Quality Control of Traditional Cannabis Tinctures: Pattern, Markers, and Stability
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Reference Standards
2.3. Preparation of Hydroethanolic Tinctures
2.3.1. First Batch—Flower Tinctures (20%/40%/80% EtOH)
2.3.2. Second Batch—Flower Tinctures and Leaf Tinctures (40%/60%/90% EtOH)
2.4. HPLC Analysis
2.5. Determination and Calculation of Single, Group, and Ratio Markers
2.5.1. Peak Identification and Qualification
2.5.2. Assay
2.5.3. Group and Ratio Marker Calculation
2.6. Stability Tests
2.7. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl Tetrazolium Bromide (MTT) Assay
2.8. NF-κB IL-6/Luciferase Assay
3. Results
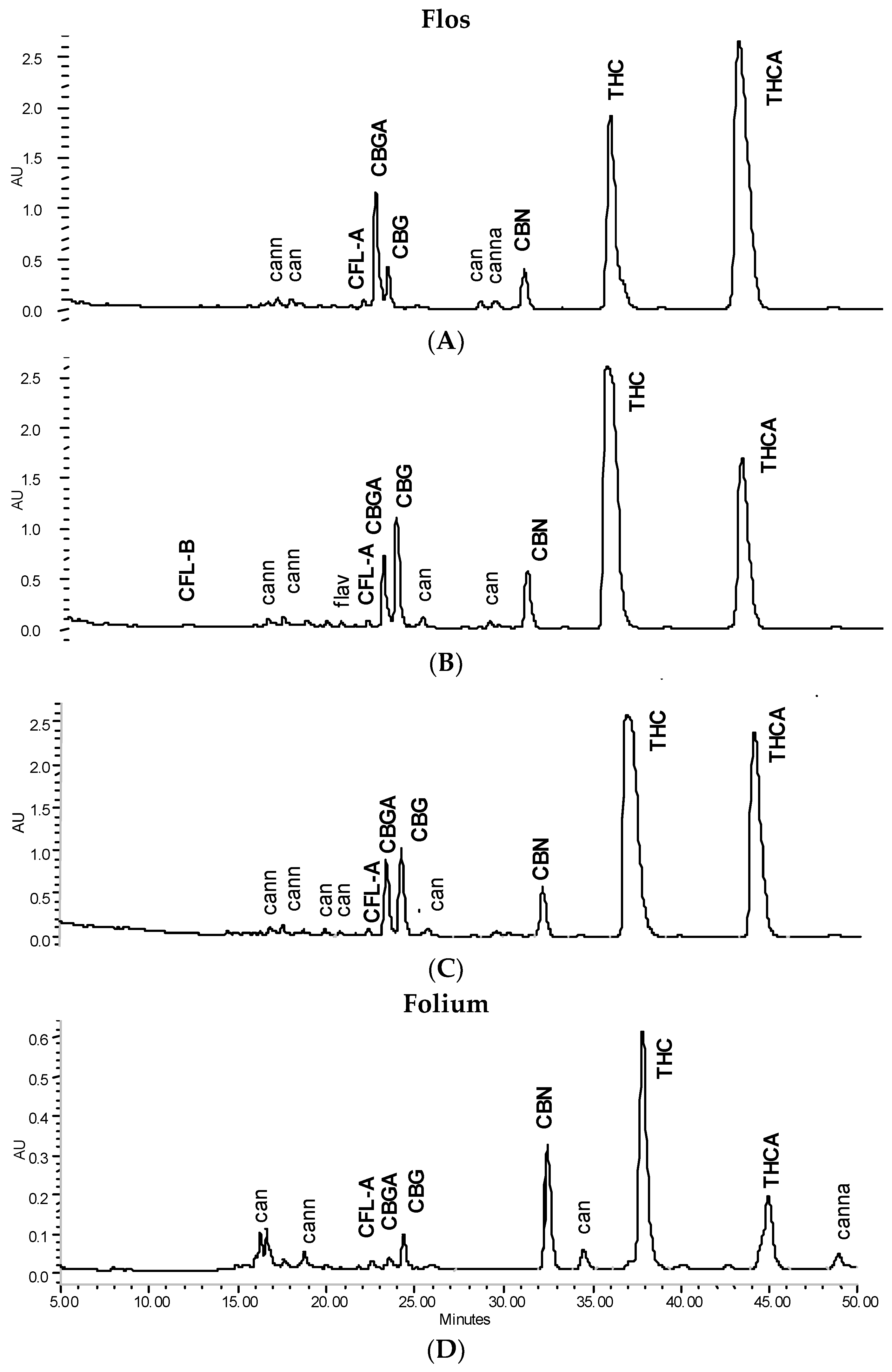
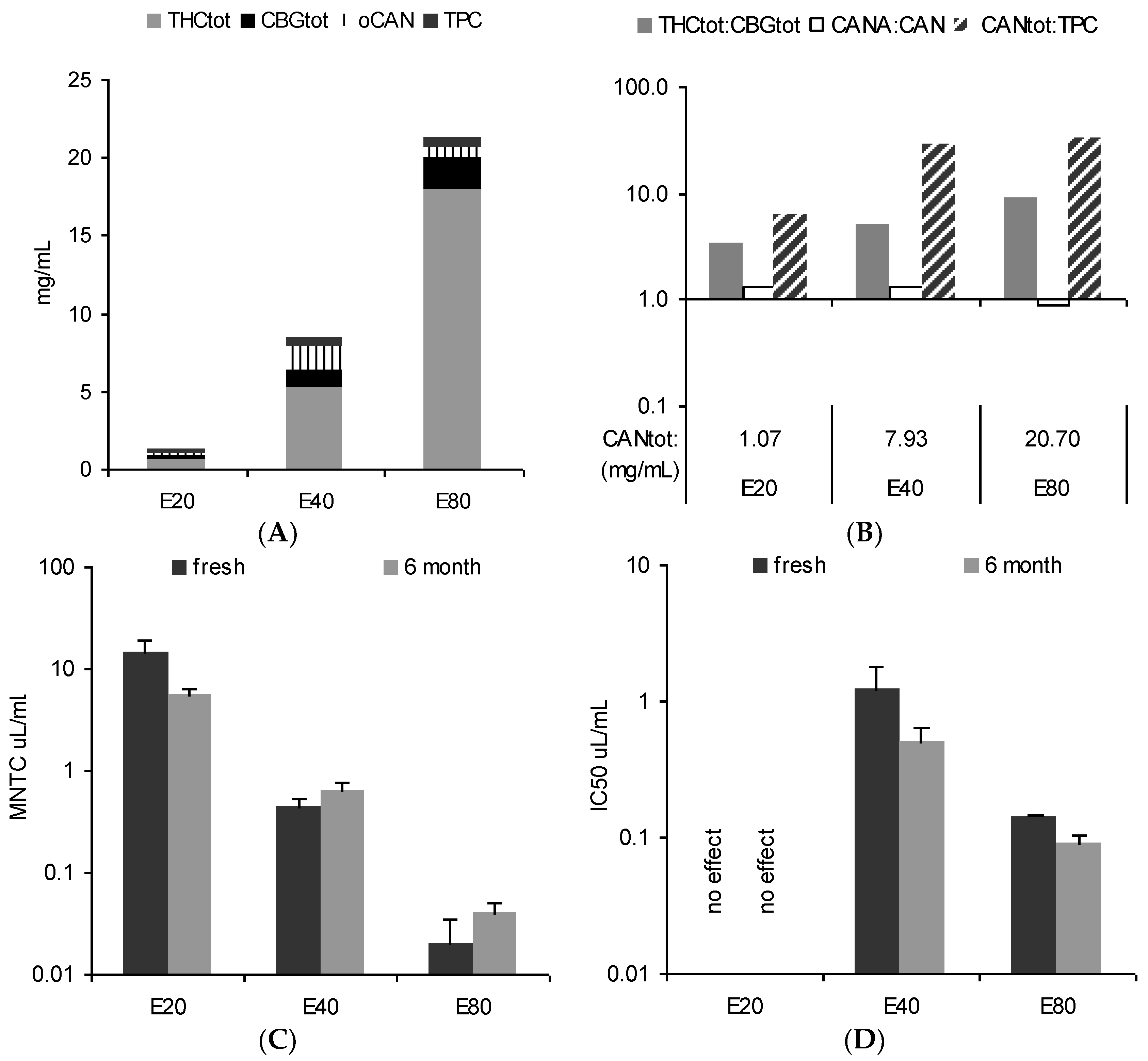
3.1. Main Pattern and in Vitro Activity of Tinctures—Influence of the Solvent
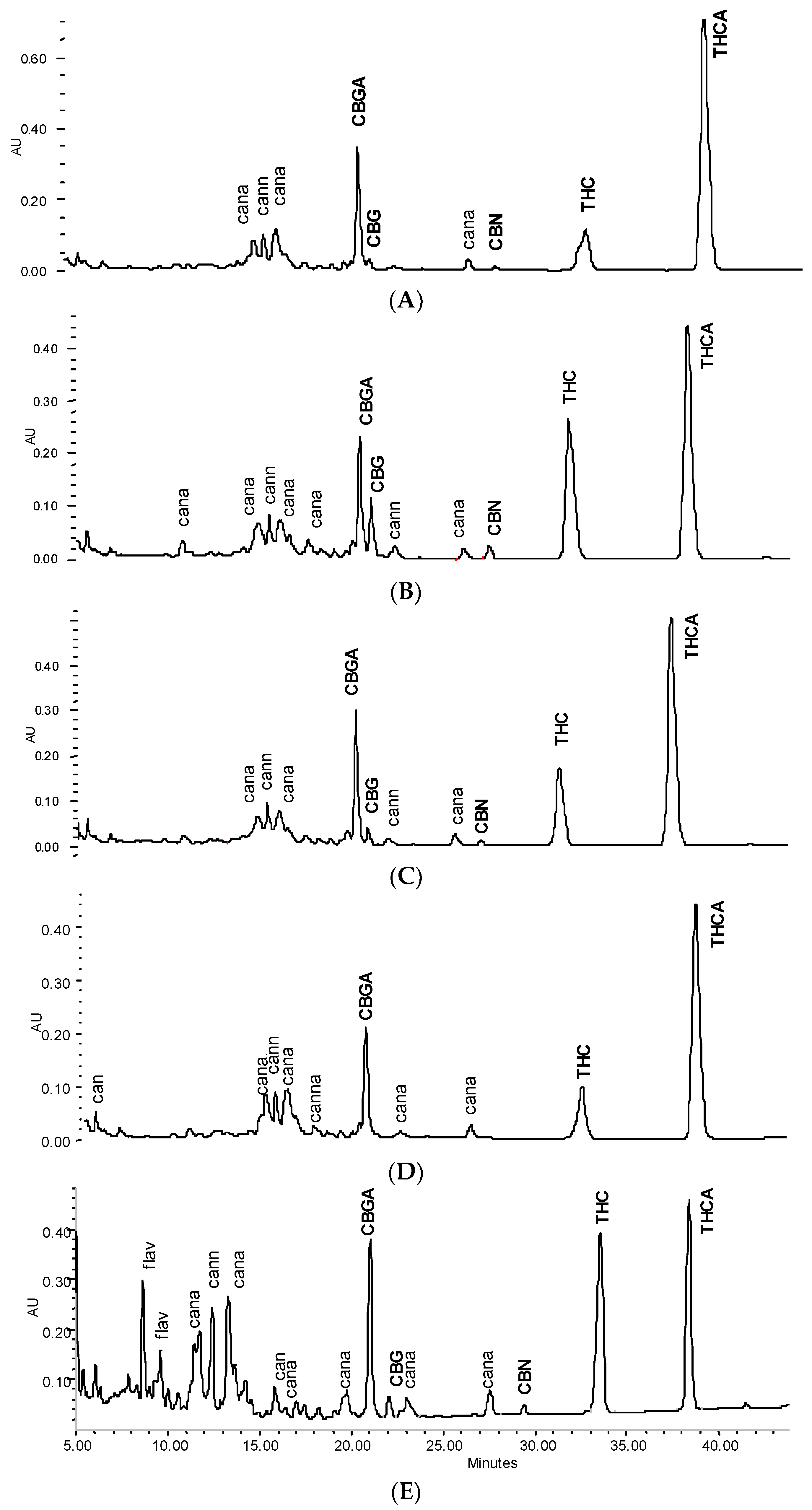
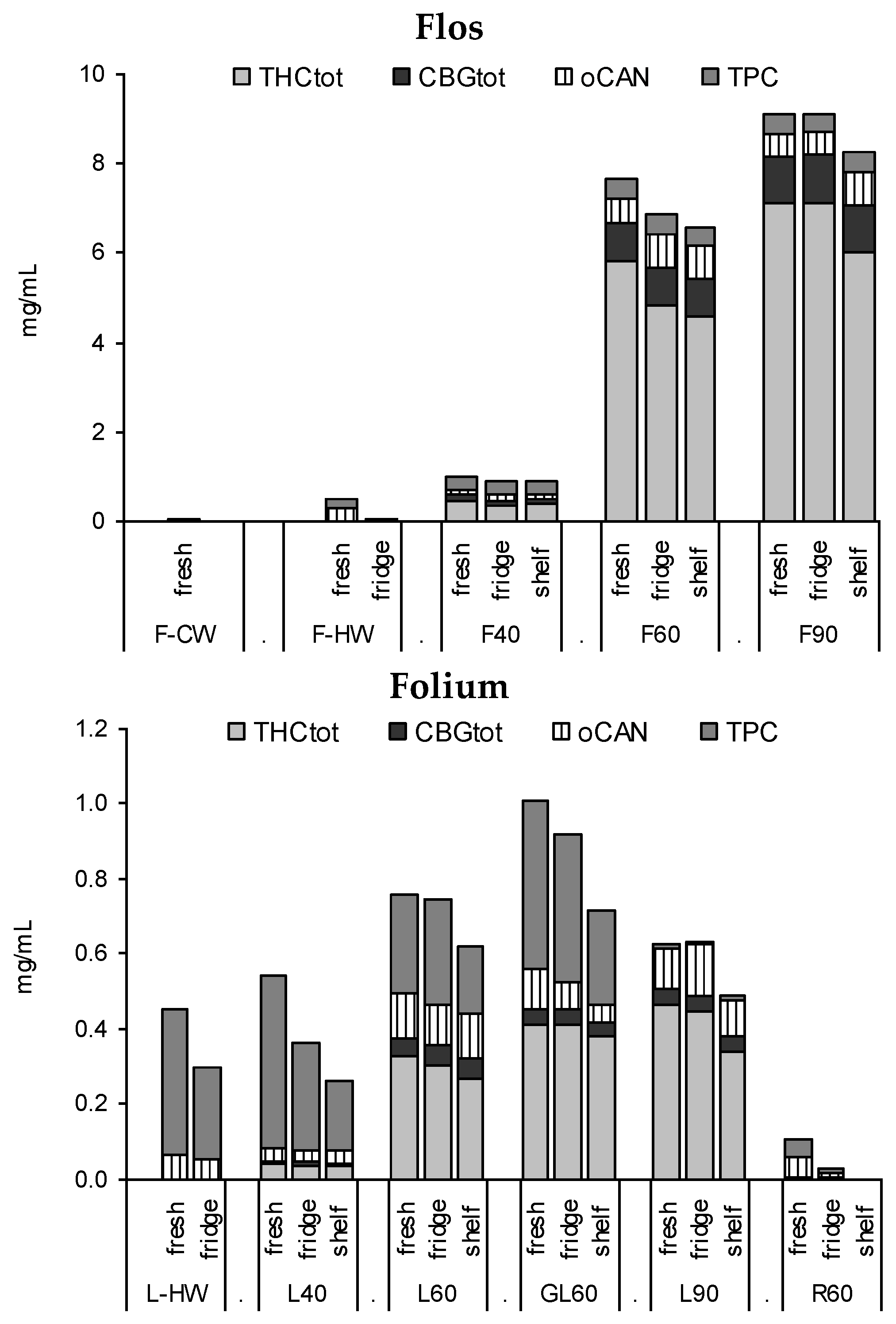
3.2. Profile of Leaf and Flower Tinctures According to Storage Conditions (3 Months)
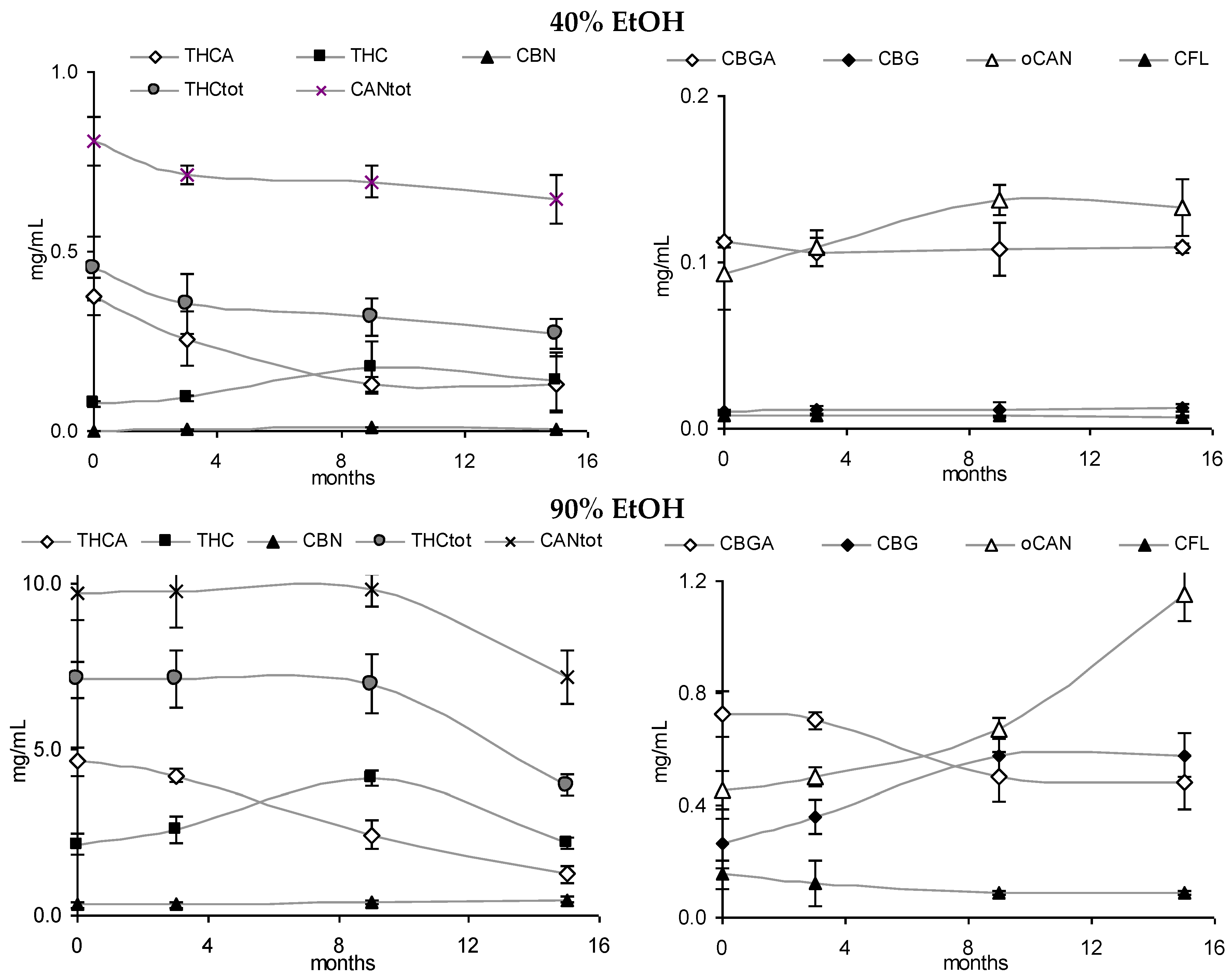
3.3. Stability of Leaf and Flower Tinctures in Cool, Dark Conditions (15 Months)
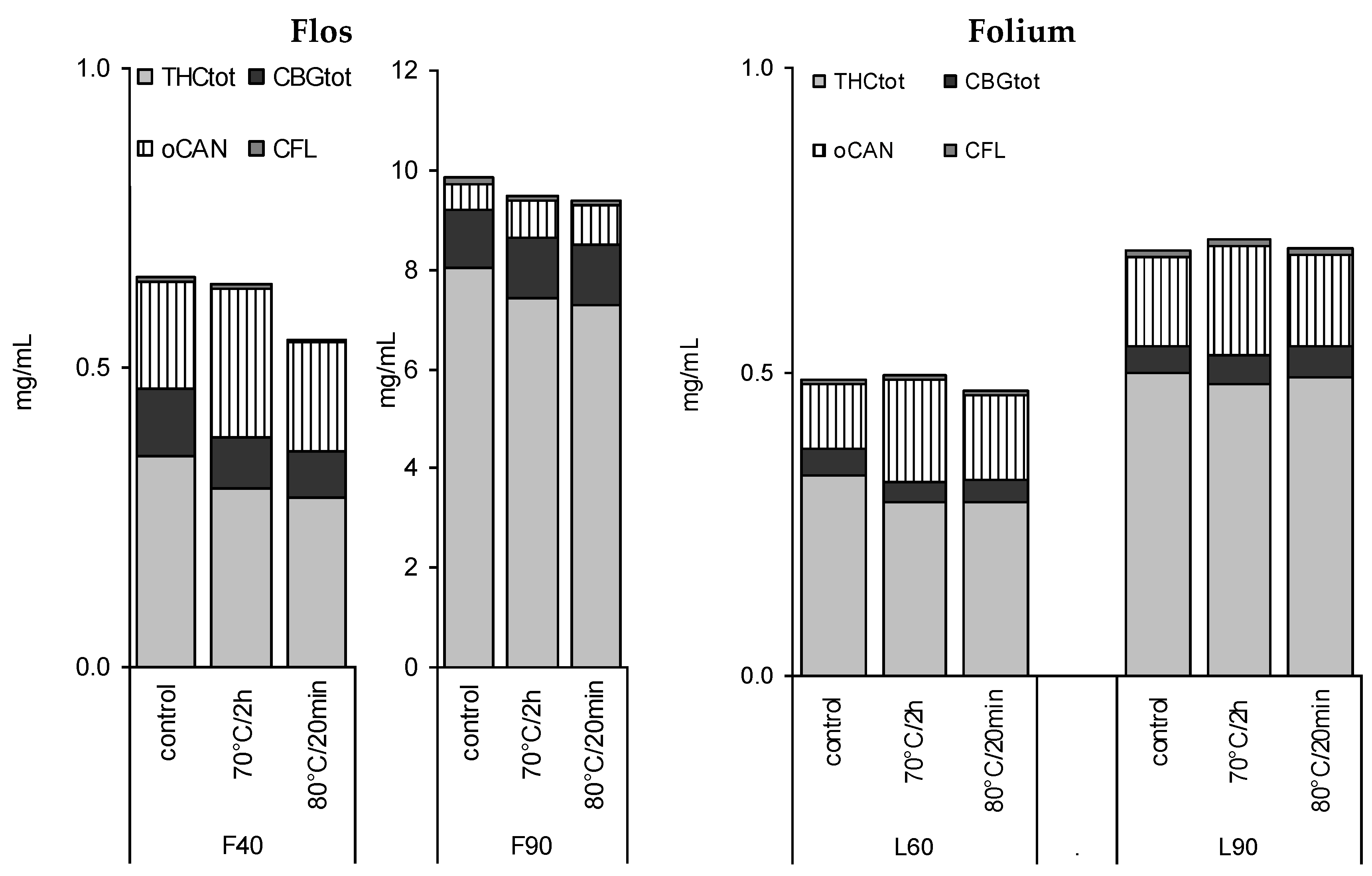
