CVD Risk Stratification in the PCSK9 Era: Is There a Role for LDL Subfractions?
Abstract
1. Introduction
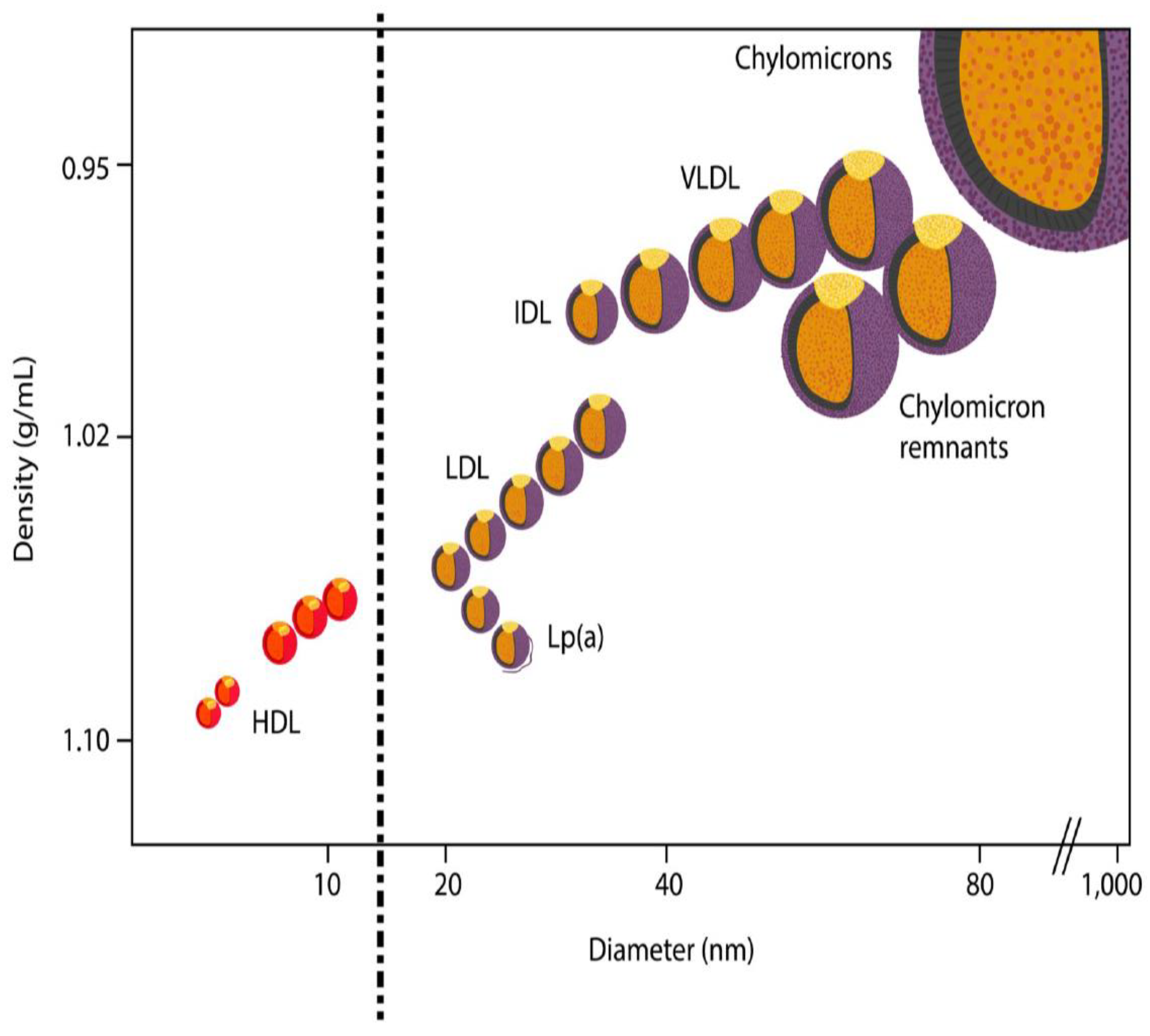
2. LDL Subfractions—And How to Separate Them
3. Comparability of Methods for LDL Subfractioning
4. The Atherogenicity of Small, Dense LDL Particles
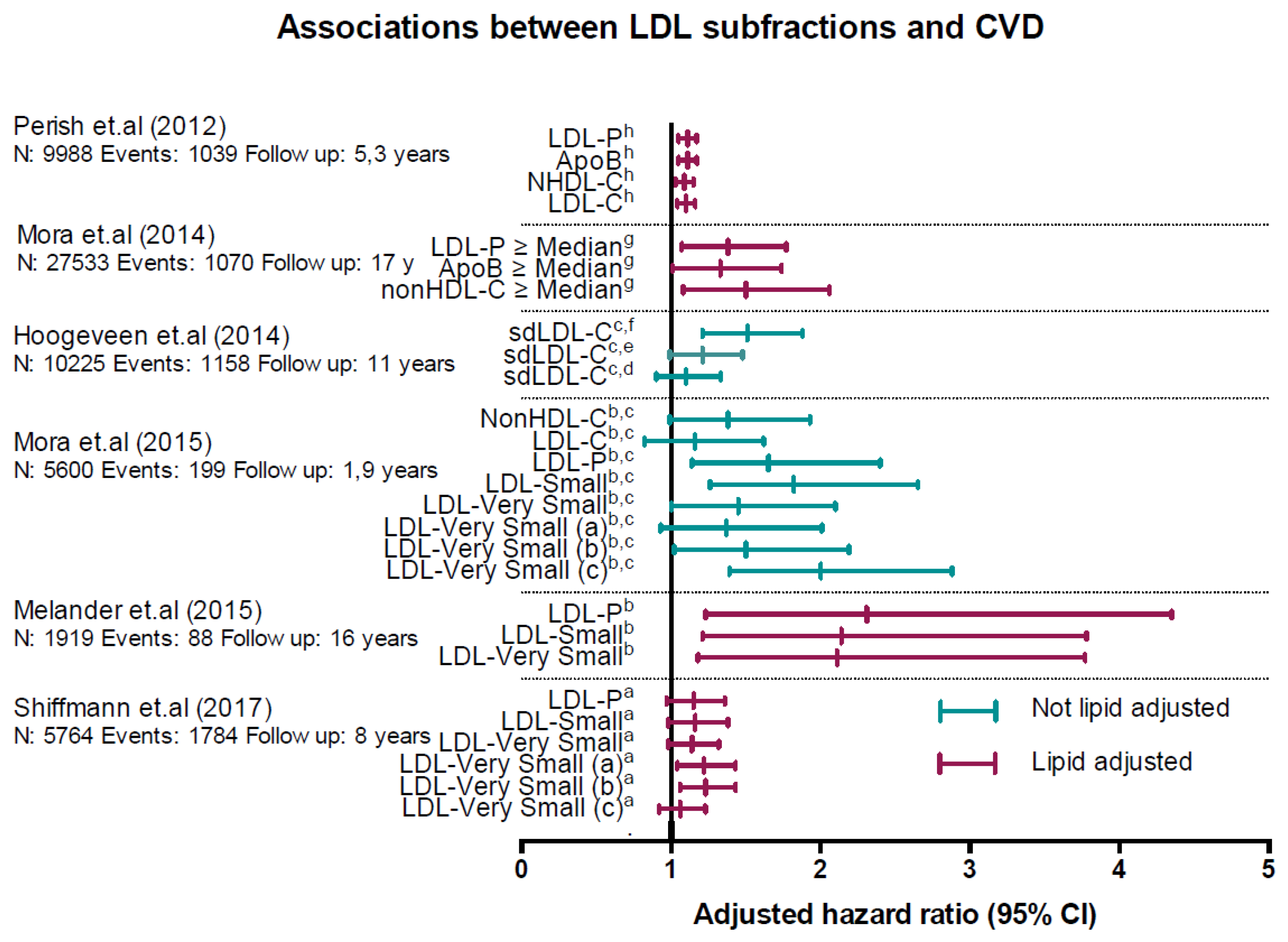
5. LDL Subfractions and Associations with Increased CVD Risk
6. LDL Subfractions and PCSK9 Inhibitors
7. Conclusions
Author Contributions
Conflicts of Interest
References
- WHO. Cardiovascular Disease; WHO: Geneva, Switzerland; Available online: http://www.who.int/cardiovascular_diseases/en/ (accessed on 26 May 2018).
