Korea J. Herbol. 2014, 29, 85–93. [Google Scholar] [CrossRef]
- Li, L.; Du, J.; Liyi, Z.; Xia, H.; Wu, T.; Kim, Y.; Lee, Y. The neuroprotective effects of decursin isolated from Angelica gigas Nakai against amyloid β-protein-induced apoptosis in PC12 cells via a mitochondria-related caspase pathway. Neurochem. Res. 2015, 40, 1555–1562. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Lee, K.Y.; Koo, K.A.; Yoon, J.S.; Lim, S.W.; Kim, Y.C.; Sung, S.H. ESP-102, a standardized combined extract of Angelica gigas, Saururus chinensis and Schizandra chinensis, significantly improved scopolamine-induced memory impairment in mice. Life Sci. 2005, 76, 1691–1705. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.J.; Kim, S.H.; Lee, K.Y.; Oh, T.; Kim, S.Y.; Sung, S.H.; Kim, Y.C. ESP-102, a combined extract of Angelica gigas, Saururus chinensis and Schizandra chinensis, protects against glutamate-induced toxicity in primary cultures of rat cortical cells. Phytother. Res. 2009, 23, 1587–1591. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.B.; Hwang, E.S.; Choi, G.Y.; Lee, S.; Park, T.S.; Lee, C.W.; Lee, E.S.; Kim, Y.C.; Kim, S.S.; Lee, S.O.; et al. ESP-102, a combined herbal extract of Angelica gigas, Saururus chinensis, and Schisandra chinensis, changes synaptic plasticity and attenuates scopolamine-induced memory impairment in rat hippocampus tissue. Evid. Based Complement. Altern. Med. 2016, 2016, 8793095. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.W.; Kim, S.T.; Son, H.J.; Han, P.L.; Cho, H.K.; Lee, Y.J.; Lyu, Y.S. Standardization of quality and inhibitory effect of Alzheimer in Aβ oligomer-induced H19-7 Cells by LMK02. Korean J. Orient. Physiol. Pathol. 2009, 23, 397–404. [Google Scholar]
- Seo, J.S.; Yun, J.H.; Baek, I.S.; Leem, Y.H.; Kang, H.W.; Cho, H.K.; Lyu, Y.S.; Son, H.J.; Han, P.L. Oriental medicine Jangwonhwan reduces Aβ(1-42) level and β-amyloid deposition in the brain of Tg-APPswe/PS1dE9 mouse model of Alzheimer disease. J. Ethnopharmacol. 2010, 128, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.S.; Jung, E.Y.; Kim, J.H.; Lyu, Y.S.; Han, P.L.; Kang, H.W. A modified preparation (LMK03) of the oriental medicine Jangwonhwan reduces Aβ1–42 level in the brain of Tg-APPswe/PS1dE9 mouse model of Alzheimer disease. J. Ethnopharmacol. 2010, 130, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Weon, J.B.; Lee, B.; Yun, B.R.; Lee, J.; Ma, J.Y.; Ma, C.J. Neuroprotective and cognitive enhancing activity of the fermented Bozhougyiqi-Tang. Pharmacogn. Mag. 2014, 10 (Suppl. 2), S249–S255. [Google Scholar] [PubMed]
- Kim, Y.O.; Ha, N.N.; Bu, Y.M.; Park, S.Y.; Park, J.Y.; Yu, Y.B.; Sin, J.S.; Kim, H.C.; An, D.G. Neuroprotective effect of Angelica gigas extracts on the brain ischemia induced by four-vessel occlusion in rats. Korea J. Herbol. 2002, 17, 151. [Google Scholar]
- Park, S.J.; Jung, J.M.; Lee, H.E.; Lee, Y.W.; Kim, D.H.; Kim, J.M.; Hong, J.G.; Lee, C.H.; Jung, I.H.; Cho, Y.B.; et al. The memory ameliorating effects of INM-176, an ethanolic extract of Angelica gigas, against scopolamine- or Aβ1–42-induced cognitive dysfunction in mice. J. Ethnopharmacol. 2012, 143, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Jung, H.J.; Son, M.S.; Jung, J.M.; Kim, D.H.; Jung, I.H.; Cho, Y.B.; Lee, E.H.; Ryu, J.H. Neuroprotective effects of INM-176 against lipopolysaccharide-induced neuronal injury. Pharmacol. Biochem. Behav. 2012, 101, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Lee, K.Y.; Park, M.J.; Kim, Y.C.; Markelonis, G.J.; Oh, T.H.; Kim, Y.C. Decursin from Angelica gigas mitigates amnesia induced by scopolamine in mice. Neurobiol. Learn. Mem. 2003, 79, 11–18. [Google Scholar] [CrossRef]
