Model to Measure the Readiness of University Testing Laboratories to Fulfill ISO/IEC 17025 Requirements (A Case Study)
Abstract
1. Introduction
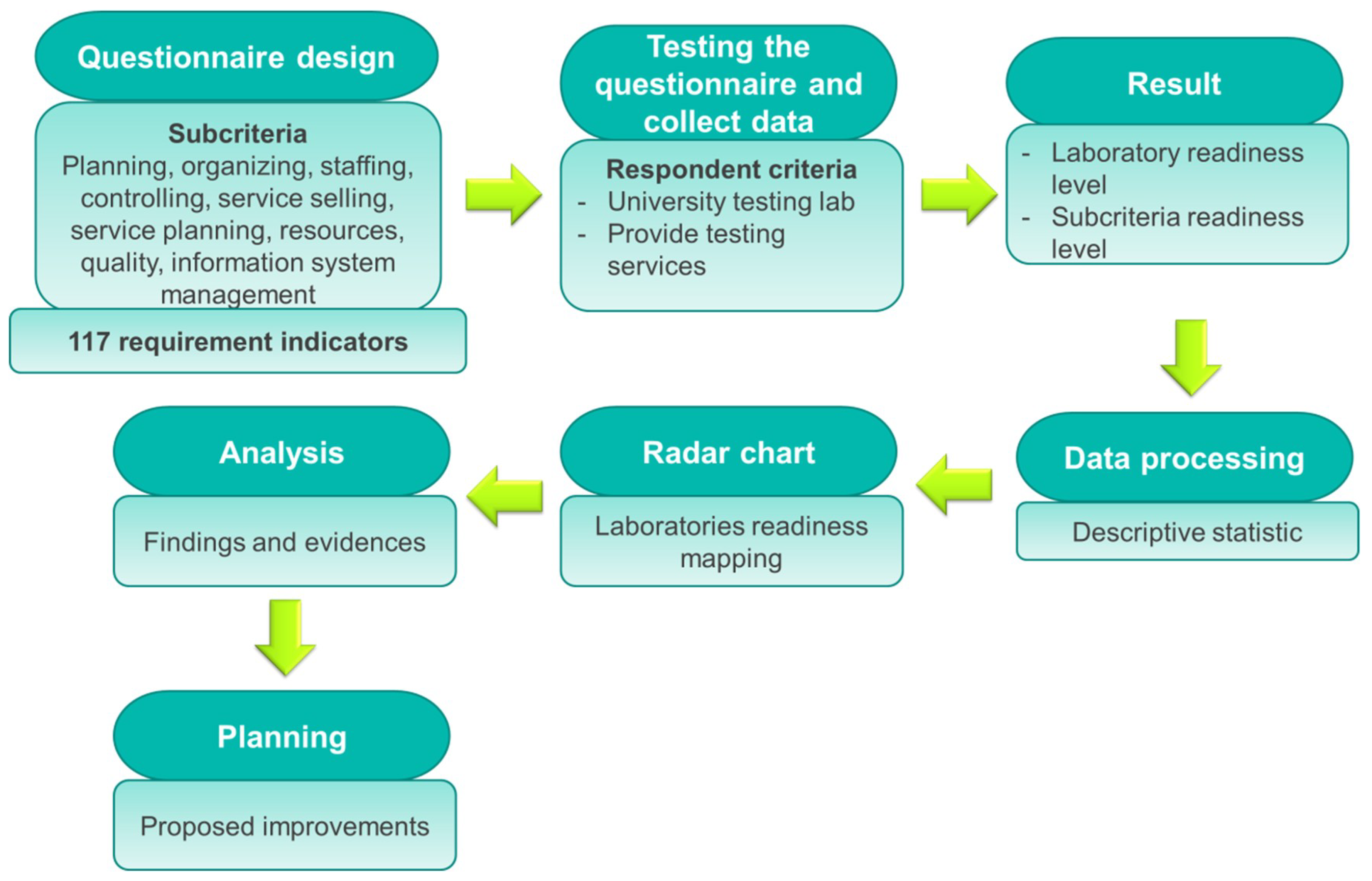
2. Methods
2.1. Designing Laboratory Readiness Measurement Instrument
2.1.1. Generating Sets of Matching Criteria, Subcriteria, and Activities
2.1.2. Generating Sets of Indicators
2.2. Instrument Testing
3. Results
3.1. Laboratory Readiness Instrument Framework
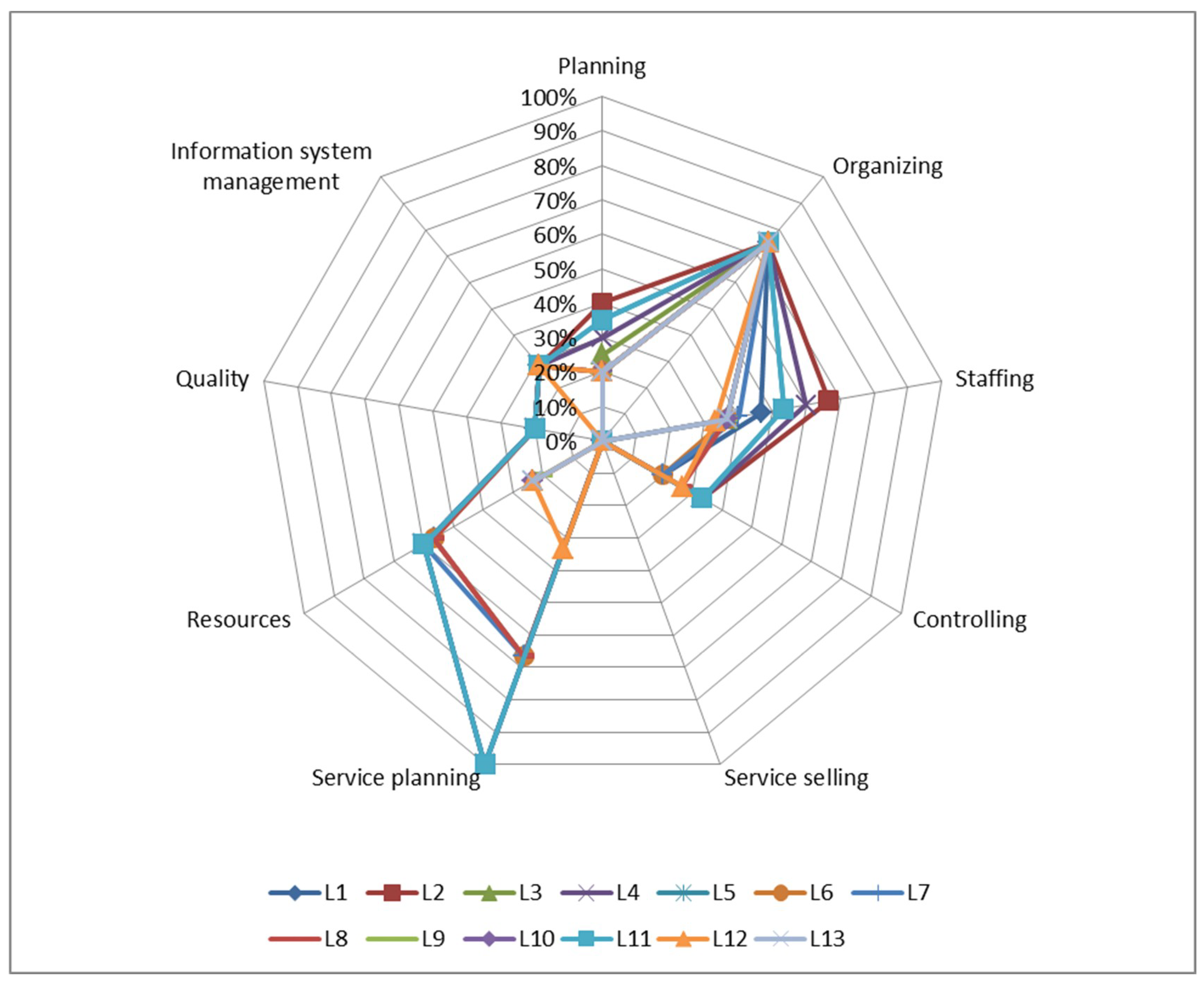
3.2. Laboratory Readiness Level Measurement
4. Discussion
- IEC 62660-2, Secondary lithium-ion cells for the propulsion of electric road vehicles—contains two test parameters (i.e., reliability and abuse testing)
- ISO 12405-1, Road vehicles: Electrically propelled road vehicles—contains test specification for lithium-ion battery packs and systems in high-power applications
- ISO 12405-2, Road vehicles: Electrically propelled road vehicles—contains test specification for lithium-ion battery packs and systems and high-energy application that defines tests and related requirements for battery systems
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grochau, I.H.; Ferreira, C.A.; Ferreira, J.Z.; Caten, C.S. Implementation of a quality management system in university test laboratories: A brief review and new proposals. Accredit. Qual. Assur. 2010, 15, 681–689. [Google Scholar] [CrossRef]
- Giuri, P.; Munari, F.; Scandura, A.; Toschi, L. The strategic orientation of universities in knowledge transfer activities. Technol. Forecast. Soc. Chang. 2010, 15, 681–689. [Google Scholar] [CrossRef]
- Perkmann, M.; Tartari, V.; McKelvey, M.; Autio, E.; Broström, A.; D’Este, P.; Fini, R.; Geuna, A.; Grimaldi, R.; Hughes, A.; et al. Academic engagement and commercialisation: A review of the literature on university–industry relations. Res. Policy 2013, 42, 423–442. [Google Scholar] [CrossRef]
- Zemguliene, J.; Valukonis, M. Structured literature review on business process performance analysis and evaluation. Entrep. Sustain. Issues 2018, 6, 226–252. [Google Scholar] [CrossRef]
- Radwan, A. Science and innovation policies in North African Countries: Exploring challenges and opportunities. Entrep. Sustain. Issues 2018, 6, 268–282. [Google Scholar] [CrossRef]
- Shim, S.O.; Park, K.B.; Choi, S.Y. Sustainable Production Scheduling in Open Innovation Perspective under the Fourth Industrial Revolution. J. Open Innov. Technol. Mark. Complex. 2018, 4, 42. [Google Scholar] [CrossRef]
- Kodama, F.; Shibata, T. Beyond fusion towards IoT by way of open innovation: An investigation based on the Japanese machine tool industry 1975–2015. J. Open Innov. Technol. Mark. Complex. 2017, 3, 23. [Google Scholar] [CrossRef]
- Park, E.S.; Kim, B.R.; Park, S.H.; Kim, D.C. Analysis of the Effects of the Home Energy Management System from an Open Innovation Perspective. J. Open Innov. Technol. Mark. Complex. 2018, 4, 31. [Google Scholar] [CrossRef]
- Lee, M.H.; Yun, J.J.; Pyka, A.; Won, D.K.; Kodama, F.; Schiuma, G.; Park, H.S.; Jeon, J.H.; Park, K.B.; Jung, K.H.; et al. How to Respond to the Fourth Industrial Revolution, or the Second Information Technology Revolution? Dynamic New Combinations between Technology, Market, and Society through Open Innovation. J. Open Innov. Technol. Mark. Complex. 2018, 4, 21. [Google Scholar] [CrossRef]
- Erkut, B. The Emergence of the ERP Software Market between Product Innovation and Market Shaping. J. Open Innov. Technol. Mark. Complex. 2018, 4, 23. [Google Scholar] [CrossRef]
- Leydesdorff, L. Synergy in Knowledge-Based Innovation Systems at National and Regional Levels: The Triple-Helix Model and the Fourth Industrial Revolution. J. Open Innov. Technol. Mark. Complex. 2018, 4, 16. [Google Scholar] [CrossRef]
- Hosseini, A.S.; Soltani, S.; Mehdizadeh, M. Competitive Advantage and Its Impact on New Product Development Strategy (Case Study: Toos Nirro Technical Firm). J. Open Innov. Technol. Mark. Complex. 2018, 4, 17. [Google Scholar] [CrossRef]
- Schiuma, G.; Carlucci, D. Managing Strategic Partnerships with Universities in Innovation Ecosystems: A Research Agenda. J. Open Innov. Technol. Mark. Complex. 2018, 4, 25. [Google Scholar] [CrossRef]
- Tani, M.; Papaluca, O.; Sasso, P. The System Thinking Perspective in the Open-Innovation Research: A Systematic Review. J. Open Innov. Technol. Mark. Complex. 2018, 4, 38. [Google Scholar] [CrossRef]