- Morris, P.B.; Ballantyne, C.M.; Birtcher, K.K.; Dunn, S.P.; Urbina, E.M. Review of clinical practice guidelines for the management of LDL-related risk. J. Am. Coll. Cardiol. 2014, 64, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Tibrewala, A.; Jivan, A.; Oetgen, W.J.; Stone, N.J. A comparative analysis of current lipid treatment guidelines: Nothing stands still. J. Am. Coll. Cardiol. 2018, 71, 794–799. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the interheart study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the european atherosclerosis society consensus panel. Eur. Heart J. 2017, 38, 2459–2472. [Google Scholar] [CrossRef] [PubMed]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corra, U.; Cosyns, B.; Deaton, C.; et al. 2016 european guidelines on cardiovascular disease prevention in clinical practice. The sixth joint task force of the european society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of 10 societies and by invited experts. Developed with the special contribution of the european association for cardiovascular prevention & rehabilitation. Giorn. Ital. Cardiol. (Rome) 2017, 18, 547–612. [Google Scholar] [PubMed]
- Ioannidis, J.P.A. Inconsistent guideline recommendations for cardiovascular prevention and the debate about zeroing in on and zeroing LDL-C levels with PCSK9 inhibitors. JAMA 2017, 318, 419–420. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.S.; Blaha, M.J.; Elshazly, M.B.; Brinton, E.A.; Toth, P.P.; McEvoy, J.W.; Joshi, P.H.; Kulkarni, K.R.; Mize, P.D.; Kwiterovich, P.O.; et al. Friedewald-estimated versus directly measured low-density lipoprotein cholesterol and treatment implications. J. Am. Coll. Cardiol. 2013, 62, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, A.; Cannon, C.P.; Deedwania, P.C.; Labresh, K.A.; Smith, S.C., Jr.; Dai, D.; Hernandez, A.; Fonarow, G.C. Lipid levels in patients hospitalized with coronary artery disease: An analysis of 136,905 hospitalizations in get with the guidelines. Am. Heart J. 2009, 157, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Otvos, J.D.; Mora, S.; Shalaurova, I.; Greenland, P.; Mackey, R.H.; Goff, D.C., Jr. Clinical implications of discordance between low-density lipoprotein cholesterol and particle number. J. Clin. Lipidol. 2011, 5, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Buring, J.E.; Ridker, P.M. Discordance of low-density lipoprotein (LDL) cholesterol with alternative ldl-related measures and future coronary events. Circulation 2014, 129, 553–561. [Google Scholar] [CrossRef] [PubMed]
- McGettigan, P.; Ferner, R.E. PCSK9 inhibitors for hypercholesterolaemia. BMJ 2017, 356, j188. [Google Scholar] [CrossRef] [PubMed]
- Landmesser, U.; Chapman, M.J.; Stock, J.K.; Amarenco, P.; Belch, J.J.F.; Boren, J.; Farnier, M.; Ference, B.A.; Gielen, S.; Graham, I.; et al. 2017 update of ESC/EAS task force on practical clinical guidance for proprotein convertase subtilisin/kexin type 9 inhibition in patients with atherosclerotic cardiovascular disease or in familial hypercholesterolaemia. Eur. Heart J. 2018, 39, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, D.M.; Morris, P.B.; Ballantyne, C.M.; Birtcher, K.K.; Daly, D.D., Jr.; DePalma, S.M.; Minissian, M.B.; Orringer, C.E.; Smith, S.C., Jr. 2017 focused update of the 2016 ACC expert consensus decision pathway on the role of non-statin therapies for LDL-cholesterol lowering in the management of atherosclerotic cardiovascular disease risk: A report of the american college of cardiology task force on expert consensus decision pathways. J. Am. Coll. Cardiol. 2017, 70, 1785–1822. [Google Scholar] [PubMed]