- Kang, S.Y.; Kim, Y.C. Decursinol and decursin protect primary cultured rat cortical cells from glutamate-induced neurotoxicity. J. Pharm. Pharmacol. 2007, 59, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, W.; Jung, S.W.; Lee, Y.W.; Kim, Y.H. Protective effects of decursin and decursinol angelate against amyloid β-protein-induced oxidative stress in the PC12 cell line: The role of Nrf2 and antioxidant enzymes. Biosci. Biotechnol. Biochem. 2011, 75, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Kim, D.Y.; Kim, Y.C.; Jung, J.W.; Lee, S.; Yoon, B.H.; Cheong, J.H.; Kim, Y.S.; Kang, S.S.; Ko, K.H.; et al. Nodakenin, a coumarin compound, ameliorates scopolamine-induced memory disruption in mice. Life Sci. 2007, 80, 1944–1950. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Kim, Y.C. Neuroprotective coumarins from the root of Angelica gigas: Structure-activity relationships. Arch. Pharm. Res. 2007, 30, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Lee, K.Y.; Sung, S.H.; Kim, Y.C. Four new neuroprotective dihydropyranocoumarins from Angelica gigas. J. Nat. Prod. 2005, 68, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, L.; Jiang, C.; Xing, C.; Kim, S.H.; Lu, J. Anti-cancer and other bioactivities of Korean Angelica gigas Nakai (AGN) and its major pyranocoumarin compounds. Anticancer Agents Med. Chem. 2012, 12, 1239–1254. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Noh, E.M.; Kim, M.S.; Hwang, J.K.; Hwang, H.Y.; Ryu, D.G.; Kim, H.J.; Yu, H.N.; You, Y.O.; Kim, J.S.; et al. Decursin prevents TPA-induced invasion through suppression of PKCα/p38/NF-κB-dependent MMP-9 expression in MCF-7 human breast carcinoma cells. Int. J. Oncol. 2014, 44, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, L.; Tang, S.; Hale, T.W.; Xing, C.; Jiang, C.; Lu, J. Cytochrome P450 isoforms in the metabolism of decursin and decursinol angelate from Korean Angelica. Am. J. Chin. Med. 2015, 43, 1211–1230. [Google Scholar] [CrossRef] [PubMed]
| Compound/Extract | Model | Mechanism | Dose | References |
|---|---|---|---|---|
| ESP-102 (a combined ethanol extract, consists of A. gigas, Saururus chinensis and Schisandra chinensis) | Scopolamine-induced memory impairment in mice. Aβ25–35 or glutamate-induced neurotoxicity in cortical neurons of rats. | Enhances cognitive performances in the passive avoidance and Morris water maze tests. Protects against neuronal damage. | Mice: 1 to 100 mg/kg; Cell line: 0.001, 0.01 and 0.1 μg/mL | [32] |
| Glutamate-induced toxicity in rat cortical cells. | Decreases [Ca2+]i, inhibits the production of NO and ROS. Increases SOD, GSH-px and GR. Maintains mitochondrial membrane potential. | 0.5 mg/mL to 50 mg/mL | [33] | |
| Scopolamine-induced memory impairment in rat hippocampus tissue | Antagonizes the preventative long-term potentiation effect. Increases BDNF and GluR-2 expressions. Acts on the AMPA/NMDA receptors. | 10 μg/mL | [34] | |