- Kodama, F. Learning Mode and Strategic Concept for the 4th Industrial Revolution. J. Open Innov. Technol. Mark. Complex. 2018, 4, 32. [Google Scholar] [CrossRef]
- Lee, J.H.; Sung, T.E.; Kim, E.D.; Shin, K.S. Evaluating Determinant Priority of License Fee in Biotech Industry. J. Open Innov. Technol. Mark. Complex. 2018, 4, 30. [Google Scholar] [CrossRef]
- Lara, A.P.; Costa, E.M.D.; Furlani, T.Z.; Yigitcanla, T. Smartness that matters: Towards a comprehensive and human-centred characterisation of smart cities. J. Open Innov. Technol. Mark. Complex. 2016, 2, 8. [Google Scholar] [CrossRef]
- Yusr, M.M. Innovation capability and its role in enhancing the relationship between TQM practices and innovation performance. J. Open Innov. Technol. Mark. Complex. 2016, 2, 6. [Google Scholar] [CrossRef]
- Ryu, J.; Byeon, S.C. Technology level evaluation methodology based on the technology growth curve. Technol. Forecast. Soc. Chang. 2011, 78, 1049–1059. [Google Scholar] [CrossRef]
- Brown, J.R.; Martinsson, G.; Petersen, B.C. Stock markets, credit markets, and technology-led growth. J. Financ. Intermed. 2017, 32, 45–59. [Google Scholar] [CrossRef]
- Manzhirov, A.V.; Gupta, N.K. Fundamentals of Continuous Growth Processes in Technology and Nature. Procedia IUTAM 2017, 23, 1–12. [Google Scholar] [CrossRef]
- Liu, W.; Zhan, J.; Wang, C.; Li, S.; Zhang, F. Environmentally sensitive productivity growth of industrial sectors in the Pearl River Delta Resources. Conserv. Recycl. 2018, 139, 50–63. [Google Scholar] [CrossRef]
- Rahman, M.M.; Kashem, M.A. Carbon emissions, energy consumption and industrial growth in Bangladesh: Empirical evidence from ARDL cointegration and Granger causality analysis. Energy Policy 2017, 110, 600–608. [Google Scholar] [CrossRef]
- Lin, J.Y.; Sun, X.; Wu, H.X. Banking structure and industrial growth: Evidence from China. J. Bank. Financ. 2015, 58, 131–143. [Google Scholar] [CrossRef]
- Sutopo, W.; Nizam, M.; Purwanto, A.; Atikah, N.; Putri, A.S. A Cost Estimation Application for Determining Feasibility Assessment of Li-Ion Battery in Mini Plant Scale. Int. J. Electr. Eng. Inform. 2016, 8, 189–199. [Google Scholar] [CrossRef]
- Sutopo, W.; Kadir, E.A. An Indonesian Standard of Lithium-ion Battery Cell Ferro Phosphate for Electric Vehicle Application. Telkomnika 2017, 15, 584–589. [Google Scholar] [CrossRef]
- Sutopo, W.; Kadir, E.A. Designing Framework for Standardization Case Study: Lithium-Ion Battery Module in Electric Vehicle Application. Int. J. Electr. Comput. Eng. 2018, 8, 220–226. [Google Scholar] [CrossRef]
- Sutopo, W.; Fauzan, M.I.R.; Yuniaristanto. A Comparative Analysis of Natural Fiber Reinforced for an Interior Electric Car. J. Mek. 2016, 39, 47–55. [Google Scholar]
- Sutopo, W.; Erliza, A.; Ardiansyah, R. Yuniaristanto, Nizam, M. Parametric Cost Estimation for Controlling the Development of Electric Vehicle Prototype. J. Mek. 2016, 39, 56–68. [Google Scholar]
- Kadir, E.A.; Shamsuddin, S.M.; Supriyanto, E.; Sutopo, W.; Rosa, S.L. Food Traceability in Supply Chain Based on EPCIS Standard and RFID Technology. Indones. J. Electr. Eng. Comput. Sci. 2015, 13, 187–194. [Google Scholar] [CrossRef]
- Nugraheni, D.D.; Sutopo, W.; Hisjam, M.; Priyandari, Y. Preliminary study of Benefits and Barriers-Costs RFID Technology Implementation on Traceability System of Banana in Indonesia. J. Mek. 2016, 39, 1–10. [Google Scholar]
- Shakeel, S.R.; Takala, J.; Zhu, L.D. Commercialization of renewable energy technologies: A ladder building approach. Renew. Sustain. Energy Rev. 2017, 78, 855–867. [Google Scholar] [CrossRef]
- Rahim, N.A.; Mohamed, Z.B.; Amrin, A. Commercialization of Emerging Technology: The Role of Academic Entrepreneur. Procedia Soc. Behav. Sci. 2015, 169, 53–60. [Google Scholar] [CrossRef][Green Version]
- Marx, M.; Hsu, D.H. Strategic switchbacks: Dynamic commercialization strategies for technology entrepreneurs. Res. Policy 2015, 44, 1815–1826. [Google Scholar] [CrossRef]
- Huang, M.H.; Chen, D.Z. How can academic innovation performance in university–industry collaboration be improved? Technol. Forecast. Soc. Chang. 2017, 123, 210–215. [Google Scholar] [CrossRef]
- Berbegal-Mirabent, J.; García, J.L.S.; Ribeiro-Soriano, D.E. University–industry partnerships for the provision of R&D services. J. Bus. Res. 2015, 68, 1407–1413. [Google Scholar] [CrossRef]