- Ramasamy, I. Update on the laboratory investigation of dyslipidemias. Clin. Chim. Acta 2018, 479, 103–125. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Fujimura, M.; Ohta, M.; Hirano, T. Development of a homogeneous assay for measurement of small dense LDL cholesterol. Clin. Chem. 2011, 57, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Hoefner, D.M.; Hodel, S.D.; O’Brien, J.F.; Branum, E.L.; Sun, D.; Meissner, I.; McConnell, J.P. Development of a rapid, quantitative method for LDL subfractionation with use of the quantimetrix lipoprint LDL system. Clin. Chem. 2001, 47, 266–274. [Google Scholar] [PubMed]
- Lipoprint, Q.L. Available online: https://quantimetrix.com/lipoprint/lipoprint-profile/ (accessed on 26 May 2018).
- LipoProfile, N. Available online: http://www.clevelandheartlab.com/tests/nmr-lipoprofile/ (accessed on 26 May 2018).
- Fractionation, Q.D.C.I. Available online: https://education.questdiagnostics.com/faq/FAQ134 (accessed on 26 May 2018).
- Sniderman, A.; Shapiro, S.; Marpole, D.; Skinner, B.; Teng, B.; Kwiterovich, P.O., Jr. Association of coronary atherosclerosis with hyperapobetalipoproteinemia [increased protein but normal cholesterol levels in human plasma low density (beta) lipoproteins]. Proc. Natl. Acad. Sci. USA 1980, 77, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Austin, M.A.; King, M.C.; Vranizan, K.M.; Newman, B.; Krauss, R.M. Inheritance of low-density lipoprotein subclass patterns: Results of complex segregation analysis. Am. J. Hum. Genet. 1988, 43, 838–846. [Google Scholar] [PubMed]
- Chung, M.; Lichtenstein, A.H.; Ip, S.; Lau, J.; Balk, E.M. Comparability of methods for LDL subfraction determination: A systematic review. Atherosclerosis 2009, 205, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Caulfield, M.P.; Wohlgemuth, J.; Chen, Z.; Superko, H.R.; Rowland, C.M.; Glynn, R.J.; Ridker, P.M.; Krauss, R.M. Atherogenic lipoprotein subfractions determined by ion mobility and first cardiovascular events after random allocation to high-intensity statin or placebo: The justification for the use of statins in prevention: An intervention trial evaluating rosuvastatin (jupiter) trial. Circulation 2015, 132, 2220–2229. [Google Scholar] [PubMed]
- Shiffman, D.; Louie, J.Z.; Caulfield, M.P.; Nilsson, P.M.; Devlin, J.J.; Melander, O. LDL subfractions are associated with incident cardiovascular disease in the malmo prevention project study. Atherosclerosis 2017, 263, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, P.N.; Pottala, J.V.; Nanjee, M.N. A comparative study of four independent methods to measure LDL particle concentration. Atherosclerosis 2015, 243, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Sninsky, J.J.; Rowland, C.M.; Baca, A.M.; Caulfield, M.P.; Superko, H.R. Classification of LDL phenotypes by 4 methods of determining lipoprotein particle size. J. Investig. Med. 2013, 61, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.T.; Zhao, X.Q.; Marcovina, S.M.; Otvos, J.D.; Brown, B.G.; Krauss, R.M. Comparison of four methods of analysis of lipoprotein particle subfractions for their association with angiographic progression of coronary artery disease. Atherosclerosis 2014, 233, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.G. What is the role of advanced lipoprotein analysis in practice? J. Am. Coll. Cardiol. 2012, 60, 2607–2615. [Google Scholar] [CrossRef] [PubMed]
