Evaluation of Wet Digestion Methods for Quantification of Metal Content in Electronic Scrap Material
Abstract
:1. Introduction
2. Materials and Methods
2.1. Equipment
2.2. Digestion Reagents and Electronic Scrap
2.3. Characterization of ESM
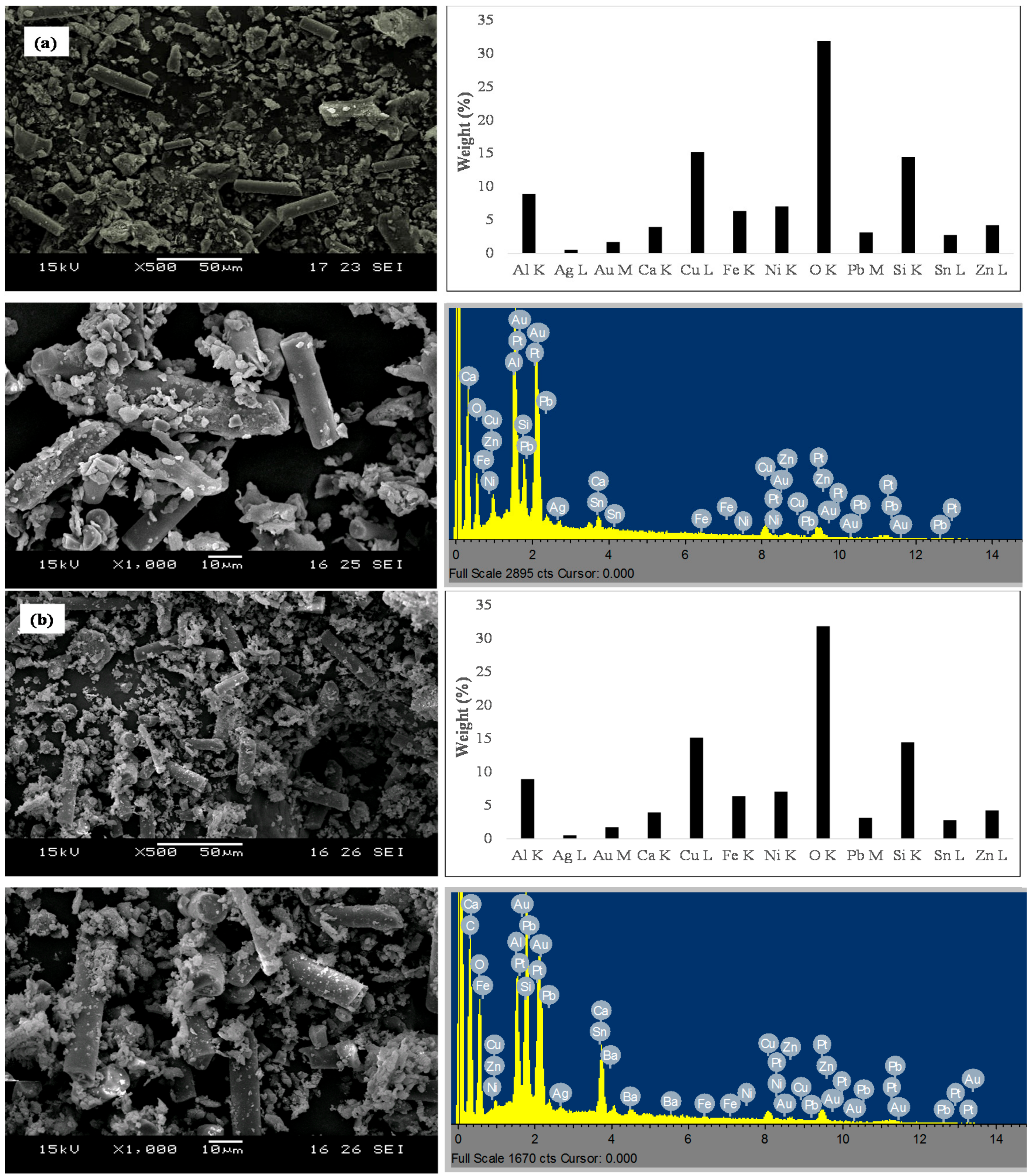
2.3.1. Scanning Electron Microscopy (SEM) and Energy-Dispersive X-ray Spectroscopy (EDX)
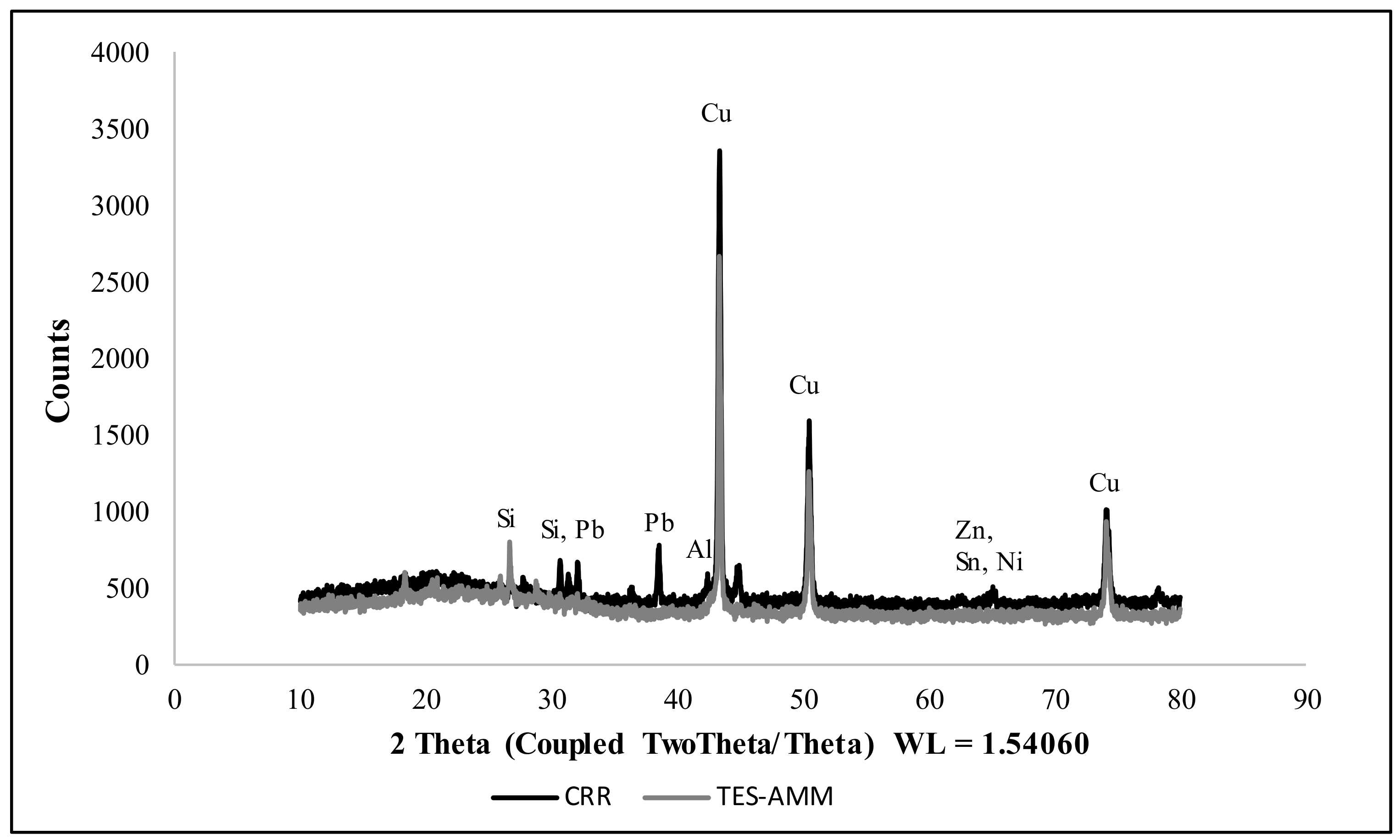
2.3.2. X-ray Diffraction (XRD)
2.4. Digestion Protocols
2.4.1. US EPA Solid Waste 846 Method 3050 B
2.4.2. ASTM D6357-11
2.4.3. Digestion Reactions in Microwave Digesters
- Microwave-Assisted Digestion Method 1 (MWD-1)
- Microwave-Assisted Digestion Method 2 (MWD-2)
- Microwave-Assisted Digestion Method 3 (MWD-3)
2.4.4. Digestion Reactions in an Ultrasonic System
- Ultrasound-Assisted Digestion Method 1 (UD-1)
- Ultrasound-Assisted Digestion Method 2 (UD-2)
- Ultrasound-Assisted Digestion Method 3 (UD-3)
- Ultrasound-Assisted Digestion Method 4 (UD-4)
2.4.5. Sequential Extraction Method (SExM)
2.4.6. Digestion Using Different Acids
2.5. Digestate Analysis
2.6. Loss on Ignition
2.7. Statistical Analysis
3. Results
3.1. ESM Characterization
3.1.1. Morphology of ESM
3.1.2. X-ray Diffraction Analysis (XRD) of ESM
3.2. Comparison of Elemental Metal Compositions of ESM after Different Acid Digestion Protocols
3.3. Efficacy of Different Acids for Metal Dissolution from ESM
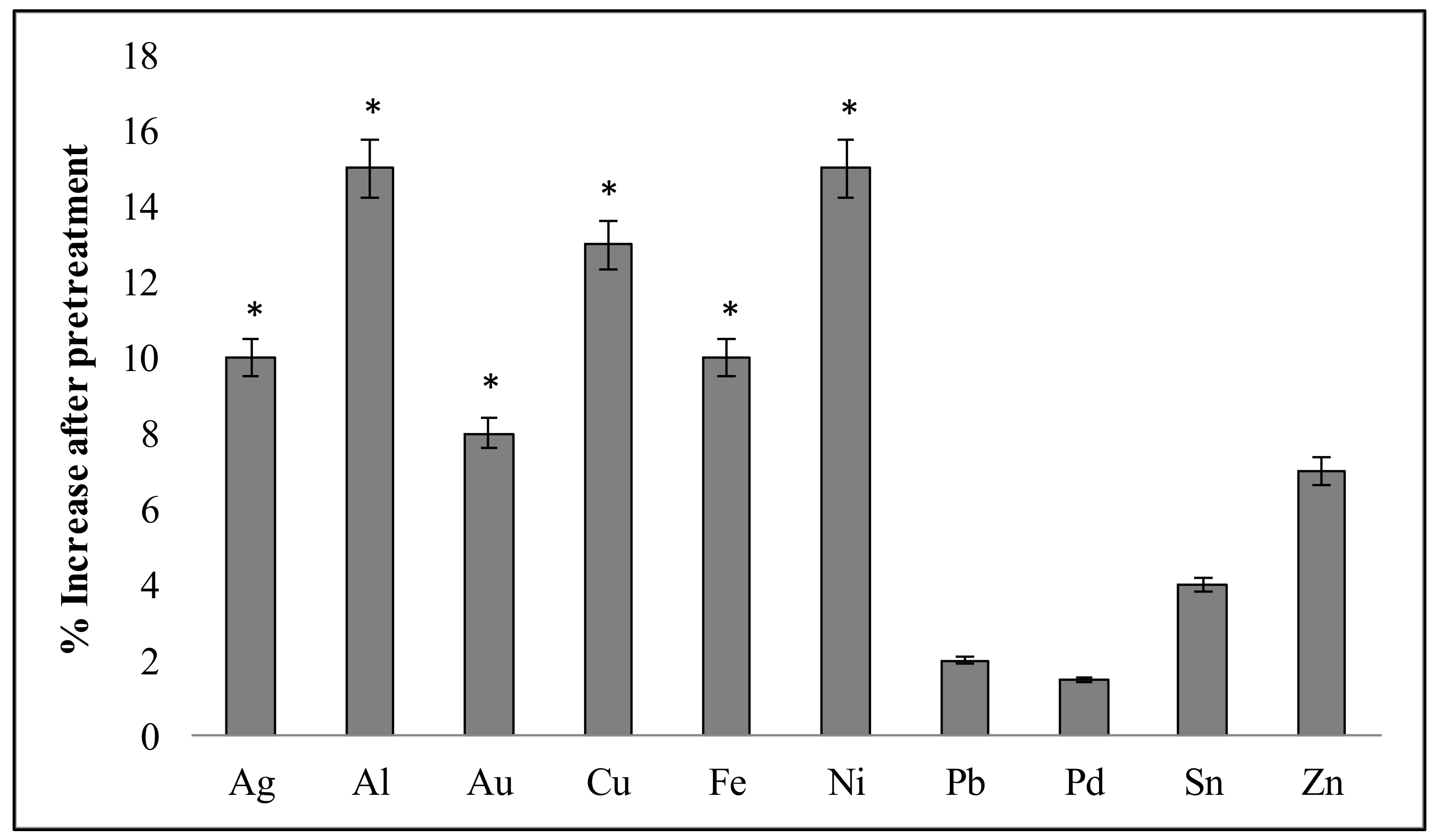
3.4. Improved Metal Solubilization through Pre-Treatment of ESM