- Dalmarco, G.; Hulsink, W.; Blois, G.V. Creating entrepreneurial universities in an emerging economy: Evidence from Brazil. Technol. Forecast. Soc. Chang. 2018, 135, 99–111. [Google Scholar] [CrossRef]
- Testing Labs—RMIT University. Available online: https://www.rmit.edu.au/industry/facilities-equipment-and-services/testing-labs (accessed on 20 September 2018).
- Environmental Testing Lab—Loyola University Chicago. Available online: www.luc.edu/sustainability/initiatives/environmentaltestinglab/ (accessed on 20 September 2018).
- Testing Labs | Ball State University. Available online: https://www.bsu.edu/about/administrativeoffices/labs/testinglabs (accessed on 20 September 2018).
- Testing Laboratory—University of Chemistry and Technology, Prague. Available online: https://www.vscht.cz/testing-laboratory (accessed on 20 September 2018).
- Structural Testing Laboratory | University of Moratuwa. Available online: https://www.mrt.ac.lk/web/civil/resources/lab-facilities/structural-testing (accessed on 20 September 2018).
- Bates, I.; Maganga, O. Light Tracking System for Teaching Laboratory Use. IFAC-PapersOnLine 2017, 50, 15698–15703. [Google Scholar] [CrossRef]
- Gallion, L.A.; Samide, M.J.; Wilson, A.M. Demonstrating the importance of cleanliness and safety in an undergraduate teaching laboratory. J. Chem. Health Saf. 2015, 22, 28–31. [Google Scholar] [CrossRef]
- El-Gilany, A.H.; El-shaer, S.; Khashaba, E.; El-dakroory, S.A.; Omar, N. Knowledge, attitude, and practice (KAP) of ‘teaching laboratory’ technicians towards laboratory safety and waste management: A pilot interventional study. J. Hosp. Infect. 2017, 96, 192–194. [Google Scholar] [CrossRef]
- Wu, T. Safety leadership in the teaching laboratories of electrical and electronic engineering departments at Taiwanese Universities. J. Saf. Res. 2008, 39, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Sandi-Urena, S.; Gatlin, T.A. Experimental Chemistry Teaching: Understanding Teaching Assistants’ Experience in the Academic Laboratory. Educ. Quím. 2012, 23, 141–148. [Google Scholar] [CrossRef]
- Hicks, S.F.; Kovash, M.A. Research at the University of Kentucky Accelerator Laboratory. Phys. Procedia 2017, 90, 440–447. [Google Scholar] [CrossRef]
- Noga, P.; Dobrovodsky´, J.; Vanˇa, D.; Benˇo, M.; Závacká, A.; Muška, M.; Halgaš, R.; Minárik, S.; Riedlmajer, R. A new ion-beam laboratory for materials research at the Slovak University of Technology. Nucl. Instrum. Methods Phys. Res. Sect. B 2017, 409, 264–267. [Google Scholar] [CrossRef]
- Zhao, S.; Xiang, C.; Su, L.; Jiang, Z.; Liao, C. Study on the Eye Movement Characteristics of Fire Hazard Identification in University Laboratories. Procedia Eng. 2018, 211, 433–440. [Google Scholar] [CrossRef]
- Chiware, E.R.T.; Becker, D.A. Research trends and collaborations by applied science researchers in South African universities of technology: 2007–2017. J. Acad. Librariansh. 2018, 44, 468–476. [Google Scholar] [CrossRef]
- Meisel, Z.; Brune, C.R.; Grimes, S.M.; Ingram, D.C.; Massey, T.N.; Voinov, A.V. The Edwards Accelerator Laboratory at Ohio University. Phys. Procedia 2017, 90, 448–454. [Google Scholar] [CrossRef]
- Sterzinger, C.H. Technology Marketing Plan for Laboratory Services to Sustain a Translational Research Centre. Ph.D. Thesis, Simon Fraser University, Burnaby, BC, Canada, 2006. [Google Scholar]
- Sadikoglu, E.; Temur, T. The Relationship Between ISO 17025 Quality Management System Accreditation and Laboratory Performance. Qual. Manag. Pract. 2012, 221–230. [Google Scholar]
- ISO/IEC 17025:2017. General Requirements for the Competence of Testing and Calibration Laboratories; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- Beckett, J.; Slay, J. Scientific underpinnings and background to standards and accreditation in digital forensics. Digit. Investig. 2011, 8, 114–121. [Google Scholar] [CrossRef]
- Honsa, J.D.; McIntyre, D.A. ISO 17025: Practical Benefits of Implementing a Quality System. J. AOAC Int. 2003, 86, 1038–1044. [Google Scholar]
- Vlachos, N.A.; Michail, C.; Sotiropoulou, D. Is ISO/IEC 17025 Accreditation a Benefit or Hindrance to Testing Laboratories? The Greek Experience. J. Food Compos. Anal. 2002, 15, 749–757. [Google Scholar] [CrossRef]
- New Initiative for Indonesian Higher Education toward World-Class Status. Available online: https://qswownews.com/new-initiative-indonesian-higher-education-toward-world-class-status/ (accessed on 21 September 2018).
