Scaling of Metabolic Scaling within Physical Limits
Abstract
:1. Introduction
2. Overview of the Metabolic-Level Boundaries Hypothesis (MLBH)
2.1. Conceptual Foundation of the MLBH
2.1.1. Body Volume and Surface Area as Physical Boundary Limits
2.1.2. Covariation of the Elevation (L) and Slope (b) of Metabolic Scaling Relationships
2.1.3. Internal and External Influences on L and b
2.2. Predictive Power of the MLBH
3. Evidence Appearing to Support or Contradict the MLBH
3.1. Effects of Physiological or Developmental State
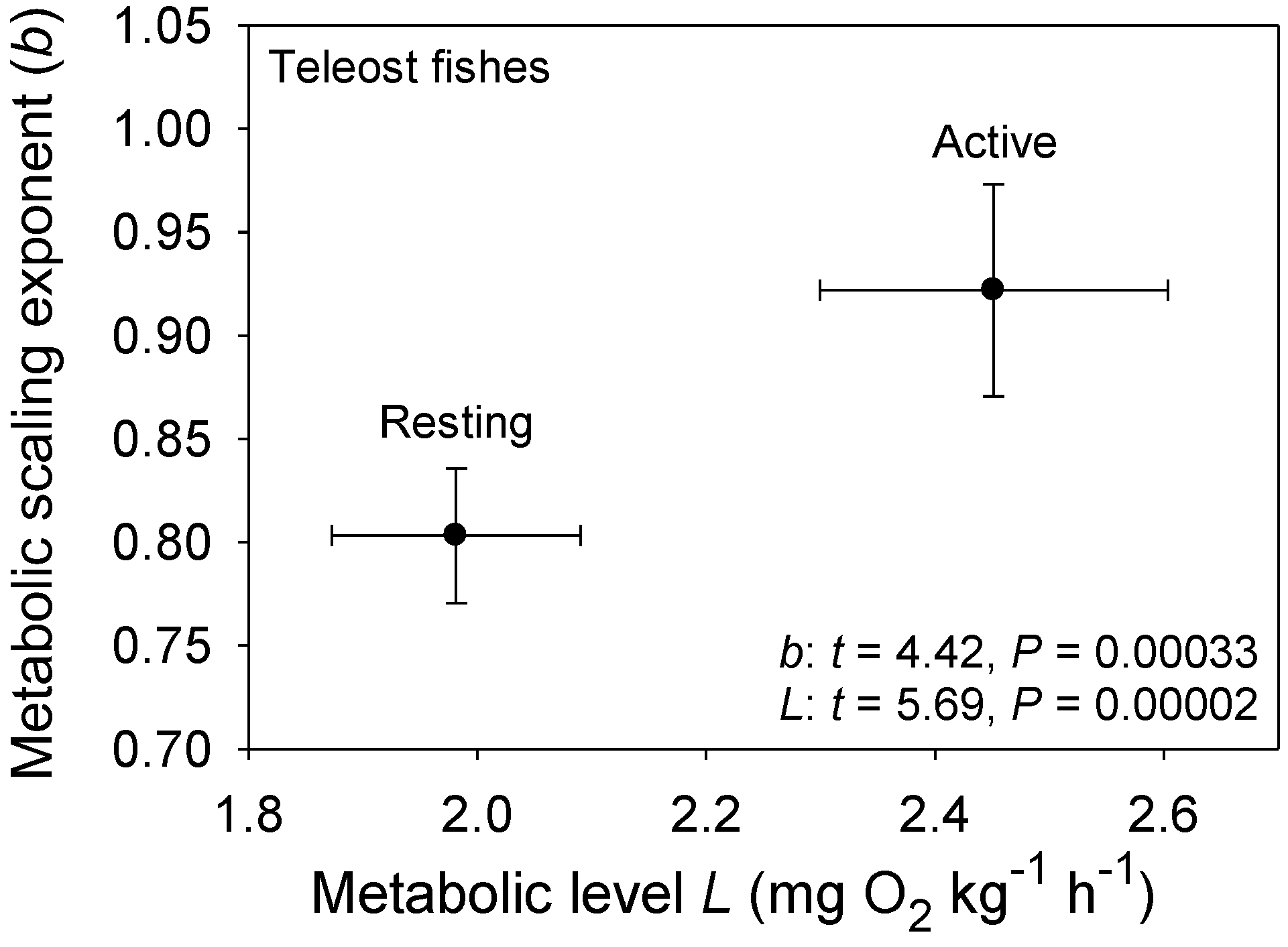
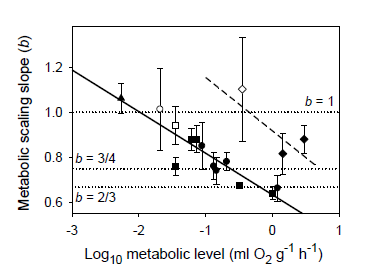
3.2. Effects of Taxonomic or Ecological Lifestyle Differences
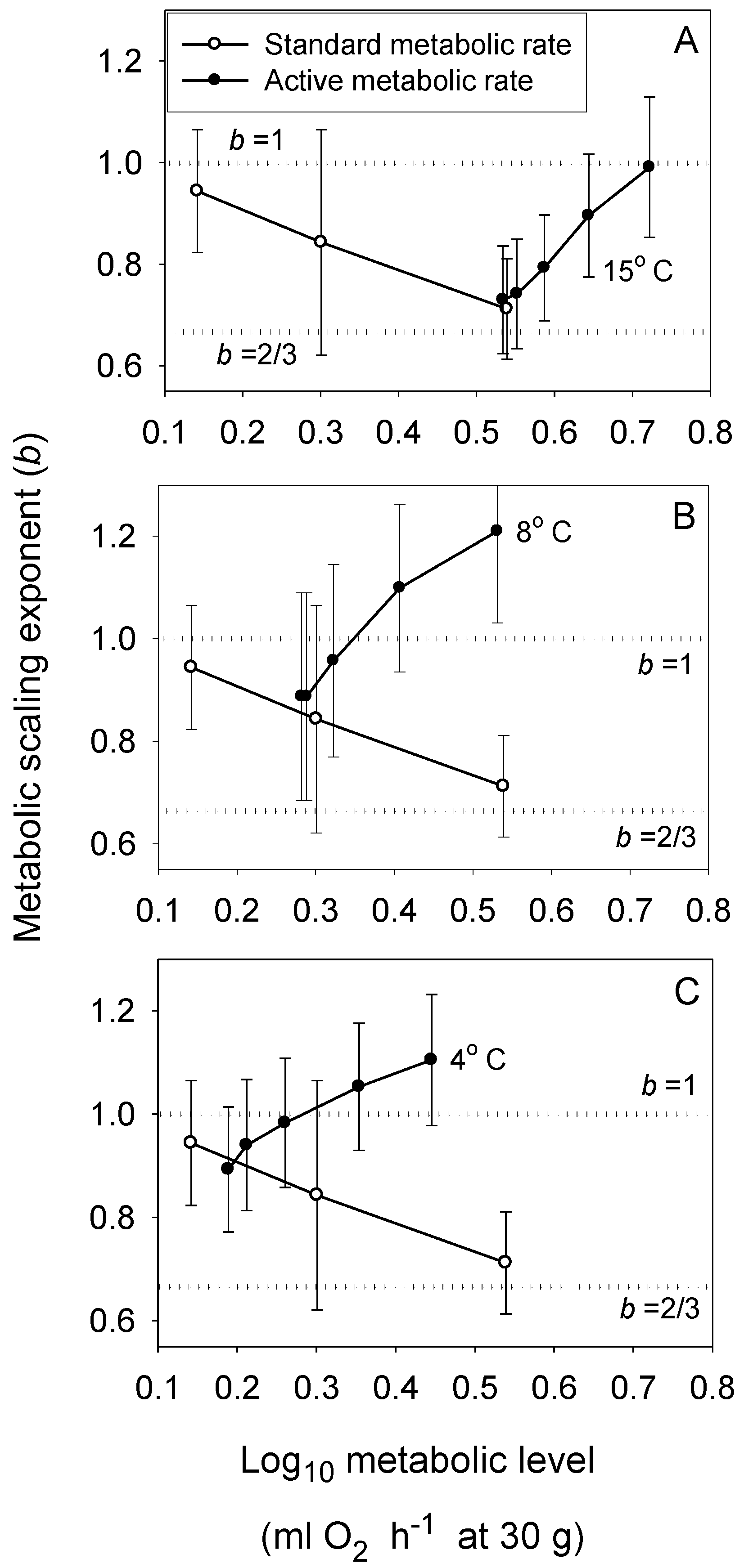

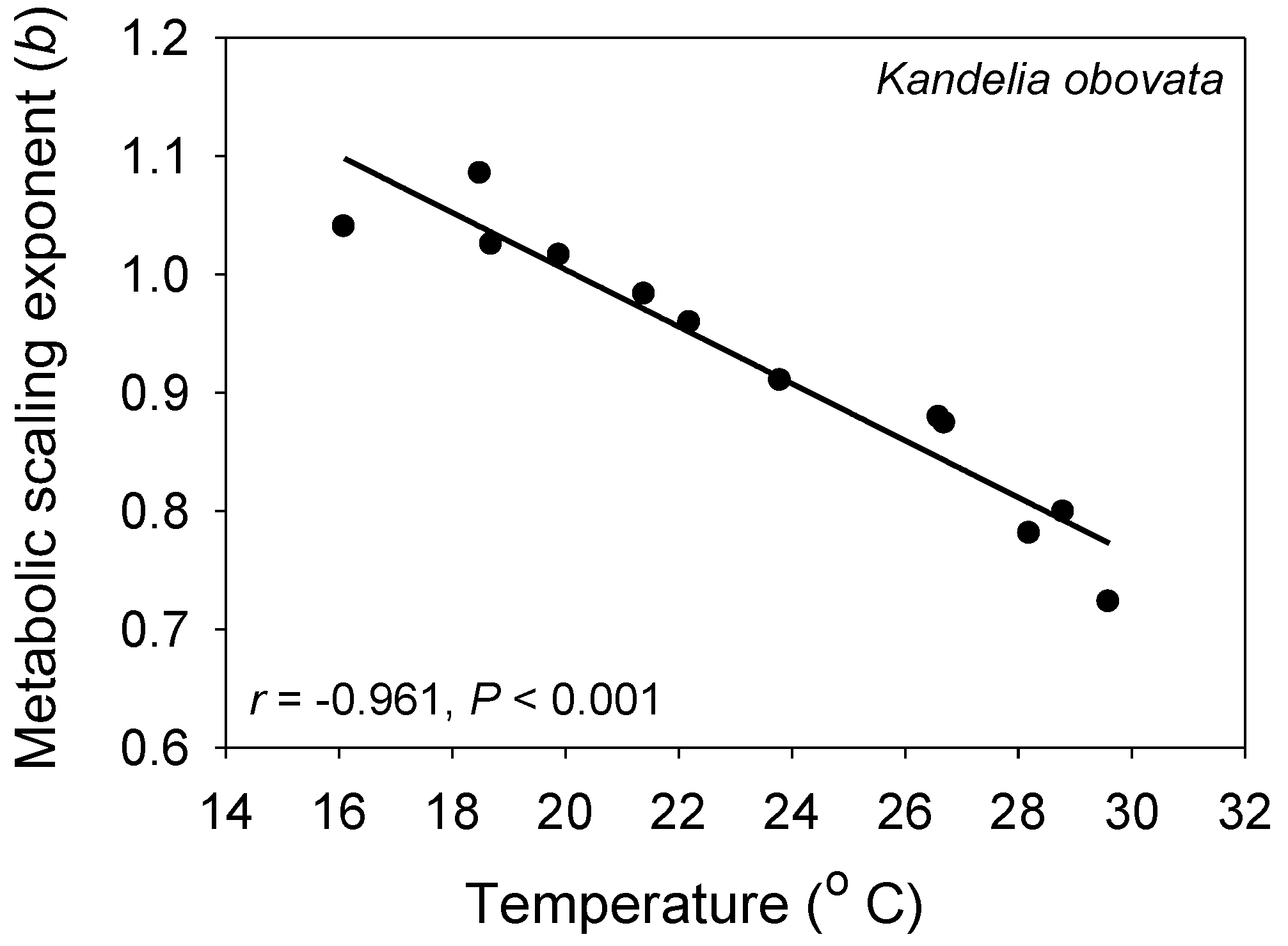
3.3. Effects of Environmental Conditions
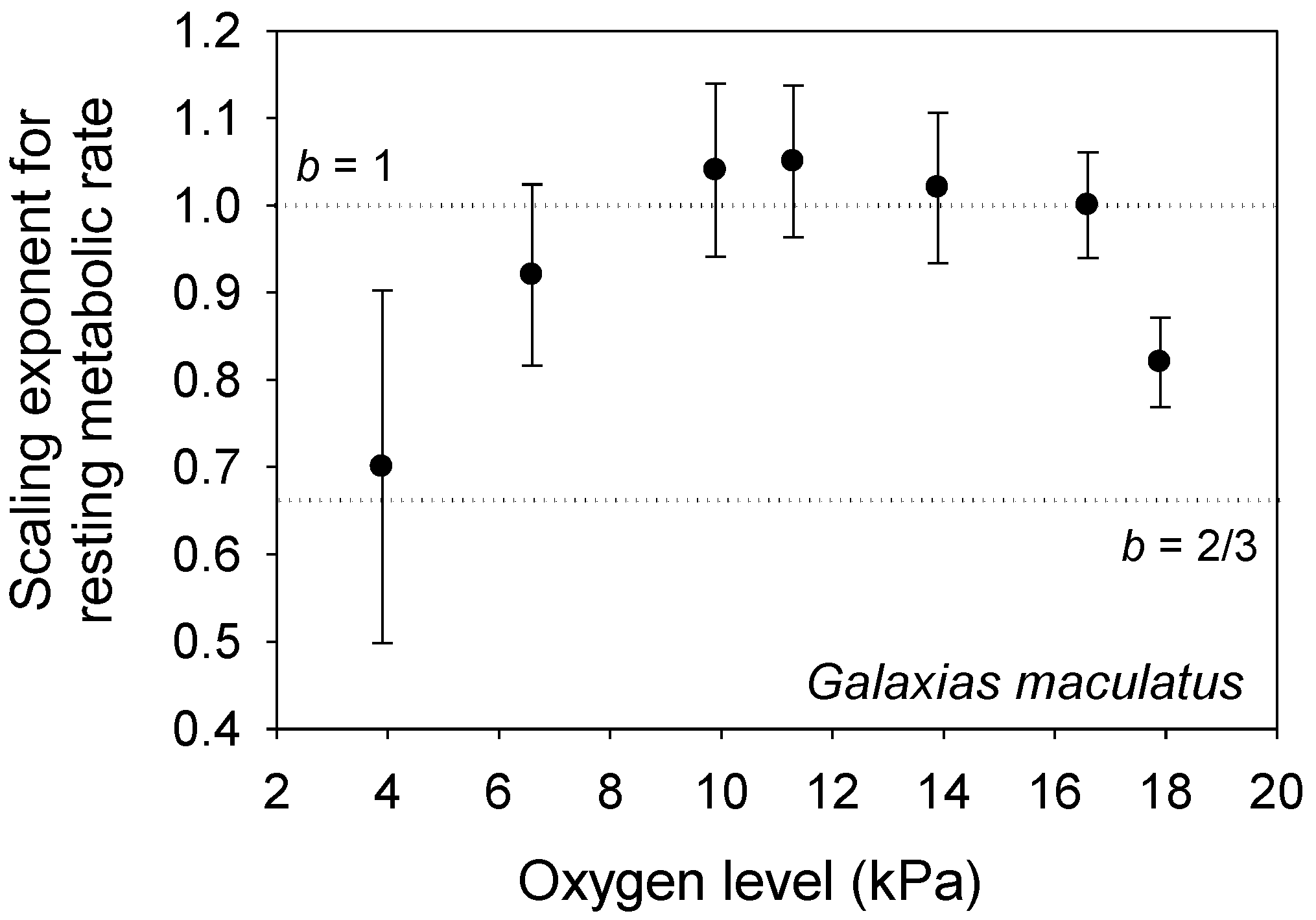
4. Reassessing Predictions of the MLBH in the Context of the CMT
5. Outlook for the MLBH: Future Research Directions
5.1. Factors Affecting L and b and Their Mechanistic Basis
5.2. Quantitative Extensions of the MLBH
5.3. Hierarchical Expansion of the MLBH
5.4. Synthesizing the MLBH with Other Models and Theories

5.5. Applying the MLBH to Other Biological Processes
6. Conclusions
Acknowledgements
Conflicts of Interest
References
- Kleiber, M. Body size and metabolism. Hilgardia 1932, 6, 315–353. [Google Scholar] [CrossRef]
- Hemmingsen, A.M. Energy metabolism as related to body size and respiratory surfaces, and its evolution. Rep. Steno Mem. Hosp. Nord. Insulin Lab. 1960, 9, 1–110. [Google Scholar]
- Peters, R.H. The Ecological Implications of Body Size; Cambridge University Press: Cambridge, UK, 1983. [Google Scholar]
- Schmidt-Nielsen, K. Scaling: Why Is Animal Size So Important? Cambridge University Press: New York, NY, USA, 1984. [Google Scholar]
- Savage, V.M.; Gillooly, J.F.; Woodruff, W.H.; West, G.B.; Allen, A.P.; Enquist, B.J.; Brown, J.H. The predominance of quarter-power scaling in biology. Funct. Ecol. 2004, 18, 257–282. [Google Scholar] [CrossRef]
- McMahon, T.A. Size and shape in biology. Science 1973, 179, 1201–1204. [Google Scholar] [CrossRef] [PubMed]
- Barenblatt, G.I.; Monin, A.S. Similarity principles for the biology of pelagic animals. Proc. Natl. Acad. Sci. USA 1983, 80, 3540–3542. [Google Scholar] [CrossRef] [PubMed]
- Patterson, M.R. A mass transfer explanation of metabolic scaling relations in some aquatic invertebrates and algae. Science 1992, 255, 1421–1423. [Google Scholar] [CrossRef] [PubMed]
- West, G.B.; Brown, J.H.; Enquist, B.J. A general model for the origin of allometric scaling laws in biology. Science 1997, 276, 122–126. [Google Scholar] [CrossRef] [PubMed]
- West, G.B.; Brown, J.H.; Enquist, B.J. The fourth dimension of life: Fractal geometry and allometric scaling of organisms. Science 1999, 284, 1677–1679. [Google Scholar] [CrossRef] [PubMed]
- Banavar, J.R.; Maritan, A.; Rinaldo, A. Size and form in efficient transportation networks. Nature 1999, 399, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Banavar, J.R.; Moses, M.E.; Brown, J.H.; Damuth, J.; Rinaldo, A.; Sibly, R.M.; Maritan, A. A general basis for quarter-power scaling in animals. Proc. Natl. Acad. Sci. USA 2010, 107, 15816–15820. [Google Scholar] [CrossRef] [PubMed]
- Banavar, J.R.; Cooke, T.J.; Rinaldo, A.; Maritan, A. Form, function, and evolution of living organisms. Proc. Natl. Acad. Sci. USA 2014, 111, 3332–3337. [Google Scholar] [CrossRef] [PubMed]
- Bejan, A. The tree of convective heat streams: Its thermal insulation function and the predicted 3/4-power relation between body heat loss and body size. Int. J. Heat Mass Transf. 2001, 44, 699–704. [Google Scholar] [CrossRef]
- Da Silva, J.K.L.; Garcia, G.J.; Barbosa, L.A. Allometric scaling laws of metabolism. Phys. Life Rev. 2006, 3, 229–261. [Google Scholar] [CrossRef]
