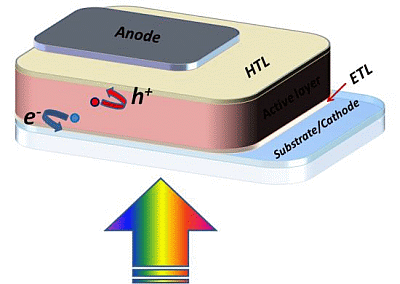
Electron and Hole Transport Layers: Their Use in Inverted Bulk Heterojunction Polymer Solar Cells
Abstract
:1. Introduction
2. The Bulk Heterojunction Solar Cell: Working Principles
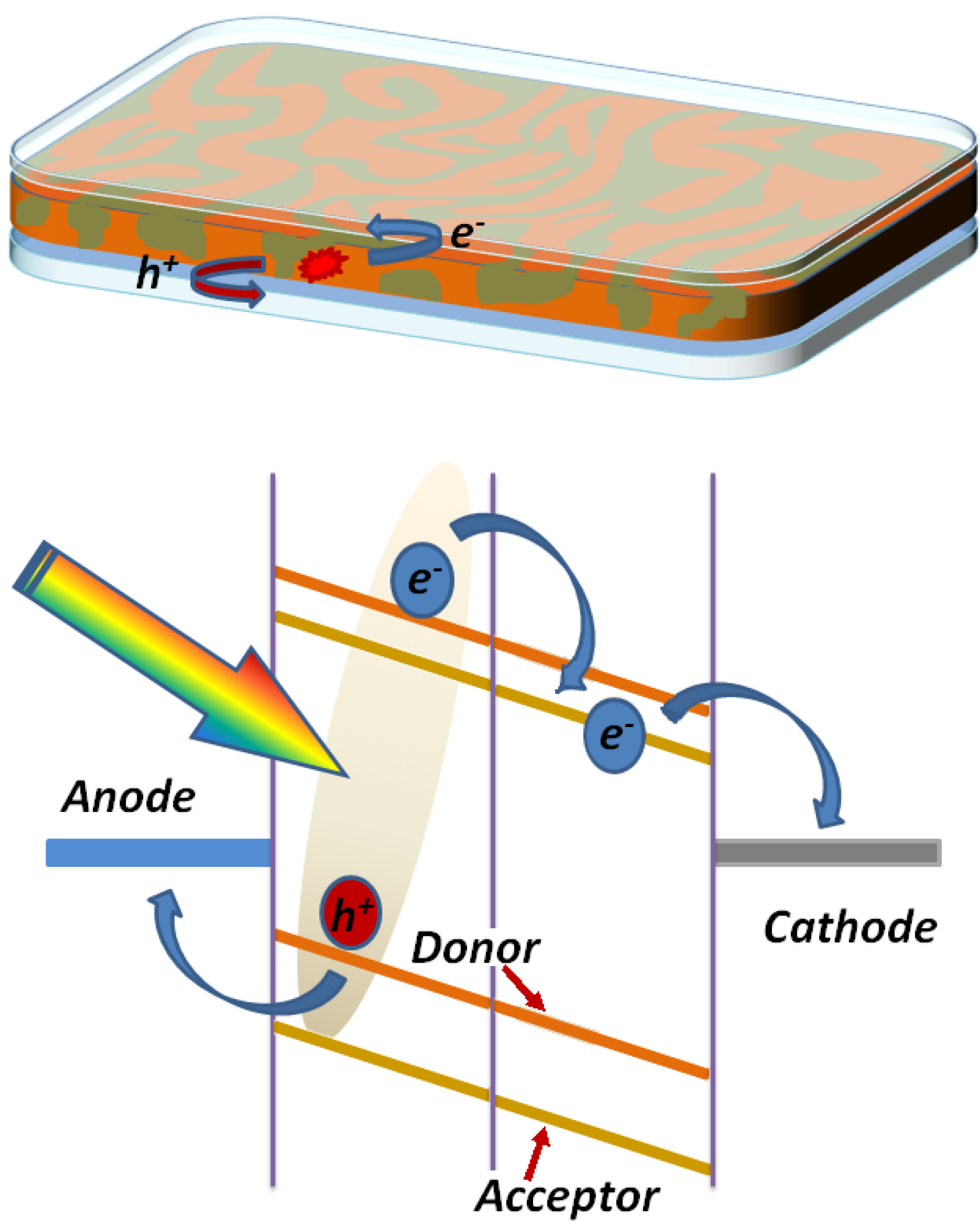
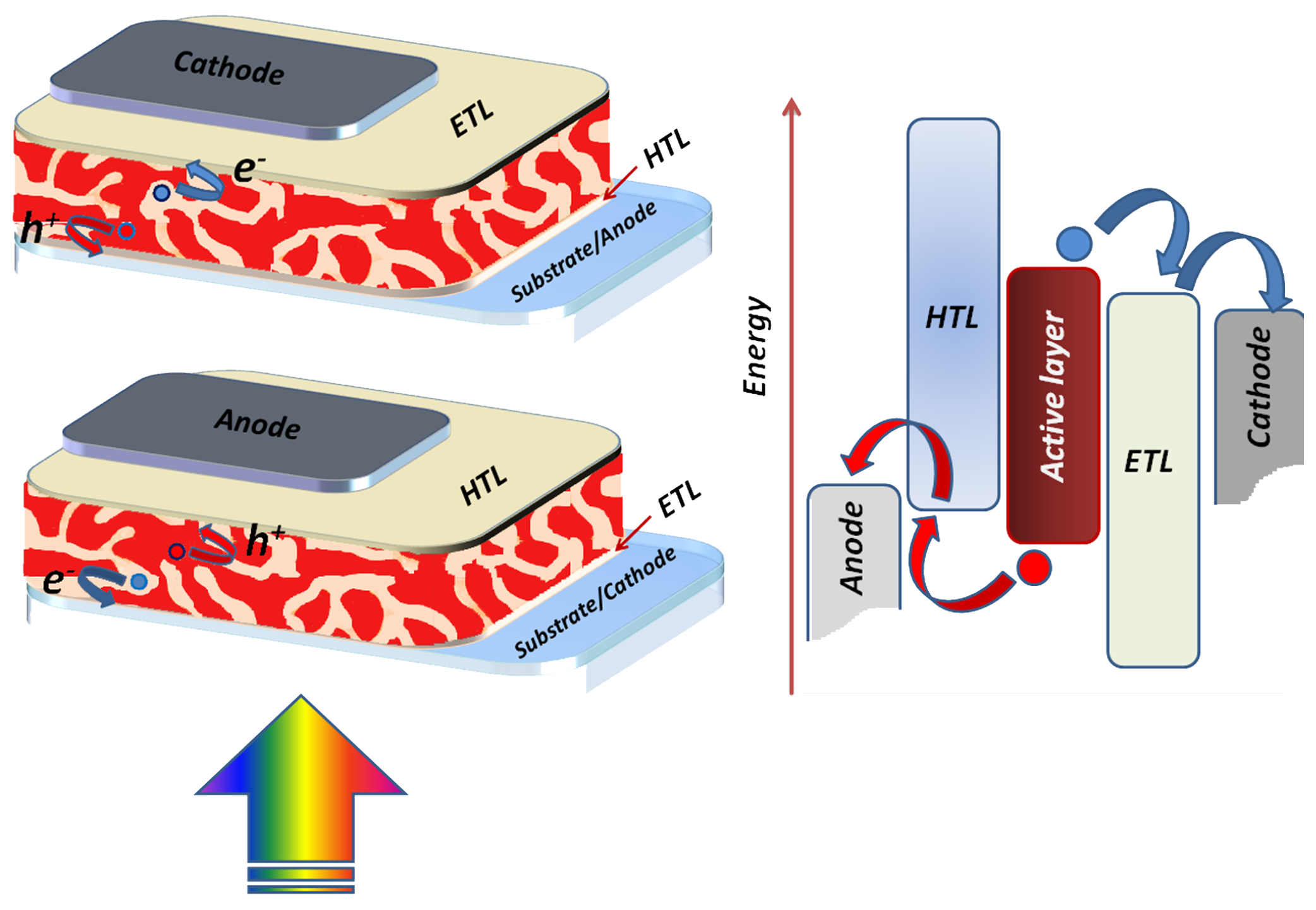
2.1. Light Absorption and Electromagnetic Field Distribution: Comparison between Standard and Inverted Structure
3. ETLs and HTLs: Recent Developments
3.1. Bottom Electrode: Electron Collection
3.1.1. ZnO

3.1.2.