- Levin, H.M.; Jeong, D.; Ou, D. What is A World Class University? Presented at the Conference of the Comparative and International Education Society, Honolulu, HI, USA, 14–18 March 2006. [Google Scholar]
- Altbach, P.G. The Costs and Benefits of World-Class Universities; International Higher Education: Chestnut Hill, MA, USA, 2003. [Google Scholar]
- Niland, J. “The Challenge of Building World Class Universities in the Asian Region,” On Line Opinion (Australia’s e-Journal of Social and Political Debate). 2000. Available online: http://www.onlineopinion.com.au/view.asp?article=997 (accessed on 21 September 2018).
- UPT Laboratorium Terpadu. Available online: https://uns.ac.id/id/laboratorium-mipa-pusat (accessed on 25 August 2018).
- Baker, A.; Gunnion, A.J.; Wang, J.; Chang, P. Advances in the proof test for certification of bonded repairs—Increasing the Technology Readiness Level. Int. J. Adhes. Adhes. 2016, 64, 128–141. [Google Scholar] [CrossRef]
- Llorach-Massana, P.; Farreny, R.; Oliver-Solà, J. Are Cradle to Cradle certified products environmentally preferable? Analysis from an LCA approach. J. Clean. Prod. 2015, 93, 243–250. [Google Scholar] [CrossRef]
- Daft, R.L. Organization Theory and Design, 10th ed.; Cengage learning EMEA: Andover, UK, 2010. [Google Scholar]
- Lopes, I.; Santos, L.; Pereira, M.F.; Vaz, P.; Alves, J.G. Implementation of the quality management system at the Laboratory of Radiological Protection and Safety (LPSR) in Portugal. Accredit. Qual. Assur. 2014, 19, 355–360. [Google Scholar] [CrossRef]
- Sutopo, W.; Astuti, R.W.; Yuniaristanto, Purwanto, A.; Nizam, M. Model to Measure University’s Readiness for Establishing Spin-Offs: Comparison Study. In IAENG Transactions on Engineering Sciences; World Scientific: Singapore, 2015; pp. 173–186. [Google Scholar]
- Lokuge, S.; Sedera, D.; Grover, V.; Xu, D. Organizational readiness for digital innovation: Development and empirical calibration of a construct. Inf. Manag. 2018. [Google Scholar] [CrossRef]
- Kobos, P.H.; Malczynski, L.A.; Walker, L.T.N.; Borns, D.J.; Klise, G.T. Timing is everything: A technology transition framework for regulatory and market readiness levels. Technol. Forecast. Soc. Chang. 2018, 137, 211–225. [Google Scholar] [CrossRef]
- Rodima, A.; Vilbaste, M.; Saks, O.; Jakobson, E.; Pihl, V.; Soovali, L.; Jalukse, L.; Traks, J.; Virro, K.; Annuk, H.; et al. ISO 17025 quality system in a university environment, Accredit. Qual. Assur. 2005, 10, 369–372. [Google Scholar] [CrossRef]
- Hullihen, K.; Fitzsimmons, V.; Fisch, M.R. Establishing an ISO 17025 compliant laboratory at a university international. J. Mod. Eng. 2009, 10, 55–64. [Google Scholar]
- Zapata-Garcıa, D.; Llaurado, M.; Rauret, G. Experience of implementing ISO 17025 for the accreditation of a university testing laboratory. Accredit. Qual. Assur. 2007, 12, 317–322. [Google Scholar] [CrossRef]
- Pratiwi, A.; Sutopo, W.; Zakaria, R.; Rasli, A. Formulating Strategy Through QSPM Based on SWOT Framework: A Case Study: Spin-Off Company in Malaysia. Adv. Sci. Lett. 2017, 23, 8646–8651. [Google Scholar] [CrossRef]
- Aslan, I.; Çınar, O.; Kumpikaitė, V. Creating strategies from TOWS matrix for strategic sustainable development of Kipaş Group. J. Bus. Econ. Manag. 2012, 13, 95–110. [Google Scholar] [CrossRef][Green Version]
- Caruana, C.J.; Wasilewska-Radwanska, M.; Aurengo, A.; Dendy, P.P.; Karenauskaite, V.; Malisan, M.R.; Mattson, S.; Meijer, J.H.; Mihov, D.; Mornstein, V.; et al. A strategic development model for the role of the biomedical physicist in the education of healthcare professionals in Europe. Phys. Med. 2012, 28, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Kash, B.A.; Deshmukh, A.A. Developing a Strategic Marketing Plan for Physical and Occupational Therapy Services: A Collaborative Project Between a Critical Access Hospital and a Graduate Program in Health Care Management. Health Mark. Q. 2013, 30, 263–280. [Google Scholar] [CrossRef] [PubMed]
- David, F.R. Strategic Management: Concepts and Cases, 13th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2011; ISBN 978-0-13-612098-8. [Google Scholar]
- Grochau, I.H.; Caten, C.S. A process approach to ISO/IEC 17025 in the implementation of a quality management system in testing laboratories. Accredit. Qual. Assur. 2012, 17, 519–527. [Google Scholar] [CrossRef]
- Aqidawati, E.F.; Sutopo, W.; Zakaria, R. Framework for Formulating Internal Environment Strengthening Strategy for Developing Testing Laboratories (Case Study: Universitas Sebelas Maret). IOP Conference Series. (in press).