- Tabas, I.; Williams, K.J.; Boren, J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: Update and therapeutic implications. Circulation 2007, 116, 1832–1844. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.L.; Brown, M.S. A century of cholesterol and coronaries: From plaques to genes to statins. Cell 2015, 161, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, E.A.; Myasoedova, V.A.; Melnichenko, A.A.; Grechko, A.V.; Orekhov, A.N. Small dense low-density lipoprotein as biomarker for atherosclerotic diseases. Oxid. Med. Cell. Longev. 2017, 2017, 1273042. [Google Scholar] [CrossRef] [PubMed]
- Nigon, F.; Lesnik, P.; Rouis, M.; Chapman, M.J. Discrete subspecies of human low density lipoproteins are heterogeneous in their interaction with the cellular LDL receptor. J. Lipid Res. 1991, 32, 1741–1753. [Google Scholar] [PubMed]
- Tribble, D.L.; Rizzo, M.; Chait, A.; Lewis, D.M.; Blanche, P.J.; Krauss, R.M. Enhanced oxidative susceptibility and reduced antioxidant content of metabolic precursors of small, dense low-density lipoproteins. Am. J. Med. 2001, 110, 103–110. [Google Scholar] [CrossRef]
- Tertov, V.V.; Kaplun, V.V.; Sobenin, I.A.; Boytsova, E.Y.; Bovin, N.V.; Orekhov, A.N. Human plasma trans-sialidase causes atherogenic modification of low density lipoprotein. Atherosclerosis 2001, 159, 103–115. [Google Scholar] [CrossRef]
- Berneis, K.K.; Krauss, R.M. Metabolic origins and clinical significance of LDL heterogeneity. J. Lipid Res. 2002, 43, 1363–1379. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.J.; Guerin, M.; Bruckert, E. Atherogenic, dense low-density lipoproteins. Pathophysiology and new therapeutic approaches. Eur. Heart J. 1998, 19, A24–A30. [Google Scholar] [PubMed]
- Ip, S.; Lichtenstein, A.H.; Chung, M.; Lau, J.; Balk, E.M. Systematic review: Association of low-density lipoprotein subfractions with cardiovascular outcomes. Ann. Intern Med. 2009, 150, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Danielson, E.; Fonseca, F.A.; Genest, J.; Gotto, A.M., Jr.; Kastelein, J.J.; Koenig, W.; Libby, P.; Lorenzatti, A.J.; MacFadyen, J.G.; et al. Rosuvastatin to prevent vascular events in men and women with elevated c-reactive protein. N. Engl. J. Med. 2008, 359, 2195–2207. [Google Scholar] [CrossRef] [PubMed]
- Melander, O.; Shiffman, D.; Caulfield, M.P.; Louie, J.Z.; Rowland, C.M.; Devlin, J.J.; Krauss, R.M. Low-density lipoprotein particle number is associated with cardiovascular events among those not classified into statin benefit groups. J. Am. Coll. Cardiol. 2015, 65, 2571–2573. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hoogeveen, R.C.; Gaubatz, J.W.; Sun, W.; Dodge, R.C.; Crosby, J.R.; Jiang, J.; Couper, D.; Virani, S.S.; Kathiresan, S.; Boerwinkle, E.; et al. Small dense low-density lipoprotein-cholesterol concentrations predict risk for coronary heart disease: The atherosclerosis risk in communities (ARIC) study. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Parish, S.; Offer, A.; Clarke, R.; Hopewell, J.C.; Hill, M.R.; Otvos, J.D.; Armitage, J.; Collins, R.; Heart Protection Study Collaborative Group. Lipids and lipoproteins and risk of different vascular events in the mrc/bhf heart protection study. Circulation 2012, 125, 2469–2478. [Google Scholar] [CrossRef] [PubMed]
