Rising Rates of Hepatocellular Carcinoma Leading to Liver Transplantation in Baby Boomer Generation with Chronic Hepatitis C, Alcohol Liver Disease, and Nonalcoholic Steatohepatitis-Related Liver Disease
Abstract
:1. Introduction
2. Methods
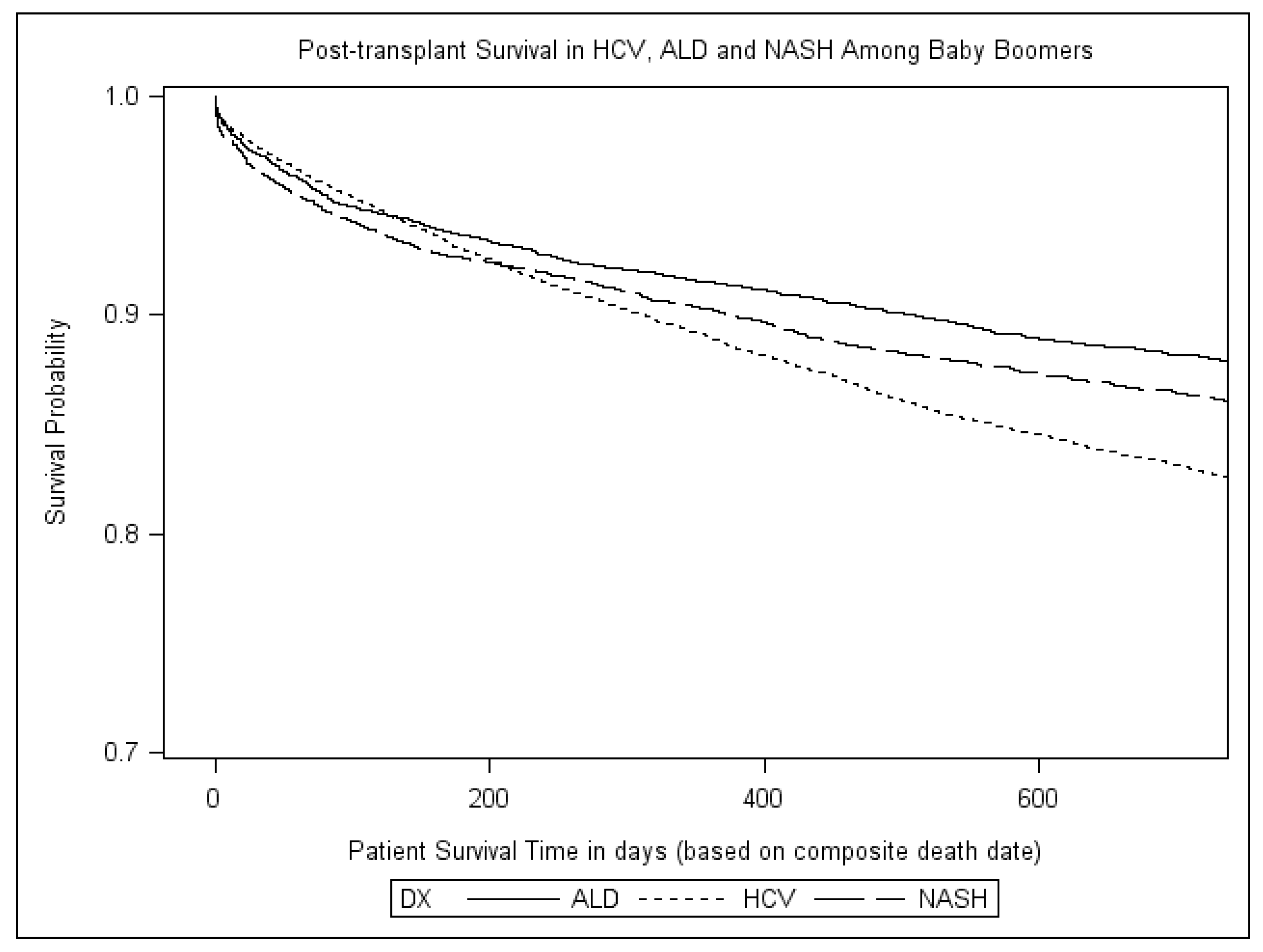
3. Results
4. Discussion
Author Contributions
Conflicts of Interest
References
- El-Serag, H.B. Hepatocellular carcinoma. N. Engl. J. Med. 2011, 365, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Altekruse, S.F.; McGlynn, K.A.; Reichman, M.E. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J. Clin. Oncol. 2009, 27, 1485–1491. [Google Scholar] [CrossRef] [PubMed]
- Chung, R.T.; Baumert, T.F. Curing chronic hepatitis C—The arc of a medical triumph. N. Engl. J. Med. 2014, 370, 1576–1578. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. Hepatitis C Fact Sheet. Available online: http://www.hhs.gov/opa/reproductive-health/stis/hepatitis-c/ (accessed on 17 July 2016).