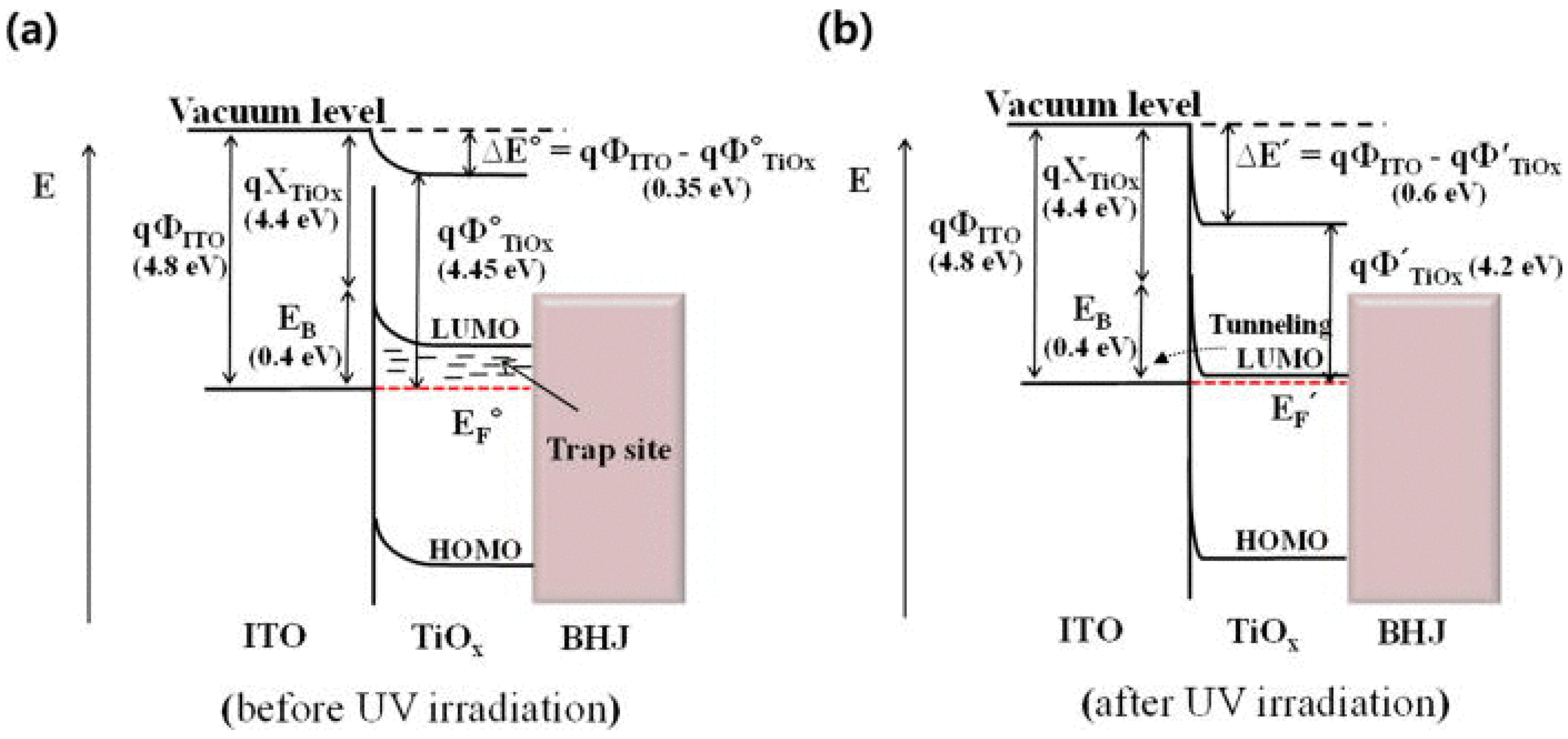
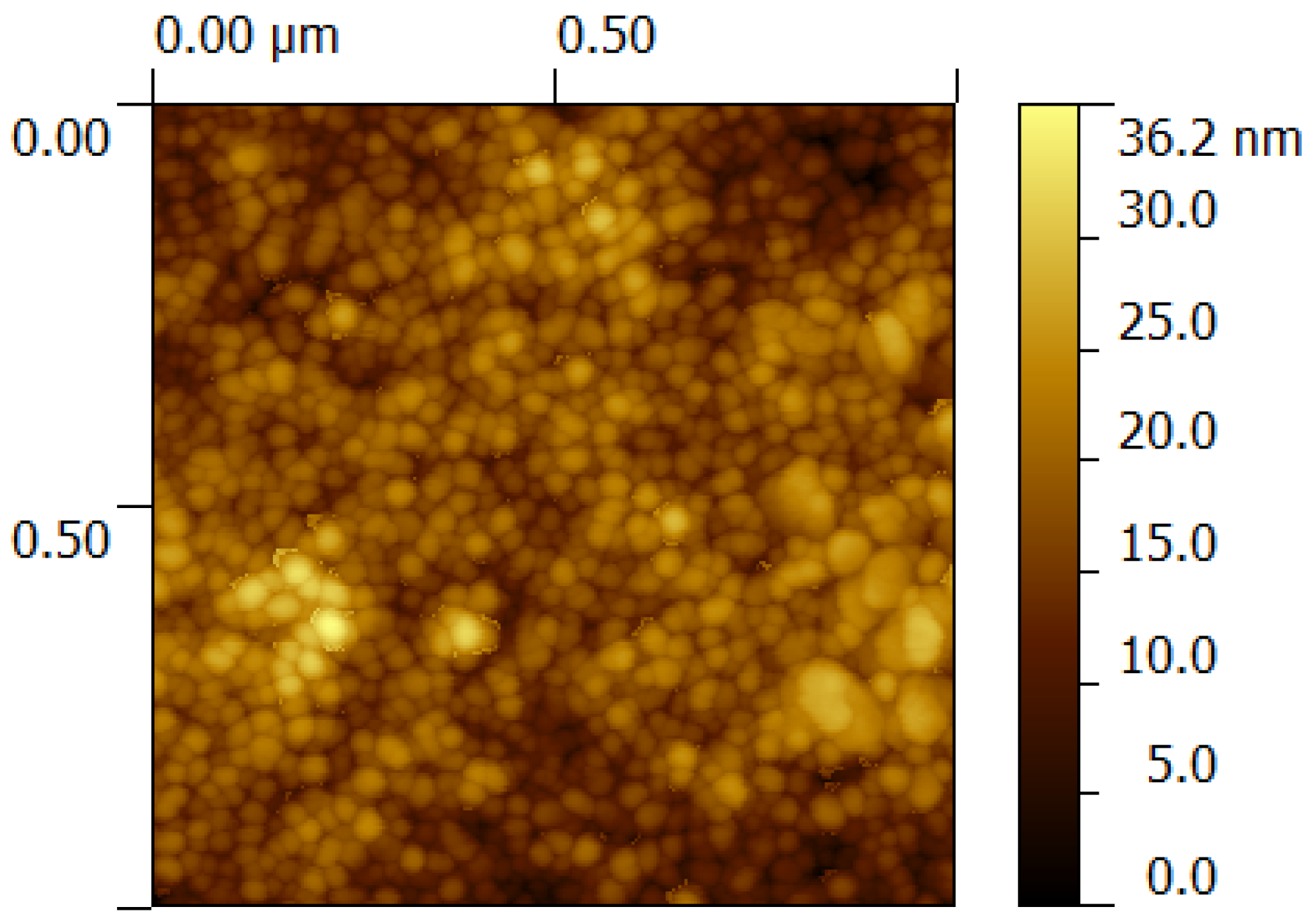
3.1.3.