3.4. Stability of Leaf and Flower Tinctures after Short-Term Heating
4. Discussion
4.1. Chemical Pattern of Tinctures
4.2. Stability of Dry Drugs and Liquid Extracts
4.3. Influence of Heating
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| CAN | neutral cannabinoids |
| CANA | cannabinoid acids |
| CANtot | total cannabinoids (CAN + CANA) |
| CBC | cannabichromene (CAS 20675-51-8) |
| CBD | cannabidiol (CAS 13956-29-1) |
| CBDA | cannabidiolic acid (CAS 1244-58-2) |
| CBDtot | cannabidiol + cannabidiolic acid |
| CBG | cannabigerol (CAS 25654-31-3) |
| CBGA | cannabigerolic acid (CAS 25555-57-1) |
| CBGtot | cannabigerol + cannabigerolic acid |
| CBN | cannabinol (CAS 521-35-7) |
| CFL-A | cannflavin A (CAS 97502-36-8) |
| CFL-B | cannflavin B (CAS 76735-58-5) |
| oCAN | other cannabinoids (CANtot—THCtot—CBDtot—CBGtot) |
| THC | Δ9-tetrahydrocannabinol (CAS 1972-08-3) |
| THCA | Δ9-tetrahydrocannabinolic acid A (CAS 23978-85-0) |
| THCtot | Δ9-tetrahydrocannabinol + Δ9-tetrahydrocannabinolic acid A + cannabinol |
| TPC | total phenolic content |
References
- Mikuriya, T.H. Marijuana: Medical Papers 1839–1972; Medi-Comp Press: Oakland, CA, USA, 1973; p. 465. [Google Scholar]
- Russo, E. Hemp for headache: An in-depth historical and scientific review of cannabis in migraine treatment. J. Cannabis Ther. 2001, 1, 21–92. [Google Scholar] [CrossRef]
- Evans, F.J. The Medicinal Chemistry of Cannabis: O’Shaughnessy’s Legacy. J. Pharm. Sci. 1997, 3, 533–537. [Google Scholar]
- Royal Pharmaceutical Society of Great Britain. Report on Cannabis. Response to the House of Lords Science and Technology Committee. 29 May 1998. Available online: http//www.parliament.the-stationery-office.co.uk/pa/ld199798/ldselect/ldsctech/151/15101.htm (accessed on 18 April 2013).