- Da Silva, J.K.; Barbosa, L.A. Non-universal interspecific allometric scaling of metabolism. Braz. J. Phys. 2009, 39, 699–706. [Google Scholar]
- Dodds, P.S. On the optimal form of branching supply and collection networks. Phys. Rev. Lett. 2010, 104. [Google Scholar] [CrossRef] [PubMed]
- Demetrius, L.; Tuszynski, J.A. Quantum metabolism explains the allometric scaling of metabolic rates. J. R. Soc. Interface 2010, 7, 507–514. [Google Scholar] [CrossRef] [PubMed]
- West, D.; West, B.J. Statistical origin of allometry. Europhys. Lett. 2011, 94. [Google Scholar] [CrossRef]
- West, B.J. A fractional probability calculus view of allometry. Systems 2014, 2, 89–118. [Google Scholar] [CrossRef]
- Glazier, D.S. Beyond the “3/4-power law”: Variation in the intra- and interspecific scaling of metabolic rate in animals. Biol. Rev. 2005, 80, 611–662. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S. A unifying explanation for diverse metabolic scaling in animals and plants. Biol. Rev. 2010, 85, 111–138. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S. Metabolic scaling in complex living systems. Systems 2014, in press. [Google Scholar]
- White, C.R.; Kearney, M.R. Determinants of inter-specific variation in basal metabolic rate. J. Comp. Physiol. B 2013, 183, 1–26. [Google Scholar] [CrossRef] [PubMed]
- White, C.R.; Kearney, M.R. Metabolic scaling in animals: Methods, empirical results, and theoretical explanations. Compr. Physiol. 2014, 4, 231–256. [Google Scholar] [PubMed]
- McNab, B.K. An analysis of the factors that influence the level and scaling of mammalian BMR. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2008, 151, 5–28. [Google Scholar] [CrossRef] [PubMed]
- McNab, B.K. Ecological factors affect the level and scaling of avian BMR. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2009, 152, 22–45. [Google Scholar] [CrossRef] [PubMed]
- Sibly, R.M.; Brown, J.H.; Kodric-Brown, A. Metabolic Ecology: A Scaling Approach; Wiley-Blackwell: Oxford, UK, 2012. [Google Scholar]
- Glazier, D.S. Is metabolic rate a universal “pacemaker” for biological processes? Biol. Rev. 2014. [Google Scholar] [CrossRef]
- Glazier, D.S. Effects of metabolic level on the body size scaling of metabolic rate in birds and mammals. Proc. R. Soc. B 2008, 275, 1405–1410. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.W. On Growth and Form, Volume I; Cambridge University Press: Cambridge, UK, 1942. [Google Scholar]
- Von Bertalanffy, L. Quantitative laws in metabolism and growth. Q. Rev. Biol. 1957, 32, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Kleiber, M. The Fire of Life; Wiley: New York, NY, USA, 1961. [Google Scholar]
- Gould, S.J. Allometry and size in ontogeny and phylogeny. Biol. Rev. 1966, 41, 587–638. [Google Scholar] [CrossRef] [PubMed]
- Davison, J. Body weight, cell surface, and metabolic rate in anuran Amphibia. Biol. Bull. 1955, 109, 407–419. [Google Scholar] [CrossRef]
- Kozłowski, J.; Konarzewski, M.; Gawelczyk, A.T. Cell size as a link between noncoding DNA and metabolic rate scaling. Proc. Natl. Acad. Sci. USA 2003, 100, 14080–14085. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S.; Powell, M.G.; Deptola, T.J. Body-size scaling of metabolic rate in the trilobite Eldredgeops rana. Paleobiology 2013, 39, 109–122. [Google Scholar] [CrossRef]
- Kooijman, S.A.L.M. Dynamic Energy Budget Theory for Metabolic Organization; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Schoombie, R.E.; Boardman, L.; Groenewald, B.; Glazier, D.S.; van Daalen, C.E.; Clusella-Trullas, S.; Terblanche, J.S. High metabolic and water-loss rates in caterpillar aggregations: Evidence against the resource-conservation hypothesis. J. Exp. Biol. 2013, 216, 4321–4325. [Google Scholar] [CrossRef] [PubMed]