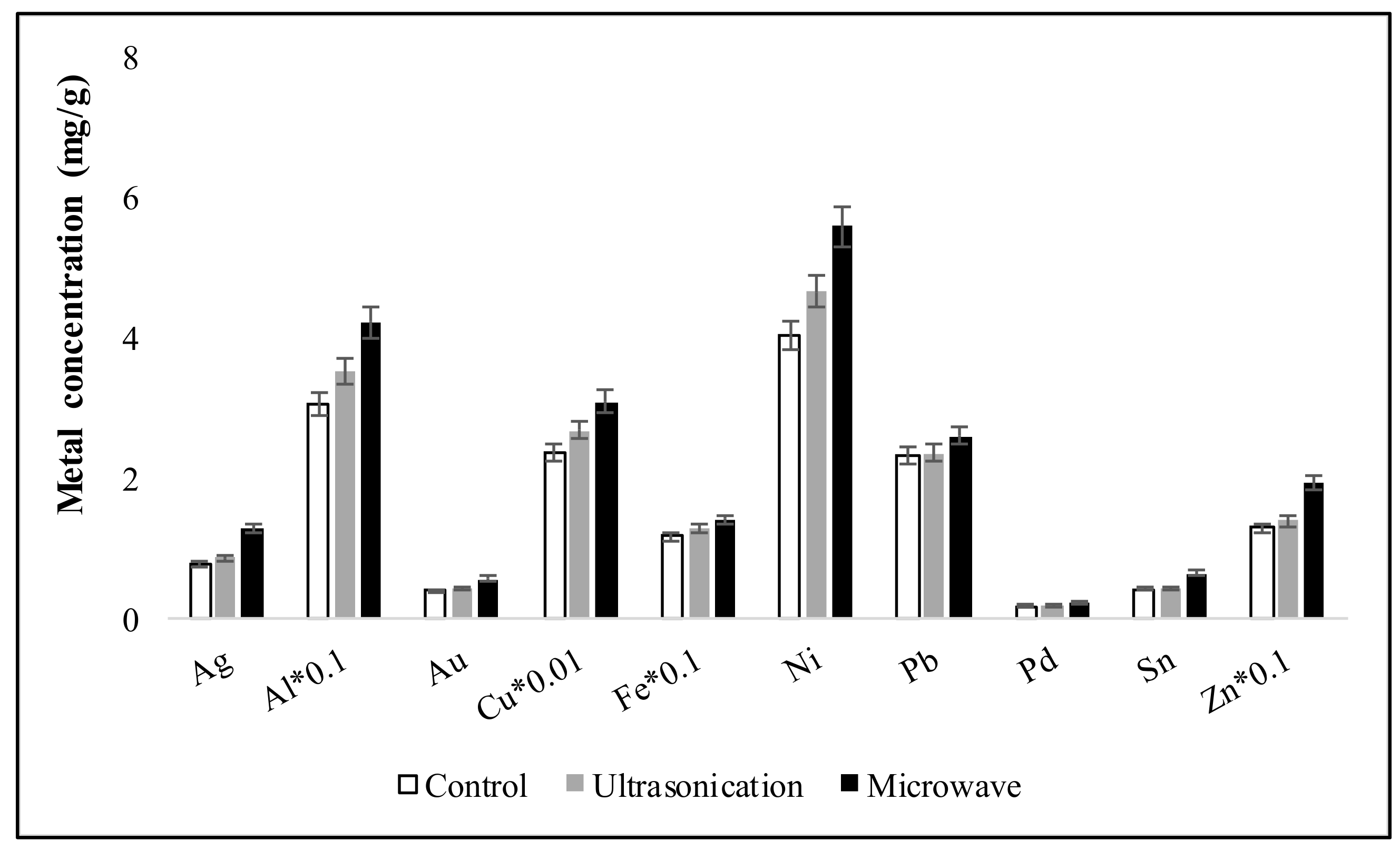
3.5. Effect of Different Driving Forces on Metal Recovery from ESM
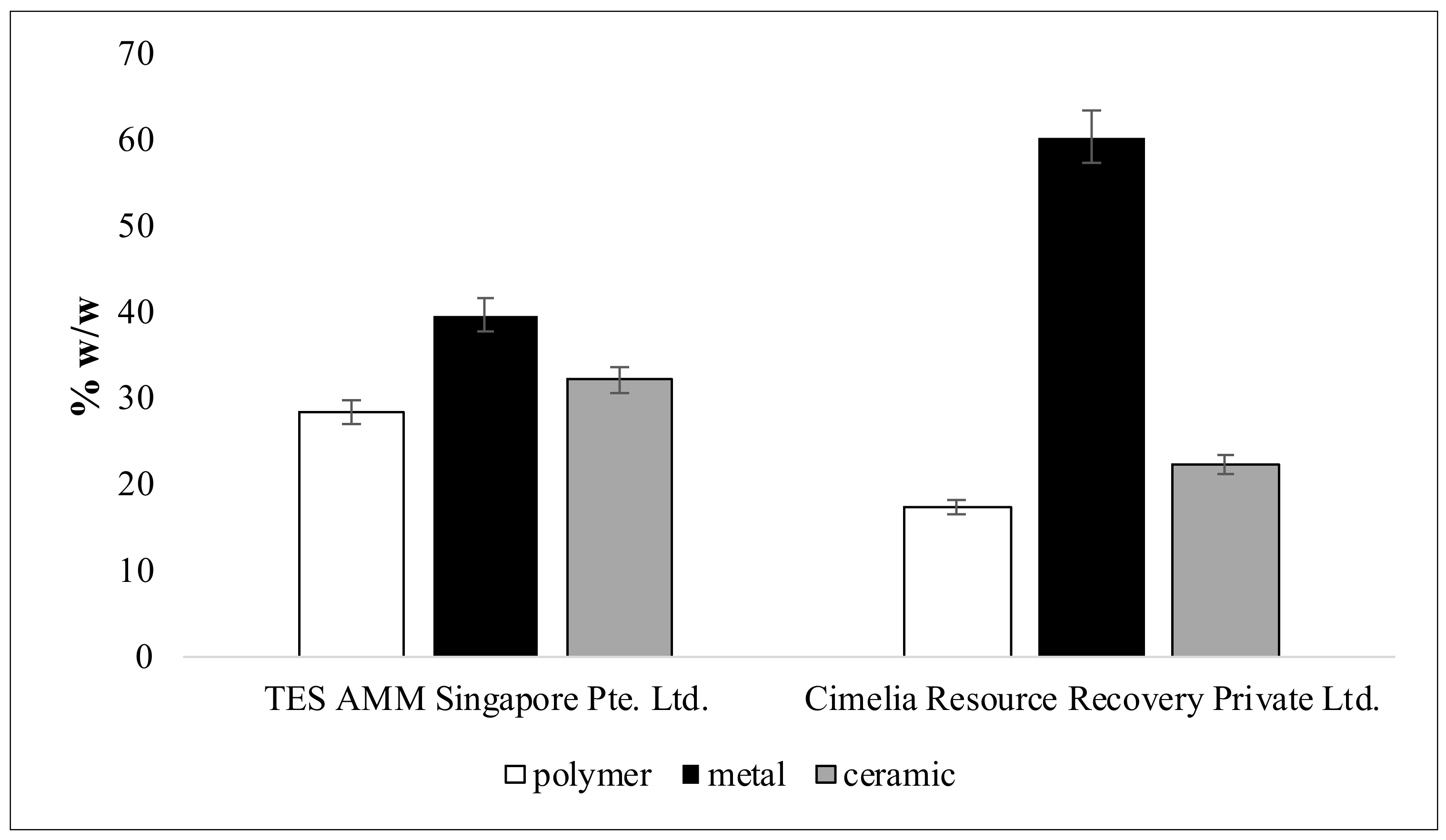
3.6. Fractional Composition of ESM
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Natarajan, G.; Ting, Y.-P. Pretreatment of e-waste and mutation of alkali-tolerant cyanogenic bacteria promote gold biorecovery. Bioresour. Technol. 2014, 152, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, T.; Ting, Y. Fly ash and the use of bioleaching for fly ash detoxification. In Fly Ash: Chemical Composition, Sources and Potential Environmental Impacts; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2014. [Google Scholar]
- Sihvonen, S.; Partanen, J. Eco-design practices with a focus on quantitative environmental targets: An exploratory content analysis within ict sector. J. Clean. Prod. 2017, 143, 769–783. [Google Scholar] [CrossRef]
- Hoornweg, D.; Bhada-Tata, P. What a Waste: A global Review of Solid Waste Management; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2012. [Google Scholar]
- Zeng, X.; Yang, C.; Chiang, J.F.; Li, J. Innovating e-waste management: From macroscopic to microscopic scales. Sci. Total Environ. 2017, 575, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Ting, Y.P. Improving gold (bio) leaching efficiency through pretreatment using hydrogen peroxide assisted sulfuric acid. CLEAN Soil Air Water 2017, 45, 1500945. [Google Scholar] [CrossRef]