| LMK02 - Jangwonhwan (a herbal mixture of boiled extracts including A. gigas) | Aβ-induced neurotoxicity in H19-7 cells from rat hippocampus. | Protects against cytotoxicity. Inhibits formation of Aβ oligomer. Activates anti-apoptosis and decreases the production of ROS. | 1 to 100 μg/mL | [35] |
| Aβ-induced neurotoxicity in SH-SY5Y cells and Tg-APPswe/PS1dE9 mice. | Protects against cytotoxicity. Reduces Aβ1–42 and Aβ1–40 levels and β-amyloid plaque deposition in the brain. | Mice: 400 mg/kg, SH-SY5Y cells: 50 to 150 mg/mL | [36] | |
| LMK03-Jangwonhwan (a combined boiled extract, consists of white Poria cocos and A. gigas) | Aβ-induced neurotoxicity in SH-SY5Y neuroblastoma cells and Tg-APPswe/PS1dE9 transgenic mice. | Protects against cytotoxicity. Reduces Aβ1–42 and Aβ1–40 levels and β-amyloid plaque deposition in the brain. | Mice: 300 mg/kg, SH-SY5Y cells: 50 and 100 mg/mL | [37] |
| Fermented Bozhougyiqi-Tang (Herbal mixture) containing A. gigas | Scopolamine-induced memory impairments in mice and glutamate induced neurotoxicity in HT22 cells. | Protects against cytotoxicity. Enhances cognitive performance in the Morris water maze test. | Mice: 30, 100 and 200 mg/kg HT22 cells: 10, 100 and 1000 μg/mL | [38] |
| Water extract of A. gigas root | Transient middle cerebral artery occlusion-induced focal cerebral ischemia in rats. | Decreases the brain infarction volumes and edema. Decreases the blood brain barrier permeability and neuronal death and inhibits the activation of astrocytes and microglia. Increases the expression of Ang-1, Tie-2, VEGF, ZO-1 and Occludin via activation of the PI3K/Akt pathway. Increases the expression of ICAM-1. | 10, 25, 50 and 100 mg/kg | [29] |
| Methanol extract of A. gigas root | Four-vessel occlusion-induced ischemia in rats. | Attenuates COX-2 induction in hippocampus. | 100, 250 and 500 mg/kg | [39] |
| Ethanol extract of A. gigas root | Aβ-induced memory impairment in mice. | Enhances cognitive performances in the passive avoidance performance and Y-maze tests. | Ethanol extract: 0.1% | [25] |
| Scopolamine/Aβ-induced cognitive dysfunction in mice. | Enhances cognitive performances in the passive avoidance and Morris water maze tests. Inhibits AChE activity. Attenuates the astrocyte activation and cholinergic neuronal damage in the hippocampus and the nucleus basalis of Meynert. | 150, 300, 600 and 1200 mg/kg | [40] | |
| LPS-induced neuronal injury in BV2 microglial cells and mice. | Inhibits NO release and suppressed the expressions of TNF-α and IL-1β, iNOS and COX-2. Attenuates neuronal damage in a hippocampal slice culture. Enhances cognitive performances in the passive avoidance and Y-maze tests. Suppresses the activation of microglia or astrocytes. | 0.05–2 μg/mL | [41] | |
| Ethanol extract of A. gigas root | Neuronal death in transient middle artery occlusion/reperfusion-induced ischemic rats and LPS-induced inflammatory response in BV2 microglia. | Decreases infarction volume in ischemic brains and inhibits the expression of iNOS, bax and caspase-3. Inhibits the production of NO, TNF-α and IL-6, and suppresses the expression of iNOS and COX-2. Attenuates phosphorylation of ERK1/2 and JNK MAPK in BV2 cells. | 50 and 100 mg/kg | [30] |