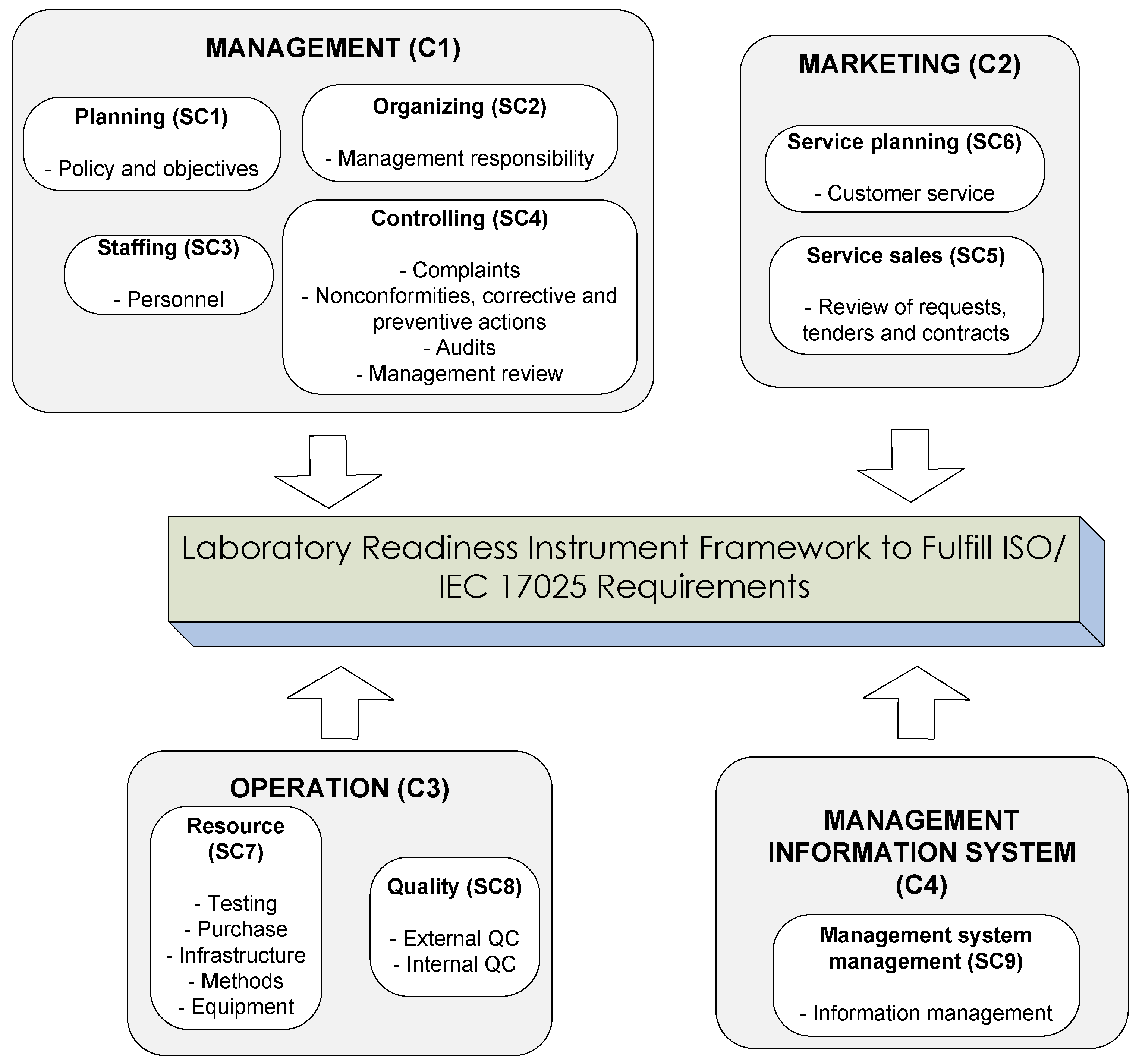
| Criteria | Subcriteria | Activities | Process Element | Standard Item in ISO 17025 |
|---|---|---|---|---|
| Management (C1) | SC1-Planning | Formulate policies and objectives of the quality management system | Policies and procedures planning | 4.2 |
| SC2-Organizing | Meet organizational requirements | Management responsibility | 4.1 | |
| SC3-Staffing |
| Personnel management | 4.1, 5.1, 5.2 | |
| SC4-Controlling |
| Complaint | 4.8 | |
| Nonconformity, corrective, and preventive actions | 4.9, 4.10, 4.11, 4.12 | ||
| Audits | 4.10, 4.14 | ||
| Management responsibility | 4.10, 4.15 | ||
| Marketing (C2) | SC5-Service sales |
| Review requests, tenders, and contracts | 4.4 |
| Marketing (C2) | SC6-Service planning |
| Customer service | 4.7 |
| Operation (C3) | SC7-Resources |
| Testing | 5.8, 4.5, 5.10 |
| Purchasing | 4.6 | ||
| Infrastructure | 5.1, 5.3 | ||
| Method | 5.1, 5.4 | ||
| Equipment | 5.1, 5.5 | ||
| SC8-Quality |
| External quality control | 5.1, 5.6, 5.9 | |
| Operation (C3) | SC8-Quality |
| Internal quality control | 5.5, 5.6, 5.9 |
| Management information system (C4) | SC9-Information system management |
| Information management | 4.3, 4.13, 5.4, 5.10 |
| Subcriteria | Indicator |
|---|---|
| SC1-Planning | 1 Determination of management systems |
| 2 Application of management systems | |
| 3 Maintenance of management systems | |
| 4 Management policy documentation | |
| 5 Management system documentation | |
| 6 Program documentation | |
| 7 Documentation procedure | |
| 8 Documentation of work instructions | |
| 9 Communication of documentation to all personnel | |
| 10 Understanding of documentation by all personnel | |
| 11 Availability of documentation for all personnel | |
| 12 Application of documentation by all personnel | |
| 13 Quality manual | |
| 14 Quality objectives | |
| 15 Issuance of quality policy | |
| SC1-Planning | 16 Laboratory management commitment |
| 17 Management statement for laboratory service standards | |
| 18 The objectives of management systems related to quality | |
| 19 Requirements for all personnel to understand and implement policies and procedures | |
| 20 Commitment to improve the effectiveness of management systems on an ongoing basis | |
| SC2-Organizing | 1 Determination of organizational structure and laboratory management |
| 2 Determination of management position within the parent organization | |
| 3 Determination of the relationship between quality management, technical activities, and supporting services | |
| 4 Determination of responsibility, authority, and relations between all personnel | |
| SC3-Staffing | 1 The presence of managerial and technical personnel |
| 2 Division of task management and technical personnel | |
| 3 Adequate supervision of testing staff | |
| 4 Technical management | |
| 5 Quality manager | |
| 6 Appointment of deputies for core management personnel | |
| 7 Educational qualifications | |
| 8 Training qualifications | |
| 9 Experience qualifications | |
| 10 Skill qualifications | |
| 11 Appropriate supervisors to staff undergoing training | |
| 12 Formulation of educational goals | |
| 13 Formulation of training objectives | |
| 14 Formulation of skill targets | |
| 15 Personnel training policies and procedures | |
| 16 Evaluate the effectiveness of training activities | |
| 17 The existence of contract/nonpermanent personnel | |
