A Galvanic Intrabody Method for Assessing Fluid Flow in Unilateral Lymphoedema
Abstract
:1. Introduction
- i
- Physical examination and investigation of the history of the affected area.
- ii
- Limb volume measurement.
- iii
- Bioelectrical impedance spectroscopy method.
- iv
- Blood test and genetic test analysis.
- v
- Imaging techniques.
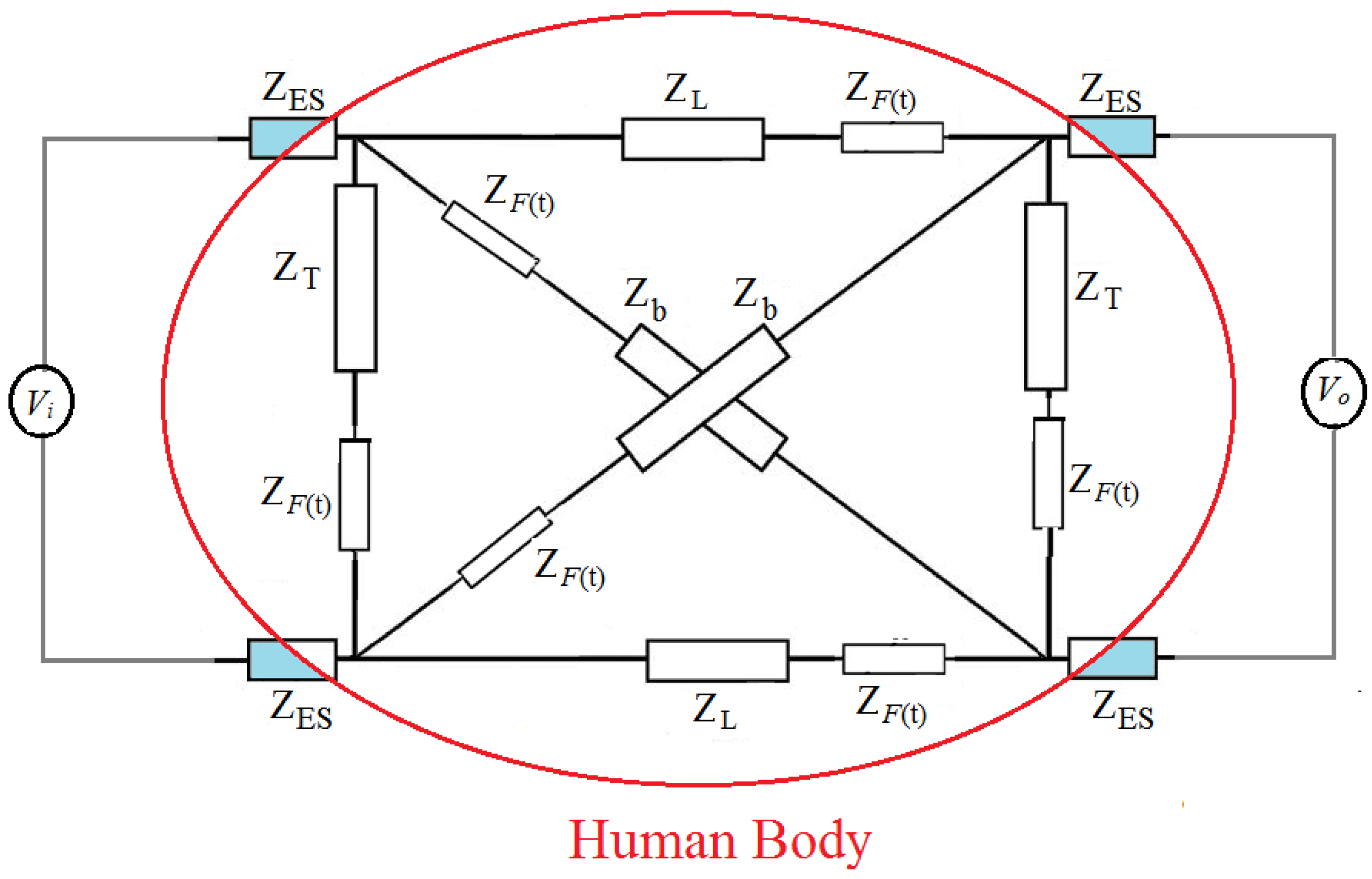
2. A New Diagnostic Method
3. Experiments
3.1. Clinical Measurements: Pathological Lymphoedema Participants
3.2. Clinical Measurements: Healthy Participants
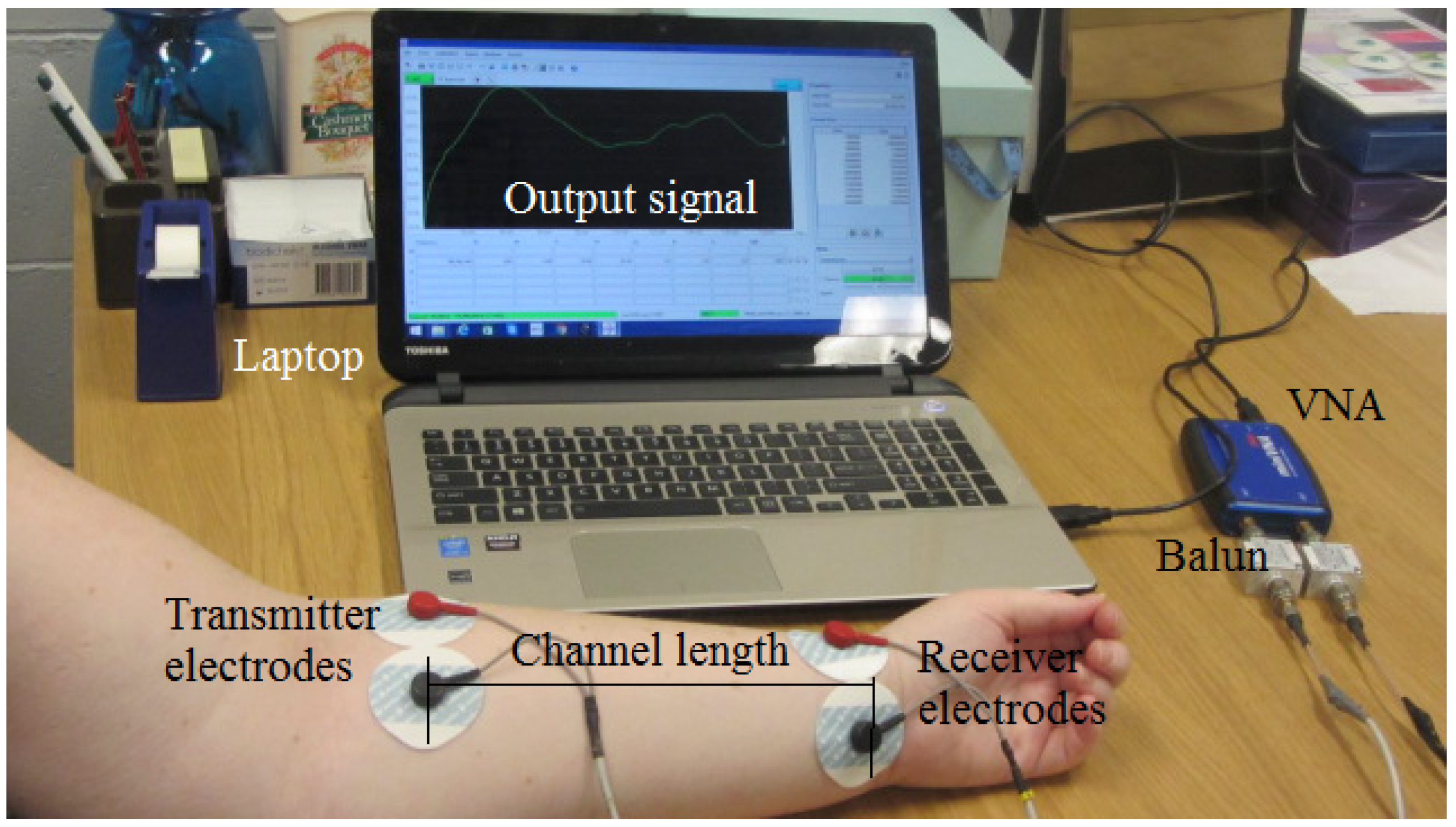
3.3. Galvanic Coupling Measurements
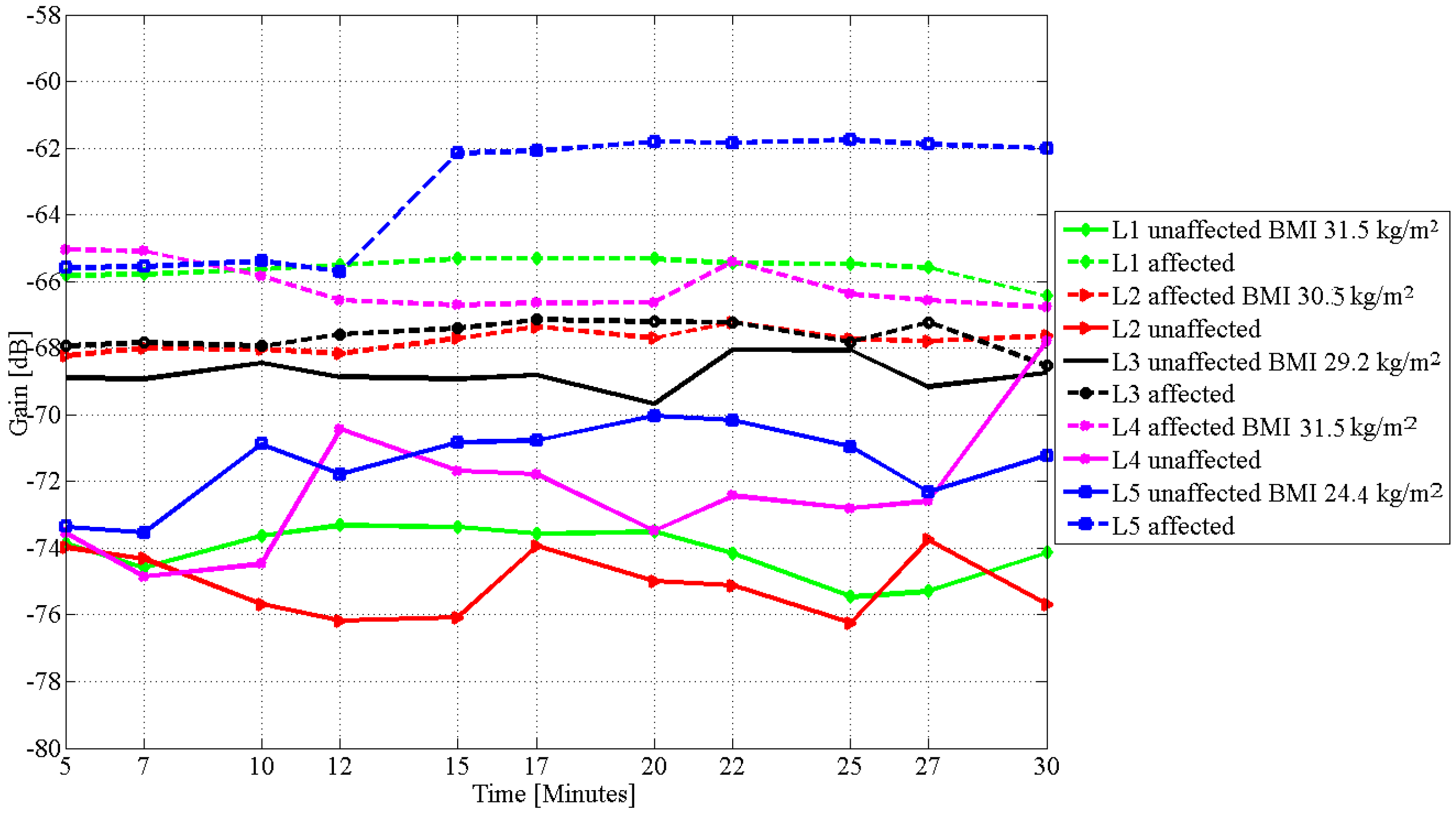
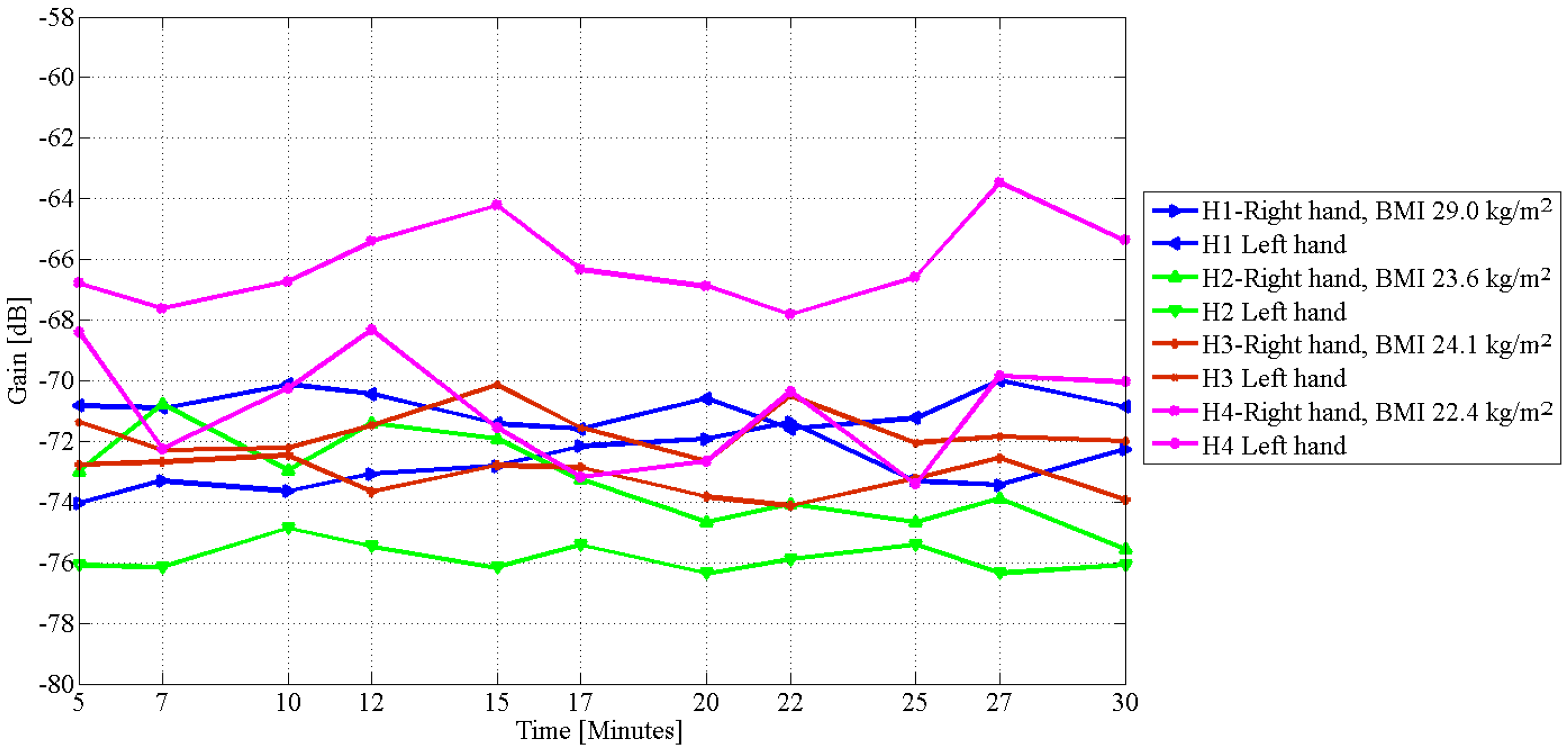
4. Results
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rockson, S.G. Lymphedema. Am. J. Med. 2001, 110, 288–295. [Google Scholar] [CrossRef]
- Hayes, S.C. Review of Research Evidence on Secondary Lymphoedema: Incidence, Prevention, Risk Factors and Treatment; National Breast and Ovarian Cancer Centre: Surry Hills, Australia, 2008. [Google Scholar]
- MacLaren, J. Skin changes in lymphoedema: Pathophysiology and management options. Int. J. Palliat. Nurs. 2001, 7, 381–388. [Google Scholar] [CrossRef]
- Foldi, E.; Jünger, M.; Partsch, H. The Science of Lymphoedema Bandaging. In European Wound Management Association (EWMA). Lymphoedema Bandaging in Practice London; MEP Ltd.: London, UK, 2005; pp. 1–4. [Google Scholar]
- Vojackova, N.; Fialova, J.; Hercogova, J. Management of lymphedema. Dermatol. Ther. 2012, 25, 352–357. [Google Scholar] [CrossRef]