3.1.4.
3.1.5. Novel Alternatives
3.1.6. ETL: Conclusions
3.2. Top Electrode: Hole Collection
3.2.1. PEDOT:PSS
3.2.2.
3.2.3.
3.2.4. and NiO
3.2.5. Novel Alternatives
3.2.6. HTLs: Conclusions
4. Device Stability: A Brief Comment
5. General Conclusions
Acknowledgments
Conflicts of Interest
References
- Shirakawa, H.; Louis, E.J.; MacDiarmid, A.G.; Chiang, C.K.; Heeger, A.J. Synthesis of electrically conducting organic polymers: Halogen derivatives of polyacetylene, (CH)x. J. Chem. Soc. Chem. Commun. 1977, 578–580. [Google Scholar] [CrossRef]
- Service, R.F. Outlook brightens for plastic solar cells. Science 2011, 332, 293. [Google Scholar] [CrossRef] [PubMed]
- Scharber, M.; Sariciftci, N.S. Efficiency of bulk-heterojunction organic solar cells. Prog. Polym. Sci. 2013, 38, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, M.; Carlé, J.E.; Sondergaard, R.R.; Lauritzen, M.; Dagnaes-Hansen, N.A.; Byskov, S.L.; Andersen, T.R.; Larsen-Olsen, T.T.; Böttiger, A.P.; Andreasen, B.; et al. The state of organic solar cells—A meta analysis. Sol. Energy Mater. Sol. Cells 2013, 119, 84–93. [Google Scholar] [CrossRef]
- Tang, C.W. Two-layer organic photovoltaic cell. Appl. Phys. Lett. 1986, 48, 183–185. [Google Scholar] [CrossRef]
- Sariciftci, N.; Smilowitz, L.; Heeger, A.; Wudl, F. Semiconducting polymers (as donors) and buckminsterfullerene (as acceptor): Photoinduced electron transfer and heterojunction devices. Synth. Met. 1993, 59, 333–352. [Google Scholar] [CrossRef]
- Sariciftci, N.S.; Braun, D.; Zhang, C.; Srdanov, V.I.; Heeger, A.J.; Stucky, G.; Wudl, F. Semiconducting polymer-buckminsterfullerene heterojunctions: Diodes, photodiodes, and photovoltaic cells. Appl. Phys. Lett. 1993, 62, 585–587. [Google Scholar] [CrossRef]
- Yu, G.; Pakbaz, K.; Heeger, A.J. Semiconducting polymer diodes: Large size, low cost photodetectors with excellent visible-ultraviolet sensitivity. Appl. Phys. Lett. 1994, 64, 3422–3424. [Google Scholar] [CrossRef]
- Hummelen, J.C.; Knight, B.W.; LePeq, F.; Wudl, F.; Yao, J.; Wilkins, C.L. Preparation and characterization of fulleroid and methanofullerene derivatives. J. Org. Chem. 1995, 60, 532–538. [Google Scholar] [CrossRef]
- Yu, G.; Gao, J.; Hummelen, J.C.; Wudl, F.; Heeger, A.J. Polymer photovoltaic cells: Enhanced efficiencies via a network of internal donor-acceptor heterojunctions. Science 1995, 270, 1789–1791. [Google Scholar] [CrossRef]
- Tremolet de Villers, B.; Tassone, C.J.; Tolbert, S.H.; Schwartz, B.J. Improving the reproducibility of P3HT:PCBM solar cells by controlling the PCBM/cathode interface. J. Phys. Chem. C 2009, 113, 18978–18982. [Google Scholar] [CrossRef]
- Sanyal, M.; Schmidt-Hansberg, B.; Klein, M.F.G.; Colsmann, A.; Munuera, C.; Vorobiev, A.; Lemmer, U.; Schabel, W.; Dosch, H.; Barrena, E. In Situ X-ray study of drying-temperature influence on the structural evolution of bulk-heterojunction polymer-fullerene solar cells processed by doctor-blading. Adv. Energy Mater. 2011, 1, 363–367. [Google Scholar] [CrossRef]
- Hoth, C.N.; Choulis, S.A.; Schilinsky, P.; Brabec, C.J. On the effect of poly(3-hexylthiophene) regioregularity on inkjet printed organic solar cells. J. Mater. Chem. 2009, 19, 5398–5404. [Google Scholar] [CrossRef]
- Nie, W.; Coffin, R.; Liu, J.; MacNeill, C.M.; Li, Y.; Noftle, R.E.; Carroll, D.L. Exploring spray-coating techniques for organic solar cell applications. Int. J. Photoenergy 2012, 2012. [Google Scholar] [CrossRef]
- Kim, Y.H.; Lee, S.H.; Noh, J.; Han, S.H. Performance and stability of electroluminescent device with self-assembled layers of poly(3,4-ethylenedioxythiophene)-poly(styrenesulfonate) and polyelectrolytes. Thin Solid Films 2006, 510, 305–310. [Google Scholar] [CrossRef]
- De Jong, M.P.; van IJzendoorn, L.J.; de Voigt, M.J.A. Stability of the interface between indium-tin-oxide and poly(3,4-ethylenedioxythiophene)/poly(styrenesulfonate) in polymer light-emitting diodes. Appl. Phys. Lett. 2000, 77, 2255–2257. [Google Scholar] [CrossRef]
- Yim, K.H.; Zheng, Z.; Friend, R.H.; Huck, W.T.S.; Kim, J.S. Surface-directed phase separation of conjugated polymer blends for efficient light-emitting diodes. Adv. Funct. Mater. 2008, 18, 2897–2904. [Google Scholar] [CrossRef]
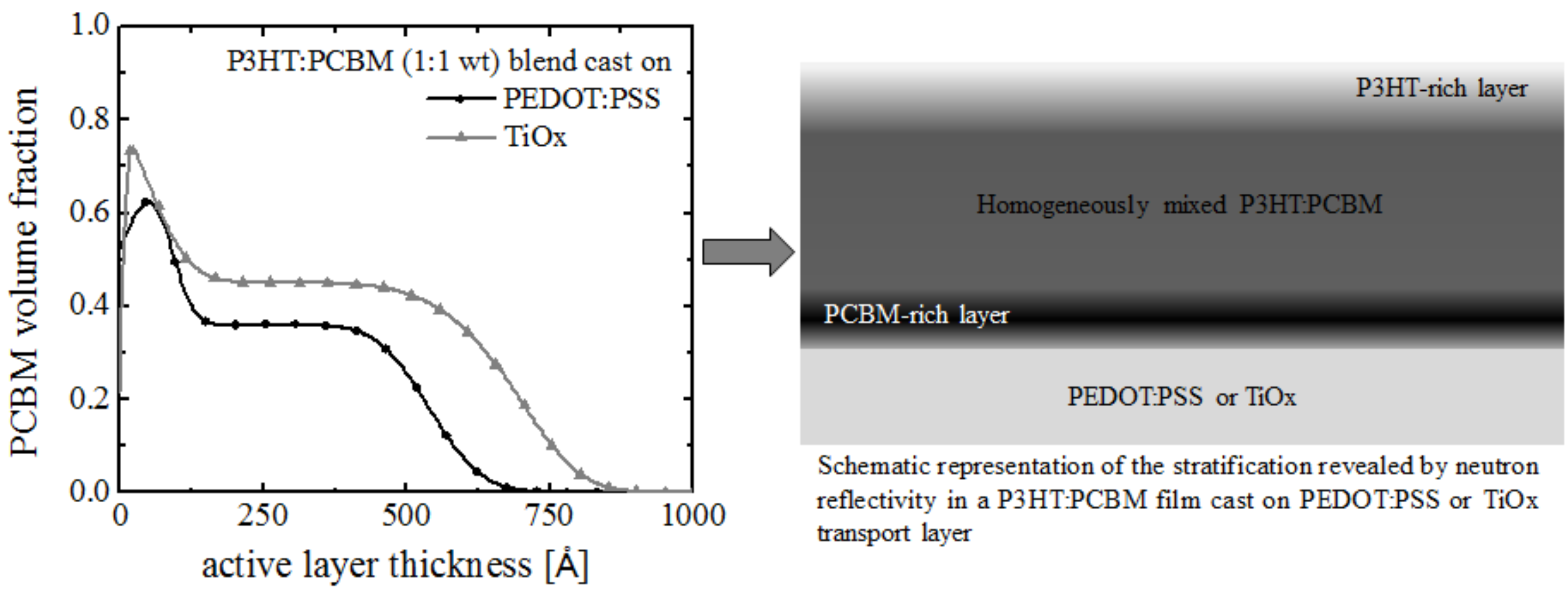
- Pavlopoulou, E.; Fleury, G.; Deribew, D.; Cousin, F.; Geoghegan, M.; Hadziioannou, G. Phase separation-driven stratification in conventional and inverted P3HT:PCBM organic solar cells. Org. Electron. 2013, 14, 1249–1254. [Google Scholar] [CrossRef]
- Baigent, D.R.; Marks, R.N.; Greenham, N.C.; Friend, R.H.; Moratti, S.C.; Holmes, A.B. Conjugated polymer light-emitting diodes on silicon substrates. Appl. Phys. Lett. 1994, 65, 2636–2638. [Google Scholar] [CrossRef]
- Arias, A.C.; Granström, M.; Thomas, D.S.; Petritsch, K.; Friend, R.H. Doped conducting-polymer–semiconducting-polymer interfaces: Their use in organic photovoltaic devices. Phys. Rev. B 1999, 60, 1854–1860. [Google Scholar] [CrossRef]
- Nyberg, T. An alternative method to build organic photodiodes. Synth. Met. 2004, 140, 281–286. [Google Scholar] [CrossRef]
- Ameri, T.; Dennler, G.; Waldauf, C.; Denk, P.; Forberich, K.; Scharber, M.C.; Brabec, C.J.; Hingerl, K. Realization, characterization, and optical modeling of inverted bulk-heterojunction organic solar cells. J. Appl. Phys. 2008, 103, 084506. [Google Scholar] [CrossRef]
- Yuan, J.; Huang, X.; Dong, H.; Lu, J.; Yang, T.; Li, Y.; Gallagher, A.; Ma, W. Structure, band gap and energy level modulations for obtaining efficient materials in inverted polymer solar cells. Org. Electron. 2013, 14, 635–643. [Google Scholar] [CrossRef]
- Hau, S.K.; Yip, H.L.; Jen, A.K.Y. A review on the development of the inverted polymer solar cell architecture. Polym. Rev. 2010, 50, 474–510. [Google Scholar] [CrossRef]