- Maino, J.L.; Kearney, M.R.; Nisbet, R.M.; Kooijman, S.A.L.M. Reconciling theories for metabolic scaling. J. Anim. Ecol. 2013, 83, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Rubner, M. Über den Einfluss der Körpergrösse auf Stoff- und Kraftwechsel. Z. Biol. 1883, 19, 535–562. [Google Scholar]
- Okie, J.G. General models for the spectra of surface area scaling strategies of cells and organisms: Fractality, geometric dissimilitude, and internalization. Am. Nat. 2013, 181, 421–439. [Google Scholar] [CrossRef] [PubMed]
- Hirst, A.G; Glazier, D.S.; Atkinson, D. Body shape-shifting during growth permits tests that distinguish between competing geometric theories of metabolic scaling. Ecol. Lett. 2014, 17, 1274–1281. [Google Scholar] [CrossRef] [PubMed]
- Killen, S.S.; Atkinson, D.; Glazier, D.S. The intraspecific scaling of metabolic rate with body mass in fishes depends on lifestyle and temperature. Ecol. Lett. 2010, 13, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Carey, N.; Sigwart, J.D.; Richards, J.G. Economies of scaling: More evidence that allometry of metabolism is linked to activity, metabolic rate and habitat. J. Exp. Mar. Biol. Ecol. 2013, 439, 7–14. [Google Scholar] [CrossRef]
- Demetrius, L. The origin of allometric scaling laws in biology. J. Theor. Biol. 2006, 243, 455–467. [Google Scholar] [CrossRef] [PubMed]
- McNab, B.K. Complications inherent in scaling the basal rate of metabolism in mammals. Q. Rev. Biol. 1988, 63, 25–54. [Google Scholar] [CrossRef] [PubMed]
- West, G.B.; Brown, J.H. The origin of allometric scaling laws in biology from genomes to ecosystems: Towards a quantitative unifying theory of biological structure and organization. J. Exp. Biol. 2005, 208, 1575–1592. [Google Scholar] [CrossRef] [PubMed]
- Price, C.A.; Enquist, B.J.; Savage, V.M. A general model for allometric covariation in botanical form and function. Proc. Natl. Acad. Sci. USA 2007, 104, 13204–13209. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S. The 3/4-power law is not universal: Evolution of isometric, ontogenetic metabolic scaling in pelagic animals. BioScience 2006, 56, 325–332. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, Y.; Liu, S.; Wang, W.; Luo, Y. Intraspecific scaling of the resting and maximum metabolic rates of the Crucian Carp (Carassius auratus). PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Killen, S.S.; Costa, I.; Brown, J.A.; Gamperl, A.K. Little left in the tank: Metabolic scaling in marine teleosts and its implications for aerobic scope. Proc. R. Soc. Lond. B 2007, 274, 431–438. [Google Scholar] [CrossRef]
- Glazier, D.S. Activity affects intraspecific body-size scaling of metabolic rate in ectothermic animals. J. Comp. Physiol. B 2009, 179, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Ohlberger, J.; Staaks, G.; Hölker, F. Effects of temperature, swimming speed and body mass on standard and active metabolic rate in vendace (Coregonus albula). J. Comp. Physiol. B 2007, 177, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Wiersma, P.; Chappell, M.A.; Williams, J.B. Cold-and exercise-induced peak metabolic rates in tropical birds. Proc. Natl. Acad. Sci. USA 2007, 104, 20866–20871. [Google Scholar] [CrossRef] [PubMed]
- Clarke, A.; Pörtner, H.-O. Temperature, metabolic power and the evolution of endothermy. Biol. Rev. 2010, 85, 703–727. [Google Scholar] [PubMed]
- Snelling, E.P.; Seymour, R.S.; Matthews, P.G.D.; Runciman, S.; White, C.R. Scaling of resting and maximum hopping rate throughout the life cycle of the locust Locusta migratoria. J. Exp. Biol. 2011, 214, 3218–3224. [Google Scholar] [CrossRef] [PubMed]
- Dlugosz, E.M.; Chappell, M.A.; Meek, T.H.; Szafrańska, P.A.; Zub, K.; Konarzewski, M.; Jones, J.H.; Bicudo, J.E.P.W.; Nespolo, R.F.; Careau, V.; et al. Phylogenetic analysis of mammalian maximal oxygen consumption during exercise. J. Exp. Biol. 2013, 216, 4712–4721. [Google Scholar] [CrossRef] [PubMed]