- Dewhurst, R.; Wheeler, J.; Chummun, K.; Mather, J.; Callaghan, A.; Crane, M. The comparison of rapid bioassays for the assessment of urban groundwater quality. Chemosphere 2002, 47, 547–554. [Google Scholar] [CrossRef]
- Balcerzak, M. Sample digestion methods for the determination of traces of precious metals by spectrometric techniques. Anal. Sci. 2002, 18, 737–750. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Tan, Q.; Chiang, J.F.; Li, J. Recovery of rare and precious metals from urban mines—A review. Front. Environ. Sci. Eng. 2017, 11, 1. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, L. Metallurgical recovery of metals from electronic waste: A review. J. Hazard. Mater. 2008, 158, 228–256. [Google Scholar] [CrossRef] [PubMed]
- Gramatyka, P.; Nowosielski, R.; Sakiewicz, P. Recycling of waste electrical and electronic equipment. J. Achiev. Mater. Manuf. Eng. 2007, 20, 535–538. [Google Scholar]
- Adie, G.U.; Sun, L.; Zeng, X.; Zheng, L.; Osibanjo, O.; Li, J. Examining the evolution of metals utilized in printed circuit boards. Environ. Technol. 2017, 38, 1696–1701. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Ding, Y.; Liu, B.; Chang, C. Supply and demand of some critical metals and present status of their recycling in weee. Waste Manag. 2017, 65, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Babu, B.R.; Parande, A.K.; Basha, C.A. Electrical and electronic waste: A global environmental problem. Waste Manag. Res. 2007, 25, 307–318. [Google Scholar]