- Zhang, F.; Xu, X.; Tang, W.; Zhang, J.; Zhuo, Z.; Wang, J.; Wang, J.; Xu, Z.; Wang, Y. Recent development of the inverted configuration organic solar cells. Sol. Energy Mater. Sol. Cells 2011, 95, 1785–1799. [Google Scholar] [CrossRef]
- Deibel, C.; Dyakonov, V. Polymer-fullerene bulk heterojunction solar cells. Rep. Prog. Phys. 2010, 73, 096401. [Google Scholar] [CrossRef]
- Chen, D.; Zhang, C.; Wang, Z.; Zhang, J.; Feng, Q.; Xu, S.; Zhou, X.; Hao, Y. Performance comparison of conventional and inverted organic bulk heterojunction solar cells from optical and electrical aspects. IEEE Trans. Electron Devices 2013, 60, 451–457. [Google Scholar] [CrossRef]
- Murphy, L.; Hong, W.; Aziz, H.; Li, Y. Organic photovoltaics with thick active layers (800 nm) using a high mobility polymer donor. Sol. Energy Mater. Sol. Cells 2013, 114, 71–81. [Google Scholar] [CrossRef]
- Litzov, I.; Brabec, C.J. Development of efficient and stable inverted bulk heterojunction (BHJ) solar cells using different metal oxide interfaces. Materials 2013, 6, 5796–5820. [Google Scholar] [CrossRef]
- Li, G.; Chu, C.W.; Shrotriya, V.; Huang, J.; Yang, Y. Efficient inverted polymer solar cells. Appl. Phys. Lett. 2006, 88, 253503. [Google Scholar] [CrossRef]
- Jin Tan, M.; Zhong, S.; Wang, R.; Zhang, Z.; Vijila, C.; Chen, W. Biopolymer as an electron selective layer for inverted polymer solar cells. Appl. Phys. Lett. 2013, 103, 063303. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, K.; Lu, J.; Zhang, J.; Yip, H.L.; Huang, F.; Cao, Y. High-efficiency polymer solar cells via the incorporation of an amino-functionalized conjugated metallopolymer as a cathode interlayer. J. Am. Chem. Soc. 2013, 135, 15326–15329. [Google Scholar] [CrossRef] [PubMed]
- Kyaw, A.K.K.; Sun, X.W.; Jiang, C.Y.; Lo, G.Q.; Zhao, D.W.; Kwong, D.L. An inverted organic solar cell employing a sol-gel derived ZnO electron selective layer and thermal evaporated MoO3 hole selective layer. Appl. Phys. Lett. 2008, 93, 221107. [Google Scholar] [CrossRef]
- Kang, Y.J.; Lim, K.; Jung, S.; Kim, D.G.; Kim, J.K.; Kim, C.S.; Kim, S.H.; Kang, J.W. Spray-coated ZnO electron transport layer for air-stable inverted organic solar cells. Sol. Energy Mater. Sol. Cells 2012, 96, 137–140. [Google Scholar] [CrossRef]
- Ibrahem, M.A.; Wei, H.Y.; Tsai, M.H.; Ho, K.C.; Shyue, J.J.; Chu, C.W. Solution-processed zinc oxide nanoparticles as interlayer materials for inverted organic solar cells. Sol. Energy Mater. Sol. Cells 2013, 108, 156–163. [Google Scholar] [CrossRef]
- Mahmood, A.; Ahmed, N.; Raza, Q.; Khan, T.M.; Mehmood, M.; Hassan, M.M.; Mahmood, N. Effect of thermal annealing on the structural and optical properties of ZnO thin films deposited by the reactive e-beam evaporation technique. Phys. Scr. 2010, 82, 065801. [Google Scholar] [CrossRef]
- Park, H.Y.; Lim, D.; Kim, K.D.; Jang, S.Y. Performance optimization of low-temperature-annealed solution-processable ZnO buffer layers for inverted polymer solar cells. J. Mater. Chem. A 2013, 1, 6327–6334. [Google Scholar] [CrossRef]
- Sun, Y.; Seo, J.H.; Takacs, C.J.; Seifter, J.; Heeger, A.J. Inverted polymer solar cells integrated with a low-temperature-annealed sol-gel-derived ZnO film as an electron transport layer. Adv. Mater. 2011, 23, 1679–1683. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Li, F.; Yuan, K.; Chen, Y. Efficiency and air-stability improvement of flexible inverted polymer solar cells using ZnO/poly(ethylene glycol) hybrids as cathode buffer layers. ACS Appl. Mater. Interfaces 2013, 5, 5763–5770. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yu, X.; Hu, Z.; Zhang, J.; Zhao, G.; Zhao, Y. Effect of sol-gel derived ZnO annealing rate on light-trapping in inverted polymer solar cells. Mater. Lett. 2013, 108, 50–53. [Google Scholar] [CrossRef]
- Ohyama, M.; Kouzuka, H.; Yoko, T. Sol-gel preparation of ZnO films with extremely preferred orientation along (002) plane from zinc acetate solution. Thin Solid Films 1997, 306, 78–85. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, J.; Liu, Y.; Hao, Z.; Zhang, X.; Zhao, Y. Influence of ZnO interlayer on the performance of inverted organic photovoltaic device. Sol. Energy Mater. Sol. Cells 2011, 95, 2126–2130. [Google Scholar] [CrossRef]
- Dhibi, O.; Ltaief, A.; Zghal, S.; Bouazizi, A. Inverted low band gap polymer solar cells integrated with a low-temperature-annealed sol-gel-derived ZnO: Active layer thickness effect on the recombination process. Superlattices Microstruct. 2013, 60, 548–560. [Google Scholar] [CrossRef]
- Nickel, F.; Sprau, C.; Klein, M.F.; Kapetana, P.; Christ, N.; Liu, X.; Klinkhammer, S.; Lemmer, U.; Colsmann, A. Spatial mapping of photocurrents in organic solar cells comprising wedge-shaped absorber layers for an efficient material screening. Sol. Energy Mater. Sol. Cells 2012, 104, 18–22. [Google Scholar] [CrossRef]
- Elumalai, N.K.; Vijila, C.; Jose, R.; Zhi Ming, K.; Saha, A.; Ramakrishna, S. Simultaneous improvements in power conversion efficiency and operational stability of polymer solar cells by interfacial engineering. Phys. Chem. Chem. Phys. 2013, 15, 19057–19064. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Zhang, J.; Zhu, Y. Inverted polymer solar cells with a boron-doped zinc oxide layer deposited by metal organic chemical vapor deposition. Sol. Energy Mater. Sol. Cells 2013, 117, 610–616. [Google Scholar] [CrossRef]
- Park, B.; Shin, J.C.; Huh, Y.H. Interface-engineering additives for inverted {BHJ} polymer solar cells. Sol. Energy Mater. Sol. Cells 2013, 110, 15–23. [Google Scholar] [CrossRef]
- Liu, Y.; Larsen-Olsen, T.T.; Zhao, X.; Andreasen, B.; Sondergaard, R.R.; Helgesen, M.; Norrman, K.; Jorgensen, M.; Krebs, F.C.; Zhan, X. All polymer photovoltaics: From small inverted devices to large roll-to-roll coated and printed solar cells. Sol. Energy Mater. Sol. Cells 2013, 112, 157–162. [Google Scholar] [CrossRef]