- Brett, J.R. The relation of size to rate of oxygen consumption and sustained swimming speed of sockeye salmon (Oncorhynchus nerka). J. Fish. Res. Board Can. 1965, 22, 1491–1501. [Google Scholar] [CrossRef]
- Rao, G.M.M. Influence of activity and salinity on the weight-dependent oxygen consumption of the rainbow trout Salmo gairdneri. Mar. Biol. 1971, 8, 205–212. [Google Scholar] [CrossRef]
- Tarby, M.J. Metabolic expenditure of walleye (Stizostedion vitreum vitreum) as determined by rate of oxygen consumption. Can. J. Zool. 1981, 59, 882–889. [Google Scholar] [CrossRef]
- Armstrong, J.D.; Priede, I.G.; Lucas, M.C. The link between respiratory capacity and changing metabolic demands during growth of northern pike, Esox lucius L. J. Fish Biol. 1992, 41, 65–75. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, Q.; Liu, S.; He, D.; Wei, G.; Luo, Y. Intraspecific mass scaling of metabolic rates in grass carp (Ctenopharyngodon idellus). J. Comp. Physiol. B 2014, 184, 347–354. [Google Scholar] [CrossRef] [PubMed]
- De Souza, S.C.R.; de Carvalho, J.E.; Abe, A.S.; Bicudo, J.E.P.; Bianconcini, M.S. Seasonal metabolic depression, substrate utilisation and changes in scaling patterns during the first year cycle of tegu lizards (Tupinambis merianae). J. Exp. Biol. 2004, 207, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Secor, S.M. Specific dynamic action: A review of the postprandial metabolic response. J. Comp. Physiol. B 2009, 179, 1–56. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.A. Field metabolic rate and body size. J. Exp. Biol. 2005, 1621, 1621–1625. [Google Scholar] [CrossRef]
- Riek, A. Relationship between field metabolic rate and body weight in mammals: Effect of the study. J. Zool. 2008, 276, 187–194. [Google Scholar] [CrossRef]
- Speakman, J.R.; Król, E. Maximal heat dissipation capacity and hyperthermia risk: Neglected key factors in the ecology of endotherms. J. Anim. Ecol. 2010, 79, 726–746. [Google Scholar] [PubMed]
- Hudson, L.N.; Isaac, N.J.; Reuman, D.C. The relationship between body mass and field metabolic rate among individual birds and mammals. J. Anim. Ecol. 2013, 82, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- White, C.R.; Phillips, N.F.; Seymour, R.S. The scaling and temperature dependence of vertebrate metabolism. Biol. Lett. 2006, 2, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.J.M.; Ruhl, H.A.; Hawkins, L.E.; Hauton, C.; Boorman, B.; Billett, D.S. Deep-sea echinoderm oxygen consumption rates and an interclass comparison of metabolic rates in Asteroidea, Crinoidea, Echinoidea, Holothuroidea and Ophiuroidea. J. Exp. Biol. 2011, 214, 2512–2521. [Google Scholar] [CrossRef] [PubMed]
- Chown, S.L.; Marais, E.; Terblanche, J.S.; Klok, C.J.; Lighton, J.R.B.; Blackburn, T.M. Scaling of insect metabolic rate is inconsistent with the nutrient supply network model. Funct. Ecol. 2007, 21, 282–290. [Google Scholar] [CrossRef]
- Swanson, D.L.; Bozinovic, F. Metabolic capacity and the evolution of biogeographic patterns in oscine and suboscine passerine birds. Physiol. Biochem. Zool. 2011, 84, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S. Metabolic level and size scaling of rates of respiration and growth in unicellular organisms. Funct. Ecol. 2009, 23, 963–968. [Google Scholar] [CrossRef]
- Lighton, J.; Brownell, P.; Joos, B.; Turner, R. Low metabolic rate in scorpions: Implications for population biomass and cannibalism. J. Exp. Biol. 2001, 204, 607–613. [Google Scholar] [PubMed]
- Clusella-Trullas, S.; Spotila, J.R.; Paladino, F.V. Energetics during hatchling dispersal of the olive ridley turtle Lepidochelys olivacea using doubly labeled water. Physiol. Biochem. Zool. 2006, 79, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Lighton, J.R.; Fielden, L.J. Mass scaling of standard metabolism in ticks: A valid case of low metabolic rates in sit-and-wait strategists. Physiol. Zool. 1995, 68, 43–62. [Google Scholar]