- Lagace, T.A. PCSK9 and ldlr degradation: Regulatory mechanisms in circulation and in cells. Curr. Opin. Lipidol. 2014, 25, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Revkin, J.; Amarenco, P.; Brunell, R.; Curto, M.; Civeira, F.; Flather, M.; Glynn, R.J.; Gregoire, J.; Jukema, J.W.; et al. Cardiovascular efficacy and safety of bococizumab in high-risk patients. N. Engl. J. Med. 2017, 376, 1527–1539. [Google Scholar] [CrossRef] [PubMed]
- Giugliano, R.P.; Mach, F.; Zavitz, K.; Kurtz, C.; Im, K.; Kanevsky, E.; Schneider, J.; Wang, H.; Keech, A.; Pedersen, T.R.; et al. Cognitive function in a randomized trial of evolocumab. N. Engl. J. Med. 2017, 377, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Korman, M.; Wisloff, T. Modelling the cost-effectiveness of pcsk9 inhibitors vs. Ezetimibe through ldl-c reductions in a norwegian setting. Eur. Heart J. Cardiovasc. Pharmacother. 2018, 4, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Bonow, R.O.; Harrington, R.A.; Yancy, C.W. Cost-effectiveness of pcsk9 inhibitors: Proof in the modeling. JAMA Cardiol. 2017, 2, 1298–1299. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.X.; Li, S.; Zhang, Y.; Li, X.L.; Guo, Y.L.; Zhu, C.G.; Li, J.J. Relation of plasma pcsk9 levels to lipoprotein subfractions in patients with stable coronary artery disease. Lipids Health Dis. 2014, 13, 188. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, R.X.; Li, S.; Zhu, C.G.; Guo, Y.L.; Sun, J.; Li, J.J. Association of plasma small dense LDL cholesterol with PCSK9 levels in patients with angiographically proven coronary artery disease. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, B.J.; Pelletier-Beaumont, E.; Almeras, N.; Tremblay, A.; Poirier, P.; Bergeron, J.; Despres, J.P. PCSK9 levels in abdominally obese men: Association with cardiometabolic risk profile and effects of a one-year lifestyle modification program. Atherosclerosis 2014, 236, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Kwakernaak, A.J.; Lambert, G.; Dullaart, R.P. Plasma proprotein convertase subtilisin-kexin type 9 is predominantly related to intermediate density lipoproteins. Clin. Biochem. 2014, 47, 679–682. [Google Scholar] [CrossRef] [PubMed]
- Baruch, A.; Mosesova, S.; Davis, J.D.; Budha, N.; Vilimovskij, A.; Kahn, R.; Peng, K.; Cowan, K.J.; Harris, L.P.; Gelzleichter, T.; et al. Effects of rg7652, a monoclonal antibody against PCSK9, on LDL-c, LDL-c subfractions, and inflammatory biomarkers in patients at high risk of or with established coronary heart disease (from the phase 2 equator study). Am. J. Cardiol. 2017, 119, 1576–1583. [Google Scholar] [CrossRef] [PubMed]
- Koren, M.J.; Kereiakes, D.; Pourfarzib, R.; Winegar, D.; Banerjee, P.; Hamon, S.; Hanotin, C.; McKenney, J.M. Effect of PCSK9 inhibition by alirocumab on lipoprotein particle concentrations determined by nuclear magnetic resonance spectroscopy. J. Am. Heart Assoc. 2015, 4, e002224. [Google Scholar] [PubMed]
- Wu, L.; Bamberger, C.; Waldmann, E.; Stark, R.; Henze, K.; Parhofer, K. The effect of PCSK9 inhibition on LDL-subfractions in patients with severe ldl-hypercholesterolemia. J. Am. Coll. Cardiol. 2017, 69, 1719. [Google Scholar] [CrossRef]