| 18 Maintenance of applicable job descriptions | |
| 19 Determination of authority to take samples | |
| 20 Determination of authority to conduct testing | |
| 21 Determination of authority to issue test reports | |
| 22 Determination of authority to give opinions and interpretation of test results | |
| 23 Determination of authority to operate certain equipment | |
| 24 Documentation of personnel authority | |
| 25 Documentation of personnel competence | |
| SC3-Staffing | 26 Documentation of personnel education |
| 27 Professional qualification documentation | |
| 28 Personnel training documentation | |
| 29 Documentation of personnel skills | |
| 30 Documentation of personnel experience | |
| SC4-Controlling | 1 Determination of policies and procedures for resolving complaints received from customers or other parties |
| 2 Documentation regarding complaints received | |
| 3 Documentation related to investigations of complaints received | |
| 4 Documentation regarding corrective actions taken | |
| 5 Determination of policies and control procedures for inappropriate testing work | |
| 6 Increasing the effectiveness of the management system on an ongoing basis | |
| 7 Determination of policies and procedures for carrying out corrective actions | |
| 8 Determination of preventive action procedures for nonconformity and policy deviation | |
| 9 Establishing schedules and procedures for internal audits | |
| 10 Periodic internal audits | |
| 11 Testing activities are included in the elements that must be audited | |
| 12 Planning and organizing audits by quality managers | |
| 13 The audit is carried out by trained personnel and independent of the audited activities | |
| 14 Schedule and procedures for laboratory management review | |
| 15 Periodic implementation of laboratory management reviews | |
| SC5-Service sales | 1 Determination and maintenance of customer request review procedures |
| 2 Determination of tender review procedures | |
| 3 Determination of procedures for reviewing test contracts | |
| SC6- Service planning | 1 Determination of test sampling procedures |
| 2 Determination of procedures for recording test sample data | |
| 3 Seek feedback from customers through customer surveys | |
| SC7-Resources | 1 Subcontract testing work |
| 2 Reporting on test results | |
| 3 Determination of transportation procedures for goods tested | |
| 4 Determination of procedures for receiving goods tested | |
| 5 Determination of procedures for handling goods tested for deterioration, loss, or damage | |
| 6 Determination of procedures for protecting goods tested | |
| SC7-Resources | 7 Determination of procedures for storing goods tested |
| 8 Determination of procedures for identifying items tested | |
| 9 Recording the condition of items tested when received by laboratory staff | |
| 10 Consultation with customers if the goods received are not in accordance with existing specifications | |
| 11 Determination of policies and procedures for selecting and buying services and supplies | |
| 12 Procedure for purchasing reagents and disposable materials | |
| 13 Procedure for receiving reagents and disposable materials | |
| 14 Procedure for storing reagents and disposable materials | |
| 15 Conduct evaluation towards supplier of disposable materials, supplies, and services | |
| 16 Energy source is adequate | |
| 17 Lighting is sufficient | |