- Cheville, A.L.; McGarvey, C.L.; Petrek, J.A.; Russo, S.A.; Taylor, M.E.; Thiadens, S.R. Lymphedema management. Semin. Radiat. Oncol. 2003, 13, 290–301. [Google Scholar] [CrossRef]
- Ramos, S.M.; O’Donnell, L.S.; Knight, G. Edema volume, not timing, is the key to success in lymphedema treatment. Am. J. Surg. 1999, 178, 311–315. [Google Scholar] [CrossRef]
- NLN Medical Advisory Committee. Position Statement of the National Lymphedema Network; National Lymphedema Network: Berkeley, CA, USA, 2011. [Google Scholar]
- World Health Organization (WHO). Global Programme to Eliminate Lymphatic Filariasis: Progress Report. 2015. Available online: http://www.who.int/mediacentre/factsheets/fs102/en/ (accessed on 17 December 2016).
- Moffatt, C.J.; Franks, P.J.; Doherty, D.C.; Williams, A.F.; Badger, C.; Jeffs, E.; Bosanquet, N.; Mortimer, P.S. Lymphoedema: An underestimated health problem. QJM 2003, 96, 731–738. [Google Scholar] [CrossRef]
- Moffatt, C.J.; Doherty, D.; Morgan, P. Best Practice for the Management of Lymphoedema; Lymphoedema Framework International Consensus; MEP Ltd.: London, UK, 2006; Volume 1, pp. 3–52. [Google Scholar]
- Kerchner, K.; Fleischer, A.; Yosipovitch, G. Lower extremity lymphedema: Update: Pathophysiology, diagnosis, and treatment guidelines. J. Am. Acad. Dermatol. 2008, 59, 324–331. [Google Scholar] [CrossRef]