- Lappegard, K.T.; Kjellmo, C.A.; Ljunggren, S.; Cederbrant, K.; Marcusson-Stahl, M.; Mathisen, M.; Karlsson, H.; Hovland, A. Lipoprotein apheresis affects lipoprotein particle subclasses more efficiently compared to the PCSK9 inhibitor evolocumab, a pilot study. Transfus. Apher. Sci. 2018, 57, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Blake, G.J.; Albert, M.A.; Rifai, N.; Ridker, P.M. Effect of pravastatin on LDL particle concentration as determined by NMR spectroscopy: A substudy of a randomized placebo controlled trial. Eur. Heart J. 2003, 24, 1843–1847. [Google Scholar] [CrossRef] [PubMed]
- Superko, H.R.; Krauss, R.M.; DiRicco, C. Effect of fluvastatin on low-density lipoprotein peak particle diameter. Am. J. Cardiol. 1997, 80, 78–81. [Google Scholar] [CrossRef]
- Frost, R.J.; Otto, C.; Geiss, H.C.; Schwandt, P.; Parhofer, K.G. Effects of atorvastatin versus fenofibrate on lipoprotein profiles, low-density lipoprotein subfraction distribution, and hemorheologic parameters in type 2 diabetes mellitus with mixed hyperlipoproteinemia. Am. J. Cardiol. 2001, 87, 44–48. [Google Scholar] [CrossRef]
- Otvos, J.D.; Shalaurova, I.; Freedman, D.S.; Rosenson, R.S. Effects of pravastatin treatment on lipoprotein subclass profiles and particle size in the PLAC-I trial. Atherosclerosis 2002, 160, 41–48. [Google Scholar] [CrossRef]
- Karalis, D.G.; Ishisaka, D.Y.; Luo, D.; Ntanios, F.; Wun, C.C. Effects of increasing doses of atorvastatin on the atherogenic lipid subclasses commonly associated with hypertriglyceridemia. Am. J. Cardiol. 2007, 100, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.J.; Orsoni, A.; Robillard, P.; Therond, P.; Giral, P. Duality of statin action on lipoprotein subpopulations in the mixed dyslipidemia of metabolic syndrome: Quantity vs. quality over time and implication of cetp. J. Clin. Lipidol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Soedamah-Muthu, S.S.; Colhoun, H.M.; Thomason, M.J.; Betteridge, D.J.; Durrington, P.N.; Hitman, G.A.; Fuller, J.H.; Julier, K.; Mackness, M.I.; Neil, H.A.; et al. The effect of atorvastatin on serum lipids, lipoproteins and NMR spectroscopy defined lipoprotein subclasses in type 2 diabetic patients with ischaemic heart disease. Atherosclerosis 2003, 167, 243–255. [Google Scholar] [CrossRef]
- Hlatky, M.A.; Greenland, P.; Arnett, D.K.; Ballantyne, C.M.; Criqui, M.H.; Elkind, M.S.; Go, A.S.; Harrell, F.E., Jr.; Hong, Y.; Howard, B.V.; et al. Criteria for evaluation of novel markers of cardiovascular risk: A scientific statement from the american heart association. Circulation 2009, 119, 2408–2416. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kjellmo, C.A.; Hovland, A.; Lappegård, K.T. CVD Risk Stratification in the PCSK9 Era: Is There a Role for LDL Subfractions? Diseases 2018, 6, 45. https://doi.org/10.3390/diseases6020045
Kjellmo CA, Hovland A, Lappegård KT. CVD Risk Stratification in the PCSK9 Era: Is There a Role for LDL Subfractions? Diseases. 2018; 6(2):45. https://doi.org/10.3390/diseases6020045
Chicago/Turabian StyleKjellmo, Christian Abendstein, Anders Hovland, and Knut Tore Lappegård. 2018. "CVD Risk Stratification in the PCSK9 Era: Is There a Role for LDL Subfractions?" Diseases 6, no. 2: 45. https://doi.org/10.3390/diseases6020045
APA StyleKjellmo, C. A., Hovland, A., & Lappegård, K. T. (2018). CVD Risk Stratification in the PCSK9 Era: Is There a Role for LDL Subfractions? Diseases, 6(2), 45. https://doi.org/10.3390/diseases6020045