- Wray, C.M.; Davis, A.M. Screening for hepatitis C. JAMA 2015, 313, 1855–1856. [Google Scholar] [CrossRef] [PubMed]
- Fattovich, G.; Stroffolini, T.; Zagni, I.; Donato, F. Hepatocellular carcinoma in cirrhosis: Incidence and risk factors. Gastroenterology 2004, 127 (Suppl. 1), S35–S50. [Google Scholar] [CrossRef] [PubMed]
- Biggins, S.W.; Bambha, K.M.; Terrault, N.A.; Inadomi, J.; Shiboski, S.; Dodge, J.L.; Gralla, J.; Rosen, H.R.; Roberts, J.P. Projected future increase in aging hepatitis C virus-infected liver transplant candidates: A potential effect of hepatocellular carcinoma. Liver Transplant. 2012, 18, 1471–1478. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.J.; Cheung, R.; Ahmed, A. Nonalcoholic steatohepatitis is the most rapidly growing indication for liver transplantation in patients with hepatocellular carcinoma in the U.S. Hepatology 2014, 59, 2188–2195. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Ha, J.; Aguilar, M.; Bhuket, T.; Liu, B.; Gish, R.G.; Cheung, R.; Wong, R.J. Birth cohort-specific disparities in hepatocellular carcinoma stage at diagnosis, treatment, and long-term survival. J. Hepatol. 2016, 64, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Northup, P.G.; Intagliata, N.M.; Shah, N.L.; Pelletier, S.J.; Berg, C.L.; Argo, C.K. Excess mortality on the liver transplant waiting list: Unintended policy consequences and Model for End-Stage Liver Disease (MELD) inflation. Hepatology 2015, 61, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Moyer, V.A. U.S. Preventive Services Task Force. Screening for hepatitis C virus infection in adults: U.S. Preventive Services Task Force recommendation statement. Ann. Intern. Med. 2013, 159, 349–357. [Google Scholar] [CrossRef] [PubMed]
| HCV | NASH | ALD | |||||||
|---|---|---|---|---|---|---|---|---|---|
| BB † | Overall | Percent | BB | Overall | Percent | BB | Overall | Percent | |
| 2003 | 149 | 216 | 69.0% | 11 | 19 | 57.9% | 22 | 37 | 59.5% |
| 2004 | 167 | 224 | 74.6% | 20 | 38 | 52.6% | 16 | 33 | 48.5% |
| 2005 | 218 | 297 | 73.4% | 22 | 47 | 46.8% | 28 | 59 | 47.5% |
| 2006 | 271 | 351 | 77.2% | 37 | 71 | 52.1% | 36 | 75 | 48.0% |
| 2007 | 373 | 461 | 80.9% | 54 | 90 | 60.0% | 38 | 85 | 44.7% |
| 2008 | 427 | 505 | 84.6% | 80 | 124 | 64.5% | 48 | 79 | 60.8% |
| 2009 | 460 | 540 | 85.2% | 96 | 151 | 63.6% | 56 | 76 | 73.7% |
| 2010 | 492 | 585 | 84.1% | 101 | 132 | 76.5% | 45 | 66 | 68.2% |
| 2011 | 567 | 632 | 89.7% | 91 | 128 | 71.1% | 81 | 107 | 75.7% |
| 2012 | 619 | 707 | 87.6% | 127 | 168 | 75.6% | 77 | 106 | 72.6% |
| 2013 | 646 | 722 | 89.5% | 153 | 191 | 80.1% | 69 | 92 | 75.0% |
| 2014 | 719 | 794 | 90.6% | 157 | 191 | 82.2% | 85 | 114 | 74.6% |