- Kumar, A.; Holuszko, M. Electronic waste and existing processing routes: A canadian perspective. Resources 2016, 5, 35. [Google Scholar] [CrossRef]
- Song, X.; Wang, J.; Yang, J.; Lu, B. An updated review and conceptual model for optimizing weee management in china from a life cycle perspective. Front. Environ. Sci. Eng. 2017, 11, 3. [Google Scholar] [CrossRef]
- Legarth, J.B.; Alting, L.; Danzer, B.; Tartler, D.; Brodersen, K.; Scheller, H.; Feldmann, K. A new strategy in the recycling of printed circuit boards. Circuit World 1995, 21, 10–15. [Google Scholar] [CrossRef]
- Ilyas, S.; Anwar, M.A.; Niazi, S.B.; Ghauri, M.A. Bioleaching of metals from electronic scrap by moderately thermophilic acidophilic bacteria. Hydrometallurgy 2007, 88, 180–188. [Google Scholar] [CrossRef]
- Yamane, L.H.; de Moraes, V.T.; Espinosa, D.C.R.; Tenório, J.A.S. Recycling of weee: Characterization of spent printed circuit boards from mobile phones and computers. Waste Manag. 2011, 31, 2553–2558. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Fray, D.J. Recovery of high purity precious metals from printed circuit boards. J. Hazard. Mater. 2009, 164, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
- Szałatkiewicz, J. Metals content in printed circuit board waste. Pol. J. Environ. Stud. 2014, 23, 2365–2369. [Google Scholar]