- Alstrup, J.; Jorgensen, M.; Medford, A.J.; Krebs, F.C. Ultra Fast and parsimonious materials screening for polymer solar cells using differentially pumped slot-die coating. ACS Appl. Mater. Interfaces 2010, 2, 2819–2827. [Google Scholar] [CrossRef] [PubMed]
- Perulli, A.; Lattante, S.; Persano, A.; Cola, A.; di Giulio, M.; Anni, M. On the spatial inhomogeneity of charge generation and collection in inverted all polymer solar cells. Appl. Phys. Lett. 2013, 103, 053305. [Google Scholar] [CrossRef]
- Ajuria, J.; Etxebarria, I.; Azaceta, E.; Tena-Zaera, R.; Fernandez-Montcada, N.; Palomares, E.; Pacios, R. Novel ZnO nanostructured electrodes for higher power conversion efficiencies in polymeric solar cells. Phys. Chem. Chem. Phys. 2011, 13, 20871–20876. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Gao, R.; Lan, J.L.; Wiranwetchayan, O.; Zhang, Q.; Li, C.; Cao, G. Growth of vertically aligned ZnO nanowalls for inverted polymer solar cells. Sol. Energy Mater. Sol. Cells 2013, 117, 34–40. [Google Scholar] [CrossRef]
- Elumalai, N.K.; Jin, T.M.; Vijila, C.; Jose, R.; Palaniswamy, S.K.; Jayaraman, S.; Raut, H.K.; Ramakrishna, S. Electrospun ZnO nanowire plantations in the electron transport layer for high-efficiency inverted organic solar cells. ACS Appl. Mater. Interfaces 2013, 5, 9396–9404. [Google Scholar] [CrossRef] [PubMed]
- Bhat, J.S.; Patil, A.S.; Swami, N.; Mulimani, B.G.; Gayathri, B.R.; Deshpande, N.G.; Kim, G.H.; Seo, M.S.; Lee, Y.P. Electron irradiation effects on electrical and optical properties of sol-gel prepared ZnO films. J. Appl. Phys. 2010, 108, 043513. [Google Scholar] [CrossRef]
- Ohyama, M.; Kozuka, H.; Yoko, T. Sol-gel preparation of transparent and conductive aluminum-doped zinc oxide films with highly preferential crystal orientation. J. Am. Ceram. Soc. 1998, 81, 1622–1632. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, J.H.; Shim, T.H.; Park, J.G. Effect of interface thickness on power conversion efficiency of polymer photovoltaic cells. Electron. Mater. Lett. 2009, 5, 47–50. [Google Scholar] [CrossRef]
- Lattante, S.; Perulli, A.; Anni, M. Study of the series resistance evolution in organic solar cells by use of the Lambert W function. Synth. Met. 2011, 161, 949–952. [Google Scholar] [CrossRef]
- Chang, J.; Lin, Z.; Zhu, C.; Chi, C.; Zhang, J.; Wu, J. Solution-processed LiF-doped ZnO films for high performance low temperature field effect transistors and inverted solar cells. ACS Appl. Mater. Interfaces 2013, 5, 6687–6693. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; He, D.; Xiao, Z.; Ding, L. The double-edged function of UV light in polymer solar cells with an inverted structure. Synth. Met. 2012, 162, 2302–2306. [Google Scholar] [CrossRef]
- Girolamo, V.B.M.; Antonucci, V.; Aricó, A.S. Influence of TiO2 film thickness on the electrochemical behaviour of dye-sensitized solar cells. Int. J. Electrochem. Sci. 2011, 6, 3375. [Google Scholar]
- Sakohara, S.; Ishida, M.; Anderson, M.A. Visible luminescence and surface properties of nanosized ZnO colloids prepared by hydrolyzing Zinc acetate. J. Phys. Chem. B 1998, 102, 10169–10175. [Google Scholar] [CrossRef]
- Monticone, S.; Tufeu, R.; Kanaev, A.V. Complex nature of the UV and visible fluorescence of colloidal ZnO nanoparticles. J. Phys. Chem. B 1998, 102, 2854–2862. [Google Scholar] [CrossRef]
- Peng, R.; Yang, F.; Ouyang, X.; Liu, Y.; Kim, Y.S.; Ge, Z. Enhanced photovoltaic performance of inverted polymer solar cells by tuning the structures of titanium dioxide. Thin Solid Films 2013, 545, 424–428. [Google Scholar] [CrossRef]
- Kuwabara, T.; Sugiyama, H.; Yamaguchi, T.; Takahashi, K. Inverted type bulk-heterojunction organic solar cell using electrodeposited titanium oxide thin films as electron collector electrode. Thin Solid Films 2009, 517, 3766–3769. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, S.H.; Lee, H.H.; Lee, K.; Ma, W.; Gong, X.; Heeger, A.J. New architecture for high-efficiency polymer photovoltaic cells using solution-based titanium oxide as an optical spacer. Adv. Mater. 2006, 18, 572–576. [Google Scholar] [CrossRef]
- Hadipour, A.; Maller, R.; Heremans, P. Room temperature solution-processed electron transport layer for organic solar cells. Org. Electron. 2013, 14, 2379–2386. [Google Scholar] [CrossRef]
- Liang, W.W.; Chang, C.Y.; Lai, Y.Y.; Cheng, S.W.; Chang, H.H.; Lai, Y.Y.; Cheng, Y.J.; Wang, C.L.; Hsu, C.S. Formation of nanostructured fullerene interlayer through accelerated self-assembly and cross-linking of trichlorosilane moieties leading to enhanced efficiency of photovoltaic cells. Macromolecules 2013, 46, 4781–4789. [Google Scholar] [CrossRef]
- Savva, A.; Neophytou, M.; Koutsides, C.; Kalli, K.; Choulis, S.A. Synergistic effects of buffer layer processing additives for enhanced hole carrier selectivity in inverted organic photovoltaics. Org. Electron. 2013, 14, 3123–3130. [Google Scholar] [CrossRef]
- Lu, H.; Akgun, B.; Russell, T.P. Morphological characterization of a low-bandgap crystalline polymer:PCBM bulk heterojunction solar cells. Adv. Energy Mater. 2011, 1, 870–878. [Google Scholar] [CrossRef]
- Waldauf, C.; Scharber, M.C.; Schilinsky, P.; Hauch, J.A.; Brabec, C.J. Physics of organic bulk heterojunction devices for photovoltaic applications. J. Appl. Phys. 2006, 99, 104503. [Google Scholar] [CrossRef]
- Steim, R.; Choulis, S.A.; Schilinsky, P.; Brabec, C.J. Interface modification for highly efficient organic photovoltaics. Appl. Phys. Lett. 2008, 92, 093303. [Google Scholar] [CrossRef]