| 18 Adequate environment | |
| 19 Procedure for guaranteeing accommodation conditions and laboratory environment | |
| 20 Procedure for access to laboratory space | |
| 21 Procedure for using laboratory space | |
| 22 Use of appropriate methods and procedures for all tests performed | |
| 23 Equipment operating instructions | |
| 24 Instructions for handling and preparing items tested | |
| 25 Procedure for estimating measurement uncertainty | |
| 26 Equipment handling procedures | |
| 27 Equipment removal procedure | |
| 28 Equipment storage procedure | |
| 29 Procedure for using tools | |
| 30 Equipment maintenance procedures | |
| SC8-Quality | 1 Calibration of equipment |
| 2 Equipment calibration programs and procedures | |
| 3 Intermediate check | |
| 4 Quality control procedures | |
| 5 Analysis of quality control data | |
| SC9-Information system management | 1 Document control procedures |
| 2 Quality and technical documentation control procedures | |
| 3 Internal audit report | |
| 4 Management review report | |
| 5 Reports on corrective actions | |
| 6 Preventive action report | |
| 7 Procedure for the protection and backup of records stored electronically | |
| 9 total subcriteria | 117 total indicators |
| Category | Laboratory |
|---|---|
| Engineering | Material Lab (L2) |
| Nano Bioenergy Lab (L3) | |
| Thermodynamic Lab (L4) | |
| Structural Lab (L5) | |
| Mechanics Lab (L11) | |
| Ergonomic Lab (L12) | |
| Basic Chemistry Lab (L13) | |
| Agriculture | Soil Fertility Lab (L6) |
| Soil Conservation Lab (L7) | |
| Biotechnology Lab (L8) | |
| Plant Breeding Lab (L9) | |
| Plant Physiology Lab (L10) | |
| Food technology | Food Biochemistry Lab (L1) |
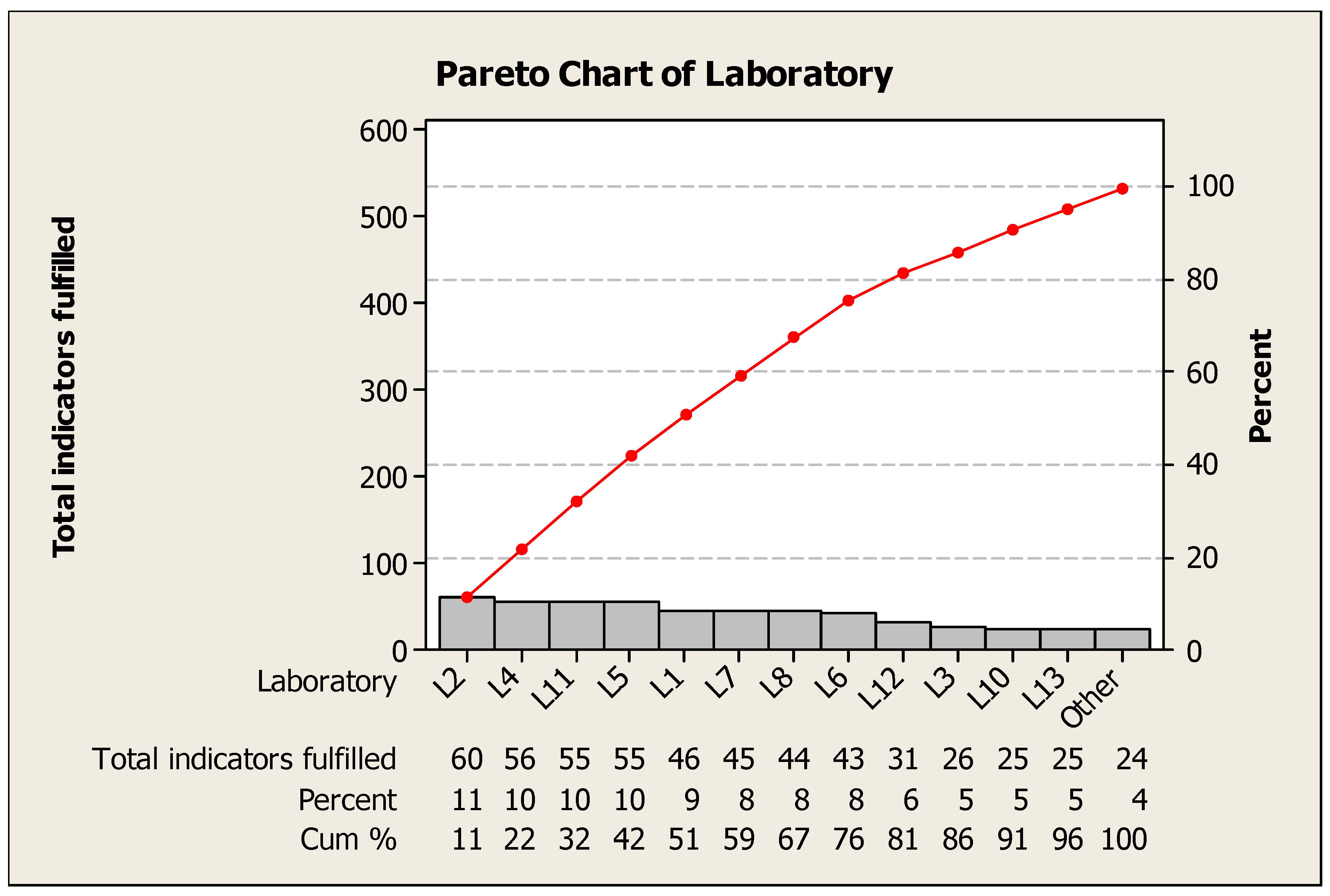
| Laboratory | Total Indicators Fulfilled | Indicators Fulfillment Percentage |
|---|---|---|
| L1 | 46 | 39% |
| L2 | 60 | 51% |
| L3 | 26 | 22% |
| L4 | 56 | 48% |
| L5 | 55 | 47% |
| L6 | 43 | 37% |
| L7 | 45 | 38% |
| L8 | 44 | 38% |
| L9 | 24 | 21% |
| L10 | 25 | 21% |
| L11 | 55 | 47% |
| L12 | 31 | 26% |
| L13 | 25 | 21% |
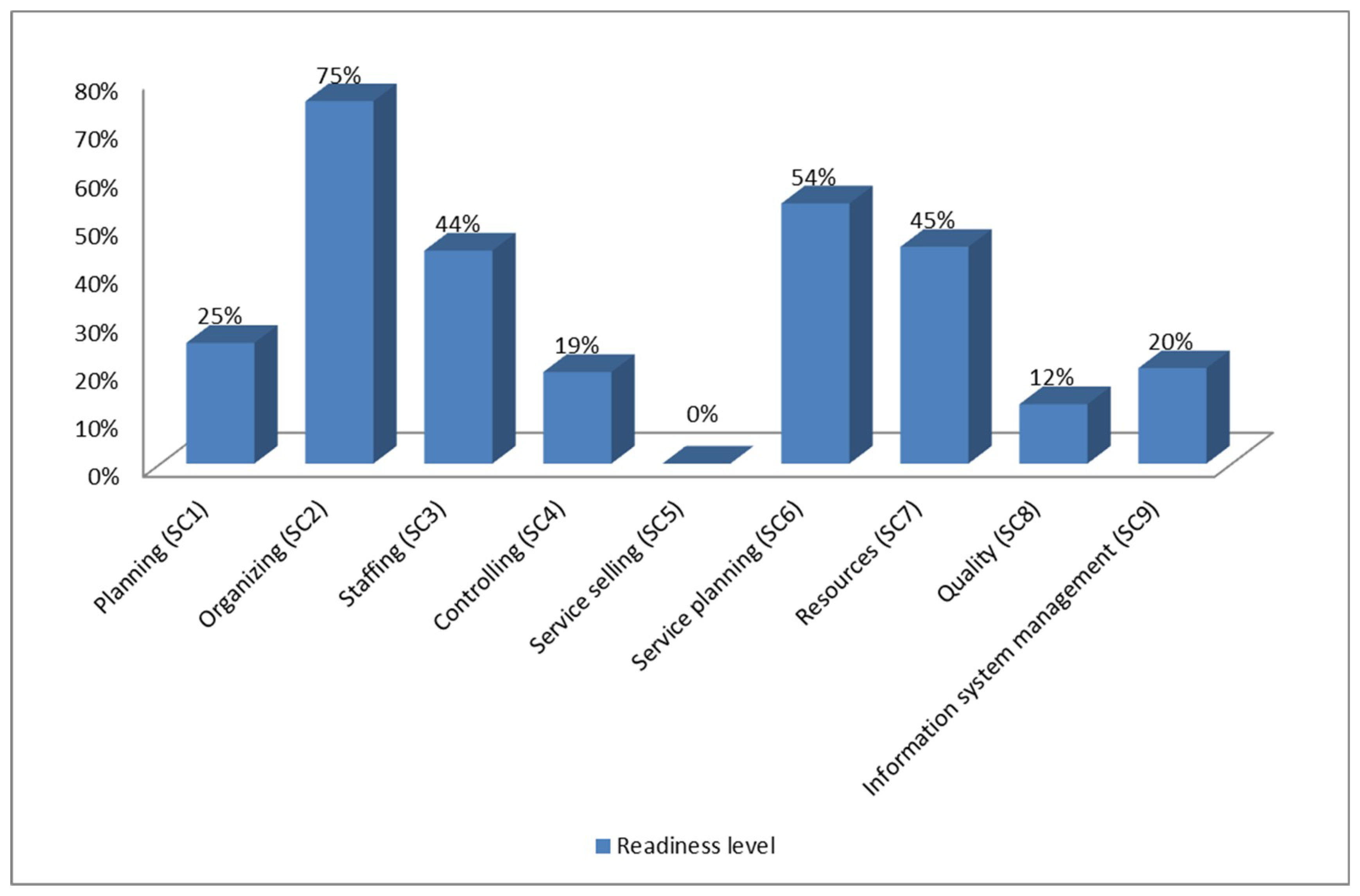
| Criterion | Subcriterion | Indicator Fulfillment Percentage |
|---|---|---|
| Management | Planning | 25% |
| Organizing | 75% | |
| Staffing | 44% | |
| Controlling | 19% | |
| Marketing | Service selling | 0% |
| Service planning | 54% | |
| Operation | Resources | 45% |
| Quality | 12% | |
| Management information system | Information system management | 20% |
| Dimension | Readiness Level | Evidence | Proposed Improvement |
|---|---|---|---|
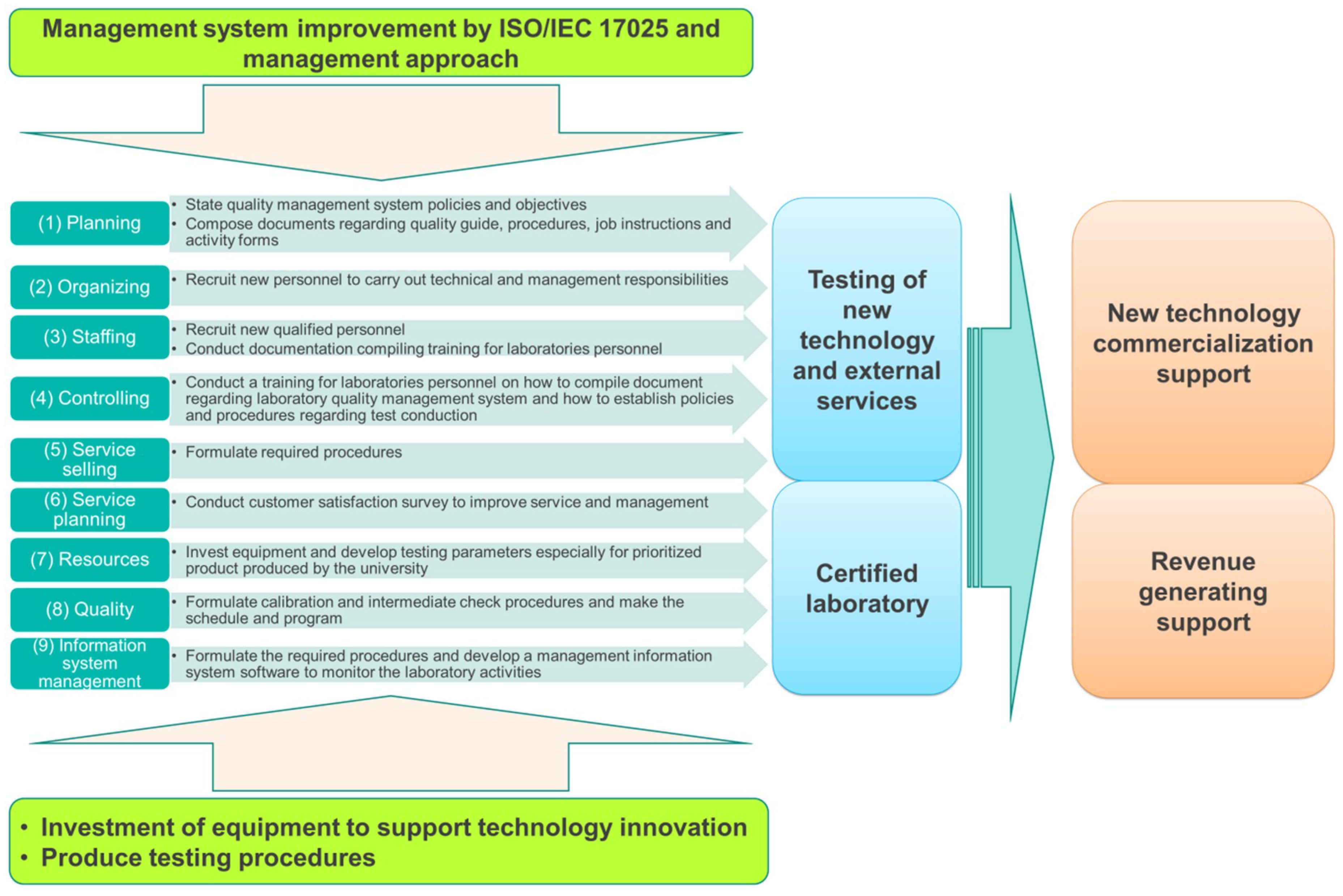
| Planning (SC1) | 25% | Lack of organized management system | State quality management system policies and objectives Compose documents regarding quality guide, procedures, job instructions, and activity forms |
| Undocumented laboratory policies, procedures, programs, and job instructions | |||
| Lack of quality manual | |||
| Unstated objective and policy regarding laboratory quality | |||
| Organizing (SC2) | 75% | Undivided responsibilities between technical and quality management | Recruit new personnel to carry out both responsibilities |
| Staffing (SC3) | 44% | Lack of quality manager | Recruit new qualified personnel |
| Lack of personnel training policy and procedure | |||
| No relevant documentation of personnel’s competency, education, qualification, training, skill, and experience | Conduct documentation compiling training for laboratories personnel | ||