- Marsden, I.D.; Shumway, S.E.; Padilla, D.K. Does size matter? The effects of body size and declining oxygen tension on oxygen uptake in gastropods. J. Mar. Biol. Assoc. UK 2012, 92, 1603–1617. [Google Scholar] [CrossRef]
- Isaac, N.J.B.; Carbone, C. Why are metabolic scaling exponents so controversial? Quantifying variance and testing hypotheses. Ecol. Lett. 2010, 13, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S. Ontogenetic body-mass scaling of resting metabolic rate covaries with species-specific metabolic level and body size in spiders and snakes. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2009, 153, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Drazen, J.C.; Yeh, J.; Friedman, J.; Condon, N. Metabolism and enzyme activities of hagfish from shallow and deep water of the Pacific Ocean. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2011, 159, 182–187. [Google Scholar] [CrossRef] [PubMed]
- DeVries, Z.C.; Appel, A.G. Standard metabolic rates of Lepisma saccharina and Thermobia domestica: Effects of temperature and mass. J. Insect Physiol. 2013, 59, 638–645. [Google Scholar] [CrossRef] [PubMed]
- DeVries, Z.C.; Kells, S.A.; Appel, A.G. Standard metabolic rate of the bed bug, Cimex lectularius: Effects of temperature, mass, and life stage. J. Insect Physiol. 2013, 59, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Koelsch, G.; Krause, A. Oxygen consumption of the aquatic leaf beetles Macroplea mutica and Macroplea appendiculata is low and not influenced by salinity. Physiol. Ent. 2011, 36, 111–119. [Google Scholar] [CrossRef]
- Šustr, V.; Pižl, V. Oxygen consumption of the earthworm species Dendrobaena mrazeki. Eur. J. Soil Biol. 2009, 45, 478–482. [Google Scholar] [CrossRef]
- Bridges, C.R.; Brand, A.R. Oxygen consumption and oxygen-independence in marine crustaceans. Mar. Ecol. Prog. Ser. 1980, 2, 133–141. [Google Scholar] [CrossRef]
- DeLong, J.P.; Okie, J.G.; Moses, M.E.; Sibly, R.M.; Brown, J.H. Shifts in metabolic scaling, production, and efficiency across major evolutionary transitions of life. Proc. Natl. Acad. Sci. USA 2010, 107, 12941–12945. [Google Scholar] [CrossRef] [PubMed]
- Glazier, D.S.; Butler, E.M.; Lombardi, S.A.; Deptola, T.J.; Reese, A.J.; Satterthwaite, E.V. Ecological effects on metabolic scaling: Amphipod responses to fish predators in freshwater springs. Ecol. Monogr. 2011, 81, 599–618. [Google Scholar] [CrossRef]
- Ohlberger, J.; Mehner, T.; Staaks, G.; Hölker, F. Intraspecific temperature dependence of the scaling of metabolic rate with body mass in fishes and its ecological implications. Oikos 2012, 121, 245–251. [Google Scholar]
- Gifford, M.E.; Clay, T.A.; Peterman, W.E. The effects of temperature and activity on intraspecific scaling of metabolic rates in a lungless salamander. J. Exp. Zool. A Ecol. Genet. Physiol. 2013, 319, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Ivleva, I.V. The dependence of crustacean respiration rate on body mass and habitat temperature. Int. Rev. Gesamten Hydrobiol. Hydrogr. 1980, 65, 1–47. [Google Scholar] [CrossRef]
- Nilsson, G.E.; Östlund‐Nilsson, S. Does size matter for hypoxia tolerance in fish? Biol. Rev. 2008, 83, 173–189. [Google Scholar]
- Hoque, A.T.M.R.; Sharma, S.; Suwa, R.; Mori, S.; Hagihara, A. Seasonal variation in the size-dependent respiration of mangroves Kandelia obovata. Mar. Ecol. Prog. Ser. 2010, 404, 31–37. [Google Scholar] [CrossRef]
- Read, K.R.H. Respiration of the bivalved molluscs Mytilus edulis L. and Brachidontes demissus plicatulus Lamarck as a function of size and temperature. Comp. Biochem. Physiol. 1962, 7, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Farmanfarmaian, A. The Respiratory Physiology of Echinoderms. In Physiology of Echinodermata; Wiley: New York, NY, USA, 1966; pp. 245–265. [Google Scholar]