- Ma, H.; Iafrati, M. Diagnosis and Management of Lymphedema. In Haimovici’s Vascular Surgery, 6th ed.; Wiley-Blackwell: Oxford, UK, 2012; pp. 1221–1235. [Google Scholar]
- Ridner, S.H.; Montgomery, L.D.; Hepworth, J.T.; Stewart, B.R.; Armer, J.M. Comparison of upper limb volume measurement techniques and arm symptoms between healthy volunteers and individuals with known lymphedema. Lymphology 2007, 40, 35–46. [Google Scholar]
- Grimnes, S.; Martinsen, Ø.G. Bioimpedance and Bioelectricity Basics; Elsevier Academic Press: London, UK, 2008. [Google Scholar]
- Jaffrin, M.Y.; Morel, H. Body fluid volumes measurements by impedance: A review of bioimpedance spectroscopy (BIS) and bioimpedance analysis (BIA) methods. Med. Eng. Phys. 2008, 30, 1257–1269. [Google Scholar] [CrossRef]
- Cornish, B.H.; Chapman, M.; Hirst, C.; Mirolo, B.; Bunce, I.H.; Ward, L.C.; Thomas, B.J. Early diagnosis of lymphedema using multiple frequency bioimpedance. Lymphology 2001, 34, 2–11. [Google Scholar]
- Matthie, J.R. Bioimpedance measurements of human body composition: Critical analysis and outlook. Expert Rev. Med. Devices 2008, 5, 239–261. [Google Scholar] [CrossRef]
- Rothlingshofer, L.; Ulbrich, M.; Hahne, S.; Leonhardt, S. Monitoring change of body fluid during physical exercise using bioimpedance spectroscopy and finite element simulations. J. Electr. Bioimped. 2011, 2, 79–85. [Google Scholar] [CrossRef]
- Asogwa, C.O.; Teshome, A.K.; Lai, D.T.H.; Collins, S.F. A Circuit Model of Real Time Human Body Hydration. IEEE Trans. Biomed. Eng. 2016, 63, 1239–1247. [Google Scholar] [CrossRef]