- Khaliq, A.; Rhamdhani, M.A.; Brooks, G.; Masood, S. Metal extraction processes for electronic waste and existing industrial routes: A review and australian perspective. Resources 2014, 3, 152–179. [Google Scholar] [CrossRef]
- Charles, R.G.; Douglas, P.; Hallin, I.L.; Matthews, I.; Liversage, G. An investigation of trends in precious metal and copper content of ram modules in weee: Implications for long term recycling potential. Waste Manag. 2017, 60, 505–520. [Google Scholar] [CrossRef] [PubMed]
- Corbu, O.; Popovici, A.; Popita, G.; Rusu, T.; Rosu, C.; Puskas, A. Ecological composite with glass from waste electrical and electronic equipment content. In Proceedings of the 14th SGEM GeoConference on Nano, Bio And Green–Technologies For A Sustainable Future, Albena, Bulgaria, 17–26 June 2014; pp. 81–88. [Google Scholar]
- Zeng, X.; Li, J. Spent rechargeable lithium batteries in e-waste: Composition and its implications. Front. Environ. Sci. Eng. 2014, 8, 792–796. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Chang, C.Y.; Chiang, H.L.; Su, Z.J.; Wang, C.F. A sequential extraction method measures the toxic metal content in fly ash from a municipal solid waste incinerator. J. Chin. Chem. Soc. 2005, 52, 921–926. [Google Scholar] [CrossRef]
- Van Herck, P.; Vandecasteele, C. Evaluation of the use of a sequential extraction procedure for the characterization and treatment of metal containing solid waste. Waste Manag. 2001, 21, 685–694. [Google Scholar] [CrossRef]