- Leung, L. Cannabis and its derivatives: Review of medical use. J. Am. Board Fam. Med. 2011, 24, 452–462. [Google Scholar] [CrossRef] [PubMed]
- Kalant, H. Clearing the Smoke on Cannabis; Medical Use of Cannabis and Cannabinoids 5; Canadian Centre on Substance Abuse: Ottawa, ON, Canada, 2014; pp. 1–10. Available online: http://www.ccsa.ca/Resource%20Library/CCSA-Medical-Use-of-Cannabis-2012-en.pdf (accessed on 12 September 2015)ISBN 978-1-77178-093-3.
- Grotenthermen, F. Pharmacokinetics and pharmacodynamics of cannabinoids. Clin. Pharmacokinet. 2003, 42, 327–360. [Google Scholar] [CrossRef] [PubMed]
- Guy, G.W.; Flint, M.E. A single centre, placebo-controlled, four period, crossover, tolerability study assessing pharmacokinetic effects, pharmacokinetic characteristics and cognitive profiles of as single dose of three formulations of cannabis based medicine extracts (CBMEs) (GWPD9901), plus a two period tolerability study comparing pharmacodynamic effects and pharmacokinetic characteristics of a single dose of a cannabis based medicine extract given via two administration routes (GWPD9901 EXT). J. Cannabis Ther. 2003, 3, 35–77. [Google Scholar]
- Karschner, E.L.; Darwin, W.D.; Goodwin, R.S.; Wright, S.; Huestis, M.A. Plasma Cannabinoid Pharmacokinetics following Controlled Oral Δ9-Tetrahydrocannabinol and Oromucosal Cannabis Extract Administration. Clin. Chem. 2011, 57, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Hornby, P.; Sharma, M.; Stegman, B. Standardized natural product cannabis in pain management and observations at a Canadian compassion society: A case report. J. Cases 2009, 2, 7487. [Google Scholar] [CrossRef] [PubMed]
- Slijkhuis, C.; Hoving, R.; Blok-Tip, L.; de Kaste, D. Kwaliteitsnormen Medicinale Cannabis Quality Standards for Medicinal Cannabis; RIVM: Bilthoven, The Netherlands, 2004. [Google Scholar]
- Bureau voor Medicinale Cannabis (BMC). Medicinale Cannabis—Informatiebrochure voor Artsen en Apothekers; Instituut voor Verantwoord Medicijngebruik van het CIBG, Ministerie van Volksgezondheid, Welzijn en Sport (VWS): Den Haag, The Netherlands, 2009. [Google Scholar]
- Peschel, W.; Politi, M. 1H-NMR and HPLC/DAD for Cannabis sativa L. chemotype distinction, extract profiling and specification. Talanta 2015, 140, 150–165. [Google Scholar] [CrossRef] [PubMed]
- Politi, M.; Peschel, W.; Wilson, N.; Zloh, M.; Prieto, J.M.; Heinrich, M. Direct NMR analysis of cannabis water extracts and tinctures and semi-quantitative data on [Delta]9-THC and [Delta]9-THC-acid. Phytochemistry 2008, 69, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Hazekamp, A.; Peltenburg, A.; Verpoorte, R.; Giroud, C. Chromatographic and Spectroscopic Data of Cannabinoids from Cannabis sativa L. J. Liq. Chromatogr. Relat. Technol. 2005, 28, 2361–2382. [Google Scholar] [CrossRef]
- Turner, J.C.; Mahlberg, P.G. Simple high-performance liquid chromatographic method for separating acidic and neutral cannabinoids in Cannabis sativa L. J. Chromatogr. A 1982, 53, 295–303. [Google Scholar] [CrossRef]
- Dussy, F.E.; Hamberg, C.; Luginbuhl, M.; Schwerzmann, T.; Briellmann, T.A. Isolation of Δ9-THCA-A from hemp and analytical aspects concerning the determination of Δ9-THC in cannabis products. Forensic Sci. Int. 2005, 149, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Fairbairn, J.W.; Rowan, M.G. Proceedings: Cannabinoid pattern in Cannabis sativa L. seedlings as an indication of chemical race. J. Pharm. Pharmacol. 1975, 27, 90. [Google Scholar]
- Holley, J.H.; Hadley, K.W.; Turner, C.E. Constituents of Cannabis sativa L. XI: Cannabidiol and Cannabichromene in Samples of Known Geographical Origin. J. Pharm. Sci. 1975, 64, 892–894. [Google Scholar] [CrossRef] [PubMed]
- Cascini, F.; Aiello, C.; Di Tanna, G.L. Increasing Delta-9-Tetrahydrocannabinol (Δ-9-THC) Content in Herbal Cannabis Over Time: Systematic Review and Meta-Analysis. Curr. Drug Abuse Rev. 2012, 5, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Czolowski, S. Timeless tinctures. Marijuana Mag. 2003, 10. Available online: http://www.cannabisculture.com/content/2003/10/02/3005 (accessed on 19 February 2016). [Google Scholar]