- Kibret, B.; Seyedi, M.; Lai, D.T.H.; Faulkner, M. Investigation of Galvanic Coupled Intrabody Communication using Human Body Circuit Model. IEEE J. Biomed. Health Inform. 2014, 18, 1196–1206. [Google Scholar] [CrossRef]
- Andreuccetti, D.; Fossi, R.; Petrucci, C. An Internet Resource for the Calculation of the Dielectric Properties of Body Tissues in the Frequency Range 10 Hz–100 GHz; Institute of Applied Physics, nello-carrara (IFAC-CNR): Florence, Italy, 2007; Available online: http://niremf.ifac.cnr.it/tissprop/ (accessed on 15 August 2015).
- Wegmueller, M.S.; Oberle, M.; Felber, N.; Kuster, N.; Fichtner, W. Signal transmission by galvanic coupling through the human body. IEEE Trans. Instrum. Meas. 2010, 59, 963–969. [Google Scholar] [CrossRef]
- Mifflin, M.D.; St Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar]
- Verbalis, J.G. Disorders of body water homeostasis. Best Pract. Res. Clin. Endocrinol. Metab. 2003, 17, 471–503. [Google Scholar] [CrossRef]
- Peronnet, F.; Mignault, D.; Du Souich, P.; Vergne, S.; Le Bellego, L.; Jimenez, L.; Rabasa-Lhoret, R. Pharmacokinetic analysis of absorption, distribution and disappearance of ingested water labelled with D2O in humans. Eur. J. Appl. Physiol. 2012, 112, 2213–2222. [Google Scholar] [CrossRef]
- Wabel, P.; Chamney, P.; Moissl, U.; Jirka, T. Importance of whole-body bioimpedance spectroscopy for the management of fluid balance. Blood Purif. 2009, 27, 75–80. [Google Scholar] [CrossRef]