- Ramanathan, T.; Ting, Y.-P. Selection of wet digestion methods for metal quantification in hazardous solid wastes. J. Environ. Chem. Eng. 2015, 3, 1459–1467. [Google Scholar] [CrossRef]
- Atiemo, S.M.; Ofosu, F.G.; Aboh, I.K.; Kuranchie-Mensah, H. Assessing the heavy metals contamination of surface dust from waste electrical and electronic equipment (e-waste) recycling site in accra, ghana. Res. J. Environ. Earth Sci. 2012, 4, 605–611. [Google Scholar]
- Hseu, Z.-Y. Evaluating heavy metal contents in nine composts using four digestion methods. Bioresour. Technol. 2004, 95, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Güven, D.; Akinci, G. Comparison of acid digestion techniques to determine heavy metals in sediment and soil samples. Gazi Univ. J. Sci. 2011, 24, 29–34. [Google Scholar]
- Edgell, K. Usepa Method Study 37 sw-846 Method 3050 Acid Digestion of Sediments, Sludges, and Soils; US Environmental Protection Agency, Environmental Monitoring Systems Laboratory: Washington, DC, USA, 1989. [Google Scholar]
- Sukandar, S.; Yasuda, K.; Tanaka, M.; Aoyama, I. Metals leachability from medical waste incinerator fly ash: A case study on particle size comparison. Environ. Pollut. 2006, 144, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Element, C. Method 3051a Microwave Assisted Acid Digestion of Sediments, Sludges, Soils, and Oils. 2007. Available online: https://www.epa.gov/hw-sw846/sw-846-test-method-3051a-microwave-assisted-acid-digestion-sediments-sludges-soils-and-oils (accessed on 15 January 2017).