- Åkerlund, G. Oxygen consumption of the ampullariid snail Marisa cornuarietis L. in relation to body weight and temperature. Oikos 1969, 20, 529–533. [Google Scholar] [CrossRef]
- Roy, A. Analyse des facteurs du taux de metabolism chez la limace Arion circumscriptus. Rev. Can. Biol. 1969, 28, 33–43. [Google Scholar] [PubMed]
- Paine, R.T. Energy flow in a natural population of the herbivorous gastropod Tegula funebralis. Limnol. Oceanogr. 1971, 16, 86–98. [Google Scholar] [CrossRef]
- Dame, R.F. The ecological energies of growth, respiration and assimilation in the intertidal American oyster Crassostrea virginica. Mar. Biol. 1972, 17, 243–250. [Google Scholar] [CrossRef]
- Armitage, K.B.; Wall, T.J. The effects of body size, starvation and temperature acclimation on oxygen consumption of the crayfish Orconectes nais. Comp. Biochem. Physiol. A Physiol. 1982, 73, 63–68. [Google Scholar] [CrossRef]
- Xie, X.; Sun, R. The bioenergetics of the southern catfish (Silurus meridionalis Chen). I. Resting metabolic rate as a function of body weight and temperature. Physiol. Zool. 1990, 63, 1181–1195. [Google Scholar]
- Al-Sadoon, M.K.; Abdo, N.M. Temperature and body mass effects on the metabolic rate of Acanthodactylus schmidti Weigmann (Reptilia: Lacertidae). J. Arid Environ. 1991, 21, 351–361. [Google Scholar]
- Vladimirova, I.G.; Kleimenov, S.Y.; Radzinskaya, L.I. The relation of energy metabolism and body weight in bivalves (Mollusca: Bivalvia). Biol. Bull. Russ. Acad. Sci. 2003, 30, 392–399. [Google Scholar] [CrossRef]
- Shimauchi, H.; Uye, S.I. Excretion and respiration rates of the scyphomedusa Aurelia aurita from the Inland Sea of Japan. J. Oceanogr. 2007, 63, 27–34. [Google Scholar] [CrossRef]
- Jiang, A.; Guo, J.; Cai, W.; Wang, C. Oxygen consumption of the ascidian Styela clava in relation to body mass, temperature and salinity. Aquacult. Res. 2008, 39, 1562–1568. [Google Scholar] [CrossRef]
- Doyle, S.R.; Momo, F.R.; Brêthes, J.C.; Ferreyra, G.A. Metabolic rate and food availability of the Antarctic amphipod Gondogeneia antarctica (Chevreux 1906): Seasonal variation in allometric scaling and temperature dependence. Polar Biol. 2012, 35, 413–424. [Google Scholar] [CrossRef]
- Weldon, C.W.; Daniels, S.R.; Clusella-Trullas, S.; Chown, S.L. Metabolic and water loss rates of two cryptic species in the African velvet worm genus Opisthopatus (Onychophora). J. Comp. Physiol. B 2013, 183, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Grigoriou, P.; Richardson, C.A. Effect of body mass, temperature and food deprivation on oxygen consumption rate of common cuttlefish Sepia officinalis. Mar. Biol. 2009, 156, 2473–2481. [Google Scholar]
- Crispin, T.S.; White, C.R. Effect of thermal acclimation on organ mass, tissue respiration, and allometry in Leichhardtian River prawns Macrobrachium tolmerum (Riek, 1951). Physiol. Biochem. Zool. 2013, 86, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.T.; Mims, S.D.; Wright, R.A. Effects of body mass and water temperature on routine metabolism of American paddlefish Polyodon spathula. J. Fish Biol. 2013, 82, 1269–1280. [Google Scholar] [CrossRef] [PubMed]
- Newell, R.C. Factors affecting the respiration of intertidal invertebrates. Am. Zool. 1973, 13, 513–528. [Google Scholar]
- Doyle, S.R.; Momo, F.R. Effects of body weight and temperature on the metabolic rate of Hyalella curvispina Shoemaker, 1942 (Amphipoda). Crustaceana 2009, 82, 1423–1439. [Google Scholar] [CrossRef]
- Luo, Y.P.; Wang, Q.Q. Effects of body mass and temperature on routine metabolic rate of juvenile largemouth bronze gudgeon Coreius guichenoti. J. Fish Biol. 2012, 80, 842–851. [Google Scholar] [CrossRef] [PubMed]