- Asogwa, C.O.; Lai, D.T.H. Effect of Changing Body Fluid Levels on Intrabody Signal Propagation. In Ambient Intelligent and Smart Environments; IOS Press: Amsterdam, The Netherlands, 2016; Volume 21, pp. 552–559. [Google Scholar]
- Ahlbom, A.; Bergqvist, U.; Bernhardt, J.; Cesarini, J.; Grandolfo, M.; Hietanem, M.; Mckinlay, A.; Repacholi, R.; Sliney, D.; Stolwijk, J. Guidelines for limiting exposure to time-varying electric, magnetic, and electromagnetic fields (up tp 300 GHz). International commission on non-ionizing radiation protection. Health Phys. 1998, 74, 494–522. [Google Scholar]
- Day, S. Important Factors in Surface EMG Measurement; Bortec Biomedical Ltd.: Calgary, AB, Canada, 2002; pp. 1–17. [Google Scholar]
- Dahlof, B.; Sever, P.S.; Poulter, N.R.; Wedel, H.; Beevers, D.G.; Caulfield, M.; Collins, R.; Kjeldsen, S.E.; Kristinsson, A.; McInnes, G.T.; et al. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA): A multicentre randomised controlled trial. Lancet 2005, 366, 895–906. [Google Scholar]
- Sands, J.M.; Layton, H.E. The physiology of urinary concentration: An update. Semin. Nephrol. 2009, 29, 178–195. [Google Scholar] [CrossRef]
- Asogwa, C.O.; Collins, S.F.; Mclaughlin, P.; Lai, D.T.H. A Galvanic Coupling Method for Assessing Hydration Rates. Electronics 2016, 5, 39. [Google Scholar] [CrossRef]
- Kloecker, R.J. Compression Garment for Selective Application for Treatment of Lymphedema and Related Illnesses Manifested at Various Locations of the Body. U.S. Patent 6436064, 20 August 2002. [Google Scholar]