- Kingston, H.; Walter, P. Microwave Assisted Acid Digestion of Siliceous and Organically Based Matrices. EPA Draft Method 1995, 3052. Available online: https://www.epa.gov/hw-sw846/sw-846-test-method-3052-microwave-assisted-acid-digestion-siliceous-and-organically-based (accessed on 15 January 2017).
- Ha, N.N.; Agusa, T.; Ramu, K.; Tu, N.P.C.; Murata, S.; Bulbule, K.A.; Parthasaraty, P.; Takahashi, S.; Subramanian, A.; Tanabe, S. Contamination by trace elements at e-waste recycling sites in Bangalore, India. Chemosphere 2009, 76, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.S.; Wu, S.; Duzgoren-Aydin, N.S.; Aydin, A.; Wong, M.H. Trace metal contamination of sediments in an e-waste processing village in china. Environ. Pollut. 2007, 145, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Robinson, B.H. E-waste: An assessment of global production and environmental impacts. Sci. Total Environ. 2009, 408, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Yafa, C.; Farmer, J.G. A comparative study of acid-extractable and total digestion methods for the determination of inorganic elements in peat material by inductively coupled plasma-optical emission spectrometry. Anal. Chim. Acta 2006, 557, 296–303. [Google Scholar] [CrossRef]
- Zhou, P.; Zheng, Z.; Tie, J. Technological Process for Extracting Gold, Silver and Palladium from Electronic Industry Waste. Chinese Patent CN1603432A, 6 April 2005. [Google Scholar]
- Wang, F.; Huisman, J.; Meskers, C.E.; Schluep, M.; Stevels, A.; Hagelüken, C. The best-of-2-worlds philosophy: Developing local dismantling and global infrastructure network for sustainable e-waste treatment in emerging economies. Waste Manag. 2012, 32, 2134–2146. [Google Scholar] [CrossRef] [PubMed]


| Sample | ESM from TES-AMM | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Method | Concentration (mg/g) | |||||||||
| Ag | Al | Au | Cu | Fe | Ni | Pb | Pd | Sn | Zn | |
| ASTM method D 6257-11 | 0.89 ± 0.01 | 33.03 ± 1.22 | 0.52 ± 0.01 | 282.41 ± 1.11 | 14.45 ± 1.42 | 5.34 ± 0.12 | 3.89 ± 0.11 | 0.27 ± 0.01 | 0.47 ± 0.02 | 16.46 ± 1.13 |
| US EPA SW 846 Method 3050B | 0.78 ± 0.03 | 30.53 ± 1.53 | 0.41 ± 0.01 | 237.13 ± 3.42 | 11.68 ± 3.12 | 4.04 ± 0.08 | 2.31 ± 0.21 | 0.18 ± 0.03 | 0.41 ± 0.04 | 12.93 ± 2.31 |
| MWD-1 | 0.54 ± 0.01 | 23.81 ± 0.91 | 0.31 ± 0.03 | 221.42 ± 2.44 | 10.11 ± 1.45 | 3.28 ± 0.31 | 2.84 ± 0.08 | 0.21 ± 0.02 | 0.32 ± 0.03 | 9.34 ± 1.12 |
| MWD-2 | 0.81 ± 0.04 | 39.11 ± 0.74 | 0.49 ± 0.02 | 247.35 ± 3.61 | 12.56 ± 3.11 | 4.43 ± 0.23 | 5.32 ± 0.18 | 0.24 ± 0.04 | 0.67 ± 0.01 | 14.57 ± 1.45 |
| MWD-3 | 0.73 ± 0.02 | 23.45 ± 1.32 | 0.36 ± 0.01 | 251.67 ± 3.15 | 11.32 ± 1.45 | 4.74 ± 0.34 | 2.46 ± 0.13 | 0.23 ± 0.05 | 0.53 ± 0.03 | 11.54 ± 1.23 |
| UD-1 | 0.67 ± 0.01 | 19.89 ± 0.65 | 0.38 ± 0.02 | 256.41 ± 1.52 | 10.11 ± 2.12 | 4.37 ± 0.31 | 4.11 ± 0.19 | 0.16 ± 0.02 | 0.55 ± 0.04 | 13.45 ± 2.34 |