| Total | 5108 | 6034 | 84.7% | 949 | 1350 | 70.3% | 601 | 929 | 64.7% |
| APC ‡ | +2.6% | +3.5% | +2.1% | ||||||
| HCV | NASH | ALD | |||||||
|---|---|---|---|---|---|---|---|---|---|
| BB n = 5108 | Non-BB n = 926 | p | BB n = 949 | Non-BB n = 401 | p | BB n = 601 | Non-BB n = 328 | p | |
| Age, median | 57 | 66 | <0.01 | 59 | 67 | <0.01 | 58 | 67 | <0.01 |
| Gender | |||||||||
| Male | 80.3% | 66.1% | <0.01 | 73.0% | 65.1% | <0.01 | 90.5% | 89.6% | 0.67 |
| Ethnicity | |||||||||
| White | 67.7% | 59.1% | <0.01 | 75.9% | 77.6% | 0.50 | 69.6% | 78.7% | <0.01 |
| Black | 13.3% | 11.1% | 0.07 | 5.4% | 3.7% | 0.20 | 3.8% | 1.2% | 0.02 |
| Hispanic | 13.6% | 17.6% | <0.01 | 14.7% | 14.5% | 0.93 | 14.7% | 14.5% | 0.14 |
| Asian | 4.1% | 11.1% | <0.01 | 2.6% | 2.2% | 0.67 | 3.0% | 1.2% | 0.89 |
| Other | 9.9% | 1.1% | <0.01 | 1.4% | 2.0% | 0.49 | 8.9% | 4.4% | <0.05 |
| HE | 40.2% | 34.8% | <0.01 | 43.4% | 41.4% | <0.50 | 50.3% | 43.9% | 0.09 |
| Diabetes | 23.6% | 28.4% | <0.01 | 47.7% | 47.7% | <0.01 | 35.1% | 33.5% | 0.63 |
| Ascites | 50.9% | 46.8% | <0.05 | 55.2% | 55.2% | 0.84 | 68.6% | 62.5% | 0.06 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cholankeril, G.; Yoo, E.R.; Perumpail, R.B.; Liu, A.; Sandhu, J.S.; Nair, S.; Hu, M.; Ahmed, A. Rising Rates of Hepatocellular Carcinoma Leading to Liver Transplantation in Baby Boomer Generation with Chronic Hepatitis C, Alcohol Liver Disease, and Nonalcoholic Steatohepatitis-Related Liver Disease. Diseases 2017, 5, 20. https://doi.org/10.3390/diseases5040020
Cholankeril G, Yoo ER, Perumpail RB, Liu A, Sandhu JS, Nair S, Hu M, Ahmed A. Rising Rates of Hepatocellular Carcinoma Leading to Liver Transplantation in Baby Boomer Generation with Chronic Hepatitis C, Alcohol Liver Disease, and Nonalcoholic Steatohepatitis-Related Liver Disease. Diseases. 2017; 5(4):20. https://doi.org/10.3390/diseases5040020
Chicago/Turabian StyleCholankeril, George, Eric R. Yoo, Ryan B. Perumpail, Andy Liu, Jeevin S. Sandhu, Satheesh Nair, Menghan Hu, and Aijaz Ahmed. 2017. "Rising Rates of Hepatocellular Carcinoma Leading to Liver Transplantation in Baby Boomer Generation with Chronic Hepatitis C, Alcohol Liver Disease, and Nonalcoholic Steatohepatitis-Related Liver Disease" Diseases 5, no. 4: 20. https://doi.org/10.3390/diseases5040020
APA StyleCholankeril, G., Yoo, E. R., Perumpail, R. B., Liu, A., Sandhu, J. S., Nair, S., Hu, M., & Ahmed, A. (2017). Rising Rates of Hepatocellular Carcinoma Leading to Liver Transplantation in Baby Boomer Generation with Chronic Hepatitis C, Alcohol Liver Disease, and Nonalcoholic Steatohepatitis-Related Liver Disease. Diseases, 5(4), 20. https://doi.org/10.3390/diseases5040020