- Brennan, M.J.; Miller, L.T. Overview of treatment options and review of the current role and use of compression garments, intermittent pumps, and exercise in the management of lymphedema. Cancer 1998, 83, 2821–2827. [Google Scholar] [CrossRef]
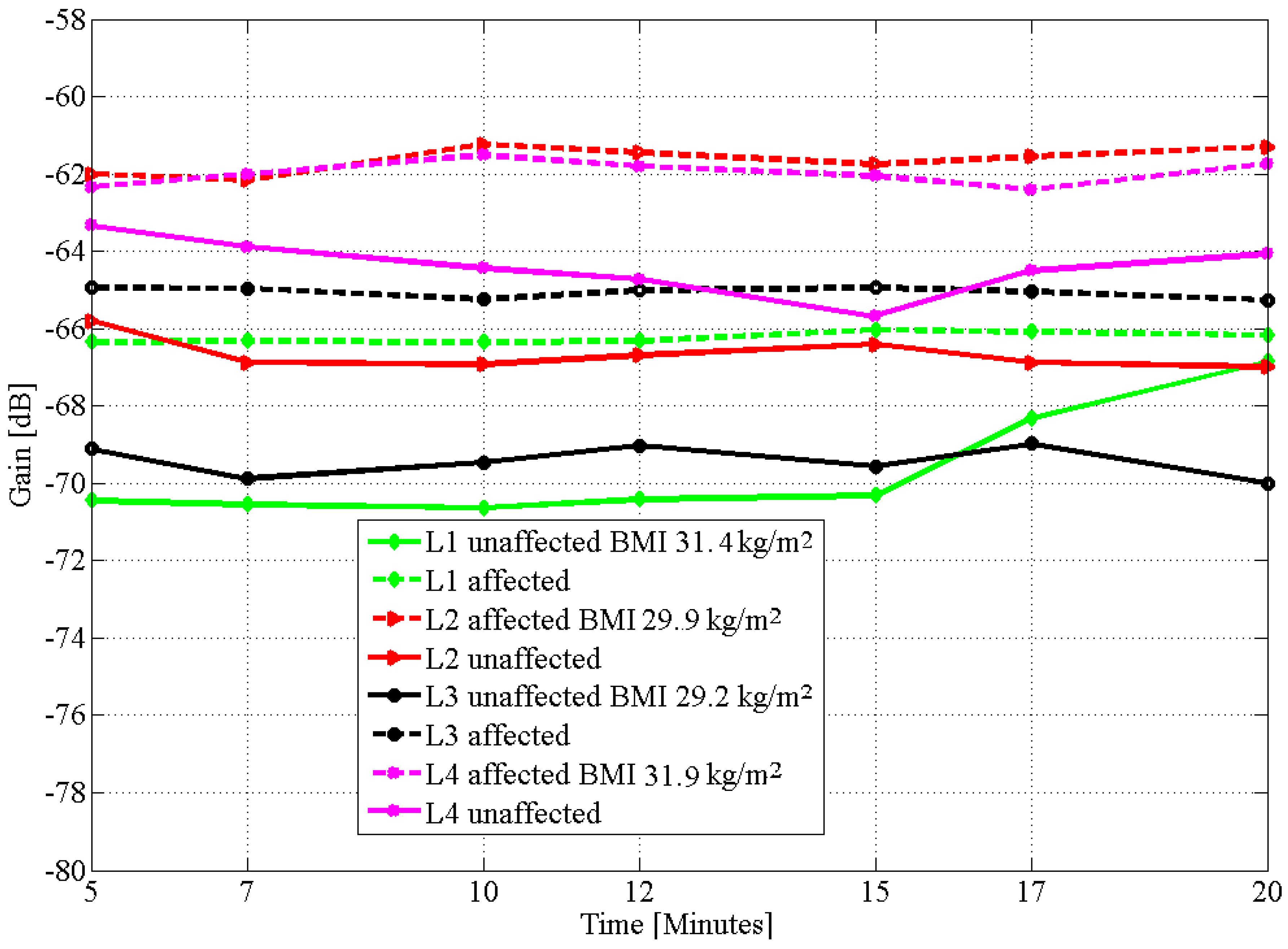
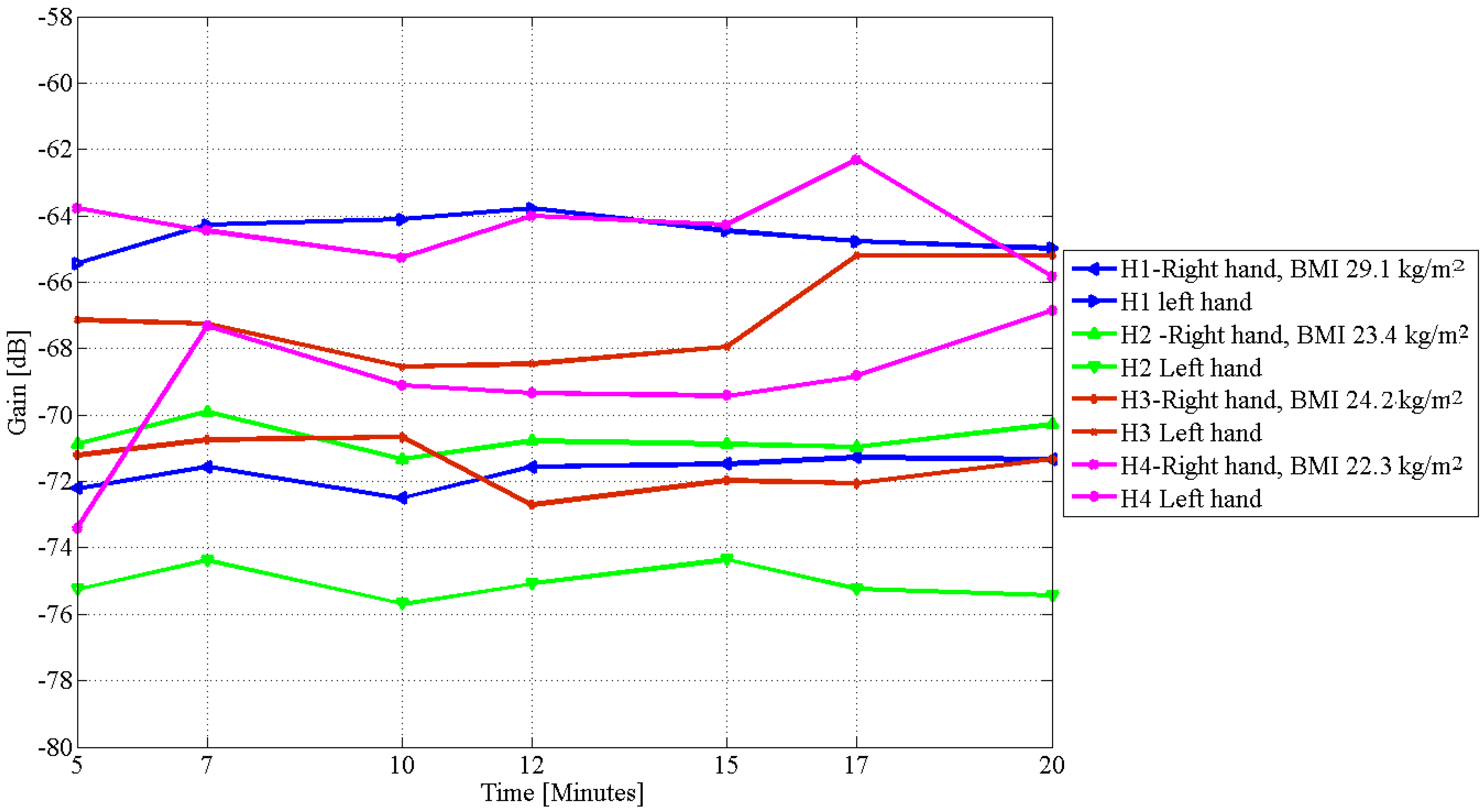
| Subject/Affected Limb | Day | BMI | Affected (CLM) | Unaffected (CLM) | % Difference | |||
|---|---|---|---|---|---|---|---|---|
| (kg/m) | Upper | Lower | Upper | Lower | Upper | Lower | ||
| L1 (left arm) | 1 | 31.7 | 39.0 | 35.0 | 34.5 | 27.0 | 13.0 | 29.6 |
| 2 | 31.5 | 39.2 | 35.0 | 34.5 | 27.0 | 13.6 | 29.6 | |
| 3 | 31.4 | 39.0 | 35.4 | 34.3 | 27.4 | 13.7 | 29.1 | |
| L2 (Right arm) | 1 | 31.3 | 37.0 | 25.0 | 29.0 | 22.5 | 27.5 | 11.1 |
| 2 | 30.5 | 36.8 | 25.3 | 29.0 | 22.7 | 26.8 | 11.4 | |
| 3 | 29.9 | 37.0 | 25.4 | 29.2 | 22.3 | 26.7 | 13.9 | |
| L3 (Left leg) | 1 | 29.1 | 43.5 | 28.8 | 38.0 | 26.0 | 14.5 | 10.7 |
| 2 | 29.2 | 43.6 | 29.2 | 38.1 | 26.3 | 14.4 | 11.0 | |
| 3 | 29.2 | 43.4 | 28.9 | 38.0 | 25.6 | 14.2 | 12.9 | |
| L4 (Right arm) | 1 | 31.7 | 36.0 | 31.0 | 34.0 | 28.0 | 5.70 | 10.7 |
| 2 | 31.5 | 36.0 | 31.0 | 34.5 | 28.0 | 4.30 | 10.7 | |
| 3 | 31.9 | 35.5 | 31.0 | 33.8 | 25.5 | 5.00 | 21.5 | |
| L5 (Left arm) | 1 | 24.5 | 31.5 | 26.0 | 27.0 | 23.7 | 16.7 | 9.7 |
| 2 | 24.4 | 31.5 | 26.0 | 27.0 | 23.2 | 16.7 | 12.1 | |
| 3 | 24.4 | 31.6 | 26.0 | 26.0 | 22.8 | 21.5 | 14.0 | |