| UD-2 | 0.57 ± 0.01 | 10.95 ± 0.21 | 0.37 ± 0.02 | 247.38 ± 5.46 | 12.56 ± 1.22 | 4.42 ± 0.41 | 4.02 ± 0.14 | 0.25 ± 0.04 | 0.51 ± 0.06 | 11.79 ± 1.87 |
| UD-3 | 0.73 ± 0.04 | 19.59 ± 1.15 | 0.38 ± 0.04 | 251.44 ± 3.82 | 10.62 ± 2.42 | 4.25 ± 0.12 | 3.89 ± 0.07 | 0.15 ± 0.01 | 0.51 ± 0.07 | 11.84 ± 2.09 |
| UD-4 | 0.61 ± 0.05 | 13.52 ± 1.46 | 0.36 ± 0.03 | 210.78 ± 2.61 | 9.56 ± 1.78 | 3.66 ± 0.37 | 4.95 ± 0.11 | 0.13 ± 0.02 | 0.53 ± 0.03 | 10.78 ± 2.34 |
| Sample | ESM from CRR | |||||||||
| Method | Concentration (mg/g) | |||||||||
| Ag | Al | Au | Cu | Fe | Ni | Pb | Pd | Sn | Zn | |
| ASTM method D 6257-11 | 1.18 ± 0.05 | 53.23 ± 1.42 | 0.18 ± 0.01 | 449.24 ± 5.32 | 31.45 ± 1.31 | 10.56 ± 0.11 | 6.45 ± 0.07 | 0.12 ± 0.01 | 1.68 ± 0.02 | 22.45 ± 1.23 |
| US EPA SW 846 Method 3050B | 1.08 ± 0.01 | 61.49 ± 2.45 | 0.18 ± 0.02 | 416.63 ± 7.82 | 28.61 ± 2.12 | 8.45 ± 1.22 | 6.34 ± 0.28 | 0.11 ± 0.05 | 1.56 ± 0.04 | 20.13 ± 2.56 |
| MWD-1 | 0.83 ± 0.07 | 42.33 ± 2.34 | 0.16 ± 0.04 | 405.35 ± 8.12 | 23.33 ± 1.13 | 9.33 ± 1.09 | 5.87 ± 0.12 | 0.10 ± 0.01 | 1.43 ± 0.01 | 18.56 ± 1.78 |
| MWD-2 | 0.96 ± 0.04 | 64.22 ± 3.12 | 0.17 ± 0.01 | 428.23 ± 4.98 | 30.45 ± 1.42 | 10.12 ± 1.13 | 7.12 ± 0.54 | 0.11 ± 0.01 | 1.96 ± 0.05 | 21.44 ± 3.09 |
| MWD-3 | 0.92 ± 0.05 | 58.78 ± 1.87 | 0.16 ± 0.03 | 407.36 ± 5.28 | 27.56 ± 3.12 | 9.67 ± 0.88 | 6.88 ± 0.15 | 0.11 ± 0.03 | 1.87 ± 0.04 | 17.56 ± 2.67 |
| UD-1 | 0.88 ± 0.07 | 50.34 ± 2.45 | 0.16 ± 0.02 | 362.53 ± 4.12 | 24.34 ± 1.15 | 8.22 ± 0.76 | 6.56 ± 0.65 | 0.09 ± 0.02 | 1.56 ± 0.03 | 16.45 ± 4.12 |
| UD-2 | 0.76 ± 0.05 | 48.56 ± 3.24 | 0.17 ± 0.02 | 387.88 ± 2.08 | 22.17 ± 0.87 | 7.85 ± 0.56 | 6.05 ± 0.34 | 0.10 ± 0.02 | 1.78 ± 0.09 | 19.78 ± 2.67 |
| UD-3 | 0.79 ± 0.03 | 49.34 ± 2.68 | 0.18 ± 0.01 | 378.65 ± 5.65 | 23.56 ± 0.98 | 8.08 ± 0.53 | 6.48 ± 0.23 | 0.11 ± 0.03 | 1.62 ± 0.06 | 21.65 ± 4.87 |
| UD-4 | 0.82 ± 0.06 | 42.46 ± 3.45 | 0.16 ± 0.02 | 364.78 ± 2.44 | 20.98 ± 1.11 | 7.45 ± 1.32 | 6.22 ± 0.12 | 0.08 ± 0.03 | 1.61 ± 0.07 | 18.12 ± 2.44 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, S.; Ting, Y.-P. Evaluation of Wet Digestion Methods for Quantification of Metal Content in Electronic Scrap Material. Resources 2017, 6, 64. https://doi.org/10.3390/resources6040064
Das S, Ting Y-P. Evaluation of Wet Digestion Methods for Quantification of Metal Content in Electronic Scrap Material. Resources. 2017; 6(4):64. https://doi.org/10.3390/resources6040064
Chicago/Turabian StyleDas, Subhabrata, and Yen-Peng Ting. 2017. "Evaluation of Wet Digestion Methods for Quantification of Metal Content in Electronic Scrap Material" Resources 6, no. 4: 64. https://doi.org/10.3390/resources6040064
APA StyleDas, S., & Ting, Y.-P. (2017). Evaluation of Wet Digestion Methods for Quantification of Metal Content in Electronic Scrap Material. Resources, 6(4), 64. https://doi.org/10.3390/resources6040064