- Lilliedal, M.R.; Medford, A.J.; Madsen, M.V.; Norrman, K.; Krebs, F.C. The effect of post-processing treatments on inflection points in current–voltage curves of roll-to-roll processed polymer photovoltaics. Sol. Energy Mater. Sol. Cells 2010, 94, 2018–2031. [Google Scholar] [CrossRef]
- Kim, J.; Kim, G.; Choi, Y.; Lee, J.; Heum Park, S.; Lee, K. Light-soaking issue in polymer solar cells: Photoinduced energy level alignment at the sol-gel processed metal oxide and indium tin oxide interface. J. Appl. Phys. 2012, 111, 114511. [Google Scholar] [CrossRef]
- Choi, H.; Lee, J.; Lee, W.; Ko, S.J.; Yang, R.; Lee, J.C.; Woo, H.Y.; Yang, C.; Kim, J.Y. Acid-functionalized fullerenes used as interfacial layer materials in inverted polymer solar cells. Org. Electron. 2013, 14, 3138–3145. [Google Scholar] [CrossRef]
- Goh, C.; Scully, S.R.; McGehee, M.D. Effects of molecular interface modification in hybrid organic-inorganic photovoltaic cells. J. Appl. Phys. 2007, 101, 114503. [Google Scholar] [CrossRef]
- Hsieh, C.H.; Cheng, Y.J.; Li, P.J.; Chen, C.H.; Dubosc, M.; Liang, R.M.; Hsu, C.S. Highly efficient and stable inverted polymer solar cells integrated with a cross-linked fullerene material as an interlayer. J. Am. Chem. Soc. 2010, 132, 4887–4893. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Wu, C.E.; Chen, S.Y.; Cui, C.; Cheng, Y.J.; Hsu, C.S.; Wang, Y.L.; Li, Y. Enhanced performance and stability of a polymer solar cell by incorporation of vertically aligned, cross-linked fullerene nanorods. Angew. Chem. Int. Ed. 2011, 50, 9386–9390. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.J.; Hsieh, C.H.; He, Y.; Hsu, C.S.; Li, Y. Combination of indene-C60 bis-adduct and cross-linked fullerene interlayer leading to highly efficient inverted polymer solar cells. J. Am. Chem. Soc. 2010, 132, 17381–17383. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.J.; Cao, F.Y.; Lin, W.C.; Chen, C.H.; Hsieh, C.H. Self-assembled and cross-linked fullerene interlayer on titanium oxide for highly efficient inverted polymer solar cells. Chem. Mater. 2011, 23, 1512–1518. [Google Scholar] [CrossRef]
- Hasegawa, T.; Miura, S.; Moriyama, T.; Kimura, T.; Takaya, I.; Osato, Y.; Mizutani, H. Novel Electron-Injection Layers for Top-Emission OLEDs. Dig. Tech. Papers—Soc. Inf. Disp. Int. Symp. 2004, 35, 154. [Google Scholar] [CrossRef]
- Liao, H.H.; Chen, L.M.; Xu, Z.; Li, G.; Yang, Y. Highly efficient inverted polymer solar cell by low temperature annealing of Cs2CO3 interlayer. Appl. Phys. Lett. 2008, 92, 173303. [Google Scholar] [CrossRef]
- Xin, Y.; Wang, Z.; Xu, L.; Xu, X.; Liu, Y.; Zhang, F. UV-Ozone treatment on Cs2CO3 interfacial layer for the improvement of inverted polymer solar cells. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- Barbot, A.; Lucas, B.; di Bin, C.; Ratier, B.; Aldissi, M. Optimized inverted polymer solar cells incorporating Cs2CO3-doped C60 as electron transport layer. Appl. Phys. Lett. 2013, 102, 193305. [Google Scholar] [CrossRef]
- Cheng, G.; Tong, W.Y.; Low, K.H.; Che, C.M. Thermal-annealing-free inverted polymer solar cells using ZnO/Cs2CO3 bilayer as electron-selective layer. Sol. Energy Mater. Sol. Cells 2012, 103, 164–170. [Google Scholar] [CrossRef]
- Barbot, A.; Bin, C.; Lucas, B.; Ratier, B.; Aldissi, M. N-type doping and thermoelectric properties of co-sublimed cesium-carbonate-doped fullerene. J. Mater. Sci. 2013, 48, 2785–2789. [Google Scholar] [CrossRef]
- Yang, H.B.; Dong, Y.Q.; Wang, X.; Khoo, S.Y.; Liu, B.; Li, C.M. Graphene quantum dots-incorporated cathode buffer for improvement of inverted polymer solar cells. Sol. Energy Mater. Sol. Cells 2013, 117, 214–218. [Google Scholar] [CrossRef]
- Wiranwetchayan, O.; Liang, Z.; Zhang, Q.; Cao, G.; Singjai, P. The role of oxide thin layer in inverted structure polymer solar cells. Mater. Sci. Appl. 2011, 2, 1697–1701. [Google Scholar] [CrossRef]
- Lenzmann, F.; Krueger, J.; Burnside, S.; Brooks, K.; Grätzel, M.; Gal, D.; Rühle, S.; Cahen, D. Surface photovoltage spectroscopy of dye-sensitized solar cells with TiO2, Nb2O5, and SrTiO3 nanocrystalline photoanodes: Indication for electron injection from higher excited dye states. J. Phys. Chem. B 2001, 105, 6347–6352. [Google Scholar] [CrossRef]
- Li, Z.; Dong, Q.; Li, Y.; Xu, B.; Deng, M.; Pei, J.; Zhang, J.; Chen, F.; Wen, S.; Gao, Y.; Tian, W. Design and synthesis of solution processable small molecules towards high photovoltaic performance. J. Mater. Chem. 2011, 21, 2159–2168. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, W.; Li, X.; Min, C.; Jiu, T.; Zhu, Y.; Dai, N.; Fang, J. Solution-processed hybrid cathode interlayer for inverted organic solar cells. ACS Appl. Mater. Interfaces 2013, 5, 10428–10432. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.H.; Jun, G.H.; Hong, S.H.; Jeon, S. Conformal coating of titanium suboxide on carbon nanotube networks by atomic layer deposition for inverted organic photovoltaic cells. Carbon 2012, 50, 4483–4488. [Google Scholar] [CrossRef]
- Kohlstadt, M.; Grein, M.; Reinecke, P.; Kroyer, T.; Zimmermann, B.; Würfel, U. Inverted ITO- and PEDOT:PSS-free polymer solar cells with high power conversion efficiency. Sol. Energy Mater. Sol. Cells 2013, 117, 98–102. [Google Scholar] [CrossRef]
- Oo, T.Z.; Chandra, R.D.; Yantara, N.; Prabhakar, R.R.; Wong, L.H.; Mathews, N.; Mhaisalkar, S.G. Zinc Tin Oxide (ZTO) electron transporting buffer layer in inverted organic solar cell. Org. Electron. 2012, 13, 870–874. [Google Scholar] [CrossRef]
- Kim, J.B.; Kim, C.S.; Kim, Y.S.; Loo, Y.L. Oxidation of silver electrodes induces transition from conventional to inverted photovoltaic characteristics in polymer solar cells. Appl. Phys. Lett. 2009, 95, 183301. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, Y.; Cheng, G.; Li, J.; Liu, C.; Zhao, Z.; Jie, Z.; Liu, S. Improved light outcoupling for phosphorescent top-emitting organic light-emitting devices. Appl. Phys. Lett. 2006, 88, 153517. [Google Scholar] [CrossRef]