| Subject | Day | BMI | CLM Left Limb | CLM Right Limb | % Difference | |||
|---|---|---|---|---|---|---|---|---|
| (kg/m) | Upper | Lower | Upper | Lower | Upper | Lower | ||
| H1 | 1 | 29.2 | 31.5 | 27.4 | 29.6 | 26.0 | 6.4 | 5.3 |
| 2 | 29.0 | 31.5 | 27.4 | 29.7 | 26.0 | 6.1 | 5.3 | |
| 3 | 29.1 | 31.6 | 27.5 | 29.6 | 26.0 | 6.7 | 5.7 | |
| H2 | 1 | 23.1 | 28.2 | 24.8 | 29.7 | 26.0 | 5.3 | 4.8 |
| 2 | 23.6 | 28.6 | 25.0 | 30.2 | 26.3 | 5.6 | 5.2 | |
| 3 | 23.4 | 28.0 | 25.8 | 29.4 | 27.0 | 5.0 | 4.6 | |
| H3 | 1 | 24.1 | 30.4 | 25.5 | 29.5 | 25.0 | 3.1 | 2.0 |
| 2 | 24.1 | 30.4 | 25.6 | 29.5 | 25.0 | 3.1 | 2.2 | |
| 3 | 24.2 | 30.5 | 25.2 | 29.5 | 25.0 | 3.4 | 2.0 | |
| H4 | 1 | 22.6 | 26.2 | 23.4 | 25.0 | 23.0 | 4.8 | 1.7 |
| 2 | 22.4 | 26.0 | 23.5 | 25.0 | 23.0 | 4.2 | 2.2 | |
| 3 | 22.2 | 26.1 | 23.4 | 25.0 | 23.2 | 4.4 | 1.3 | |
| Subject | BMI | Percentage Change | Difference in Gain |
|---|---|---|---|
| kg/m | in CLM | dB | |
| L1 | 31.5 | 29.6 | 8.0 |
| L2 | 30.5 | 27.5 | 6.0 |
| L3 | 29.2 | 14.5 | 1.2 |
| L4 | 31.5 | 21.5 | 9.0 |
| L5 | 24.4 | 13.0 | 5.0 |
| Healthy Participants | |||
| BMI | CLM | Difference in Gain | |
| kg/m2 | Left and Right | dB | |
| H1 | 29.0 | 5.6 | 3.0 |
| H2 | 23.6 | 3.8 | 4.0 |
| H3 | 24.1 | 2.0 | 2.0 |
| H4 | 22.4 | 4.4 | 4.0 |
| Subject | Day | BMI | Affected | Unaffected |
|---|---|---|---|---|
| kg/m | dB/min | dB/min | ||
| L1 | 2 | 31.5 | 0.26 | 0.81 |
| 3 | 31.4 | 0.16 | 1.83 | |
| L2 | 2 | 30.5 | 0.24 | 0.93 |
| 3 | 29.9 | 0.37 | 0.82 | |
| L3 | 2 | 29.2 | 0.34 | 0.60 |