- De Backer, B.; Debrus, B.; Lebrun, P.; Theunis, L.; Dubois, N.; Decock, L.; Verstraete, A.; Hubert, P.; Charlier, C. Innovative development and validation of an HPLC/DAD method for the qualitative and quantitative determination of major cannabinoids in cannabis plant material. J. Chromatogr. B 2009, 877, 4115–4124. [Google Scholar] [CrossRef] [PubMed]
- Ambach, L.; Penitschka, F.; Broillet, A.; König, S.; Weinmann, W.; Bernhard, W. Simultaneous quantification of delta-9-THC, THC-acid A, CBN and CBD in seized drugs using HPLC-DAD. Forensic Sci. Int. 2014, 243, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R. Marihuana chemistry. Science 1970, 168, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Turk, R.F.; Manno, J.E.; Jain, N.C.; Forney, R.B. The identification, isolation, and preservation of Δ9-tetrahydrocannabinol (Δ9-THC). J. Pharm. Pharmacol. 1971, 23, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Garret, E.R.; Tsau, J. Stability of tetrahydrocannabinols. J. Pharm. Sci. 1974, 63, 1563–1574. [Google Scholar] [CrossRef]
- Turner, C.E.; Hadley, K.W.; Fetterman, P.S.; Doorenbos, N.J.; Quimby, M.W.; Waller, C. Constituents of Cannabis sativa L. IV. Stability of cannabinoids in stored plant material. J. Pharm. Sci. 1973, 62, 1601–1605. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.S.; Turner, C.E. Constituents of Cannabis sativa L. XIII: Stability of dosage form prepared by impregnating synthetic (−)-delta 9-trans-tetrahydrocannabinol on placebo Cannabis plant material. J. Pharm. Sci. 1978, 67, 876–878. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.E.; Henry, J.T. Constituents of Cannabis sativa L. IX: Stability of synthetic and naturally occurring cannabinoids in chloroform. J. Pharm. Sci. 1975, 64, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.M.; Borke, M.L.; Block, L.H.; Cochran, T.G. Decomposition of cannabidiol in chloroform solution. J. Pharm. Sci. 1974, 63, 970–971. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.N.; Vaughan, C.G. The decomposition of acidic and neutral cannabinoids in organic solvents. J. Pharm. Pharmacol. 1977, 29, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Taschwer, M.; Schmid, M.G. Determination of the relative percentage distribution of THCA and Δ9-THC in herbal cannabis seized in Austria—Impact of different storage temperatures on stability. Forensic Sci. Int. 2015, 254, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Zoller, O.; Rhyn, P.; Zimmerli, B. High-performance liquid chromatographic determination of [Delta]9-tetrahydrocannabinol and the corresponding acid in hemp containing foods with special regard to the fluorescence properties of [Delta]9-tetrahydrocannabinol. J. Chromatogr. A 2000, 872, 101–110. [Google Scholar] [CrossRef]
- Harvey, D.J. Stability of cannabinoids in dried samples of cannabis dating from around 1896–1905. J. Ethnopharmacol. 1990, 28, 117–128. [Google Scholar] [CrossRef]
- Fairbain, J.W.; Liebmann, J.A.; Rowan, M.G. The stability of cannabis and its preparations on storage. J. Pharm. Pharmacol. 1976, 28, 1–7. [Google Scholar] [CrossRef]
- Razdan, R.K.; Puttick, A.J.; Zitko, B.A.; Handrick, G.R. Hashish VI1: Conversion of (−)-Δ1(6)-tetrahydrocannabinol to (−)-Δ1(7)-tetrahydrocannabinol. Stability of (−)-Δ1 and (−)-Δ1(6)-tetrahydrocannabinols. Experientia 1972, 28, 121–122. [Google Scholar] [CrossRef]
- Potter, D.J. A review of the cultivation and processing of cannabis (Cannabis sativa L.) for production of prescription medicines in the UK. Drug Test. Anal. 2014, 6, 31–38. [Google Scholar] [CrossRef] [PubMed]
- EMA/HMPC/41500/2010. Questions and Answers on Quality of Herbal Medicinal Products/Traditional Herbal Medicinal Products; Committee on Herbal Medicinal Products (HMPC), European Medicines Agency: London, UK, 2015; Available online: http://www.ema.europa.eu/ema/index.jsp?curl=pages/regulation/general/general_content_000365.jsp&mid=WC0b01ac0580029569 (accessed on 22 March 2015).