- Newell, R.C.; Ahsanullah, M.; Pye, V.I. Aerial and aquatic respiration in the shore crab Carcinus maenas (L.). Comp. Biochem. Physiol. A Physiol. 1972, 43, 239–252. [Google Scholar] [CrossRef]
- Mladenova, A. Importance of the temperature for the energetic metabolism of fresh-water isopod [Asellus aquaticus (L.)]. Russ. J. Aquat. Ecol. 1993, 2, 55–63. [Google Scholar]
- Downs, C.J.; Hayes, J.P.; Tracy, C.R. Scaling metabolic rate with body mass and inverse body temperature: A test of the Arrhenius fractal supply model. Funct. Ecol. 2008, 22, 239–244. [Google Scholar] [CrossRef]
- Irlich, U.M.; Terblanche, J.S.; Blackburn, T.M.; Chown, S.L. Insect rate‐temperature relationships: Environmental variation and the metabolic theory of ecology. Am. Nat. 2009, 174, 819–835. [Google Scholar] [CrossRef] [PubMed]
- Dell, A.I.; Pawar, S.; Savage, V.M. Systematic variation in the temperature dependence of physiological and ecological traits. Proc. Natl. Acad. Sci. USA 2011, 108, 10591–10596. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, D.G. Influence of temperature and duration of acclimation, time of day, sex and body weight on metabolic rates in the hylid frog, Acris crepitans. Comp. Biochem. Physiol. 1969, 31, 555–570. [Google Scholar] [CrossRef] [PubMed]
- Lease, H.M.; Klok, C.J.; Kaiser, A.; Harrison, J.F. Body size is not critical for critical PO2 in scarabaeid and tenebrionid beetles. J. Exp. Biol. 2012, 215, 2524–2533. [Google Scholar] [PubMed]
- Everett, M.V.; Crawford, D.L. Adaptation vs. allometry: Population and body mass effects on hypoxic metabolism in Fundulus grandis. Physiol. Biochem. Zool. 2010, 83, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Urbina, M.A.; Glover, C.N. Relationship between fish size and metabolic rate in the oxyconforming inanga Galaxias maculatus reveals size-dependent strategies to withstand hypoxia. Physiol. Biochem. Zool. 2013, 86, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Seibel, B.A. On the depth and scale of metabolic rate variation: Scaling of oxygen consumption rates and enzymatic activity in the Class Cephalopoda (Mollusca). J. Exp. Biol. 2007, 210, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Giguere, L.A.; Cote, B.; St-Pierre, J.F. Metabolic rates scale isometrically in larval fishes. Mar. Ecol. Prog. Ser. 1988, 50, 13–19. [Google Scholar] [CrossRef]
- Brett, J.R.; Glass, N.R. Metabolic rates and critical swimming speeds of sockeye salmon (Oncorhynchus nerka) in relation to size and temperature. J. Fish. Res. Board Can. 1973, 30, 379–387. [Google Scholar] [CrossRef]
- Paranjape, M.A. Molting and respiration of euphausiids. J. Fish. Board Can. 1967, 24, 1229–1240. [Google Scholar] [CrossRef]
- Mori, S.; Yamaji, K.; Ishida, A.; Prokushkin, S.G.; Masyagina, O.V.; Hagihara, A.; Rafiqul Hoque, A.T.M.; Suwa, R.; Osawa, A.; Nishizono, T.; et al. Mixed-power scaling of whole-plant respiration from seedlings to giant trees. Proc. Natl. Acad. Sci. USA 2010, 107, 1447–1451. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.L.; Li, T.; Zhong, Q.L.; Wang, G.X. Scaling relationship between tree respiration rates and biomass. Biol. Lett. 2010, 6, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Burton, T.; Killen, S.S.; Armstrong, J.D.; Metcalfe, N.B. What causes intraspecific variation in resting metabolic rate and what are its ecological consequences? Proc. R. Soc. Lond. B 2011, 278, 3465–3473. [Google Scholar]
- Konarzewski, M.; Książek, A. Determinants of intra-specific variation in basal metabolic rate. J. Comp. Physiol. B 2013, 183, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; O’Connor, T.P.; Heshka, S.; Heymsfield, S.B. The reconstruction of Kleiber’s law at the organ-tissue level. J. Nutr. 2001, 131, 2967–2970. [Google Scholar] [PubMed]