| Controlling (SC4) | 19% | Lack of policy and procedure to resolve customer complaints | Conduct training for laboratories personnel on how to compile documents regarding laboratory quality management system and how to establish policies and procedures regarding test conduction |
| No documentation of customer complaints, investigation, and corrective action towards them | |||
| No established policy and procedure to conduct corrective action | |||
| Lack of procedure to prevent nonconformity and policy deviation | |||
| Lack of audit planning and organizing | |||
| Lack of schedule and procedures for laboratory management review | |||
| Lack of periodic laboratory management review | |||
| Service selling (SC5) | 0% | Lack of customer request, tender, and contract review procedure | Formulate required procedures |
| Service planning (SC6) | 54% | Low interest of laboratories to seek feedback through customer survey | Conduct customer satisfaction survey to improve service and management |
| Resources (SC7) | 45% | Lack of procedure to receive, handle, protect, and identify test items | Invest in equipment and develop testing parameters especially for prioritized product produced by the university |
| Lack of policy and procedure for selecting and buying services and supplies | |||
| Lack of procedure to purchase and receive reagents and disposable materials | |||
| No evaluation towards supplier was conducted | |||
| Lack of procedure for guaranteeing accommodation conditions and laboratory environment | |||
| Lack of procedure to estimate measurement uncertainty | |||
| Quality (SC8) | 12% | Lack of equipment handling and calibration procedure | Formulate calibration and intermediate check procedures and make the schedule and program |
| No intermediate check was conducted | |||
| Lack of quality control procedure and data analysis | |||
| Information system management (SC9) | 20% | Lack of document controlling procedure, corrective and preventive action report | Formulate the required procedures and develop a management information system software to monitor the laboratory activities |
| Lack of record backup making and protection procedure |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aqidawati, E.F.; Sutopo, W.; Zakaria, R. Model to Measure the Readiness of University Testing Laboratories to Fulfill ISO/IEC 17025 Requirements (A Case Study). J. Open Innov. Technol. Mark. Complex. 2019, 5, 2. https://doi.org/10.3390/joitmc5010002
Aqidawati EF, Sutopo W, Zakaria R. Model to Measure the Readiness of University Testing Laboratories to Fulfill ISO/IEC 17025 Requirements (A Case Study). Journal of Open Innovation: Technology, Market, and Complexity. 2019; 5(1):2. https://doi.org/10.3390/joitmc5010002
Chicago/Turabian StyleAqidawati, Era Febriana, Wahyudi Sutopo, and Roni Zakaria. 2019. "Model to Measure the Readiness of University Testing Laboratories to Fulfill ISO/IEC 17025 Requirements (A Case Study)" Journal of Open Innovation: Technology, Market, and Complexity 5, no. 1: 2. https://doi.org/10.3390/joitmc5010002
APA StyleAqidawati, E. F., Sutopo, W., & Zakaria, R. (2019). Model to Measure the Readiness of University Testing Laboratories to Fulfill ISO/IEC 17025 Requirements (A Case Study). Journal of Open Innovation: Technology, Market, and Complexity, 5(1), 2. https://doi.org/10.3390/joitmc5010002