- Tao, C.; Ruan, S.; Zhang, X.; Xie, G.; Shen, L.; Kong, X.; Dong, W.; Liu, C.; Chen, W. Performance improvement of inverted polymer solar cells with different top electrodes by introducing a MoO3 buffer layer. Appl. Phys. Lett. 2008, 93, 193307. [Google Scholar] [CrossRef]
- Tao, C.; Ruan, S.; Xie, G.; Kong, X.; Shen, L.; Meng, F.; Liu, C.; Zhang, X.; Dong, W.; Chen, W. Role of tungsten oxide in inverted polymer solar cells. Appl. Phys. Lett. 2009, 94, 043311. [Google Scholar] [CrossRef]
- Servaites, J.D.; Ratner, M.A.; Marks, T.J. Organic solar cells: A new look at traditional models. Energy Environ. Sci. 2011, 4, 4410–4422. [Google Scholar] [CrossRef]
- Carter, S.A.; Angelopoulos, M.; Karg, S.; Brock, P.J.; Scott, J.C. Polymeric anodes for improved polymer light-emitting diode performance. Appl. Phys. Lett. 1997, 70, 2067–2069. [Google Scholar] [CrossRef]
- Kemerink, M.; Timpanaro, S.; de Kok, M.M.; Meulenkamp, E.A.; Touwslager, F.J. Three-dimensional inhomogeneities in PEDOT:PSS films. J. Phys. Chem. B 2004, 108, 18820–18825. [Google Scholar] [CrossRef]
- Ionescu-Zanetti, C.; Mechler, A.; Carter, S.; Lal, R. Semiconductive polymer blends: Correlating structure with transport properties at the nanoscale. Adv. Mater. 2004, 16, 385–389. [Google Scholar] [CrossRef]
- Stubhan, T.; Oh, H.; Pinna, L.; Krantz, J.; Litzov, I.; Brabec, C.J. Inverted organic solar cells using a solution processed aluminum-doped zinc oxide buffer layer. Org. Electron. 2011, 12, 1539–1543. [Google Scholar] [CrossRef]
- Lipomi, D.J.; Tee, B.C.K.; Vosgueritchian, M.; Bao, Z. Stretchable organic solar cells. Adv. Mater. 2011, 23, 1771–1775. [Google Scholar] [CrossRef] [PubMed]
- Baek, W.H.; Choi, M.; Yoon, T.S.; Lee, H.H.; Kim, Y.S. Use of fluorine-doped tin oxide instead of indium tin oxide in highly efficient air-fabricated inverted polymer solar cells. Appl. Phys. Lett. 2010, 96, 133506. [Google Scholar] [CrossRef]
- Heo, S.W.; Baek, K.H.; Lee, T.H.; Lee, J.Y.; Moon, D.K. Enhanced performance in inverted polymer solar cells via solution process: Morphology controlling of PEDOT:PSS as anode buffer layer by adding surfactants. Org. Electron. 2013, 14, 1629–1635. [Google Scholar] [CrossRef]
- Lim, F.J.; Ananthanarayanan, K.; Luther, J.; Ho, G.W. Influence of a novel fluorosurfactant modified PEDOT:PSS hole transport layer on the performance of inverted organic solar cells. J. Mater. Chem. 2012, 22, 25057–25064. [Google Scholar] [CrossRef]
- Gupta, D.; Wienk, M.M.; Janssen, R.A.J. Efficient polymer solar cells on opaque substrates with a laminated PEDOT:PSS top electrode. Adv. Energy Mater. 2013, 3, 782–787. [Google Scholar] [CrossRef]
- Suh, Y.; Lu, N.; Lee, S.H.; Chung, W.S.; Kim, K.; Kim, B.; Ko, M.J.; Kim, M.J. Degradation of a thin Ag layer induced by poly(3,4-ethylenedioxythiophene):polystyrene sulfonate in a transmission electron microscopy specimen of an inverted polymer solar cell. ACS Appl. Mater. Interfaces 2012, 4, 5118–5124. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.; Hamwi, S.; Bülow, T.; Johannes, H.H.; Riedl, T.; Kowalsky, W. Highly efficient simplified organic light emitting diodes. Appl. Phys. Lett. 2007, 91, 113506. [Google Scholar] [CrossRef]
- Wang, F.; Qiao, X.; Xiong, T.; Ma, D. The role of molybdenum oxide as anode interfacial modification in the improvement of efficiency and stability in organic light-emitting diodes. Org. Electron. 2008, 9, 985–993. [Google Scholar] [CrossRef]
- Tokito, S.; Noda, K.; Taga, Y. Metal oxides as a hole-injecting layer for an organic electroluminescent device. J. Phys. D 1996, 29, 2750–2753. [Google Scholar] [CrossRef]
- Yin, Z.; Zheng, Q.; Chen, S.C.; Cai, D. Interface control of semiconducting metal oxide layers for efficient and stable inverted polymer solar cells with open-circuit voltages over 1.0 volt. ACS Appl. Mater. Interfaces 2013, 5, 9015–9025. [Google Scholar] [CrossRef] [PubMed]
- Chambon, S.; Derue, L.; Lahaye, M.; Pavageau, B.; Hirsch, L.; Wantz, G. MoO3 thickness, thermal annealing and solvent annealing effects on inverted and direct polymer photovoltaic solar cells. Materials 2012, 5, 2521–2536. [Google Scholar] [CrossRef]
- Ng, A.; Liu, X.; To, C.H.; Djurišió, A.B.; Zapien, J.A.; Chan, W.K. Annealing of P3HT:PCBM blend film—The effect on its optical properties. ACS Appl. Mater. Interfaces 2013, 5, 4247–4259. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.R.; Oliver, M.; Krebs, F.C.; Dauskardt, R.H. Interlayer adhesion in roll-to-roll processed flexible inverted polymer solar cells. Sol. Energy Mater. Sol. Cells 2012, 97, 171–175. [Google Scholar] [CrossRef]
- Kundu, S.; Gollu, S.R.; Sharma, R.; G, S.; Ashok, A.; Kulkarni, A.; Gupta, D. Device stability of inverted and conventional bulk heterojunction solar cells with MoO3 and ZnO nanoparticles as charge transport layers. Org. Electron. 2013, 14, 3083–3088. [Google Scholar] [CrossRef]
- Elumalai, N.K.; Saha, A.; Vijila, C.; Jose, R.; Jie, Z.; Ramakrishna, S. Enhancing the stability of polymer solar cells by improving the conductivity of the nanostructured MoO3 hole-transport layer. Phys. Chem. Chem. Phys. 2013, 15, 6831–6841. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Zuo, L.; Fu, W.; Fan, C.; Andreasen, B.; Jiang, X.; Norrman, K.; Krebs, F.C.; Chen, H. MoO3–Au composite interfacial layer for high efficiency and air-stable organic solar cells. Org. Electron. 2013, 14, 797–803. [Google Scholar] [CrossRef]
- Lampande, R.; Kim, G.W.; Boizot, J.; Kim, Y.J.; Pode, R.; Kwon, J.H. A highly efficient transition metal oxide layer for hole extraction and transport in inverted polymer bulk heterojunction solar cells. J. Mater. Chem. A 2013, 1, 6895–6900. [Google Scholar] [CrossRef]