| Hot-melting extrusion -processed A. gigas/Soluplus | Scopolamine-induced memory disruption in mice. | Enhances cognitive performances in the Morris water maze and passive avoidance tests. | 200 mg/kg | [26] |
| Decursin | Scopolamine-induced amnesia in mice. | Enhances cognitive performances in the passive avoidance and Morris water maze tests. Inhibits AChE activity. | 1 and 5 mg/kg | [42] |
| Glutamate-induced toxicity in rat cortical cells. | Protects against cytotoxicity. Reduces [Ca2+]i in cortical cells. Increases the glutathione and GSH-px levels. | 0.1–10.0 μM | [43] | |
| Decursin | Aβ-induced neurotoxicity in PC12 cells. | Increases Nrf2 expression and suppresses the aggregation of Amyloid-β. | 0.01–10.0 μM | [44] |
| Aβ-induced neurotoxicity in PC12 cells. | Induces Nrf2 nuclear translocation, the upstream of HO-1 expression, ERK and dephosphorylated p38. | 0.01–10 μM | [27] | |
| Aβ-induced neurotoxicity in PC12 cells. | Protects against cytotoxicity and apoptosis. Reduces the mitochondrial membrane potential, ROS production, and mitochondrial release of cytochrome c. Suppresses the caspase-3 activity and moderated the ratio of Bcl-2/Bax. | 0.01–10 μM | [31] | |
| Decursinol | Aβ-induced memory impairment in mice. | Enhances cognitive performances in the passive avoidance and Y-maze tests. | 0.001%, 0.002%, and 0.004% | [25] |
| Glutamate-induced toxicity in rat cortical cells. | Protects against cytotoxicity. Reduces [Ca2+]i in cortical cells. Protects neurons against KA-induced neurotoxicity. Increases the glutathione and GSH-px levels. | 0.1–10.0 μM | [43] | |
| Decursinol angelate | Aβ-induced neurotoxicity in PC12 cells. | Increases Nrf2 expression and suppresses the aggregation of Aβ. | 0.1–10.0 μM | [44] |
| Nodakenin | Scopolamine-induced memory disruption in mice. | Enhances cognitive performances in the passive avoidance, Y-maze and Morris water maze tests. Inhibits AChE activity. | 10 mg/kg | [45] |
| Glutamate-induced toxicity in rat cortical cells. | Protects against cytotoxicity. | 0.1 to 10 μM | [46] | |
| 4″-Hydroxytigloyldecursinol, 4″-hydroxydecursin, (2″S,3″S)-epoxyangeloyldecursinol, (2″R,3″R)-epoxyangeloyldecursinol, Marmesinin, columbianetin-O-β-D-glucopyranoside | Glutamate-induced toxicity in rat cortical cells. | Protects against cytotoxicity. | 0.1 to 10 μM | [46,47] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sowndhararajan, K.; Kim, S. Neuroprotective and Cognitive Enhancement Potentials of Angelica gigas Nakai Root: A Review. Sci. Pharm. 2017, 85, 21. https://doi.org/10.3390/scipharm85020021
Sowndhararajan K, Kim S. Neuroprotective and Cognitive Enhancement Potentials of Angelica gigas Nakai Root: A Review. Scientia Pharmaceutica. 2017; 85(2):21. https://doi.org/10.3390/scipharm85020021
Chicago/Turabian StyleSowndhararajan, Kandhasamy, and Songmun Kim. 2017. "Neuroprotective and Cognitive Enhancement Potentials of Angelica gigas Nakai Root: A Review" Scientia Pharmaceutica 85, no. 2: 21. https://doi.org/10.3390/scipharm85020021
APA StyleSowndhararajan, K., & Kim, S. (2017). Neuroprotective and Cognitive Enhancement Potentials of Angelica gigas Nakai Root: A Review. Scientia Pharmaceutica, 85(2), 21. https://doi.org/10.3390/scipharm85020021