| Marker | Calculation | Calculated as | Description/Relevance |
|---|---|---|---|
| THCtot | Σ THC, THCA, CBN | THC, THCA, CBN (CP/FP) | Total THC (‘potency marker’) - dominant in classic plants and preparations - constituent with highest CB1/CB2 receptor activity plus its unstable parent and main degradation product - psychotropic with legal/forensic importance |
| CBDtot | Σ CBD, CBDA | CBD (CP) | Sum of CBD and CBDA - dominant in some chemotypes (intermediate, hemp) - main constituents without CB1/CB2 receptor affinity - non-psychotropic but specific pharmacological effects |
| CBGtot | Σ CBG, CBGA | CBG (CP) | Sum of CBG and CBGA - dominant in some chemotypes - main constituents without CB1/CB2 receptor affinity - non-psychotropic but specific pharmacological effects |
| CAN | Σ neutral cannabinoids | THC, CBD, CBG, CBN (CP/FP) | Neutral (decarboxylated) cannabinoids - dominant upon excessive drying, heating, age |
| CANA | Σ acidic cannabinoids | THC, CBD, CBG, CBN (CP/FP) | Acidic (carboxylated) cannabinoids - dominant in fresh plants and preparations without heating |
| CANtot | Σ neutral and acidic cannabinoids, CAN + CANA | THC, CBD, CBG, CBN (CP/FP) | Total cannabinoid content |
| oCAN | CANtot − (Σ THCtot + CBDtot + CBGtot) | THC (CP) | Other cannabinoids than those usually found as main cannabinoids in common chemotypes, (i.e., THCtot, CBDtot, CBGtot) |
| CFL | CFL-A + CFL-B | CFL-A, CFL-B (CP) | Cannflavins (cannabis specific prenylated flavones) |
| TPC | Σ flavonoids and phenolcarbonic acids | vitexin, chlorogenic acid (FP) | Total phenolic content (without CFL) |
| THCtot/(CBDtot + CBGtot) | Σ THC, THCA, CBN/Σ CBDtot, CBGtot | as for THCtot, CBGtot, CBDtot (CP/FP) | ‘chemotype marker’ - ratio main constituents in common chemotypes - ratio main CB1/CB2 active vs. inactive constituents (plus acidic pro-drug) - ratio main psychotropic/non-psychotropic constituents (plus acidic pro-drug) - legal/forensic importance |
| CANA/CAN | Σ CANA/Σ CAN | as for CANA, CAN (CP/FP) | ‘decarboxylation marker’ - ratio acidic /neutral cannabinoids - indicator for drug and extract quality and age |
| CANtot/TPC | CANtot/TPC | as for CANtot, TPC (FP) | ‘polarity marker’ - indicator for extract/solvent polarity - indicator for leave and flower portions - potential influence of phenolics on cannabinoid activity |
| Marker | Fresh | 3 Months ‘Shelf’ | 3 Months ‘Fridge’ | 3 Months ‘Fridge’ | 3 Months ‘Fridge’ | 9 Months ‘Fridge’ | 15 Months ‘Fridge’ | |
|---|---|---|---|---|---|---|---|---|
| +2 h 70 °C | +20 min 80 °C | |||||||
| F40 | THC (%) | 100.0 | 193.5 | 119.5 | 73.5 | 72.2 | 229.9 | 179.2 |
| CBN (%) | (<LoQ) 1 | (0.01 mg/mL) | (0.005 mg/mL) | (<LoQ) 1 | (<LoQ) 1 | (0.009 mg/mL) | (0.005 mg/mL) | |
| THCtot (%) | 100.0 | 85.1 | 78.3 | 66.0 | 62.6 | 70.5 | 60.1 | |
| CANtot (%) | 100.0 | 90.6 | 84.8 | 78.4 | 69.2 | 85.1 | 77.7 | |
| CANA/CAN | 5.72 | 1.57 | 3.31 | 5.75 | 5.51 | 1.22 | 1.52 | |
| F90 | THC (%) | 100.0 | 175.0 | 121.4 | 118.1 | 126.7 | 194.4 | 102.6 |
| CBN (%) | 100.0 | 118.7 | 102.7 | 101.9 | 105.5 | 118.7 | 146.4 | |
| THCtot (%) | 100.0 | 84.5 | 100.5 | 93.3 | 91.4 | 98.2 | 55.2 | |
| CANtot (%) | 100.0 | 90.1 | 100.5 | 97.6 | 96.8 | 100.8 | 70.8 | |
| CANA/CAN | 1.97 | 0.48 | 1.49 | 1.39 | 1.20 | 0.57 | 0.53 | |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peschel, W. Quality Control of Traditional Cannabis Tinctures: Pattern, Markers, and Stability. Sci. Pharm. 2016, 84, 567-584. https://doi.org/10.3390/scipharm84030567
Peschel W. Quality Control of Traditional Cannabis Tinctures: Pattern, Markers, and Stability. Scientia Pharmaceutica. 2016; 84(3):567-584. https://doi.org/10.3390/scipharm84030567
Chicago/Turabian StylePeschel, Wieland. 2016. "Quality Control of Traditional Cannabis Tinctures: Pattern, Markers, and Stability" Scientia Pharmaceutica 84, no. 3: 567-584. https://doi.org/10.3390/scipharm84030567
APA StylePeschel, W. (2016). Quality Control of Traditional Cannabis Tinctures: Pattern, Markers, and Stability. Scientia Pharmaceutica, 84(3), 567-584. https://doi.org/10.3390/scipharm84030567