- Stubhan, T.; Li, N.; Luechinger, N.A.; Halim, S.C.; Matt, G.J.; Brabec, C.J. High fill factor polymer solar cells incorporating a low temperature solution processed WO3 hole extraction layer. Adv. Energy Mater. 2012, 2, 1433–1438. [Google Scholar] [CrossRef]
- Zilberberg, K.; Trost, S.; Meyer, J.; Kahn, A.; Behrendt, A.; Lützenkirchen-Hecht, D.; Frahm, R.; Riedl, T. Inverted organic solar cells with sol-gel processed high work-function vanadium oxide hole-extraction layers. Adv. Funct. Mater. 2011, 21, 4776–4783. [Google Scholar] [CrossRef]
- Yu, W.; Shen, L.; Ruan, S.; Meng, F.; Wang, J.; Zhang, E.; Chen, W. Performance improvement of inverted polymer solar cells thermally evaporating nickel oxide as an anode buffer layer. Sol. Energy Mater. Sol. Cells 2012, 98, 212–215. [Google Scholar] [CrossRef]
- Lim, D.C.; Kim, Y.T.; Shim, W.H.; Jang, A.Y.; Lim, J.H.; Kim, Y.D.; Jeong, Y.; Kim, Y.D.; Lee, K.H. Wet-chemically prepared NiO layers as hole transport layer in the inverted organic solar cell. Bull. Korean Chem. Soc. 2011, 32, 1067–1070. [Google Scholar] [CrossRef]
- Huang, J.S.; Chou, C.Y.; Lin, C.F. Efficient and air-stable polymer photovoltaic devices with WO3-V2O5 mixed oxides as anodic modification. IEEE Electron Device Lett. 2010, 31, 332–334. [Google Scholar] [CrossRef]
- Shelton, S.W.; Chen, T.L.; Barclay, D.E.; Ma, B. Solution-processable triindoles as hole selective materials in organic solar cells. ACS Appl. Mater. Interfaces 2012, 4, 2534–2540. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xue, Y.; Gao, Y.; Yu, D.; Durstock, M.; Dai, L. Hole and electron extraction layers based on graphene oxide derivatives for high-performance bulk heterojunction solar cells. Adv. Mater. 2012, 24, 2228–2233. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.J.; Tung, V.C.; Kaner, R.B. Honeycomb carbon: A review of graphene. Chem. Rev. 2010, 110, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Yao, K.; Chen, L.; Chen, X.; Chen, Y. Self-organized hole transport layers based on polythiophene diblock copolymers for inverted organic solar cells with high efficiency. Chem. Mater. 2013, 25, 897–904. [Google Scholar] [CrossRef]
- Greiner1, M.T.; Lu, Z.H. Thin-film metal oxides in organic semiconductor devices: Their electronic structures, work functions and interfaces. NPG Asia Mater. 2013, 5, e55. [Google Scholar] [CrossRef]
- Xie, F.; Choy, W.C.H. Hydrogen metal oxide bronzes for efficient hole transport layers. SPIE Newsroom 2013. [Google Scholar] [CrossRef]
- Stubhan, T.; Ameri, T.; Salinas, M.; Krantz, J.; Machui, F.; Halik, M.; Brabec, C.J. High shunt resistance in polymer solar cells comprising a MoO3 hole extraction layer processed from nanoparticle suspension. Appl. Phys. Lett. 2011, 98, 253308. [Google Scholar] [CrossRef]
- Choi, H.; Kim, B.; Ko, M.J.; Lee, D.K.; Kim, H.; Kim, S.H.; Kim, K. Solution processed {WO3} layer for the replacement of PEDOT:PSS layer in organic photovoltaic cells. Org. Electron. 2012, 13, 959–968. [Google Scholar] [CrossRef]
- Li, N.; Stubhan, T.; Luechinger, N.A.; Halim, S.C.; Matt, G.J.; Ameri, T.; Brabec, C.J. Inverted structure organic photovoltaic devices employing a low temperature solution processed {WO3} anode buffer layer. Org. Electron. 2012, 13, 2479–2484. [Google Scholar] [CrossRef]
- Li, N.; Stubhan, T.; Baran, D.; Min, J.; Wang, H.; Ameri, T.; Brabec, C.J. Design of the solution-processed intermediate layer by engineering for inverted organic multi junction solar cells. Adv. Energy Mater. 2013, 3, 301–307. [Google Scholar] [CrossRef]
- Yip, H.L.; Jen, A.K.Y. Recent advances in solution-processed interfacial materials for efficient and stable polymer solar cells. Energy Environ. Sci. 2012, 5, 5994–6011. [Google Scholar] [CrossRef]
- Angmo, D.; Gonzalez-Valls, I.; Veenstra, S.; Verhees, W.; Sapkota, S.; Schiefer, S.; Zimmermann, B.; Galagan, Y.; Sweelssen, J.; Lira-Cantu, M.; et al. Low-cost upscaling compatibility of five different ITO-free architectures for polymer solar cells. J. Appl. Polym. Sci. 2013, 130, 944–954. [Google Scholar] [CrossRef]
- Voroshazi, E.; Cardinaletti, I.; Uytterhoeven, G.; Shan, L.; Empl, M.; Aernouts, T.; Rand, B.P. Role of electron- and hole-collecting buffer layers on the stability of inverted polymer: Fullerene photovoltaic devices. IEEE J. Photovolt. 2014, 4, 265–270. [Google Scholar] [CrossRef]
- Lizin, S.; van Passel, S.; de Schepper, E.; Maes, W.; Lutsen, L.; Manca, J.; Vanderzande, D. Life cycle analyses of organic photovoltaics: A review. Energy Environ. Sci. 2013, 6, 3136–3149. [Google Scholar] [CrossRef]
- Espinosa, N.; Hosel, M.; Angmo, D.; Krebs, F.C. Solar cells with one-day energy payback for the factories of the future. Energy Environ. Sci. 2012, 5, 5117–5132. [Google Scholar] [CrossRef]
- Espinosa, N.; Garcia-Valverde, R.; Krebs, F.C. Life-cycle analysis of product integrated polymer solar cells. Energy Environ. Sci. 2011, 4, 1547–1557. [Google Scholar] [CrossRef]
© 2014 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lattante, S. Electron and Hole Transport Layers: Their Use in Inverted Bulk Heterojunction Polymer Solar Cells. Electronics 2014, 3, 132-164. https://doi.org/10.3390/electronics3010132
Lattante S. Electron and Hole Transport Layers: Their Use in Inverted Bulk Heterojunction Polymer Solar Cells. Electronics. 2014; 3(1):132-164. https://doi.org/10.3390/electronics3010132
Chicago/Turabian StyleLattante, Sandro. 2014. "Electron and Hole Transport Layers: Their Use in Inverted Bulk Heterojunction Polymer Solar Cells" Electronics 3, no. 1: 132-164. https://doi.org/10.3390/electronics3010132
APA StyleLattante, S. (2014). Electron and Hole Transport Layers: Their Use in Inverted Bulk Heterojunction Polymer Solar Cells. Electronics, 3(1), 132-164. https://doi.org/10.3390/electronics3010132