- Nakaya, F.; Saito, Y.; Motokawa, T. Switching of metabolic-rate scaling between allometry and isometry in colonial ascidians. Proc. R. Soc. Lond. B 2003, 270, 1105–1113. [Google Scholar] [CrossRef]
- White, C.R.; Kearney, M.R.; Matthews, P.G.; Kooijman, S.A.; Marshall, D.J. A manipulative test of competing theories for metabolic scaling. Am. Nat. 2011, 178, 746–754. [Google Scholar] [CrossRef] [PubMed]
- Szarski, H. Cell size and the concept of wasteful and frugal evolutionary strategies. J. Theor. Biol. 1983, 105, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Gregory, T.R. A bird’s-eye view of the C-value enigma: Genome size, cell size, and metabolic rate in the class Aves. Evolution 2002, 56, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Starostová, Z.; Kubička, L.; Konarzewski, M.; Kozłowski, J.; Kratochvíl, L. Cell size but not genome size affects scaling of metabolic rate in eyelid geckos. Am. Nat. 2009, 174, E100–E105. [Google Scholar] [CrossRef] [PubMed]
- Maciak, S.; Janko, K.; Kotusz, J.; Choleva, L.; Boroń, A.; Juchno, D.; Kujawa, R.; Kozłowski, J.; Konarzewski, M. Standard metabolic rate (SMR) is inversely related to erythrocyte and genome size in allopolyploid fish of the Cobitis taenia hybrid complex. Funct. Ecol. 2011, 25, 1072–1078. [Google Scholar] [CrossRef]
- Starostová, Z.; Konarzewski, M.; Kozłowski, J.; Kratochvíl, L. Ontogeny of metabolic rate and red blood cell size in eyelid geckos: Species follow different paths. PLoS One 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Davison, J. An analysis of cell growth and metabolism in the crayfish (Procambarus alleni). Biol. Bull. 1956, 110, 264–273. [Google Scholar] [CrossRef]
- Riveros, A.J.; Enquist, B.J. Metabolic scaling in insects supports the predictions of the WBE model. J. Insect Physiol. 2011, 57, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Gregory, T.R. Animal Genome Size Database. 2009. Available online: http://www.genomesize.com/ (accessed on 20 May 2014).
- Gunther, B. Dimensional analysis and theory of biological similarity. Physiol. Rev. 1975, 55, 659–699. [Google Scholar] [PubMed]
- Calder, W.A. Size, Function, and Life History; Harvard University Press: Cambridge, MA, USA, 1984. [Google Scholar]
- Wilson, A.E.; Kaul, R.B.; Sarnelle, O. Growth rate consequences of coloniality in a harmful phytoplankter. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Hayward, A.; Gillooly, J.F. The cost of sex: Quantifying energetic investment in gamete production by males and females. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Falster, D.S.; Moles, A.T.; Westoby, M. A general model for the scaling of offspring size and adult size. Am. Nat. 2008, 172, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.J.; Davidson, A.D.; Sibly, R.M.; Brown, J.H. Universal scaling of production rates across mammalian lineages. Proc. R. Soc. Lond. B 2011, 278, 560–566. [Google Scholar] [CrossRef]
- Meiri, S.; Brown, J.H.; Sibly, R.M. The ecology of lizard reproductive output. Glob. Ecol. Biogeogr. 2012, 21, 592–602. [Google Scholar] [CrossRef]
- Sibly, R.M.; Witt, C.C.; Wright, N.A.; Venditti, C.; Jetz, W.; Brown, J.H. Energetics, lifestyle, and reproduction in birds. Proc. Natl. Acad. Sci. USA 2012, 109, 10937–10941. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glazier, D.S. Scaling of Metabolic Scaling within Physical Limits. Systems 2014, 2, 425-450. https://doi.org/10.3390/systems2040425
Glazier DS. Scaling of Metabolic Scaling within Physical Limits. Systems. 2014; 2(4):425-450. https://doi.org/10.3390/systems2040425
Chicago/Turabian StyleGlazier, Douglas S. 2014. "Scaling of Metabolic Scaling within Physical Limits" Systems 2, no. 4: 425-450. https://doi.org/10.3390/systems2040425
APA StyleGlazier, D. S. (2014). Scaling of Metabolic Scaling within Physical Limits. Systems, 2(4), 425-450. https://doi.org/10.3390/systems2040425