| 3 | 29.2 | 0.15 | 0.44 | |
| L4 | 2 | 31.5 | 0.81 | 3.79 |
| 3 | 31.9 | 0.35 | 0.83 | |
| L5 | 2 | 24.4 | 1.93 | 1.25 |
| 3 | - | - | - |
| Subject | Day | BMI | Left Limb | Right Limb |
|---|---|---|---|---|
| kg/m | (dB/min) | (dB/min) | ||
| H1 | 2 | 29.0 | 0.52 | 0.82 |
| 3 | 29.1 | 1.22 | 1.32 | |
| H2 | 2 | 23.6 | 1.16 | 1.54 |
| 3 | 23.4 | 0.59 | 0.72 | |
| H3 | 2 | 24.1 | 0.48 | 0.69 |
| 3 | 24.2 | 0.66 | 0.87 | |
| H4 | 2 | 22.4 | 2.02 | 2.06 |
| 3 | 22.3 | 2.14 | 2.31 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asogwa, C.O.; Aryo Bay, J.; Mclaughlin, P.; Collins, S.F.; Lai, D.T.H. A Galvanic Intrabody Method for Assessing Fluid Flow in Unilateral Lymphoedema. Electronics 2017, 6, 47. https://doi.org/10.3390/electronics6020047
Asogwa CO, Aryo Bay J, Mclaughlin P, Collins SF, Lai DTH. A Galvanic Intrabody Method for Assessing Fluid Flow in Unilateral Lymphoedema. Electronics. 2017; 6(2):47. https://doi.org/10.3390/electronics6020047
Chicago/Turabian StyleAsogwa, Clement Ogugua, Johanis Aryo Bay, Patrick Mclaughlin, Stephen F. Collins, and Daniel T. H. Lai. 2017. "A Galvanic Intrabody Method for Assessing Fluid Flow in Unilateral Lymphoedema" Electronics 6, no. 2: 47. https://doi.org/10.3390/electronics6020047
APA StyleAsogwa, C. O., Aryo Bay, J., Mclaughlin, P., Collins, S. F., & Lai, D. T. H. (2017). A Galvanic Intrabody Method for Assessing Fluid Flow in Unilateral Lymphoedema. Electronics, 6(2), 47. https://doi.org/10.3390/electronics6020047






